Abstract
Thioredoxin-interacting protein (TXNIP) is commonly considered a master regulator of cellular oxidation, regulating the expression and function of Thioredoxin (Trx). Recent work has identified that TXNIP has a far wider range of additional roles: from regulating glucose and lipid metabolism, to cell cycle arrest and inflammation. Its expression is increased by stressors commonly found in neoplastic cells and the wider tumor microenvironment (TME), and, as such, TXNIP has been extensively studied in cancers. In this review, we evaluate the current literature regarding the regulation and the function of TXNIP, highlighting its emerging role in modulating signaling between different cell types within the TME. We then assess current and future translational opportunities and the associated challenges in this area. An improved understanding of the functions and mechanisms of TXNIP in cancers may enhance its suitability as a therapeutic target.
Similar content being viewed by others
Introduction
Reduction-oxidation (redox) reactions, where the exchange of electrons from one compound to another occurs within the same reaction, are critical to cellular life. They are required for multiple biological processes from metabolism to enzymic function. These potent reactions carry risk, with dysregulated redox management being shown to be involved in the initiation and progression of multiple pathologies, including metabolic, neurodegenerative, cardiovascular, and neoplastic diseases [1,2,3]. Thioredoxins (TXN and TXN2), together with glutathione, constitute the major thiol antioxidants that ensure these reactions take place with limited local damage [4]. Thioredoxin-interacting protein (TXNIP) has been shown to bind and inhibit thioredoxins (Trx) [5,6,7]. Consequently, dysregulation of this TXNIP-Trx axis is strongly associated with metabolic diseases [8,9,10].
In addition to being a major redox regulator, TXNIP has also been identified as a tumor suppressor gene (TSG), and its expression is reduced in a wide range of primary tumors and cancer cell lines compared to normal tissue and cell lines, respectively [11,12,13,14,15,16]. Its function as a TSG is also supported by the observation that Txnip-deficient mice have a 40% higher incidence of spontaneously developing hepatocellular carcinoma (HCC) [17]. However, different studies in a variety of different cancers, utilising a variety of different techniques have reached different conclusions, suggesting that the role of TXNIP is complex in cancer and may have different implications depending on cancer type and stage of disease (Table 1). Moreover, single-cell RNA sequencing of T-cell lymphoma reveals that low levels of TXNIP expression correlate with malignancy and disease progression [18]. Indeed, accumulating evidence suggests that downregulation of TXNIP is associated with poorer prognosis in diffuse large B-cell lymphoma [19].
In contrast, other reports show that high TXNIP levels can also correlate with poor clinical prognosis in some cancers. For example, lung cancer patients with high levels of TXNIP exhibit decreased progression-free survival compared to counterparts with low TXNIP levels (18.0 vs. 23.0 months) [20]. To be noted, just 70 samples were collected for analysis. Thus, more samples need to be used for further investigation. In HCC and conventional (clear cell) renal cell carcinoma, TXNIP overexpression promotes angiogenesis and metastasis [21, 22]. Similarly, even though high TXNIP expression is associated with favorable prognosis in breast, liver, and lung cancers, it correlates with poor prognosis in gastric cancer in a pan-cancer analysis. These data indicate that the roles of TXNIP in cancers show tumor specificity [14]. In addition to tumor-specific functions, TXNIP may also exert opposite functions at different stages during cancer progression. When analysing early vs. late-stage cases separately in ovarian cancer, TXNIP expression is associated with different clinical outcomes, namely, improved survival in early-stage disease but poor survival in late-stage disease [23].
In this review, our focus is less on deciphering TXNIP’s prognostic impact but more on the role of TXNIP within the tumor microenvironment (TME), including both tumor cells and host cells, and its impact on different cancer hallmarks. Led by the literature, we pay particular attention to the roles of TXNIP in redox, metabolic and immunological control of tumor biology. Papers that we consider to be key in understanding the role of TXNIP in cancer biology are highlighted in italic throughout the review.
Regulation of TXNIP
The expression of TXNIP is regulated by a variety of biological processes and associated pathways/factors. These pathways include common regulatory mechanisms (such as transcriptional factors, microRNAs and circular RNAs, epigenetic regulators and regulators of mRNA and protein stability), oncogenes and TSGs, ER stress signaling, cytokines and growth factors. Notably, many of these regulatory signaling pathways are bi-directional. Indeed TXNIP has been reported to regulate the expression of >10 factors that regulate TXNIP’s own expression: microRNAs (miRNA-204 and miR-124a) [24,25,26], tumor suppressors (p53 [17, 27] and PTEN [28]), ER components, (protein disulfide isomerases [29] or apoptosis signal-regulating kinase 1 [30]), cytokines (IL-1β, IL-18 [31], TNFα and COX-2 [32]). These and other mono-directional regulators of TXNIP expression are summarised in Table 2.
TXNIP expression has been strongly associated with glucose-sensing transcriptional complexes, especially the ChREBP/MondoA:Mlx complex [33]. As a result, MondoA/TXNIP signaling has been linked to the regulation of cellular glucose [34]. The factors involved in TXNIP regulation (Table 2) constitute a comprehensive regulatory network that can be broadly divided into four classes [35]: 1) transcription factors (MondoA [36], ChREBP [37], PTEN [38], MLX [33], FoxO1 [37], STAT3 [39]); 2) microRNAs and circular RNAs (miR-21 [40], miR-148a [41], miR-135b-5p [42], miR-152-5p [43], miR-204 [24]); 3) epigenetic regulators (EZH2 [44], UHRF1 [12]) and 4) regulators of mRNA and protein stability (LncRNA Gm15441 [45], LncRNA SNHG15 [46]).
Oncogenes
TXNIP expression can also be suppressed by oncogenes [47]. For instance, in breast cancer, c-Myc has been exhibited to antagonise TXNIP expression in MondoA-dependent pathway [48]. By binding to TXNIP promoter in E-box -containing region, c-Myc competes with MondoA and represses TXNIP expression in TNBC, indeed a c-Mychigh/TXNIPlow signature correlates with poor OS specifically in this subclass of breast cancer [36]. When compared with iAP mice (mice harbouring conditional null alleles of Apc and Trp53), iKAP mice (engineered with a doxycycline-inducible oncogenic Kras allele and conditional null alleles of Apc and Trp53) exhibit reduced TXNIP expression, suggesting that oncogenic KRAS is capable of TXNIP regulation [49]. Laio et al. go on to demonstrate that KRAS inhibits interferon regulatory factor 2 which in turn inhibits CXCL3 expression and the recruitment of CXCR2+ myeloid cells, an axis that can be used to stratify patients for efficacious anti-PD1 therapy [50]. The oncogenic GTPase Ras has also been shown to inhibit TXNIP expression by suppressing the translation of TXNIP mRNA [50]. Additionally, in a study of 788 node-negative patients (which showed that TXNIP expression is associated with better prognosis [HR 0.642; p < 0.001]), oncogenic activation of HER2 is associated with decreased TXNIP expression and a concomitant increase in reactive oxygen species (ROS) production in breast cancer [51].
ER stress signaling
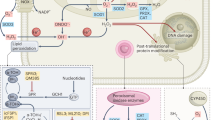
ER stress signaling is regulated by three major functional sensors: activating transcription factor 6 (ATF6), inositol-requiring enzyme 1α (IRE1α) and protein kinase R-like ER kinase (PERK) [52]. Under homeostatic conditions, the luminal ER master chaperone protein Binding immunoglobulin Protein (BiP) is bound to these sensors, maintaining sensors in an inactive state. Under ER stress conditions, misfolded proteins accumulate in the ER lumen and bind with high affinity to BiP, resulting in displacement of BiP and the activation of ER sensors. This ultimately leads to transcriptional reprogramming to maintain ER homeostasis, a process known as the unfolded protein response (UPR) [52]. The UPR is an evolutionarily conserved cell stress response, but aberration in the activation of ER stress is a key driver of tumorigenesis and reprogramming of the TME [53]. TXNIP signaling is implicated in ER stress, participating in the different branches of the UPR. Both PERK and IRE-1 are required for TXNIP induction in ER-stress-induced β-cell death [31], while activation of ATF6 signaling fails to induce TXNIP expression. Importantly, PERK and IRE-1α are able to regulate the expression of TXNIP via eIF2α-ATF5 and XBP1 signaling pathways, respectively [54,55,56,57] (Fig. 1).
Both PERK and IRE-1 are required for TXNIP induction in ER-stress-induced β-cell death [31]. Notably, PERK on its own can also regulate TXNIP [57]. IRE1α and its downstream effector XBP1 are also shown to be responsible for TXNIP-induced mitochondrial dysfunction, without involvement of PERK signaling [56]. Recently, IRE1α-microRNA signaling axis (miR-17) has been described to control TXNIP expression [55].
Cytokines and growth factors
Cytokines play a crucial part in immunity and the TME by mediating cell-to-cell communication [58]. The signaling driven by inflammatory, regenerative, and anti-inflammatory cytokines modulate the recruitment, development, and behavior of different cell types from the innate and adaptive immune pools. TXNIP expression is regulated by cytokines to achieve various functions. In naïve T cells, TNFα triggers TXNIP downregulation leading to increased glucose uptake and further T cell stimulation [59]. Insulin-like growth factor 1, a growth factor known to promote cancer development, negatively regulates TXNIP expression enhancing its antiapoptotic effects [60]. In addition, IL-1β and TGFβ1 suppress TXNIP activation in fibroblasts and mesenchymal progenitors, respectively [61, 62]. However, TGF-β1 can also induce TXNIP expression to achieve transcriptional repression in HL-60 cells [63].
Other regulatory conditions
Additional endogenous and environmental factors have been reported to induce TXNIP expression. In energy-depleted conditions, AMP-activated protein kinase induces the degradation of TXNIP [64], while under hypoxic conditions [65, 66], HIF-1α induction has been shown to increase TXNIP expression. Inversely, TXNIP also causes the degradation and export of HIF-1 α, suggestive of another bi-directional regulatory loop [67]. The CISD2 (NAF-1, nutrient-deprivation autophagy factor-1) protein is reported to regulate TXNIP expression through a process that involves the perturbation of mitochondrial labile iron, mitochondrial ROS and triggered ferroptosis in breast cancer cells [68]. Retinoic acid-mediated TXNIP suppression is found to de-activate hepatic stellate cells and thereby help prevent liver fibrosis and carcinogenesis [69].
In conclusion, TXNIP expression and, therefore, function is regulated by diverse factors associated with different tissues and conditions (Fig. 2), and a complex network of positive and negative regulatory loops.
TXNIP expression is regulated by a variety of different signaling pathways, including microRNAs, oncogenes and TSGs, cytokines and growth factors, endoplasmic reticulum and some specific environmental conditions (e.g. hypoxia). Additionally, TXNIP also regulates these pathways as part of a feedback loop to attenuate or amplify signaling. For example, oncogenes, including Kras, HER2 and c-Myc, induce TXNIP expression, while TXNIP can regulate the expression of p53 and PTEN. Moreover, HIF-1a and TXNIP can regulate each other under different conditions.
Biological roles of TXNIP
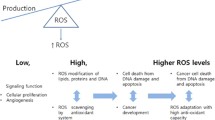
TXNIP has been seen to be involved in a myriad of cellular responses, including oxidative stress, differentiation, angiogenesis, apoptosis and glycolysis (Fig. 3).
a TXNIP can positively or negatively regulate oxidative stress via binding with either Trx or p53; b the activation of TXNIP leads to tumor suppression by affecting cell differentiation, cell stemness and cell death (such as apoptosis, autophagy, and senescence); c TXNIP mediates drug-induced cell death via ROS-dependent/- independent pathways; d TXNIP impacts on cellular metabolism, transforming cells from glycolytic to reliant on oxidative phosphorylation, by regulating the expression of GLUT1/4; e TXNIP increases the expression of VEGFA, PDGF, and ANG2.
TXNIP and oxidative stress
As discussed, TXNIP was originally identified as a key regulator of cellular redox and its expression has subsequently and consequently been shown to be closely associated with intracellular ROS levels [14]. This regulation is mediated by its antagonistic effects on Trx by an intermolecular disulfide interaction, meaning TXNIP-Trx binding increases the presence of ROS [44, 70]. A study, which assessed blood samples from chronic lymphocytic leukemia patients, demonstrated that TXNIP levels robustly correlated with ROS production [71]. Moreover, silencing of TXNIP has been demonstrated to decrease ROS levels in macrophages [72], but overexpressed TXNIP causes high oxidative stress, leading to DNA damage, cell death [73], and autophagy-related apoptosis [74]. However, TXNIP has also been shown to bind and stabilise p53 protein, showing antioxidant effects and further maintaining the cell survival of the hematopoietic cells [75]. Taken together, TXNIP can either promote or inhibit the production of ROS by binding to either Trx or p53, leading to cell death or cell survival, respectively. Notably, the dual role of ROS in cancer could partially be the reason for diverse functions of TXNIP in cancer [76].
TXNIP and tumor suppression
TXNIP can induce cell death and inhibit proliferation, thus being regarded as a TSG. TXNIP activation leads to G1/S phase arrest by modulating cell cycle regulatory proteins (such as p27kip1, JAB1, CDK2, and cyclinE) [77]. In contrast, loss of TXNIP facilitates rapid cell division and activation of DNA replication, leading to cell proliferation in breast and lung cancer models [78, 79]. After shuttling into the mitochondria, TXNIP binds to thioredoxin and abolishes its inhibitory effect on ASK1-mediated apoptosis [30]. In addition, TXNIP is also involved in autophagy and senescence [80,81,82]. Mechanistically, TXNIP interacts with REDD1 to promote mitochondrial rearrangement and ROS production, suppressing ATG4B catalytic activity and inducing autophagy [83]. Moreover, TXNIP can promote the differentiation of leukemia-initiating cells and CRC cells in glycolysis-independent and glycolysis-dependent manners, respectively. TXNIP-dependent cell differentiation in leukemia and CRC promotes the suppression of leukemogenesis and reduces CRC cell viability [84,85,86]. Additionally, a reduction in TXNIP induced by M2 macrophage-derived exosomes has been observed to be critical for maintaining cancer “stemness” and promoting tumor progression in HCC [87].
TXNIP has also been reported to reduce the migratory capacity of tumor cells. Downregulation of TXNIP maintains the Trx/Trx reductase (Trx/TrxR) system in an active state, driving epithelial-mesenchymal transition and increasing the metastatic potential of cancer cells [88]. In pancreatic cancer, elevated TXNIP expression leads to repression of malignant transcripts and impairment of metastatic tumorigenesis through the epigenetic reprogramming of chromatin [89]. Similarly, albeit through a different mechanism, TXNIP mediates the internalisation and degradation of EGFR, decreasing the migratory capacity of breast cancer cells [90]. Interestingly, breast cancer cell-derived exosomes negatively regulate TXNIP expression, resulting in the activation of the WNT/β-catenin pathway in fibroblasts and induction of cancer-associated fibroblasts (CAFs) [91]. These CAFs then promote cancer cell invasion and metastasis [91]. However, another study, this time in HCC, observes that TXNIP expression is positively associated with the migratory and invasive ability of hepatocellular cancer cells [92], stressing the importance of underlying tissue and cell type in determining the impact of TXNIP function on migration.
TXNIP can also affect tumorigenesis through its association with metabolic disorders. Epidemiological and clinical studies highlight that cancer patients with diabetes have a higher morbidity and mortality [93]. The mechanisms of diabetic stress-associated tumor progression and metastasis include inhibition of antitumor immune responses [94, 95], metabolic transcriptional modulation of cancer cells [96], decellularization of extracellular matrix scaffolds [97], and even vascular dysfunction [98]. The master roles of TXNIP in fasting, insulin sensitivity, and β-cell apoptosis are well known, and these functions have been linked to an increased risk of diabetes and other metabolic disorders [99,100,101,102]. These data collectively suggest TXNIP acts as a driver of metabolic diseases, contributing to the development of cancers [35, 103].
TXNIP and chemotherapy
Interestingly, cancer cells, displaying high baseline levels of ROS, are vulnerable to further damage caused by ROS accumulation. In this vein, a number of studies have shown that increased TXNIP expression can enhance the cytotoxicity of chemotherapeutic reagents by manipulating ROS levels, as the levels of ROS in cancer cells provide a potential therapeutic vulnerability. This antitumor strategy has already been exploited by several agents, including dBET-3, vorinostat, pterostilbene, and resveratrol [104,105,106,107]. Additionally, platinum-based drugs can also inhibit the activity of TrxR; a process that has been demonstrated to be critical in promoting antitumor effects [108,109,110].
TXNIP can also promote treatment efficacy in a ROS-independent fashion. In esophageal cancer, cisplatin treatment leads to TXNIP upregulation, mediating its cytotoxicity via an unknown mechanism [111]. In oral cancer models, overexpression of TXNIP enhances the effectiveness of radiotherapy via the DNA repair pathway [112]. Compared to cisplatin-sensitive cells, cisplatin-resistant cells exhibit downregulation of TXNIP mRNA mediated by UCA1, suggesting a role of UCA1 and TXNIP in contributing to cisplatin resistance in lung adenocarcinoma [113]. In support of these findings, exogeneous overexpression of TXNIP in glioma cells decreases the median inhibitory concentration (IC50) of cisplatin [114]. Combining a TXNIP agonist, D-Allose, with chemotherapy or radiotherapy results in enhanced antitumor effects in both head and neck and lung cancer models [115, 116]. These studies collectively suggest that increased TXNIP expression mediates or enhances the cytotoxicity of chemo-radio therapies.
TXNIP and glycolysis
Metabolic reprogramming is a hallmark of cancer development and metastasis and TXNIP-dependent metabolic phenotypes are associated with patient prognosis. Elevated glycolysis is closely associated with the initiation of cancer, producing glucose-dependent adenosine triphosphate (ATP) and glycolytic intermediates for macromolecular biosynthesis. c-Myc, a well-known modulator of metabolism, mediates metabolic and phenotypic changes in cancer [117]. TXNIP is reported to both regulate lipid and glucose metabolism directly [118, 119] and mediate c-Myc-driven metabolic changes indirectly [20, 120,121,122]. For instance, a study in TNBC identified that TXNIP suppression by MYC can reprogramme the metabolic phenotype of cancer cells [36]. Additionally, in ER+ breast cancer, the levels of TXNIP expression in tumor cells are associated with different metabolic subtypes [54]. In MCF7 cells, which have high basal TXNIP expression, an elevated mitochondrial oxidative phosphorylation (OXPHOS) phenotype is observed. In contrast, T47D cells, which have low expression of TXNIP, display an aerobic glycolysis phenotype [54]. Interestingly, estrogen has been shown to repress TXNIP expression and drive the Warburg effect [54]. In pancreatic cancer, the tumor suppressor FBW7 (F-box and WD Repeat Domain-Containing 7) exerts its antitumor effects by controlling glucose metabolism and oxygen consumption in a TXNIP-dependent manner [123]. More importantly, it should be mentioned that genetic deletion of TXNIP increases the uptake of glucose by regulating the expression of HIF-1a or c-Myc, which leads to the metabolic reprogramming towards aerobic glycolysis [119, 124, 125]. Collectively, these data highlight the dominant role of TXNIP in controlling glucose homeostasis [48].
Further understanding reveals one of the molecular mechanisms is the association between TXNIP and GLUT family. The GLUT membrane transporter family is crucial in facilitating glucose transportation and includes class I (GLUT1-4), class II (GLUT7, GLUT11) and class III (GLUT6, GLUT8, GLUT12) transporters [126]. TXNIP inhibits the influx of glucose and lactate by decreasing the expression of class I glucose transporters like GLUT1 and GLUT4 via both endocytosis and degradation of protein levels and reduction of mRNA levels [64, 127, 128]. Recently, a class III transporter, GLUT8, a central regulator of metabolism, has also been identified to interact with TXNIP to enable hexosamine homeostasis [129]. Extracellular matrix remodelling is another critical factor governing extrinsic metabolic regulation. Defects in matrix attachment affect cellular metabolism, resulting in a reduction in glucose uptake and subsequent ATP deficiencies [130]. Matrix digestion reportedly destabilises TXNIP and enriches GLUT1 transporter at the plasma membrane to promote glycolysis; a process which is fundamental for both embryogenesis and tumorigenesis [130, 131]. All these observations emphasise the critical role of TXNIP in metabolic reprograming.
TXNIP and tumor angiogenesis
Angiogenesis, another hallmark of cancer, enables tumors to meet nutrient and oxygen needs to sustain proliferative and metabolic requirements. In conventional RCC, immunohistochemical staining of 691 patient samples revealed that patients with high TXNIP expression have a marked reduction in tumor free survival and a higher occurrence of metastasis. Interestingly, this study showed a significantly positive correlation between TXNIP expression and inefficient vascularisation favouring tumor cell survival in RCC [22]. Notably, this study was an observational study, lacking in-vitro and in-vivo experiments. Thus, this report might not be very evident regarding the impact of TXNIP on angiogenesis. But we have to admit that it brings great significance by providing clinical support. Mechanistically, TXNIP overexpression leads to upregulation of angiogenesis-related proteins (VEGFA, PDGF and ANG2), along with an angiogenic phenotype [132]. Moreover, in osteosarcoma, single-cell RNA sequencing analysis identifies different functional subtypes in the myeloid compartment [121]. Among them, TXNIP+ macrophages tend to be M2-like (a broadly anti-inflammatory phenotype) and express M2 signature markers, including MERTK, MRC1, STAB1 and CD163. Furthermore, ligand-receptor interaction analysis identifies an association between TXNIP+ macrophages and angiogenic endothelial cells, suggesting TXNIP+ macrophages may facilitate angiogenesis [121]. However, exogenous TXNIP expression in CRC lines (LoVo and HT29) represses angiogenesis [42]. Similarly, inhibition of a cyclin-dependent kinase transcriptionally represses TXNIP expression and promotes endothelial cell invasion, migration and vascular sprouting in breast, lung and prostate cancer cell lines [122]. Thus, TXNIP’s role in regulating angiogenesis is context dependent.
Immune regulation by TXNIP
An increasing number of studies are unveiling the impact of TXNIP expression on the immune system. A pan-cancer study recently reported a correlation between TXNIP and infiltration of immune cells, supporting the idea that TXNIP may be an important player in determining the immunological makeup of the TME [14]. In addition to its regulation of immune-related signaling pathways and cytokine production, TXNIP is also demonstrably involved in the development and maturation of innate and adaptive immune cells (Fig. 4). By impacting different immune cell in different ways, TXNIP can drive both antitumor and pro-tumor effects.
Schematic summarising the impacts of TXNIP on different arms of the immune system. TXNIP can maintain the survival and promote the activation of NK and dendritic cells (DC), leading to increase cytotoxicity [148, 151]. Meanwhile, TXNIP facilitates the differentiation of monocytes to M2 macrophages, creating a pro-tumoral microenvironment [154]. Moreover, TXNIP is involved in the development of various T and B cell subsets. It is essential in maintaining the identity of Tregs [163], while inhibiting the formation and activation of memory T cells and CD8+ T cells [156, 159]. Through inhibition of BCL-6, TXNIP can promote the formation of the germinal center [168].
TXNIP, NF-κB, and NLRP3 inflammasome signaling
TXNIP can exert effects on the immune system in several ways. As an intracellular amplifier of oxidative stress and inflammasome activation [133], TXNIP is detected in different cell types (such as tumor cells, immune cells and stromal cells). In endothelial cells, for example, nuclear translocation of TXNIP leads to NF-κB activation, which facilitates the expression of pro-inflammatory cytokines such as IL-1β [134, 135]. However in tumor cells, TXNIP suppresses TNF-α-induced NF-κB activity and subsequently inhibits hepatocarcinogenesis [79, 136].
Activation of the NOD-like receptor (NLR) family pyrin domain containing 3 (NLRP3) inflammasome is observed under diverse physiological and pathological conditions, such as caloric restriction [45], type 2 diabetes [137], preeclampsia [138], Alzheimer’s disease [139] and cancer [140]. It has been established that the NLRP3 inflammasome is involved in many cancer-immune relationships [141], with both antitumorigenic and pro-tumorigenic roles. On one hand, NLRP3 contributes to various types of cell death, like pyroptosis, apoptosis, necroptosis, and ferroptosis [142]; on the other, although inflammasome-inducing IL-1β can activate DCs to facilitate adaptive antitumor immune activation [143], it also expands myeloid-derived suppressor cells (MDSCs) [144].
Numerous studies have uncovered a link between TXNIP and NLRP3 inflammasome activation, mostly due to the functions of the Trx1/TXNIP axis in ROS regulation [100, 145]. However, this is not always the case, as Trx1 can lead to NLRP3 inflammasome activation independently of TXNIP [146]. STING triggers the TXNIP-NLRP3 interaction, leading to inflammasome activation without the involvement of Trx1 [2]. Similarly, CXCR4 can also directly bind to TXNIP and induce NLRP3 inflammasome activation without affecting the activity of Trx1 [147]. UPR signaling is another system that regulates inflammasome activation via TXNIP-dependent mitochondrial dysfunction, rather than through direct modulation of ROS levels [56]. Together, these findings indicate both Trx1 and TXNIP can also induce the activation of the NLRP3 inflammasome independently of the Trx1/TXNIP relationship and ROS regulation.
TXNIP and innate immunity
In addition to its roles in NF-kB and inflammasome-mediated inflammation, TXNIP is also involved in regulating the generation, development and functionality of innate immune cells. Txnip−/− mice carry a severely reduced number of NK cells [140]. There was also a decreased expression of IL2RB (CD122), but, intriguingly, the development of T and B cell populations was not impaired [148]. The reduced number of Txnip−/− NK cells observed in this study were also shown to be functionally impaired when considering cytotoxicity and IFN-γ production [148]. Another study has also demonstrated a similar requirement of TXNIP in the effective differentiation of NK cells [149]. Moreover, given that the core of tumors contains high levels of ROS that are associated with the presence of NK cells, TXNIP has been suggested to be an important factor governing the infiltration of NK cells into the TME [150]. The mechanism by which tumor-infiltrating NK cells gain resistance to oxidative stress is through retention of nuclear TXNIP leading to higher Trx-1 activity [150].
TXNIP is also reported to regulate the development of myeloid lineage. A study using Txnip-deficient mice demonstrated the requirement for TXNIP in maintaining normal functions of DCs, including secretion of the cytokines IL-12 and IL-6 and subsequent activation of T cells [151]. When comparing gene signatures between non-activated polymorphonuclear myeloid-derived suppressor cells (PMN-MDSCs) and activated ones from the same murine models, TXNIP expression is a significantly upregulated differential in the activated group. The authors felt these findings may tentatively indicate that TXNIP may have a role in maintaining immune-suppressive activity [152]. Tumor-associated macrophages (TAMs) are abundant in the TME of solid tumors and promote tumor development by suppressing immune responses and facilitating tumor growth and metastasis [153]. In PDAC, TXNIP expression is upregulated in TAMs, and this is driven by KRAS activity in cancer cells [154]. The study goes on to demonstrate that the high expression of TXNIP in TAMs contributes to metabolic changes which are required for macrophage polarisation and the promotion of pro-tumor responses [154]. Collectively, these studies suggest a requirement for TXNIP/txnip in NK cell development and function, along with TXNIP-mediated promotion of suppressive myeloid phenotypes.
TXNIP and adaptive immunity
The role of TXNIP in adaptive immunity appears to be more complex than in the innate compartment. In melanoma, TXNIP expression is enriched in memory T cells [155]. This may be due to TXNIP’s role in regulating CPT1a’s metabolic functions which are essential for protective memory T cell generation [156, 157], however, TXNIP has been observed to inhibit CPT1a expression, resulting in inhibited generation of memory T cells [157]. Dual anti-CD3/anti-CD28 stimulation on T cells suppresses TXNIP expression, and this has been attributed mainly to anti-CD3, suggesting anti-CD28 co-stimulation has minor effects [157, 158]. The activation of T cells may, at least to some extent, involve anti-CD3-mediated suppression of TXNIP, which potentially abolishes inhibitory impacts of TXNIP on transcriptional activation of targeted genes involved with T cell activation, differentiation, cytokine signaling as well as cell death pathways [159]. Notably, despite T cells showing higher levels of glucose uptake with anti-CD3/anti-CD28 stimulation, these metabolic changes are independent of TXNIP-mediated regulation of glycolysis [158].
Co-stimulatory signals are required for full TXNIP-dependent activation of T cells after TCR-MHC complex engagement, including signals from the tumor necrosis factor receptor superfamily (TNFRSF) members [157]. In a similar manner, TLR2, 4, and 5 agonists partially inhibit TXNIP expression through TNFα production [59]. The potential mechanism is likely to involve the downregulation of TXNIP, cell cycle entry and metabolic changes which are optimal for T cell proliferation and activation [59]. TXNIP also appears to be indispensable in the restriction of T cell (mainly in CD4+ T cells) and germinal center B cell expansion following viral infections, a process that relies on Trx1/TXNIP balance [158]. Additionally, similarly to the study reported by Yang et al. [150], this study also observed that ablation of TXNIP does not affect the development and homeostatic maintenance of T cells, B cells and myeloid cells [158]. Importantly, with regards checkpoint therapy, the levels of TXNIP have recently been reported to be negatively associated with the expression of PD-L1, indicating the potential impacts of TXNIP on immunomodulatory proteins [160]. However, whether or not other immune checkpoints are regulated by TXNIP needs further elucidation.
Regulatory T cells (Treg) are immunosuppressive cells which regulate multiple arms of the immune system with a particular emphasis on cytotoxic T cell responses. The impact of Tregs in tumorigenesis varies depending on the specific subtype of Tregs; consequently they are predictive of a variety clinical outcomes with an overall strong trend towards the prognostically negative [161, 162]. The plasticity and stability of Tregs are regulated, at least in part, by cellular metabolism [162]. A recent study highlights the requirement of the MondoA-TXNIP axis in maintaining the identity and functionality of Tregs by repressing glycolysis in CRC [163]. Inhibition of MondoA or TXNIP in Tregs leads to the upregulation of glycolytic genes and the increase of glycolytic activities, which compromises immuno-suppressive functions in these cells [163]. Fascinatingly, glycolysis reduces FOXP3 and RORγτ expression in Tregs, promoting a switch to a Th17-like effector phenotype, which can be reversed by TXNIP activation [163]. Accordingly, intra-tumoral Tregs generally present with increased induction of glycolytic pathways, resulting in a pro-tumor immune microenvironment [163, 164] (Fig. 5).
Germinal centers (GCs) are the main sites of antigen-stimulated B cell proliferation and differentiation. In GCs, antigen-activated B cells not only produce high-affinity antibodies through somatic hypermutation (SHM) on immunoglobulin genes, but also produce antibodies with specialised functions via class-switch recombination (CSR). GC B cells express high levels of BCL-6 which modulates GC formation through several different mechanisms, including inducing the GC to undergo SHM and CSR, supressing premature B cell activation prior to GC formation and inhibiting B cell differentiation [165,166,167]. TXNIP is reported to promote GC development by suppressing BCL-6 activity [168]. Txnip−/− mice are reported to exhibit large secondary follicles with a GC-like structure and a higher population of Ki-67+ B cells in the spleen [168]. TXNIP has also been shown to be expressed at different stages of B cell development [118]. As a central metabolic gatekeeper, TXNIP restricts glucose and energy supply, which are essential for pre-B cell development [118]. Additionally, deletion of TXNIP provides strong survival advantage and rescues prednisolone-induced cell death in pre-B Acute Lymphoblastic Leukemia (ALL) cells due to removal of ATP production [118]. Collectively, TXNIP is involved in the maintenance and activation of different adaptive immune cell types, with its main impact potentially being on metabolic and subsequent phenotypic changes.
TXNIP-targeting therapeutics
As we have discussed, it is clear that TXNIP is associated with multiple biological functions that are critical for the development of several pathological processes. Consequently, there are a number of therapeutic strategies currently aimed at modulating TXNIP expression/functions for clinical application.
TXNIP can contribute to disease by regulating oxidative and glycolytic stress, inflammation, and by inhibiting the cell cycle. These notions are supported by cumulative evidence that loss of TXNIP by pharmacological inhibition or genetic perturbations results in amelioration from neurological disease and diabetes in murine models [3, 169]. TXNIP antagonists have been comprehensively reviewed by Qayyum et al. [170], and consist of small-molecule drug, phytochemicals, and peptides. Two well-characterised drugs, verapamil (NCT02372253) and Taurine (NCT01226537), that modulate TXNIP levels are currently being tested in clinical trials for the treatment of type 1 and 2 diabetes. Verapamil, a non-dihydropyridine L-type calcium channel blocker traditionally used orally for the treatment of hypertension, inhibits TXNIP expression [171]. A recent high-throughput screen has identified another TXNIP inhibitor SRI-37330, which significantly decreased TXNIP expression, glucagon secretion, and hepatic glucose output, thereby being championed as a potential treatment for diabetes [172]. Interestingly and in contrast to these inhibitors, taurine, used for glycaemic control in diabetic patients, is reported to increase TXNIP expression [173].
In the context of cancer treatment, TXNIP agonists hold great potential as antitumor agents. Vorinostat, a pan histone-deacetylase inhibitor, and rapamycin, an mTORC1 inhibitor, have been shown to limit disease progression in Ras-driven cancers, with the ability to induce TXNIP expression [105]. Targeted therapies in breast cancer such as trastuzumab, cetuximab, and lapatinib, which block the Her-1/2 pathway, can cause G1 cell cycle arrest and also highly increase TXNIP expression [174]. Additionally, in TNBC, silibinin, commonly used in the treatment of toxic liver damage, has been shown to upregulate TXNIP, which suppresses glycolysis and cell proliferation [175]. BRAFi, which has been approved to treat advanced melanoma and proved to show strong clinical benefit in BRAFv600 melanoma [48, 176, 177], can also induce TXNIP expression through regulating the association between MondoA and TXNIP promoter [176]. Notably, the expression level of TXNIP is associated with favorable clinical response to BRAFi [176]. With a renewed emphasis on therapies which modulate the tumor metabolome, these and additional TXNIP agonists may show great potential.
Conclusion
TXNIP is a multifaceted protein involved in several fundamental biological processes and therefore is potential pharmacological target for multiple applications. Its ability to regulate glycolytic stress, oxidative stress, ER stress and the cell cycle gives it a central role in balancing different cell states, leading to various cell fates. Accordingly, TXNIP can drive both beneficial and detrimental effects in different pathologies, like metabolic diseases and cancer. Consequently, TXNIP antagonists are candidates for treating diabetes and neurological diseases, whereas TXNIP agonists hold potential for cancer treatment.
TXNIP regulatory networks are complex and feedback loops render them mutually regulatory. Apart from the mechanisms reviewed by Masutani et al. [35], oncogenic and tumor suppressor genes, ER stress signaling, cytokines, and other conditions like hypoxia also modulate TXNIP expression. Oncogene-mediated downregulation of TXNIP is mostly associated with inhibition of cell death and an increase in cellular metabolism, which favour tumor proliferative abilities and resistance to anticancer treatment [48]. Cellular recovery from ER stress via UPR signaling alters TXNIP as well. So far, two of three UPR transducers are mainly reported in TXNIP regulation, namely PERK and IRE-1a [31]. In certain conditions, they work together or separately for TXNIP alteration. Several effectors lie at the downstream signaling of PERK-eIF2a axis to determine cell fate [178]. ATF4 is a well-known early acute UPR and terminal-UPR mediator and reported to be required for TXNIP-mediated NLRP1 inflammasome activation instead of NLRP3 inflammasome activation [178, 179]. The literature reviewed here suggests that the stability of TXNIP serves as key switch between terminal UPR and adaptive UPR, with clear integrative mechanisms requiring further elucidation. Cytokines not only regulate survival, proliferation, differentiation and the function of immune cells, but also contribute to reshaping the TME [180], including through their exosomal binding [181]. Exosomes and TXNIP also have an intimate relationship as several miRNAs shuttled in exosomes mediate TXNIP downregulation, which can complement the effects of cytokines in the tumor microenvironment [42, 87, 91]. The regulation of TXNIP expression, therefore, appears to be under the control of a plethora of inter-cellular signals (multiple cytokines and exosomal miRNAs), which makes it complex to establish the exact role of TXNIP in tumor microenvironment-driven tumor progression [182].
The molecular mechanisms of TXNIP regulation of cell cycle, inflammation and glycolysis can have tremendous consequences on both tumor and immune cells. With important roles in several cancer types, TXNIP affects cell proliferation and death, drug sensitivity, angiogenesis, and glycolysis [16]. Consequently, TXNIP is closely involved in the remodelling of the TME, especially the immune compartment. The importance of immune contexture has been emphasised in cancer control in recent years [182]. Other than the interaction between TXNIP and NF-κB or inflammasome signaling, the roles of TXNIP in both innate and adaptive immune modulation suggest its potential role as a target for drug discoveries. NK cells and DCs are two specialised innate immune cell types, acting as the main effector and antigen-presenting cells, respectively [183, 184]. Deletion of TXNIP restrains the development and maturation of NK cells and functions of DCs, which causes the dysfunction of antitumor immunity [148, 151]. However, TXNIP is also observed in activated PMD-MDSCs and potentially associated with their suppressive activity [152]. As well, TXNIP inhibits the generation of protective memory T cells via degradation of CPT1a [157]. Moreover, TXNIP affects the proliferation and activation of T cells, and is crucial to maintain Tregs identity and its immune-suppressive function [163]. In addition, TXNIP is involved in GCs formation and development of B cells at different stages [118, 168].
In summary, as a central element receiving inputs from multiple extracellular signals, and acting as an intracellular hub for ROS homeostasis, metabolic responses, stress integration, immune functions, and cellular outcomes, TXNIP holds a crucial and pivotal role in health and disease. TXNIP offers an attractive point of pharmacological intervention. Future studies and clinical trials in humans will eventually translate the vast scientific research in the field of TXNIP regulation, into tangible outcomes for the benefit of multiple patient groups.
Authors’ comment
We set out to write as comprehensive a review as possible, and although there are contradictory reports, a consensus view as to TXNIP’s role in cellular and tissue homeostasis emerges. It’s central role can be clearly seen in maintaining cellular health in a supportive tissue environment, post-stress, and although not yet robustly demonstrated, it seems reasonable to hypothesise that under normal physiological conditions, the return of TXNIP to resting levels occurs quickly in co-ordination with resolution.
Interestingly, in cancers, frequently considered diseases of chronic epithelial stress, TXNIP is more commonly seen to be decreased in expression compared to healthy tissue at clinical presentation (Table 1), with additional stressors such as chemotherapy, hypoxia, or acidification seen to increase expression back towards ‘healthy’ expression levels. Given TXNIP’s central role, its loss being a positive for cancer survival is prima face contradictory, however, the key here is perhaps TXNIP’s impact on its environment.
This current “age of the TME” is increasingly revealing that successful tumors subvert their proximal, and sometimes distal, environments, indeed there is a live debate concerning whether or not certain tumors emerge because of their local environment rather than create it. Here we see that the loss TXNIP in epithelial cells has been shown to inhibit the inflammasome, but beyond this, the loss of environmental TXNIP, something that can be driven by a shared stressor or crosstalk (via common mediators), decreases NK cell generation and myeloid (including DC) activation.
Given the importance of the innate immune system in developing and maintaining the TME, this suggests to us that low environmental TXNIP is important in allowing for tumor development. Although not in cancer, an example of this “shared movement” of TXNIP across multiple cells types in the systemic environment can be seen in type 2 diabetes where high TXNIP can be seen in the PBMCs of diabetic patients, with correlations with ER stress and a common stressor, alongside inflammasome induction [185].
When viewed in the round, it is striking that TXNIP increases inflammation and antigenicity (if one accepts the PDAC TAM paper as evidence of macrophage differentiation per se) via the epithelial and innate compartments whilst simultaneously inhibiting the adaptive arms. To us, this resembles an immunological program designed for the innate-driven clearance of stressed cells whilst limiting the likelihood of adaptive-driven autoimmunity. There is an additional clue in the GC data, in that TXNIP promotes development, suggesting to us, that TXNIP may prime the adaptive arm through, for example the enhancement of local ectopic lymphoid structures, allowing adaptive immunity to react as soon as local TXNIP levels drop, if required.
Intriguingly, when considering the adaptive arm, low TXNIP is associated with increased memory T cell (CD4 and CD8) efficacy and lower numbers of Tregs, however without effective APCs or neoantigen presentation (e.g., IFNγ dependent immunoproteosomal switch) the positive impacts of these changes are minimal. A sudden increase in TXNIP expression however, through for example chemotherapy, may alter these dynamics dramatically, allowing for innate responses. If the increase is temporary, and cyclical, as with most chemotherapeutic regimes, we hypothesise that the cycles of innate stimulation/adaptive inhibition, innate inhibition/adaptive stimulation in part through TXNIP regulation may promote tumor destruction and a return to normal tissue homeostasis.
For these authors, the next steps in trying to understand the role of TXNIP in cancer, are to understand which functions of TXNIP are important in each different biological context. For example, although crudely speaking most primary carcinomas express low levels of TXNIP, what is the key reason for this—metabolism, the inflammasome, angiogenesis, or immune crosstalk? To help in this endeavor, there are variants, for example the TXNIP-T variant, that associate with an increased propensity to develop diabetes [38]. The critical cysteines for thioredoxin binding have been identified on TXNIP [186], and recent data shows that the C247S mutation protects against myocardial infarction in mice [187] whilst also regulating adipogenesis [188]. As such, the stage is now set for the field to assess the importance of thioredoxin binding in cancer models, and in so doing begin to decipher the impact of TXNIP’s diverse roles more specifically.
References
Yoshihara E, Masaki S, Matsuo Y, Chen Z, Tian H, Yodoi J. Thioredoxin/Txnip: redoxisome, as a redox switch for the pathogenesis of diseases. Front Immunol. 2014;4:514.
Li N, Zhou H, Wu H, Wu Q, Duan M, Deng W, et al. STING-IRF3 contributes to lipopolysaccharide-induced cardiac dysfunction, inflammation, apoptosis and pyroptosis by activating NLRP3. Redox Biol. 2019;24:101215.
Nasoohi S, Ismael S, Ishrat T. Thioredoxin-interacting protein (TXNIP) in cerebrovascular and neurodegenerative diseases: regulation and implication. Mol Neurobiol. 2018;55:7900–20.
Ren X, Zou L, Zhang X, Branco V, Wang J, Carvalho C, et al. Redox signaling mediated by thioredoxin and glutathione systems in the central nervous system. Antioxid Redox Signal. 2017;27:989–1010.
Nishiyama A, Matsui M, Iwata S, Hirota K, Masutani H, Nakamura H, et al. Identification of thioredoxin-binding protein-2/vitamin D(3) up-regulated protein 1 as a negative regulator of thioredoxin function and expression. J Biol Chem. 1999;274:21645–50.
Junn E, Han SH, Im JY, Yang Y, Cho EW, Um HD, et al. Vitamin D3 up-regulated protein 1 mediates oxidative stress via suppressing the thioredoxin function. J Immunol. 2000;164:6287–95.
Chen KS, DeLuca HF. Isolation and characterization of a novel cDNA from HL-60 cells treated with 1,25-dihydroxyvitamin D-3. Biochim Biophys Acta. 1994;1219:26–32.
Otaki Y, Takahashi H, Watanabe T, Funayama A, Netsu S, Honda Y, et al. HECT-type ubiquitin E3 ligase ITCH interacts with thioredoxin-interacting protein and ameliorates reactive oxygen species-induced cardiotoxicity. J Am Heart Assoc. 2016;5:e002485.
Yoshioka J, Imahashi K, Gabel SA, Chutkow WA, Burds AA, Gannon J, et al. Targeted deletion of thioredoxin-interacting protein regulates cardiac dysfunction in response to pressure overload. Circ Res. 2007;101:1328–38.
Yoshioka J, Schulze PC, Cupesi M, Sylvan JD, MacGillivray C, Gannon J, et al. Thioredoxin-interacting protein controls cardiac hypertrophy through regulation of thioredoxin activity. Circulation. 2004;109:2581–6.
Nishizawa K, Nishiyama H, Matsui Y, Kobayashi T, Saito R, Kotani H, et al. Thioredoxin-interacting protein suppresses bladder carcinogenesis. Carcinogenesis. 2011;32:1459–66.
Jiao D, Huan Y, Zheng J, Wei M, Zheng G, Han D, et al. UHRF1 promotes renal cell carcinoma progression through epigenetic regulation of TXNIP. Oncogene. 2019;38:5686–99.
Morrison JA, Pike LA, Sams SB, Sharma V, Zhou Q, Severson JJ, et al. Thioredoxin interacting protein (TXNIP) is a novel tumor suppressor in thyroid cancer. Mol Cancer. 2014;13:62.
Guo X, Huang M, Zhang H, Chen Q, Hu Y, Meng Y, et al. A pan-cancer analysis of thioredoxin-interacting protein as an immunological and prognostic biomarker. Cancer Cell Int. 2022;22:230.
Schroder J, Schumacher U, Bockelmann LC. Thioredoxin Interacting Protein (TXNIP) is differentially expressed in human tumor samples but is absent in human tumor cell line xenografts: implications for its use as an immunosurveillance marker. Cancers. 2020;12:3028.
Chen Y, Ning J, Cao W, Wang S, Du T, Jiang J, et al. Research progress of TXNIP as a tumor suppressor gene participating in the metabolic reprogramming and oxidative stress of cancer cells in various cancers. Front Oncol. 2020;10:568574.
Sheth SS, Bodnar JS, Ghazalpour A, Thipphavong CK, Tsutsumi S, Tward AD, et al. Hepatocellular carcinoma in Txnip-deficient mice. Oncogene. 2006;25:3528–36.
Rindler K, Jonak C, Alkon N, Thaler FM, Kurz H, Shaw LE, et al. Single-cell RNA sequencing reveals markers of disease progression in primary cutaneous T-cell lymphoma. Mol Cancer. 2021;20:124.
Tome ME, Johnson DB, Rimsza LM, Roberts RA, Grogan TM, Miller TP, et al. A redox signature score identifies diffuse large B-cell lymphoma patients with a poor prognosis. Blood. 2005;106:3594–601.
Li Y, Miao LY, Xiao YL, Huang M, Yu M, Meng K, et al. Hypoxia induced high expression of thioredoxin interacting protein (TXNIP) in non-small cell lung cancer and its prognostic effect. Asian Pac J Cancer Prev. 2015;16:2953–8.
Gunes A, Bagirsakci E, Iscan E, Cakan-Akdogan G, Aykutlu U, Senturk S, et al. Thioredoxin interacting protein promotes invasion in hepatocellular carcinoma. Oncotarget. 2018;9:36849–66.
Meszaros M, Yusenko M, Domonkos L, Peterfi L, Kovacs G, Banyai D. Expression of TXNIP is associated with angiogenesis and postoperative relapse of conventional renal cell carcinoma. Sci Rep. 2021;11:17200.
Spaeth-Cook D, Burch M, Belton R, Demoret B, Grosenbacher N, David J, et al. Loss of TXNIP enhances peritoneal metastasis and can be abrogated by dual TORC1/2 inhibition. Oncotarget. 2018;9:35676–86.
Xu G, Chen J, Jing G, Shalev A. Thioredoxin-interacting protein regulates insulin transcription through microRNA-204. Nat Med. 2013;19:1141–6.
Jing G, Westwell-Roper C, Chen J, Xu G, Verchere CB, Shalev A. Thioredoxin-interacting protein promotes islet amyloid polypeptide expression through miR-124a and FoxA2. J Biol Chem. 2014;289:11807–15.
Parikh H, Carlsson E, Chutkow WA, Johansson LE, Storgaard H, Poulsen P, et al. TXNIP regulates peripheral glucose metabolism in humans. PLoS Med. 2007;4:e158.
Ji Cho M, Yoon SJ, Kim W, Park J, Lee J, Park JG, et al. Oxidative stress-mediated TXNIP loss causes RPE dysfunction. Exp Mol Med. 2019;51:1–13.
Hui ST, Andres AM, Miller AK, Spann NJ, Potter DW, Post NM, et al. Txnip balances metabolic and growth signaling via PTEN disulfide reduction. Proc Natl Acad Sci USA. 2008;105:3921–6.
Lee S, Min Kim S, Dotimas J, Li L, Feener EP, Baldus S, et al. Thioredoxin-interacting protein regulates protein disulfide isomerases and endoplasmic reticulum stress. EMBO Mol Med. 2014;6:732–43.
Chen CL, Lin CF, Chang WT, Huang WC, Teng CF, Lin YS. Ceramide induces p38 MAPK and JNK activation through a mechanism involving a thioredoxin-interacting protein-mediated pathway. Blood. 2008;111:4365–74.
Shen L, O’Shea JM, Kaadige MR, Cunha S, Wilde BR, Cohen AL, et al. Metabolic reprogramming in triple-negative breast cancer through Myc suppression of TXNIP. Proc Natl Acad Sci USA. 2015;112:5425–30.
Kwon HJ, Won YS, Nam KT, Yoon YD, Jee H, Yoon WK, et al. Vitamin D(3) upregulated protein 1 deficiency promotes N-methyl-N-nitrosourea and Helicobacter pylori-induced gastric carcinogenesis in mice. Gut. 2012;61:53–63.
Stoltzman CA, Peterson CW, Breen KT, Muoio DM, Billin AN, Ayer DE. Glucose sensing by MondoA:Mlx complexes: a role for hexokinases and direct regulation of thioredoxin-interacting protein expression. Proc Natl Acad Sci USA. 2008;105:6912–7.
Wilde BR, Ye Z, Lim TY, Ayer DE. Cellular acidosis triggers human MondoA transcriptional activity by driving mitochondrial ATP production. Elife. 2019;8:e40199.
Masutani H. Thioredoxin-interacting protein in cancer and diabetes. Antioxid Redox Signal. 2022;36:1001–22.
Shen L, O’Shea JM, Kaadige MR, Cunha S, Wilde BR, Cohen AL, et al. Metabolic reprogramming in triple-negative breast cancer through Myc suppression of TXNIP. Proc Natl Acad Sci USA. 2015;112:5425–5430.
Noblet B, Benhamed F, O-Sullivan I, Zhang W, Filhoulaud G, Montagner A, et al. Dual regulation of TxNIP by ChREBP and FoxO1 in liver. iScience. 2021;24:102218.
Tang JY, Li DY, He L, Qiu XS, Wang EH, Wu GP. HPV 16 E6/E7 promote the glucose uptake of GLUT1 in lung cancer through downregulation of TXNIP due to inhibition of PTEN phosphorylation. Front Oncol. 2020;10:559543.
Ganguly D, Sims M, Cai C, Fan M, Pfeffer LM. Chromatin remodeling factor BRG1 regulates stemness and chemosensitivity of glioma initiating cells. Stem Cells. 2018;36:1804–15.
Dai Q, Li N, Zhou X. Increased miR-21a provides metabolic advantages through suppression of FBP1 expression in non-small cell lung cancer cells. Am J Cancer Res. 2017;7:2121–30.
Okumura S, Hirano Y, Komatsu Y. Stable duplex-linked antisense targeting miR-148a inhibits breast cancer cell proliferation. Sci Rep. 2021;11:11467.
Yin H, Yu S, Xie Y, Dai X, Dong M, Sheng C, et al. Cancer-associated fibroblasts-derived exosomes upregulate microRNA-135b-5p to promote colorectal cancer cell growth and angiogenesis by inhibiting thioredoxin-interacting protein. Cell Signal. 2021;84:110029.
Li K, Tang M, Tong S, Wang C, Sun Q, Lv M, et al. BRAFi induced demethylation of miR-152-5p regulates phenotype switching by targeting TXNIP in cutaneous melanoma. Apoptosis. 2020;25:179–91.
Yi S, Sun J, Qiu L, Fu W, Wang A, Liu X, et al. Dual role of EZH2 in cutaneous anaplastic large cell lymphoma: promoting tumor cell survival and regulating tumor microenvironment. J Investig Dermatol. 2018;138:1126–36.
Brocker CN, Kim D, Melia T, Karri K, Velenosi TJ, Takahashi S, et al. Long non-coding RNA Gm15441 attenuates hepatic inflammasome activation in response to PPARA agonism and fasting. Nat Commun. 2020;11:5847.
Zhu QQ, Lai MC, Chen TC, Wang X, Tian L, Li DL, et al. LncRNA SNHG15 relieves hyperglycemia-induced endothelial dysfunction via increasing ubiquitination of thioredoxin-interacting protein. Life Sci. 2021;101:1142–52.
Elgort MG, O’Shea JM, Jiang Y, Ayer DE. Transcriptional and translational downregulation of thioredoxin interacting protein is required for metabolic reprogramming during G(1). Genes Cancer. 2010;1:893–907.
Wilde BR, Ayer DE. Interactions between Myc and MondoA transcription factors in metabolism and tumourigenesis. Br J Cancer. 2015;113:1529–33.
Liao W, Overman MJ, Boutin AT, Shang X, Zhao D, Dey P, et al. KRAS-IRF2 axis drives immune suppression and immune therapy resistance in colorectal cancer. Cancer Cell. 2019;35:559–72.
Ye Z, Ayer DE. Ras suppresses TXNIP expression by restricting ribosome translocation. Mol Cell Biol. 2018;38:e00178–18.
Cadenas C, Franckenstein D, Schmidt M, Gehrmann M, Hermes M, Geppert B, et al. Role of thioredoxin reductase 1 and thioredoxin interacting protein in prognosis of breast cancer. Breast Cancer Res. 2010;12:R44.
Avril T, Vauleon E, Chevet E. Endoplasmic reticulum stress signaling and chemotherapy resistance in solid cancers. Oncogenesis. 2017;6:e373.
Chen X, Cubillos-Ruiz JR. Endoplasmic reticulum stress signals in the tumour and its microenvironment. Nat Rev Cancer. 2021;21:71–88.
Wang Y, Chen S. TXNIP links anticipatory unfolded protein response to estrogen reprogramming glucose metabolism in breast cancer cells. Endocrinology. 2022;163:bqab212.
Lerner AG, Upton JP, Praveen PV, Ghosh R, Nakagawa Y, Igbaria A, et al. IRE1alpha induces thioredoxin-interacting protein to activate the NLRP3 inflammasome and promote programmed cell death under irremediable ER stress. Cell Metab. 2012;16:250–64.
Bronner DN, Abuaita BH, Chen X, Fitzgerald KA, Nunez G, He Y, et al. Endoplasmic reticulum stress activates the inflammasome via NLRP3- and caspase-2-driven mitochondrial damage. Immunity. 2015;43:451–62.
Zhao Q, Che X, Zhang H, Tan G, Liu L, Jiang D, et al. Thioredoxin-interacting protein mediates apoptosis in early brain injury after subarachnoid haemorrhage. Int J Mol Sci. 2017;18:854.
West NR, McCuaig S, Franchini F, Powrie F. Emerging cytokine networks in colorectal cancer. Nat Rev Immunol. 2015;15:615–29.
Levring TB, Kongsbak-Wismann M, Rode AKO, Al-Jaberi FAH, Lopez DV, Met O, et al. Tumor necrosis factor induces rapid down-regulation of TXNIP in human T cells. Sci Rep. 2019;9:16725.
Nagaraj K, Lapkina-Gendler L, Sarfstein R, Gurwitz D, Pasmanik-Chor M, Laron Z, et al. Identification of thioredoxin-interacting protein (TXNIP) as a downstream target for IGF1 action. Proc Natl Acad Sci USA. 2018;115:1045–50.
Kanari Y, Sato Y, Aoyama S, Muta T. Thioredoxin-interacting protein gene expression via MondoA is rapidly and transiently suppressed during inflammatory responses. PLoS One. 2013;8:e59026.
Contreras O, Cruz-Soca M, Theret M, Soliman H, Tung LW, Groppa E, et al. Cross-talk between TGF-beta and PDGFRalpha signaling pathways regulates the fate of stromal fibro-adipogenic progenitors. J Cell Sci. 2019;132:jcs232157.
Han SH, Jeon JH, Ju HR, Jung U, Kim KY, Yoo HS, et al. VDUP1 upregulated by TGF-beta1 and 1,25-dihydorxyvitamin D3 inhibits tumor cell growth by blocking cell-cycle progression. Oncogene. 2003;22:4035–46.
Wu N, Zheng B, Shaywitz A, Dagon Y, Tower C, Bellinger G, et al. AMPK-dependent degradation of TXNIP upon energy stress leads to enhanced glucose uptake via GLUT1. Mol Cell. 2013;49:1167–75.
Baker AF, Koh MY, Williams RR, James B, Wang H, Tate WR, et al. Identification of thioredoxin-interacting protein 1 as a hypoxia-inducible factor 1alpha-induced gene in pancreatic cancer. Pancreas. 2008;36:178–86.
Le Jan S, Le Meur N, Cazes A, Philippe J, Le Cunff M, Leger J, et al. Characterization of the expression of the hypoxia-induced genes neuritin, TXNIP and IGFBP3 in cancer. FEBS Lett. 2006;580:3395–400.
Wang P, Zheng D, Qi H, Gao Q. Thioredoxin-interacting protein is a favored target of miR-125b, promoting metastasis and progression of pancreatic cancer via the HIF1alpha pathway. J Biochem Mol Toxicol. 2021;35:e22782.
Karmi O, Sohn YS, Zandalinas SI, Rowland L, King SD, Nechushtai R, et al. Disrupting CISD2 function in cancer cells primarily impacts mitochondrial labile iron levels and triggers TXNIP expression. Free Radic Biol Med. 2021;176:92–104.
Shimizu H, Tsubota T, Kanki K, Shiota G. All-trans retinoic acid ameliorates hepatic stellate cell activation via suppression of thioredoxin interacting protein expression. J Cell Physiol. 2018;233:607–16.
Hwang J, Suh HW, Jeon YH, Hwang E, Nguyen LT, Yeom J, et al. The structural basis for the negative regulation of thioredoxin by thioredoxin-interacting protein. Nat Commun. 2014;5:2958.
Koshy NV, Friday E, Turturro F. Thioredoxin interacting protein (TXNIP) correlates with production of reactive oxygen species (ROS) and underlines differences in stress response gene profile in patients with chronic lymphocytic leukemia (CLL). Blood. 2009;114:4375.
Hu H, Tian M, Li P, Guan X, Lian Z, Yin Y, et al. Brucella infection regulates thioredoxin-interacting protein expression to facilitate intracellular survival by reducing the production of nitric oxide and reactive oxygen species. J Immunol. 2020;204:632–43.
Oberacker T, Bajorat J, Ziola S, Schroeder A, Roth D, Kastl L, et al. Enhanced expression of thioredoxin-interacting-protein regulates oxidative DNA damage and aging. FEBS Lett. 2018;592:2297–307.
Gao C, Wang R, Li B, Guo Y, Yin T, Xia Y, et al. TXNIP/Redd1 signalling and excessive autophagy: a novel mechanism of myocardial ischaemia/reperfusion injury in mice. Cardiovasc Res. 2020;116:645–57.
Jung H, Kim MJ, Kim DO, Kim WS, Yoon SJ, Park YJ, et al. TXNIP maintains the hematopoietic cell pool by switching the function of p53 under oxidative stress. Cell Metab. 2013;18:75–85.
Reczek CR, Chandel NS. The two faces of reactive oxygen species in cancer. Annu Rev Cancer Biol 2017;1:79–98.
Kamitori K, Yamaguchi F, Dong Y, Hossain A, Katagi A, Noguchi C, et al. Both Ser361 phosphorylation and the C-arrestin domain of thioredoxin interacting protein are important for cell cycle blockade at the G1/S checkpoint. FEBS Open Bio. 2018;8:1804–19.
Jeon JH, Lee KN, Hwang CY, Kwon KS, You KH, Choi I. Tumor suppressor VDUP1 increases p27(kip1) stability by inhibiting JAB1. Cancer Res. 2005;65:4485–9.
Kim MJ, Kim WS, Kim DO, Byun JE, Huy H, Lee SY, et al. Macrophage migration inhibitory factor interacts with thioredoxin-interacting protein and induces NF-kappaB activity. Cell Signal. 2017;34:110–20.
Riahi Y, Kaiser N, Cohen G, Abd-Elrahman I, Blum G, Shapira OM, et al. Foam cell-derived 4-hydroxynonenal induces endothelial cell senescence in a TXNIP-dependent manner. J Cell Mol Med. 2015;19:1887–99.
Sharma C, Wang HX, Li Q, Knoblich K, Reisenbichler ES, Richardson AL, et al. Protein acyltransferase DHHC3 regulates breast tumor growth, oxidative stress, and senescence. Cancer Res. 2017;77:6880–90.
Huang C, Zhang Y, Kelly DJ, Tan CY, Gill A, Cheng D, et al. Thioredoxin interacting protein (TXNIP) regulates tubular autophagy and mitophagy in diabetic nephropathy through the mTOR signaling pathway. Sci Rep. 2016;6:29196.
Qiao S, Dennis M, Song X, Vadysirisack DD, Salunke D, Nash Z, et al. A REDD1/TXNIP pro-oxidant complex regulates ATG4B activity to control stress-induced autophagy and sustain exercise capacity. Nat Commun. 2015;6:7014.
Zhou J, Bi C, Cheong LL, Mahara S, Liu SC, Tay KG, et al. The histone methyltransferase inhibitor, DZNep, up-regulates TXNIP, increases ROS production, and targets leukemia cells in AML. Blood. 2011;118:2830–9.
Hu J, Feng L, Ren M, Zhao Y, Lu G, Lu X, et al. Colorectal cancer cell differentiation is dependent on the repression of aerobic glycolysis by NDRG2-TXNIP axis. Dig Dis Sci. 2022;67:3763–72.
Zeng H, Gu H, Chen C, Li M, Xia F, Xie L, et al. ChREBP promotes the differentiation of leukemia-initiating cells to inhibit leukemogenesis through the TXNIP/RUNX1 pathways. Oncotarget. 2016;7:38347–58.
Li W, Xin X, Li X, Geng J, Sun Y. Exosomes secreted by M2 macrophages promote cancer stemness of hepatocellular carcinoma via the miR-27a-3p/TXNIP pathways. Int Immunopharmacol. 2021;101:107585.
Ogata FT, Simoes Sato AY, Coppo L, Arai RJ, Stern AI, Pequeno Monteiro H. Thiol-based antioxidants and the epithelial/mesenchymal transition in cancer. Antioxid Redox Signal. 2022;36:1037–50.
Bechard ME, Smalling R, Hayashi A, Zhong Y, Word AE, Campbell SL, et al. Pancreatic cancers suppress negative feedback of glucose transport to reprogram chromatin for metastasis. Nat Commun. 2020;11:4055.
Yang KH, Kim GT, Choi S, Yoon SY, Kim JW. 1‑Palmitoyl‑2‑linoleoyl‑3‑acetyl‑rac‑glycerol ameliorates EGF‑induced MMP‑9 expression by promoting receptor desensitization in MDA‑MB‑231 cells. Oncol Rep. 2020;44:241–51.
Yang SS, Ma S, Dou H, Liu F, Zhang SY, Jiang C, et al. Breast cancer-derived exosomes regulate cell invasion and metastasis in breast cancer via miR-146a to activate cancer associated fibroblasts in tumor microenvironment. Exp Cell Res. 2020;391:111983.
Gunes A, Iscan E, Topel H, Avci ST, Gumustekin M, Erdal E, et al. Heparin treatment increases thioredoxin interacting protein expression in hepatocellular carcinoma cells. Int J Biochem Cell Biol. 2015;65:169–81.
Song M. Cancer overtakes vascular disease as leading cause of excess death associated with diabetes. Lancet Diabetes Endocrinol. 2021;9:131–3.
Shang S, Yang YW, Chen F, Yu L, Shen SH, Li K, et al. TRIB3 reduces CD8(+) T cell infiltration and induces immune evasion by repressing the STAT1-CXCL10 axis in colorectal cancer. Sci Transl Med. 2022;14:f992.
Ringel AE, Drijvers JM, Baker GJ, Catozzi A, Garcia-Canaveras JC, Gassaway BM, et al. Obesity shapes metabolism in the tumor microenvironment to suppress anti-tumor immunity. Cell. 2020;183:1848–66.
Maguire OA, Ackerman SE, Szwed SK, Maganti AV, Marchildon F, Huang X, et al. Creatine-mediated crosstalk between adipocytes and cancer cells regulates obesity-driven breast cancer. Cell Metab. 2021;33:499–512.
Wishart AL, Conner SJ, Guarin JR, Fatherree JP, Peng Y, McGinn RA, et al. Decellularized extracellular matrix scaffolds identify full-length collagen VI as a driver of breast cancer cell invasion in obesity and metastasis. Sci Adv. 2020;6:eabc3175.
McDowell SAC, Luo RBE, Arabzadeh A, Dore S, Bennett NC, Breton V, et al. Neutrophil oxidative stress mediates obesity-associated vascular dysfunction and metastatic transmigration. Nat Cancer. 2021;2:545–62.
Chen J, Saxena G, Mungrue IN, Lusis AJ, Shalev A. Thioredoxin-interacting protein: a critical link between glucose toxicity and beta-cell apoptosis. Diabetes. 2008;57:938–44.
Zhou R, Tardivel A, Thorens B, Choi I, Tschopp J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat Immunol. 2010;11:136–40.
Hui TY, Sheth SS, Diffley JM, Potter DW, Lusis AJ, Attie AD, et al. A. Mice lacking thioredoxin-interacting protein provide evidence linking cellular redox state to appropriate response to nutritional signals. J Biol Chem. 2004;279:24387–93.
Sheth SS, Castellani LW, Chari S, Wagg C, Thipphavong CK, Bodnar JS, et al. Thioredoxin-interacting protein deficiency disrupts the fasting-feeding metabolic transition. J Lipid Res. 2005;46:123–34.
Esposito K, Chiodini P, Colao A, Lenzi A, Giugliano D. Metabolic syndrome and risk of cancer: a systematic review and meta-analysis. Diabetes Care. 2012;35:2402–11.
Kregel S, Malik R, Asangani IA, Wilder-Romans K, Rajendiran T, Xiao L, et al. Functional and mechanistic interrogation of BET bromodomain degraders for the treatment of metastatic castration-resistant prostate cancer. Clin Cancer Res. 2019;25:4038–48.
Malone CF, Emerson C, Ingraham R, Barbosa W, Guerra S, Yoon H, et al. mTOR and HDAC inhibitors converge on the TXNIP/thioredoxin pathway to cause catastrophic oxidative stress and regression of RAS-Driven tumors. Cancer Discov. 2017;7:1450–63.
Rodriguez-Garcia A, Hevia D, Mayo JC, Gonzalez-Menendez P, Coppo L, Lu J, et al. Thioredoxin 1 modulates apoptosis induced by bioactive compounds in prostate cancer cells. Redox Biol. 2017;12:634–47.
Schmidt L, Baskaran S, Johansson P, Padhan N, Matuszewski D, Green LC, et al. Case-specific potentiation of glioblastoma drugs by pterostilbene. Oncotarget. 2016;7:73200–15.
Cai W, Zhang L, Song Y, Wang B, Zhang B, Cui X, et al. Small molecule inhibitors of mammalian thioredoxin reductase. Free Radic Biol Med. 2012;52:257–65.
Kelland L. The resurgence of platinum-based cancer chemotherapy. Nat Rev Cancer. 2007;7:573–84.
Jia JJ, Geng WS, Wang ZQ, Chen L, Zeng XS. The role of thioredoxin system in cancer: strategy for cancer therapy. Cancer Chemother Pharm. 2019;84:453–70.
Di Y, Jiang Y, Shen X, Liu J, Gao Y, Cai H, et al. Downregulation of miR-135b-5p suppresses progression of esophageal cancer and contributes to the effect of cisplatin. Front Oncol. 2021;11:679348.
Lin CH, Lee HH, Chang WM, Lee FP, Chen LC, Lu LS, et al. FOXD1 repression potentiates radiation effectiveness by downregulating G3BP2 expression and promoting the activation of TXNIP-related pathways in oral cancer. Cancers. 2020;12:2690.
Zhou H, Shen Q, Fu J, Jiang F, Wang L, Wang Y. Analysis of lncRNA UCA1-related downstream pathways and molecules of cisplatin resistance in lung adenocarcinoma. J Clin Lab Anal. 2020;34:e23312.
Haas B, Schutte L, Wos-Maganga M, Weickhardt S, Timmer M, Eckstein N. Thioredoxin confers intrinsic resistance to cytostatic drugs in human glioma cells. Int J Mol Sci. 2018;19:2874.
Kanaji N, Kamitori K, Hossain A, Noguchi C, Katagi A, Kadowaki N, et al. Additive antitumour effect of D‑allose in combination with cisplatin in non-small cell lung cancer cells. Oncol Rep. 2018;39:1292–8.
Hoshikawa H, Kamitori K, Indo K, Mori T, Kamata M, Takahashi T, et al. Combined treatment with D-allose, docetaxel and radiation inhibits the tumor growth in an in vivo model of head and neck cancer. Oncol Lett. 2018;15:3422–8.
Dang CV. A time for MYC: metabolism and therapy. Cold Spring Harb Symp Quant Biol. 2016;81:79–83.
Chan LN, Chen Z, Braas D, Lee JW, Xiao G, Geng H, et al. Metabolic gatekeeper function of B-lymphoid transcription factors. Nature. 2017;542:479–83.
Alhawiti NM, Al Mahri S, Aziz MA, Malik SS, Mohammad S. TXNIP in metabolic regulation: physiological role and therapeutic outlook. Curr Drug Targets. 2017;18:1095–103.
Shen S, Yao T, Xu Y, Zhang D, Fan S, Ma J. CircECE1 activates energy metabolism in osteosarcoma by stabilizing c-Myc. Mol Cancer. 2020;19:151.
Liu Y, Feng W, Dai Y, Bao M, Yuan Z, He M, et al. Single-cell transcriptomics reveals the complexity of the tumor microenvironment of treatment-naive osteosarcoma. Front Oncol. 2021;11:709210.
Kuljaca S, Liu T, Dwarte T, Kavallaris M, Haber M, Norris MD, et al. The cyclin-dependent kinase inhibitor, p21(WAF1), promotes angiogenesis by repressing gene transcription of thioredoxin-binding protein 2 in cancer cells. Carcinogenesis. 2009;30:1865–71.
Ji S, Qin Y, Liang C, Huang R, Shi S, Liu J, et al. FBW7 (F-box and WD repeat domain-containing 7) negatively regulates glucose metabolism by targeting the c-Myc/TXNIP (Thioredoxin-Binding Protein) axis in pancreatic cancer. Clin Cancer Res. 2016;22:3950–60.
Feng L, Ding R, Qu X, Li Y, Shen T, Wang L, et al. BCR-ABL triggers a glucose-dependent survival program during leukemogenesis through the suppression of TXNIP. Cell Death Dis. 2023;14:287.
Farrell MR, Rogers LK, Liu Y, Welty SE, Tipple TE. Thioredoxin-interacting protein inhibits hypoxia-inducible factor transcriptional activity. Free Radic Biol Med. 2010;49:1361–7.
Navale AM, Paranjape AN. Glucose transporters: physiological and pathological roles. Biophys Rev. 2016;8:5–9.
Waldhart AN, Dykstra H, Peck AS, Boguslawski EA, Madaj ZB, Wen J, et al. Phosphorylation of TXNIP by AKT mediates acute influx of glucose in response to insulin. Cell Rep. 2017;19:2005–13.
Muraleedharan R, Gawali MV, Tiwari D, Sukumaran A, Oatman N, Anderson J, et al. AMPK-regulated astrocytic lactate shuttle plays a non-cell-autonomous role in neuronal survival. Cell Rep. 2020;32:108092.
Alexander CM, Martin JA, Oxman E, Kasza I, Senn KA, Dvinge H. Alternative splicing and cleavage of GLUT8. Mol Cell Biol. 2020;41:e00480–20.
Sullivan WJ, Mullen PJ, Schmid EW, Flores A, Momcilovic M, Sharpley MS, et al. Extracellular matrix remodeling regulates glucose metabolism through TXNIP destabilization. Cell. 2018;175:117–32.
Schafer ZT, Grassian AR, Song L, Jiang Z, Gerhart-Hines Z, Irie HY, et al. Antioxidant and oncogene rescue of metabolic defects caused by loss of matrix attachment. Nature. 2009;461:109–13.
Guo J, Xiao F, Ren W, Zhu Y, Du Q, Li Q, et al. Circular ribonucleic acid circFTO promotes angiogenesis and impairs blood-retinal barrier via targeting the miR-128-3p/thioredoxin interacting protein axis in diabetic retinopathy. Front Mol Biosci. 2021;8:685466.
Nasoohi S, Parveen K, Ishrat T. Metabolic syndrome, brain insulin resistance, and Alzheimer’s disease: thioredoxin interacting protein (TXNIP) and inflammasome as core amplifiers. J Alzheimers Dis. 2018;66:857–85.
Mohamed IN, Ishrat T, Fagan SC, El-Remessy AB. Role of inflammasome activation in the pathophysiology of vascular diseases of the neurovascular unit. Antioxid Redox Signal. 2015;22:1188–206.
Perrone L, Devi TS, Hosoya K, Terasaki T, Singh LP. Thioredoxin interacting protein (TXNIP) induces inflammation through chromatin modification in retinal capillary endothelial cells under diabetic conditions. J Cell Physiol. 2009;221:262–72.
Kwon HJ, Won YS, Suh HW, Jeon JH, Shao Y, Yoon SR, et al. Vitamin D3 upregulated protein 1 suppresses TNF-alpha-induced NF-kappaB activation in hepatocarcinogenesis. J Immunol. 2010;185:3980–9.
Schroder K, Zhou R, Tschopp J. The NLRP3 inflammasome: a sensor for metabolic danger? Science. 2010;327:296–300.
Cheng SB, Nakashima A, Huber WJ, Davis S, Banerjee S, Huang Z, et al. Pyroptosis is a critical inflammatory pathway in the placenta from early onset preeclampsia and in human trophoblasts exposed to hypoxia and endoplasmic reticulum stressors. Cell Death Dis. 2019;10:927.
Li L, Ismael S, Nasoohi S, Sakata K, Liao FF, McDonald MP, et al. Thioredoxin-interacting protein (TXNIP) associated NLRP3 inflammasome activation in human Alzheimer’s disease brain. J Alzheimer’s Dis. 2019;68:255–65.
Sharma BR, Kanneganti TD. NLRP3 inflammasome in cancer and metabolic diseases. Nat Immunol. 2021;22:550–9.
Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39:1–10.
Huang Y, Xu W, Zhou R. NLRP3 inflammasome activation and cell death. Cell Mol Immunol. 2021;18:2114–27.
Han C, Godfrey V, Liu Z, Han Y, Liu L, Peng H, et al. The AIM2 and NLRP3 inflammasomes trigger IL-1-mediated antitumor effects during radiation. Sci Immunol. 2021;6:eabc6998.
Tengesdal IW, Menon DR, Osborne DG, Neff CP, Powers NE, Gamboni F, et al. Targeting tumor-derived NLRP3 reduces melanoma progression by limiting MDSCs expansion. Proc Natl Acad Sci USA. 2021;118:e2000915118.
Lane T, Flam B, Lockey R, Kolliputi N. TXNIP shuttling: missing link between oxidative stress and inflammasome activation. Front Physiol. 2013;4:50.
Muri J, Thut H, Feng Q, Kopf M. Thioredoxin-1 distinctly promotes NF-kappaB target DNA binding and NLRP3 inflammasome activation independently of Txnip. Elife. 2020;9:e53627.
Pan Z, Shan Q, Gu P, Wang XM, Tai LW, Sun M, et al. miRNA-23a/CXCR4 regulates neuropathic pain via directly targeting TXNIP/NLRP3 inflammasome axis. J Neuroinflammation. 2018;15:29.
Lee KN, Kang HS, Jeon JH, Kim EM, Yoon SR, Song H, et al. VDUP1 is required for the development of natural killer cells. Immunity. 2005;22:195–208.
Taveirne S, Wahlen S, Van Loocke W, Kiekens L, Persyn E, Van Ammel E, et al. The transcription factor ETS1 is an important regulator of human NK cell development and terminal differentiation. Blood. 2020;136:288–98.
Yang Y, Neo SY, Chen Z, Cui W, Chen Y, Guo M, et al. Thioredoxin activity confers resistance against oxidative stress in tumor-infiltrating NK cells. J Clin Investig. 2020;130:5508–22.
Son A, Nakamura H, Okuyama H, Oka S, Yoshihara E, Liu W, et al. Dendritic cells derived from TBP-2-deficient mice are defective in inducing T cell responses. Eur J Immunol. 2008;38:1358–67.
Veglia F, Hashimoto A, Dweep H, Sanseviero E, De Leo A, Tcyganov E, et al. Analysis of classical neutrophils and polymorphonuclear myeloid-derived suppressor cells in cancer patients and tumor-bearing mice. J Exp Med. 2021;218:e20201803.
Xiang X, Wang J, Lu D, Xu X. Targeting tumor-associated macrophages to synergize tumor immunotherapy. Signal Transduct Target Ther. 2021;6:75.
Boyer S, Lee HJ, Steele N, Zhang L, Sajjakulnukit P, Andren A, et al. Multiomic characterization of pancreatic cancer-associated macrophage polarization reveals deregulated metabolic programs driven by the GM-CSF-PI3K pathway. Elife. 2022;11:e73796.
Li H, van der Leun AM, Yofe I, Lubling Y, Gelbard-Solodkin D, van Akkooi ACJ, et al. Dysfunctional CD8 T cells form a proliferative, dynamically regulated compartment within human melanoma. Cell. 2019;176:775–89.
Raud B, Roy DG, Divakaruni AS, Tarasenko TN, Franke R, Ma EH, et al. Etomoxir actions on regulatory and memory T cells are independent of Cpt1a-mediated fatty acid oxidation. Cell Metab. 2018;28:504–15.
Klein Geltink RI, O’Sullivan D, Corrado M, Bremser A, Buck MD, Buescher JM, et al. Mitochondrial priming by CD28. Cell. 2017;171:385–97.
Muri J, Thut H, Kopf M. The thioredoxin-1 inhibitor Txnip restrains effector T-cell and germinal center B-cell expansion. Eur J Immunol. 2021;51:115–24.
Dünnbier, MSS. The role of Thioredoxin-interacting protein in T cell receptor signalling. Dissertation 2019.
Jandova J, Wondrak GT. Genomic GLO1 deletion modulates TXNIP expression, glucose metabolism, and redox homeostasis while accelerating human A375 malignant melanoma tumor growth. Redox Biol. 2021;39:101838.
Saito T, Nishikawa H, Wada H, Nagano Y, Sugiyama D, Atarashi K, et al. Two FOXP3(+)CD4(+) T cell subpopulations distinctly control the prognosis of colorectal cancers. Nat Med. 2016;22:679–84.
Shi H, Chi H. Metabolic control of treg cell stability, plasticity, and tissue-specific heterogeneity. Front Immunol. 2019;10:2716.
Lu Y, Li Y, Liu Q, Tian N, Du P, Zhu F, et al. MondoA-thioredoxin-interacting protein axis maintains regulatory T-cell identity and function in colorectal cancer microenvironment. Gastroenterology. 2021;161:575–91.
Gerriets VA, Kishton RJ, Johnson MO, Cohen S, Siska PJ, Nichols AG, et al. Foxp3 and Toll-like receptor signaling balance T(reg) cell anabolic metabolism for suppression. Nat Immunol. 2016;17:1459–66.
Klein U, Dalla-Favera R. Germinal centres: role in B-cell physiology and malignancy. Nat Rev Immunol. 2008;8:22–33.
Tunyaplin C, Shaffer AL, Angelin-Duclos CD, Yu X, Staudt LM, Calame KL. Direct repression of prdm1 by Bcl-6 inhibits plasmacytic differentiation. J Immunol. 2004;173:1158–65.
Basso K, Dalla-Favera R. BCL6: master regulator of the germinal center reaction and key oncogene in B cell lymphomagenesis. Adv Immunol. 2010;105:193–210.
Shao Y, Kim SY, Shin D, Kim MS, Suh HW, Piao ZH, et al. TXNIP regulates germinal center generation by suppressing BCL-6 expression. Immunol Lett. 2010;129:78–84.
Chen J, Hui ST, Couto FM, Mungrue IN, Davis DB, Attie AD, et al. Thioredoxin-interacting protein deficiency induces Akt/Bcl-xL signaling and pancreatic beta-cell mass and protects against diabetes. FASEB J. 2008;22:3581–94.
Qayyum N, Haseeb M, Kim MS, Choi S. Role of thioredoxin-interacting protein in diseases and its therapeutic outlook. Int J Mol Sci. 2021;22:2754.
Malayeri A, Zakerkish M, Ramesh F, Galehdari H, Hemmati AA, Angali KA. The effect of verapamil on TXNIP gene expression, GLP1R mRNA, FBS, HbA1c, and Lipid Profile in T2DM patients receiving metformin and sitagliptin. Diabetes Ther. 2021;12:2701–13.
Thielen LA, Chen J, Jing G, Moukha-Chafiq O, Xu G, Jo S, et al. Identification of an anti-diabetic, orally available small molecule that regulates TXNIP expression and glucagon action. Cell Metab. 2020;32:353–65.
Gondo Y, Satsu H, Ishimoto Y, Iwamoto T, Shimizu M. Effect of taurine on mRNA expression of thioredoxin interacting protein in Caco-2 cells. Biochem Biophys Res Commun. 2012;426:433–7.
Nie W, Huang W, Zhang W, Xu J, Song W, Wang Y, et al. TXNIP interaction with the Her-1/2 pathway contributes to overall survival in breast cancer. Oncotarget. 2015;6:3003–12.
Iqbal MA, Chattopadhyay S, Siddiqui FA, Ur Rehman A, Siddiqui S, Prakasam G, et al. Silibinin induces metabolic crisis in triple-negative breast cancer cells by modulating EGFR-MYC-TXNIP axis: potential therapeutic implications. FEBS J. 2021;288:471–85.
Parmenter TJ, Kleinschmidt M, Kinross KM, Bond ST, Li J, Kaadige MR, et al. Response of BRAF-mutant melanoma to BRAF inhibition is mediated by a network of transcriptional regulators of glycolysis. Cancer Discov. 2014;4:423–33.
Carvalho LAC, Queijo RG, Baccaro ALB, Siena ADD, Silva WA Jr, Rodrigues T, et al. Redox-related proteins in melanoma progression. Antioxidation. 2022;11:438.
Anthony TG, Wek RC. TXNIP switches tracks toward a terminal UPR. Cell Metab. 2012;16:135–7.
D’Osualdo A, Anania VG, Yu K, Lill JR, Kaufman RJ, Matsuzawa S, et al. Transcription factor ATF4 Induces NLRP1 inflammasome expression during endoplasmic reticulum stress. PLoS One. 2015;10:e130635.
Luther SA, Cyster JG. Chemokines as regulators of T cell differentiation. Nat Immunol. 2001;2:102–7.
Lima LG, Ham S, Shin H, Chai EPZ, Lek ESH, Lobb RJ, et al. Tumor microenvironmental cytokines bound to cancer exosomes determine uptake by cytokine receptor-expressing cells and biodistribution. Nat Commun. 2021;12:3543.
Galon J, Bruni D. Tumor immunology and tumor evolution: intertwined histories. Immunity. 2020;52:55–81.
Myers JA, Miller JS. Exploring the NK cell platform for cancer immunotherapy. Nat Rev Clin Oncol. 2021;18:85–100.
Wculek SK, Cueto FJ, Mujal AM, Melero I, Krummel MF, Sancho D. Dendritic cells in cancer immunology and immunotherapy. Nat Rev Immunol. 2020;20:7–24.
Szpigel A, Hainault I, Carlier A, Venteclef N, Batto AF, Hajduch E, et al. Lipid environment induces ER stress, TXNIP expression and inflammation in immune cells of individuals with type 2 diabetes. Diabetologia. 2018;61:399–412.
Patwari P, Higgins LJ, Chutkow WA, Yoshioka J, Lee RT. The interaction of thioredoxin with Txnip. Evidence for formation of a mixed disulfide by disulfide exchange. J Biol Chem. 2006;281:21884–91.
Nakayama Y, Mukai N, Wang BF, Yang K, Patwari P, Kitsis RN, et al. Txnip C247S mutation protects the heart against acute myocardial infarction. J Mol Cell Cardiol. 2021;155:36–49.
Chutkow WA, Lee RT. Thioredoxin regulates adipogenesis through thioredoxin-interacting protein (Txnip) protein stability. J Biol Chem. 2011;286:29139–45.
Ho AS, Kannan K, Roy DM, Morris LG, Ganly I, Katabi N, et al. The mutational landscape of adenoid cystic carcinoma. Nat Genet. 2013;45:791–8.
Noura M, Matsuo H, Koyama A, Adachi S, Masutani H. TXNIP induces growth arrest and enhances ABT263-induced apoptosis in mixed-lineage leukemia-rearranged acute myeloid leukemia cells. FEBS Open Bio. 2020;10:1532–41.
Ghashghaei M, Le CT, Shaalan H, Escano L, Yue M, Arsalan A, et al. miR-148a-3p and DDX6 functional link promotes survival of myeloid leukemia cells. Blood Adv. 2023;7:3846–61.
Huy H, Kim TD, Kim WS, Kim DO, Byun JE, Kim MJ, et al. TLR4/NF-kappaB axis induces fludarabine resistance by suppressing TXNIP expression in acute myeloid leukemia cells. Biochem Biophys Res Commun. 2018;506:33–40.
Tohi Y, Taoka R, Zhang X, Matsuoka Y, Yoshihara A, Ibuki E, et al. Antitumor effects of orally administered rare sugar D-allose in bladder cancer. Int J Mol Sci. 2022;23:6771.
Yao A, Storr SJ, Al-Hadyan K, Rahman R, Smith S, Grundy R, et al. Thioredoxin system protein expression is associated with poor clinical outcome in adult and paediatric gliomas and medulloblastomas. Mol Neurobiol. 2020;57:2889–901.
Chen Y, Feng X, Yuan Y, Jiang J, Zhang P, Zhang B. Identification of a novel mechanism for reversal of doxorubicin-induced chemotherapy resistance by TXNIP in triple-negative breast cancer via promoting reactive oxygen-mediated DNA damage. Cell Death Dis. 2022;13:338.
Kim MJ, Lee HJ, Choi MY, Kang SS, Kim YS, Shin JK, et al. UHRF1 induces methylation of the TXNIP promoter and down-regulates gene expression in cervical cancer. Mol Cells. 2021;44:146–59.
Zhu J, Han S. Histone deacetylase 10 exerts anti-tumor effects on cervical cancer via a novel microRNA-223/TXNIP/Wnt/beta-catenin pathway. IUBMB Life. 2021.
Zhang J, Tian X, Yin H, Xiao S, Yi S, Zhang Y, et al. TXNIP induced by MondoA, rather than ChREBP, suppresses cervical cancer cell proliferation, migration and invasion. J Biochem. 2020;167:371–7.
Frullanti E, Colombo F, Falvella FS, Galvan A, Noci S, De Cecco L, et al. Association of lung adenocarcinoma clinical stage with gene expression pattern in noninvolved lung tissue. Int J Cancer. 2012;131:E643–E648.
Park KH, Yang JW, Kwon JH, Lee H, Yoon YD, Choi BJ, et al. Targeted induction of endogenous VDUP1 by small activating RNA inhibits the growth of lung cancer cells. Int J Mol Sci. 2022;23:7743.
Cheng Z, Lu C, Wang H, Wang N, Cui S, Yu C, et al. Long noncoding RNA LHFPL3-AS2 suppresses metastasis of non-small cell lung cancer by interacting with SFPQ to regulate TXNIP expression. Cancer Lett. 2022;531:1–13.
Kim GT, Kim EY, Shin SH, Lee H, Lee SH, Sohn KY, et al. Suppression of tumor progression by thioredoxin-interacting protein-dependent adenosine 2B receptor degradation in a PLAG-treated Lewis lung carcinoma-1 model of non-small cell lung cancer. Neoplasia. 2022;31:100815.
Woolston CM, Madhusudan S, Soomro IN, Lobo DN, Reece-Smith AM, Parsons SL, et al. Thioredoxin interacting protein and its association with clinical outcome in gastro-oesophageal adenocarcinoma. Redox Biol. 2013;1:285–91.
Li L, Liu Y, Zhao Y, Feng R, Li Y, Yu X, et al. Deubiquitinase USP8 increases ID1 stability and promotes esophageal squamous cell carcinoma tumorigenesis. Cancer Lett. 2022;542:215760.
Feingold PL, Surman DR, Brown K, Xu Y, McDuffie LA, Shukla V, et al. Induction of thioredoxin-interacting protein by a histone deacetylase inhibitor, entinostat, is associated with DNA damage and apoptosis in esophageal adenocarcinoma. Mol Cancer Ther. 2018;17:2013–23.
Yuan Y, Liu Q, Wu Z, Zhong W, Lin Z, Luo W. TXNIP inhibits the progression of osteosarcoma through DDIT4-mediated mTORC1 suppression. Am J Cancer Res. 2022;12:3760–79.
Li YH, Tong KL, Lu JL, Lin JB, Li ZY, Sang Y, et al. PRMT5-TRIM21 interaction regulates the senescence of osteosarcoma cells by targeting the TXNIP/p21 axis. Aging. 2020;12:2507–29.
Demokan S, Chuang AY, Chang X, Khan T, Smith IM, Pattani KM, et al. Identification of guanine nucleotide-binding protein gamma-7 as an epigenetically silenced gene in head and neck cancer by gene expression profiling. Int J Oncol. 2013;42:1427–36.
Hoshikawa H, Indo K, Mori T, Mori N. Enhancement of the radiation effects by D-allose in head and neck cancer cells. Cancer Lett. 2011;306:60–66.
Chen Q, Liu T, Bao Y, Zhao T, Wang J, Wang H, et al. CircRNA cRAPGEF5 inhibits the growth and metastasis of renal cell carcinoma via the miR-27a-3p/TXNIP pathway. Cancer Lett. 2020;469:68–77.
Lu Y, Liu Y, Lan J, Chan YT, Feng Z, Huang L, et al. Thioredoxin-interacting protein-activated intracellular potassium deprivation mediates the anti-tumour effect of a novel histone acetylation inhibitor HL23, a fangchinoline derivative, in human hepatocellular carcinoma. J Adv Res. 2023;51:181–96.
Zhang Y, Yan Q, Gong L, Xu H, Liu B, Fang X, et al. C-terminal truncated HBx initiates hepatocarcinogenesis by downregulating TXNIP and reprogramming glucose metabolism. Oncogene. 2021;40:1147–61.
Knoll S, Furst K, Kowtharapu B, Schmitz U, Marquardt S, Wolkenhauer O, et al. E2F1 induces miR-224/452 expression to drive EMT through TXNIP downregulation. EMBO Rep. 2014;15:1315–29.
Zhou J, Li T, Chen H, Jiang Y, Zhao Y, Huang J, et al. ADAMTS10 inhibits aggressiveness via JAK/STAT/c-MYC pathway and reprograms macrophage to create an anti-malignant microenvironment in gastric cancer. Gastric Cancer. 2022;25:1002–16.
Lin F, Zhang P, Zuo Z, Wang F, Bi R, Shang W, et al. Thioredoxin-1 promotes colorectal cancer invasion and metastasis through crosstalk with S100P. Cancer Lett. 2017;401:1–10.
Erkeland SJ, Palande KK, Valkhof M, Gits J, Danen-van Oorschot A, Touw IP. The gene encoding thioredoxin-interacting protein (TXNIP) is a frequent virus integration site in virus-induced mouse leukemia and is overexpressed in a subset of AML patients. Leuk Res. 2009;33:1367–71.
Jin P, Zhou Q, Xi S. Low-dose arsenite causes overexpression of EGF, TGFalpha, and HSP90 through Trx1-TXNIP-NLRP3 axis mediated signaling pathways in the human bladder epithelial cells. Ecotoxicol Environ Saf. 2022;247:114263.
Rolver MG, Holland LKK, Ponniah M, Prasad NS, Yao J, Schnipper J, et al. Chronic acidosis rewires cancer cell metabolism through PPARalpha signaling. Int J Cancer. 2023;152:1668–84.
Zhang J, An H, Ni K, Chen B, Li H, Li Y, et al. Glutathione prevents chronic oscillating glucose intake-induced beta-cell dedifferentiation and failure. Cell Death Dis. 2019;10:321.
Yu FX, Chai TF, He H, Hagen T, Luo Y. Thioredoxin-interacting protein (Txnip) gene expression: sensing oxidative phosphorylation status and glycolytic rate. J Biol Chem. 2010;285:25822–30.
Qi W, Chen X, Holian J, Tan CY, Kelly DJ, Pollock CA. Transcription factors Kruppel-like factor 6 and peroxisome proliferator-activated receptor-gamma mediate high glucose-induced thioredoxin-interacting protein. Am J Pathol. 2009;175:1858–67.
He X, Ma Q. Redox regulation by nuclear factor erythroid 2-related factor 2: gatekeeping for the basal and diabetes-induced expression of thioredoxin-interacting protein. Mol Pharm. 2012;82:887–97.
Lee H, Lee S, Bae H, Kang HS, Kim SJ. Genome-wide identification of target genes for miR-204 and miR-211 identifies their proliferation stimulatory role in breast cancer cells. Sci Rep. 2016;6:25287.
Zhu G, Zhou L, Liu H, Shan Y, Zhang X. MicroRNA-224 promotes pancreatic cancer cell proliferation and migration by targeting the TXNIP-mediated HIF1alpha pathway. Cell Physiol Biochem. 2018;48:1735–46.
Chen D, Dang BL, Huang JZ, Chen M, Wu D, Xu ML, et al. MiR-373 drives the epithelial-to-mesenchymal transition and metastasis via the miR-373-TXNIP-HIF1alpha-TWIST signaling axis in breast cancer. Oncotarget. 2015;6:32701–12.
Zhang C, Wang H, Liu X, Hu Y, Ding L, Zhang X, et al. Oncogenic microRNA-411 promotes lung carcinogenesis by directly targeting suppressor genes SPRY4 and TXNIP. Oncogene. 2019;38:1892–904.
Fan HP, Zhu ZX, Xu JJ, Li YT, Guo CW, Yan H. The lncRNA CASC9 alleviates lipopolysaccharide-induced acute kidney injury by regulating the miR-424-5p/TXNIP pathway. J Int Med Res. 2021;49:675868873.
Liang Y, Wang H, Chen B, Mao Q, Xia W, Zhang T, et al. circDCUN1D4 suppresses tumor metastasis and glycolysis in lung adenocarcinoma by stabilizing TXNIP expression. Mol Ther Nucleic Acids. 2021;23:355–68.
Chen Z, Lai X, Ding H, Zhang A, Sun Y, Ling J, et al. ATF4/TXNIP/REDD1/mTOR signaling mediates the antitumor activities of liver X receptor in pancreatic cancers. Cancer Innov. 2022;1:55–69.
Park SJ, Kim Y, Li C, Suh J, Sivapackiam J, Goncalves TM, et al. Blocking CHOP-dependent TXNIP shuttling to mitochondria attenuates albuminuria and mitigates kidney injury in nephrotic syndrome. Proc Natl Acad Sci USA. 2022;119:e2116505119.
Acknowledgements
The authors apologise to the authors of the many outstanding publications not referenced here owing to space restrictions. This work was supported by the China Scholarship Council Award (No. 201806010012 to JD, No. 202006940028 to TP); National Key R&D Programs (NKPs) of China (2022YFC3601800 to MY and JD); CRUK Early Detection and Diagnosis Committee (Project grant, C1519/A27375). RB is supported by MR/R000026/1 and UCLH/UCL BRC, who received a proportion of funding from the Department of Health’s NIHR Biomedical Research Centers funding scheme. Giovanna Alfano and Jose Vicencio are supported by CRUK Early Detection and Diagnosis Committee Project grant (C7675/A29313). AAM is supported by a CRUK City of London Clinical Research Fellowship (SEBSTF-2021\100007).
Author information
Authors and Affiliations
Contributions
JD and TN contributed to the discussion of the content. JD, TP, and RB wrote the article. JD and TP depicted the figures and summarised the tables. All authors reviewed the manuscript. RB and TN edited the manuscript before submission.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Deng, J., Pan, T., Liu, Z. et al. The role of TXNIP in cancer: a fine balance between redox, metabolic, and immunological tumor control. Br J Cancer 129, 1877–1892 (2023). https://doi.org/10.1038/s41416-023-02442-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41416-023-02442-4
- Springer Nature Limited
This article is cited by
-
MYG1 drives glycolysis and colorectal cancer development through nuclear-mitochondrial collaboration
Nature Communications (2024)
-
Nobiletin regulates the proliferation and migration of ovarian cancer A2780 cells via DPP4 and TXNIP
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
The MondoA-dependent TXNIP/GDF15 axis predicts oxaliplatin response in colorectal adenocarcinomas
EMBO Molecular Medicine (2024)