Abstract
ZUMA-1 safety management cohort 6 investigated the impact of prophylactic corticosteroids and earlier corticosteroids and/or tocilizumab on the incidence and severity of cytokine release syndrome (CRS) and neurologic events (NEs) following axicabtagene ciloleucel (axi-cel) in patients with relapsed/refractory large B-cell lymphoma (R/R LBCL). Prior analyses of cohort 6 with limited follow-up demonstrated no Grade ≥3 CRS, a low rate of NEs, and high response rates, without negatively impacting axi-cel pharmacokinetics. Herein, long-term outcomes of cohort 6 (N = 40) are reported (median follow-up, 26.9 months). Since the 1-year analysis (Oluwole, et al. Blood. 2022;138[suppl 1]:2832), no new CRS was reported. Two new NEs occurred in two patients (Grade 2 dementia unrelated to axi-cel; Grade 5 axi-cel–related leukoencephalopathy). Six new infections and eight deaths (five progressive disease; one leukoencephalopathy; two COVID-19) occurred. Objective and complete response rates remained at 95% and 80%, respectively. Median duration of response and progression-free survival were reached at 25.9 and 26.8 months, respectively. Median overall survival has not yet been reached. Eighteen patients (45%) remained in ongoing response at data cutoff. With ≥2 years of follow-up, prophylactic corticosteroids and earlier corticosteroids and/or tocilizumab continued to demonstrate CRS improvement without compromising efficacy outcomes, which remained high and durable.
Similar content being viewed by others
Introduction
Axicabtagene ciloleucel (axi-cel), an autologous anti-CD19 chimeric antigen receptor (CAR) T-cell therapy, was approved for relapsed/refractory large B-cell lymphoma (R/R LBLC) after ≥2 lines of systemic therapy based on the ZUMA-1 registrational study in refractory LBCL (NCT02348216) [1,2,3]. ZUMA-1 pivotal cohorts 1 + 2 (N = 101) demonstrated high, durable responses (83% objective response rate [ORR]; 58% complete response [CR] rate), and a manageable safety profile with long-term follow-up (median, 27.1 months) [4]. Grade ≥3 cytokine release syndrome (CRS) and neurologic events (NEs) were reported in 11% and 31% of patients, respectively. With 63.1-months median follow-up, median overall survival (OS) was 25.8 months (95% CI, 12.8–not estimable) and the 5-year OS rate was 43% (95% CI, 33–52%) [5].
The clinical promise of CAR T-cell therapy is challenged by CRS and NEs, which are acute toxicities that can be life-threatening, requiring careful management and monitoring [6, 7]. Therefore, CRS and NE management have been evaluated to optimize safety outcomes without compromising efficacy [6, 7], and several exploratory safety management cohorts were added to ZUMA-1 [8,9,10]. Cohort 6 evaluated the impact of prophylactic corticosteroids and earlier corticosteroid and/or tocilizumab intervention on the incidence and severity of CRS and NEs [10]. With 14.9-months median follow-up, cohort 6 demonstrated lower rates of Grade ≥3 CRS and NEs (no Grade ≥3 CRS; 15% Grade ≥3 NEs) than cohorts 1 + 2, and high, durable response rates (95% ORR, 80% CR, and 53% ongoing responses) [11]. Here, long-term data from ZUMA-1 cohort 6 are reported with at least 2 years of follow-up for all patients, including a competing risk analysis of OS performed after propensity score matching (PSM) of patients in cohort 6 and cohorts 1 + 2.
Methods
Patients and study design
Full study procedures for ZUMA-1 cohort 6 were previously reported [10]. Patient eligibility and additional study design details are noted within the Supplemental methods. Patients in cohort 6 received conditioning chemotherapy for 3 days (cyclophosphamide 500 mg/m2/day and fludarabine 30 mg/m2/day on days –5, –4, and –3) prior to a single intravenous infusion of axi-cel (target dose, 2 × 106 CAR T cells/kg) on day 0. Patients received once-daily corticosteroid prophylaxis (oral dexamethasone 10 mg) on days 0 (before axi-cel), 1, and 2, and earlier corticosteroids and/or tocilizumab for CRS and NE management (Fig. S1). Unlike cohorts 1 + 2, patients in cohort 6 could receive optional bridging therapy after leukapheresis at the investigator’s discretion [10].
Endpoints and assessments
The primary endpoints were incidence and severity of CRS and NEs, which were identified and graded as previously reported [10]. Briefly, severity of CRS was graded per modified Lee 2014 criteria [12]. NEs were identified using a Medical Dictionary for Regulatory Activities version 24.1 search term list that was developed based on a modification of the specific search strategy by Topp et al [13], with severity graded per National Cancer Institute Common Terminology Criteria for Adverse Events version 4.03. Secondary endpoints included adverse event (AE) incidence (Supplemental methods), ORR (partial response [PR] or CR as assessed by investigator per revised International Working Group Response Criteria for Malignant Lymphoma) [14], duration of response (DOR), progression-free survival (PFS), OS, and CAR T-cell levels in blood. The cumulative incidence of non–lymphoma-related mortality was assessed in a post hoc analysis. The associations between pharmacokinetic parameters (median peak CAR T-cell levels and area under the curve within the first 28 days after treatment [AUC0-28]) and severity of CRS and NEs were also examined.
Statistical analysis
For cohort 6, all endpoints were analyzed descriptively; no formal hypothesis was tested [10]. Disease assessment after initiation of new anticancer therapy (excluding stem cell transplant) was not included in DOR or PFS derivations. Descriptive P values, calculated by Wilcoxon 2-sample test, were generated to compare pharmacokinetic parameters with toxicity severity. As previously reported [10], an exploratory PSM analysis [15, 16] was performed to retrospectively compare outcomes for patients in cohort 6 and cohorts 1 + 2 (Supplemental methods). Matched cohorts were identified after balancing for the following key baseline disease characteristics: tumor burden, International Prognostic Index score, number of prior lines of chemotherapy, disease stage, and lactate dehydrogenase level. Patients were selected using 1:1 nearest neighbor propensity score matching and caliper option. The cumulative incidence function was compared between competing risks of lymphoma-related and non–lymphoma-related deaths for OS on matched patients. Additional statistical methods can be found in the Supplement.
Results
Patients
Forty-two patients were enrolled and leukapheresed; 40 received conditioning chemotherapy and axi-cel treatment [10]. Of 21 patients (53%) who received bridging therapy, the most common regimens (used in three or more patients) were corticosteroids (9 [23%]); rituximab with bendamustine and corticosteroids (4 [10%]); and rituximab with bendamustine (3 [8%]) [10]. As of 16 December 2021, median follow-up was 26.9 months (range, 24.0–30.1). Patient and disease characteristics at baseline were previously reported [10].
Safety
All 40 patients reported Grade ≥3 treatment-emergent AEs (TEAEs; Table 1). The most common Grade ≥3 TEAEs were neutropenia (80%), leukopenia (40%), and thrombocytopenia (28%). Serious any-grade TEAEs occurred in 24 patients (60%), and 20 patients (50%) reported Grade ≥3 events (Table S1). Prolonged Grade ≥3 cytopenias (i.e., those present on or after 30 days from axi-cel infusion) were reported in 21 patients (53%) (Table S2).
To date for cohort 6, 24 patients (60%) had any-grade infections (11 [28%] Grade ≥3). Five patients (13%) had COVID-19 infections (3 [8%] Grade ≥3), none related to axi-cel treatment per investigator assessment (Table S3). One additional death due to COVID-19 was reported; the COVID-19 infection was not reported as a Grade 5 AE given that it occurred outside of the protocol-specified AE reporting period. Since the 1-year analysis [11], six new infections were reported, including COVID-19 (Grades 1, 2, and 5 [each n = 1]), Grade 3 Pneumocystis jirovecii pneumonia, Grade 3 unknown infectious episode with inflammatory syndrome, and Grade 2 herpes zoster. The latter three events were axi-cel–related per investigator assessment (Table S4). At month 3, the first assessment of B-cell levels post–axi-cel, 1/18 evaluable patients (6%) in ongoing response had detectable B cells. At 2 years, 5/16 evaluable patients (31%) had detectable B cells (Table S5). To date, 8 patients (20%) had hypogammaglobulinemia; all were Grade 1 (n = 2) or 2 (n = 6). Seven patients (18%) received intravenous immunoglobulin (IVIG) therapy per investigator’s discretion, and all uses were for AE treatment, although one patient also received IVIG for prophylaxis.
Since the 1-year analysis [11], the incidence of CRS was unchanged (Table 2; Supplementary results). No Grade ≥3 CRS events have occurred to date in cohort 6. Two new treatment-emergent NEs were observed in 2 patients since the 1-year analysis [11] (Table 2) and were ongoing at time of data cutoff (Table S6). One patient had Grade 2 dementia that was unrelated to axi-cel per investigator assessment (onset on day 685). The second patient had Grade 5 axi-cel–related (per investigator assessment) leukoencephalopathy that was ultimately fatal on day 815. A brain biopsy performed on day 802 suggested that the underlying etiology of the leukoencephalopathy was infection versus other malignancy. Serology testing of cerebrospinal fluid indicated the presence of antibodies to JC virus, suggesting the event may have been caused by JC virus. However, an autopsy was not performed. The patient was in CR at time of death and died in hospice care. Gamma globulin level was low at 238 mg/dL on day 734; the patient received chronic IVIG support until day 734. Since the Grade 5 event was coded as a NE, the incidence of Grade ≥3 NEs increased from 15% to 18% since the 1-year analysis.
Eight deaths occurred since the 1-year analysis, including five due to progressive disease and three from aforementioned AEs (leukoencephalopathy [n = 1] and COVID-19 [n = 2]). No cases of replication-competent retroviruses or secondary malignancies have been reported thus far in cohort 6.
Efficacy
The ORR was 95% (95% CI, 83–99%) and the CR rate was 80% (95% CI, 64–91%), both unchanged from the 1-year analysis [11]. Among patients who received corticosteroids for prophylaxis only (n = 15) versus for prophylaxis and toxicity management (n = 25), the ORR was 100% (95% CI, 78–100%) versus 92% (95% CI, 74–99%) and CR rates were 73% (95% CI, 45–92%) versus 84% (95% CI, 64–95%), respectively. As previously reported [10], median cumulative cortisone-equivalent corticosteroid doses were 1252 mg and 2504 mg among those who received corticosteroids for prophylaxis only versus prophylaxis and AE management, respectively.
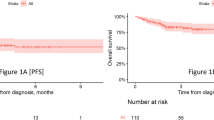
Since the 1-year analysis [11], one responder developed progressive disease and two responders died of AEs (patients’ last disease assessments were CR). Median DOR and PFS were reached at 25.9 months (95% CI, 7.8–not estimable) and 26.8 months (95% CI, 8.7–not estimable), respectively (Figs. 1–2). Median PFS in patients who achieved a best response of CR (n = 32) or PR (n = 6) was 26.8 months (95% CI, 12.2–not estimable) and 6.2 months (95% CI, 2.8–not estimable), respectively (Fig. 2b). Median OS was still not reached (95% CI, 18.9 months–not estimable; Fig. 3). Kaplan–Meier estimates of the 2-year DOR, PFS, and OS rates were 53% (95% CI, 36–68%), 53% (95% CI, 36–67%), and 62% (95% CI, 45–75%), respectively. Cumulative incidence rates of non–lymphoma-related mortality at 1 year and 2 years were 7.7% (95% CI, 1.9–18.9) and 15.4% (95% CI, 6.1–28.5), respectively. Of 18 patients (45%) in ongoing response at data cutoff, all achieved CR as best response.
a Progression-free survival. b Progression-free survival by best overall response subgroup. Disease assessment after initiation of new anticancer therapy (not including stem cell transplant) was not included in the progression-free survival derivation. CI confidence interval, CR complete response, PFS progression-free survival, PR partial response.
Of the 40 patients in cohort 6, 32 were matched to those in cohorts 1 + 2 during PSM analysis. Eight patients from cohort 6 were not included due to nonavailability of matched patients in cohorts 1 + 2. Among the 32 matched patients in cohort 6 and cohorts 1 + 2, the 2-year cumulative incidence rate of lymphoma-related death was 26% (95% CI, 12–42%) and 25% (95% CI, 12–41%), respectively (Fig. S2) [10]. Among matched patients, non–lymphoma-related deaths were observed in 6 patients from cohort 6 and no patients in cohorts 1 + 2. Specifically, 2 patients in cohort 6 died of other reasons, and 4 died due to Grade 5 AEs (Table S7).
Biomarker analyses
By month 24, 14/20 patients (70%) with evaluable blood samples had detectable levels of gene-marked anti-CD19 CAR T cells (Fig. S3A) versus 23/36 patients (64%) in cohorts 1 + 2. Similar to the 1-year analysis of cohort 6 [11], median peak CAR T-cell levels were higher in patients with ongoing response (61 cells/µl [n = 18]) or who relapsed by 2 years (68 cells/µl [n = 18]) versus nonresponders (18 cells/µl [n = 2]; Fig. S3B). A similar trend was observed with CAR T-cell expansion by AUC0-28. Notably, positive associations between median AUC0-28 and CRS severity, and median CAR T-cell peak and AUC0-28 with NE severity were observed (Supplementary results).
Discussion
Based on the cohort 6 primary analysis [10], the United States Food and Drug Administration approved an update to the axi-cel prescribing information to include use of prophylactic corticosteroids for toxicity management across indications [2]. Moreover, the nursing guidelines on CAR T-cell therapy by the European Society for Bone Marrow Transplantation recommend the use of corticosteroids to manage therapy-related CRS and neurotoxicity [17]. With a median follow-up of 26.9 months, the cohort 6 findings reported herein demonstrate the long-term safety profile of axi-cel in R/R LBCL.
Similar to prior analyses [10, 11], the incidence, severity, and duration of CRS were decreased in cohort 6 versus cohorts 1 + 2, and the time to onset was delayed. Although 2 additional NEs were reported in 2 patients since the 1-year analysis for cohort 6 (one dementia and one infection as suspected etiologies) [11], the incidence and severity of NEs remain numerically lower than that reported in cohorts 1 + 2 [4, 10]. It should be noted that cohort 6 allowed for the use of bridging therapy after leukapheresis and before axi-cel, whereas cohorts 1 + 2 did not. Thus, it could be argued that the lower incidence of CRS and NEs in cohort 6 versus cohorts 1 + 2 could be attributable, in part, to better disease control prior to CAR T-cell therapy. However, median tumor burden (as assessed per sum of product diameters of target lesions) and lactate dehydrogenase levels were generally balanced in cohort 6 compared with cohorts 1 + 2 after PSM, where the differences in CRS and NEs remained apparent [18].
Beyond CRS and NEs, generally acute toxicities with early onset, cytopenia and immune deficiency are more frequently reported as late-onset AEs associated with CD19-targeted CAR T-cell therapy [19, 20]. Prolonged cytopenia is commonly reported, indicating a potential class effect [19]. B-cell aplasia represents an on-target/off-tumor effect of anti-CD19 CAR T-cell therapies, and patients may present with hypogammaglobulinemia, potentially requiring IVIG replacement to help mitigate infection risk [4, 19, 20]. Furthermore, corticosteroid use is independently associated with higher risk of infection [21, 22], which, when taken together, highlights infection as a key clinical consideration following CAR T-cell therapy. Incidence of Grade ≥3 prolonged cytopenia was numerically higher between patients in cohort 6 and cohorts 1 + 2 [4] in this unmatched analysis. However, B-cell recovery was observed over time in cohort 6 patients in ongoing response, similar to observations in cohorts 1 + 2 [4], and incidence of Grade ≥3 infections occurred at similar frequency between these patient groups [4]. These findings suggest that the cohort 6 toxicity management strategy did not lead to an added risk of infections versus that previously observed with the ZUMA-1 pivotal cohorts.
Clinically meaningful outcomes were observed in cohort 6 through ≥2 years of follow-up, and responses were consistent with prior analyses [10, 11]. Further, peak CAR T-cell levels were comparable in cohort 6 versus cohorts 1 + 2, suggesting no negative impact of corticosteroids on CAR T-cell pharmacokinetics [10]. A retrospective single-center experience with commercial axi-cel suggested that corticosteroid use may have prognostic impact, as higher cumulative doses (above the median cumulative dexamethasone-equivalent dose of 186 mg, n = 60) were associated with shorter PFS and OS, with no negative impact on CAR T-cell pharmacokinetics [23]. The median cumulative cortisone-equivalent corticosteroid dose including prophylaxis was 1252 mg for cohort 6 and 7418 mg for cohorts 1 + 2 [10], equal to 40 mg and 240 mg dexamethasone-equivalent doses, respectively. Although a greater proportion of patients in cohort 6 received corticosteroids compared with cohorts 1 + 2 (100% [as required per protocol] versus 22%, respectively) [10], patients in cohort 6 received less cumulative corticosteroids and median DOR and PFS were notably longer versus cohorts 1 + 2 [4]. Furthermore, the dexamethasone-equivalent corticosteroid dose in cohort 6 was less than one-third of the median dose reported in the aforementioned single-center experience [23], supporting the argument that prophylactic and earlier corticosteroid use may be associated with notably lower cumulative corticosteroid doses, and by that am improvement in the overall safety profile.
Median OS was not reached in cohort 6 or cohorts 1 + 2 [4] at 2 years, and lymphoma-related mortality was similar between these patient groups in this analysis (Fig. S2). These findings suggest that the cohort 6 toxicity management strategy did not increase likelihood of disease-related mortality. Indeed, all but 3 patients in cohort 6 who were in response at 1 year remained in response as of the 2-year data cutoff date, speaking to response durability [11]. Notably, non–lymphoma-related mortality was low in cohorts 1 + 2, with only 7 deaths reported among the 101 axi-cel–treated patients (3 deaths due to AEs and 4 for other reasons). None of these 7 patients were included in the PSM analysis set. Conversely, 6 patients in cohort 6 died of non–lymphoma-related reasons, and all were included in PSM. This difference may account for the lower overall mortality observed in cohorts 1 + 2 versus cohort 6 (Fig. S2).
Limitations of this study include the low number of patients enrolled, the lack of direct comparator arm, and those inherent with conducting an exploratory cohort study versus a randomized controlled trial. These factors may limit the interpretability of the results and, thus, additional confirmatory studies may be needed to further understand the impact of prophylactic corticosteroids on clinical practice and patient outcomes.
Collectively, with ≥2 years of follow-up, the ZUMA-1 cohort 6 toxicity management strategy continued to demonstrate reduced Grade ≥3 CRS without adversely affecting CAR T-cell pharmacokinetics or compromising efficacy outcomes, which remain high and durable, for patients with R/R LBCL treated with axi-cel.
Data availability
Kite is committed to sharing clinical trial data with external medical experts and scientific researchers in the interest of advancing public health, and access can be requested by contacting medinfo@kitepharma.com.
References
YESCARTTA® (axicabtagene ciloleucel) [summary of product characteristics]. Hoofddorp, The Netherlands: Kite Pharma EU B.V; 2022.
YESCARTA® (axicabtagene ciloleucel) [package insert]. Santa Monica, CA: Kite Pharma, Inc; 2022.
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, et al. Axicabtagene Ciloleucel CAR T-Cell therapy in refractory large B-Cell lymphoma. N Engl J Med 2017;377:2531–44. https://doi.org/10.1056/NEJMoa1707447.
Locke FL, Ghobadi A, Jacobson CA, Miklos DB, Lekakis LJ, Oluwole OO, et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1-2 trial. Lancet Oncol. 2019;20:31–42. https://doi.org/10.1016/S1470-2045(18)30864-7.
Neelapu SS, Jacobson CA, Ghobadi A, Miklos DB, Lekakis LJ, Oluwole OO, et al. 5-Year follow-up supports curative potential of axicabtagene ciloleucel in refractory large B-Cell Lymphoma (ZUMA-1). Blood. 2023;141:2307–15. https://doi.org/10.1182/blood.2022018893.
Neelapu SS. Managing the toxicities of CAR T-cell therapy. Hematol Oncol 2019;37:48–52. https://doi.org/10.1002/hon.2595.
Neelapu SS, Tummala S, Kebriaei P, Wierda W, Gutierrez C, Locke FL, et al. Chimeric antigen receptor T-cell therapy - assessment and management of toxicities. Nat Rev Clin Oncol 2018;15:47–62. https://doi.org/10.1038/nrclinonc.2017.148.
Locke FL, Neelapu SS, Bartlett NL, Lekakis LJ, Jacobson CA, Braunschweig I, et al. Preliminary results of prophylactic tocilizumab after axicabtagene ciloleucel (axi-cel; KTE-C19) treatment for patients with refractory, aggressive non-Hodgkin lymphoma (NHL). Blood. 2017;130:1547–1547.
Topp MS, van Meerten T, Houot R, Minnema MC, Bouabdallah K, Lugtenburg PJ, et al. Earlier corticosteroid use for adverse event management in patients receiving axicabtagene ciloleucel for large B-cell lymphoma. Br J Haematol 2021;195:388–98. https://doi.org/10.1111/bjh.17673.
Oluwole OO, Bouabdallah K, Munoz J, De Guibert S, Vose JM, Bartlett NL, et al. Prophylactic corticosteroid use in patients receiving axicabtagene ciloleucel for large B-cell lymphoma. Br J Haematol 2021;194:690–700. https://doi.org/10.1111/bjh.17527.
Oluwole OO, Forcade E, Muñoz J, de Guibert S, Vose JM, Bartlett NL, et al. Prophylactic Corticosteroid Use with Axicabtagene Ciloleucel (Axi-Cel) in Patients (Pts) with Relapsed/Refractory Large B-Cell Lymphoma (R/R LBCL): One-Year Follow-up of ZUMA-1 Cohort 6 (C6). Blood. 2021;138:2832 https://doi.org/10.1182/blood-2021-147403.
Lee DW, Gardner R, Porter DL, Louis CU, Ahmed N, Jensen M, et al. Current concepts in the diagnosis and management of cytokine release syndrome. Blood. 2014;124:188–95. https://doi.org/10.1182/blood-2014-05-552729.
Topp MS, Gokbuget N, Stein AS, Zugmaier G, O’Brien S, Bargou RC, et al. Safety and activity of blinatumomab for adult patients with relapsed or refractory B-precursor acute lymphoblastic leukaemia: a multicentre, single-arm, phase 2 study. Lancet Oncol. 2015;16:57–66. https://doi.org/10.1016/S1470-2045(14)71170-2.
Cheson BD, Pfistner B, Juweid ME, Gascoyne RD, Specht L, Horning SJ, et al. Revised response criteria for malignant lymphoma. J Clin Oncol 2007;25:579–86. https://doi.org/10.1200/JCO.2006.09.2403.
Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70:41–55.
Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivar Behav Res. 2011;46:399–424. https://doi.org/10.1080/00273171.2011.568786.
Ellard R, Kenyon M, Hutt D, Aerts E, de Ruijter M, Chabannon C, et al. The EBMT immune effector cell nursing guidelines on CAR-T therapy: a framework for patient care and managing common toxicities. Clin Hematol Int. 2022;4:75–88. https://doi.org/10.1007/s44228-022-00004-8.
Oluwole OO, Bouabdallah K, Muñoz J, De Guibert S, Vose JM, Bartlett NL, et al. Prophylactic corticosteroid use in patients receiving axicabtagene ciloleucel for large B‐cell lymphoma. Br J Haematol 2021;194:690–700. https://doi.org/10.1111/bjh.17527.
Strati P, Varma A, Adkins S, Nastoupil LJ, Westin J, Hagemeister FB, et al. Hematopoietic recovery and immune reconstitution after axicabtagene ciloleucel in patients with large B-cell lymphoma. Haematologica. 2021;106:2667–72. https://doi.org/10.3324/haematol.2020.254045.
Stewart AG, Henden AS. Infectious complications of CAR T-cell therapy: a clinical update. Ther Adv Infect Dis. 2021;8:20499361211036773 https://doi.org/10.1177/20499361211036773.
Logue JM, Zucchetti E, Bachmeier CA, Krivenko GS, Larson V, Ninh D, et al. Immune reconstitution and associated infections following axicabtagene ciloleucel in relapsed or refractory large B-cell lymphoma. Haematologica. 2021;106:978–86. https://doi.org/10.3324/haematol.2019.238634.
Neill L, Mackenzie SC, Marzolini MAV, Townsend W, Ardeshna KM, Cwynarski K, et al. Steroid use, advanced stage disease and ≥3 lines of prior chemotherapy are associated with a higher risk of infection following CD19 CAR T-Cell therapy for B-NHL: real world data from a large UK center. Blood. 2020;136:20.
Strati P, Ahmed S, Furqan F, Fayad LE, Lee HJ, Iyer SP, et al. Prognostic impact of corticosteroids on efficacy of chimeric antigen receptor T-cell therapy in large B-cell lymphoma. Blood. 2021;137:3272–6. https://doi.org/10.1182/blood.2020008865.
Acknowledgements
This study was funded by Kite, a Gilead Company. The authors thank the patients who participated in the trial, and their families, caregivers, and friends, and the study investigators, coordinators, and health care staff at each study site. The authors thank Andrew Lee, MD, of Kite, a Gilead Company, for critical review of safety data. Medical writing support was provided by Ashley Skorusa, PhD, and Alberto Moldón, PhD, of Nexus Global Group Science LLC, funded by Kite, a Gilead Company.
Author information
Authors and Affiliations
Contributions
OOO, JJK and TvM designed the study. OOO, EF, JM, SdG, JMV, NLB, YL, AD, PMS, AHG, MJK, CAJ, UF, MCM, CT, JMT, PS, IA, DT and TvM enrolled and treated patients. All authors collected or assembled data, participated in the data interpretation and manuscript writing, had full access to the data, approved of the final version of the manuscript, and are accountable for all aspects of this work.
Corresponding author
Ethics declarations
Competing interests
OOO: honoraria from Kite, a Gilead Company, Pfizer, and Gilead; consulting/advisory role for Pfizer, Kite, a Gilead Company, Gilead, AbbVie, Janssen, TG Therapeutics, ADC Therapeutics, Novartis, Epizyme, Curio Science, Nektar Therapeutics, Cargo Therapeutics, and Caribou Biosciences; institutional funding from Kite, a Gilead Company, Pfizer, Daiichi Sankyo, and Allogene. EF: speakers’ bureau participation for Gilead, Jazz, Novartis, and GSK; and travel support from Gilead, Jazz, Sanofi, and MSD. JM: honoraria from Curio, Kyowa Kirin, OncView, Physicians’ Education Resource, Seagen, and Targeted Oncology; consulting/advisory role for ADC Therapeutics, Alexion, Bayer, BeiGene, Bristol Myers Squibb, Debiopharm, Epizyme, Fosun Kite, Genmab, Innovent, Janssen, Juno/Celgene, Karyopharm, Kite, a Gilead Company, Kyowa Kirin, MorphoSys/Incyte, Novartis, Pfizer, Pharmacyclics/AbbVie, Seagen, and Servier; speakers’ bureau participation for Acrotech/Aurobindo, AstraZeneca, Bayer, BeiGene, Celgene/Bristol Myers Squibb, Genentech/Roche, Kite, a Gilead Company, Kyowa, Pharmacyclics/Janssen, Seagen, and Verastem; and research funding (paid to institution) from Bayer, Celgene, Genentech, Gilead/Kite, Incyte, Janssen, Merck, Millennium, Pharmacyclics, Portola, and Seagen. SdG: honoraria from and consultancy or advisory role for Gilead, AbbVie, and Janssen. JMV: honoraria from AstraZeneca, Janssen, Lilly, and AbbVie; consulting or advisory role for Johnson and Johnson, Daiichi Sankyo, Pharmacyclics, and MorphoSys; and research funding from Kite, a Gilead Company. NLB: consultancy or advisory role for ADC Therapeutics, Roche/Genentech, and Seagen; and research funding from ADC Therapeutics, Autolus, Bristol Myers Squibb, Celgene, Forty Seven, Genentech, Janssen, Kite, a Gilead Company, Merck, Millennium, Pharmacyclics, and Seagen. YL: consultancy or advisory role for bluebird bio, Celgene, Gamida Cell, Janssen, Novartis, Juno, Kite, a Gilead Company, Legend, Sorrento, and Vineti; research funding from bluebird bio, Celgene, Janssen, Kite, a Gilead Company, Merck, and Takeda. AD: consultancy or advisory role for Adicet, Janssen, and Kite, a Gilead Company. PMS: honoraria from, consulting or advisory role for, and speakers’ bureau participation for Kite, a Gilead Company, and research funding from AlloVir, Autolus, Kite, a Gilead Company, and Novartis. AHG: employment with Regional Cancer Care Associates, OMI; leadership role and stock or other ownership at COTA (Cancer Outcome Tracking Analysis) and Genomic Testing Cooperative, Resilience; honoraria from AstraZeneca, Bristol Myers Squibb, Celgene, Elsevier PracticeUpdate: Oncology, Incyte, Janssen, Kite, a Gilead Company, MorphoSys, Novartis, OncLive Peer Exchange, Pharmacyclics, Vincerx, and Xcenda; consultancy or advisory role for AbbVie, Bristol Myers Squibb, Celgene, Elsevier PracticeUpdate: Oncology, Janssen, Kite, a Gilead Company, Medscape, Michael J. Hennessy Associates, Inc., Novartis, Pharmacyclics, and Physicians’ Education Resource; research funding from Acerta, AstraZeneca, Celgene, Genentech, Hoffmann-La Roche, Infinity Pharmaceuticals, Janssen, Karyopharm, and Pharmacyclics; and other relationships with MorphoSys and Incyte Steering Committee, AstraZeneca MCL Steering Committee, Vincerx Scientific Advisory Board. MJK: honoraria from and consultancy or advisory role for BMS/Celgene, Kite, a Gilead Company, Miltenyi Biotec, Novartis, Adicet Bio and Roche; research funding from Kite, a Gilead Company, Roche, Takeda, and Celgene; and travel support from Kite, a Gilead Company, Miltenyi Biotec, Novartis, and Roche. CAJ: honoraria from Kite, a Gilead Company, Novartis, BMS/Celgene, Instil Bio, ImmPACT Bio, Lonza, Ipsen, Epizyme, bluebird bio, and Daiichi Sankyo; consulting or advisory role for Kite, a Gilead Company, Novartis, BMS/Celgene, Instil Bio, ImmPACT Bio, Lonza, Ipsen, Epizyme, bluebird bio, and Daiichi Sankyo; and research funding from Kite, a Gilead Company, and Pfizer. UF: honoraria from Caribou and Kite, a Gilead Company, and consulting or advisory role for MorphoSys. MCM: consultancy or advisory role for Gilead, Bristol Myers Squibb, GSK, CDR-life, and Janssen-Cilag; speakers’ bureau participation for WebMD; and research support from BeiGene. CT: honoraria from BMS, Roche AbbVie, Novartis, Kite, a Gilead Company, Incyte, Amgen, and Takeda; consulting or advisory role for BMS, Roche, AbbVie, Novartis, Kite, a Gilead Company, Incyte, Amgen, and Takeda; and research funding from Gilead and Novartis. JMT: consultancy or advisory role for Kite, a Gilead Company; and research funding from Bristol Myers Squibb, Kite, a Gilead Company, Merck, and Spectrum. PS: honoraria from and consultancy or advisory role for MorphoSys and CRISPR Therapeutics; and research funding from Amgen, Pfizer, Karyopharm, Gilead, Incyte, Gamida Cell, Seagen, and Cellectar. IA: speakers’ bureau participation for Kite, a Gilead Company, and Novartis. DT: consultancy or advisory role for BMS, EUSA, Partner, and Takeda, and research funding from BMS. JJK, YZ, JN, HM: employment with Kite, a Gilead Company; and stock or other ownership in Gilead Sciences. RRS: employment with, leadership role with, stock or other ownership in, and patents, royalties, and other intellectual property from Atara and Kite, a Gilead Company. SV: employment with, research funding from Kite, a Gilead Company; and stock or other ownership in Gilead Sciences. TvM: honoraria from Kite, a Gilead Company; consultancy or advisory role for Janssen and Kite, a Gilead Company; and research funding from Celgene/BMS and Genentech.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Oluwole, O.O., Forcade, E., Muñoz, J. et al. Long-term outcomes of patients with large B-cell lymphoma treated with axicabtagene ciloleucel and prophylactic corticosteroids. Bone Marrow Transplant 59, 366–372 (2024). https://doi.org/10.1038/s41409-023-02169-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41409-023-02169-z
- Springer Nature Limited
This article is cited by
-
Advancements and challenges in CAR T cell therapy in autoimmune diseases
Nature Reviews Rheumatology (2024)