Abstract
Background
Cancer and its treatments can lead to excess body fat, decreases in lean mass, cardiotoxicity, and other side effects. The Mediterranean diet (MED-diet) has the potential to improve clinical and supportive care outcomes. The aim of this systematic review was to evaluate the feasibility, safety, and efficacy of the MED-diet on health outcomes in adults with cancer.
Methods
Three databases were searched from inception to February 2023. Eligible studies included randomised controlled trials testing a MED-diet intervention among adults with cancer. Within- and between-group differences for adherence, dietary intake and health outcomes were extracted.
Results
Fifteen studies describing fourteen interventions were included, and there were considerable differences in study design and implementation of the MED-diet. Studies were predominately in women with a history of breast cancer. The MED-diet was safe with no adverse events reported, and feasible with high adherence and/or increases in MED-diet-compliant foods. The MED-diet when applied with an energy restriction below estimated requirements for weight loss demonstrated reductions in body weight (range: −3.9 kg to −0.7 kg). Interventions that showed significant reductions in body weight also improved quality of life. There is limited evidence to evaluating the MED-diet on cardiovascular and inflammatory markers, and heterogenous MED-diet prescriptions impede definitive conclusions on these health outcomes.
Conclusion
The MED-diet was feasible and safe for adults with cancer. There were reported benefits for weight loss following a MED-diet when an energy restriction was applied, however further evaluation to determine the effects on cardiometabolic biomarkers and other outcomes are required.
Similar content being viewed by others
Introduction
Globally, 19.3 million people were diagnosed with cancer in 2020 and cancer is now the leading cause of morbidity and mortality worldwide [1]. After cancer treatment, many adults are faced with adverse treatment-related side effects that accelerate aging, increase risk of co-morbidities, and reduce quality of life [2]. These side effects include cardiomyopathy, persistent fatigue, reduced muscle mass with simultaneous increases in fat mass, cognitive impairment, and early menopause for women with breast cancer (which includes risks for bone health and cardiovascular disease) [2,3,4]. Several guidelines suggest nutrition and exercise interventions are important strategies to address persistent treatment-related toxicities and late side effects from cancer treatment [5, 6]. However, despite clear evidence for nutritional interventions in adults with cancer who are at risk of malnutrition [7], the optimal nutrition prescription to address other treatment-related toxicities (i.e., body composition, cardiovascular and metabolic health) after cancer treatment is yet to be determined.
The Mediterranean style diet (MED-diet) is a high-quality pattern of eating, with consistent observational evidence associating the MED-diet with reduced risk of chronic disease [8, 9]. There are many forms of the MED-diet, but it is typically characterised by (i) a high intake of fish, vegetables, fruits, legumes, nuts, and extra virgin olive oil; (ii) moderate intake of dairy products and red wine, and (iii) low consumption of added sugar, processed foods, and red meats [10]. Traditional versions of the MED-diet specify that dairy products are to be fermented, however, recent interpretations have been updated to include ‘low-fat’ dairy products [11]. The high antioxidant and anti-inflammatory properties of the MED-diet are proposed to offer synergistic benefits to cardiometabolic and body composition health [12, 13]. Observational evidence suggests the MED-diet can extend cancer survivorship, where high adherence to a MED-diet has shown a 22% and 13% reduction in prostate cancer and breast cancer mortality, respectively [14, 15]. The survivorship benefits from the MED-diet are potentially clinically important given the negative cardiometabolic and body composition changes from hormone therapy in breast and prostate cancer [16, 17]. Nutrition interventions in general, usually in combination with physical exercise, have shown promising results, particularly for patients with breast or prostate cancer by reducing body weight and fat mass [18] and improving quality of life [19]. However, despite several studies indicating the MED-diet may offer improvements to body composition and cardiometabolic health predominately after cancer treatment, the potential health benefits from the MED-diet in adults with cancer is yet to be systematically evaluated [13]. The aim of this review was to determine the feasibility (i.e., prescription, intervention design and support, participant adherence, safety) of delivering a MED-diet in adults with cancer, during or after treatment, and synthesise health outcomes from MED-diet intervention trials.
Methods
Protocol and registration
This systematic review was conducted in compliance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines [20]. The protocol was registered in the PROSPERO database (registration ID: 376985).
Information sources and search strategy
Embase, CINAHL, and Scopus databases were systematically searched on 17/02/2023. The search strategy was based on the Population Intervention Comparison and Outcomes (PICO) framework [21] and the specific search strategy was adjusted according to each database outlined in Supplementary Material 1. Each search concept included search terms relating to: (i) Mediterranean-style diet, (ii) adults with cancer, and (iii) randomised controlled trials. To summarise the evidence as a collective, outcome measures were not included in the search term strategy. Keyword search terms and Medical Subject Headings (MeSH) were applied to all search engines where applicable. Reference lists of identified articles were manually searched. There were no search restrictions on dates or language.
Eligibility criteria
The inclusion criteria follows the PICO framework: (i) population: participants aged ≥18 years, of any sex, had a histological cancer diagnosis (any cancer type) prior to the intervention, and underwent any type of cancer treatment or were about to start treatment; (ii) intervention: any lifestyle intervention that included the MED-diet prescribed alone or in conjunction with exercise and/or psychosocial support; (iii) study design: randomised controlled trials where the control group received either no intervention or another intervention, a different dietary intervention, or a supplement, and (iv) reported the adherence, dietary intake, and/or any health-related outcome. Importantly, the nutrition intervention had to self-identify as the MED-diet or be based on the MED-diet principles outlined in the methods. Articles with similar dietary goals or nutritional targets to the MED-diet, yet did not explicitly mention the MED-diet, were excluded. Articles were also excluded if the intervention included the MED-diet plus a dietary supplement or medication, and/or both the intervention and control group received the MED-diet.
Study selection
Four independent reviewers screened titles and abstracts for inclusion (AM, ES, BG, and PR). Studies that were not clearly excluded progressed to full text screening where the study was reviewed in detail against the inclusion criteria. Where discrepancies in inclusion occurred between reviewers, a fifth reviewer was sourced (BB) and issues were reconciled with discussion. Covidence systematic review software (Veritas Health Innovation, Melbourne, Australia, available at www.covidence.org) was used to organize the paper identification and complete data extraction.
Data extraction
Data extracted included author(s), sample characteristics, trial length and design, data collection time points, control group, intervention methodology, and MED-diet prescription (macro and micronutrients and food groups). The primary outcome was extracted along with the intent of the MED-diet intervention on body weight. Descriptive data were extracted to report the feasibility of the MED-diet interventions (i.e., completion, consult attendance). Mean, standard deviation (SD) and/or 95% confidence interval for baseline values (where applicable), change within the intervention group, and between-group differences were extracted for changes in diet, health-related outcomes (weight and body composition), cardiovascular biomarkers, and quality of life.
Quality assessment
The methodological quality was assessed using the Cochrane Risk of Bias Tool [21]. This tool looks at risk of bias in the following domains: sequence generation, allocation concealment, blinding, incomplete outcome data, selective outcome reporting, and other potential sources of systematic bias. Each study was rated against these domains and ranked as low, high, or unclear risk of bias. Blinding of participants to the intervention was assessed as “low risk” for all studies because it is not possible for the participant to be blinded to a dietary intervention supported by a health professional.
Data synthesis
Continuous variables are presented as means and deviation from the mean (i.e., SD, Standard Error of Mean, range). Feasibility data are reported by the percentage of completion and attendance. Outcome variables (MED-diet adherence, body composition, biomarkers, quality of life) are reported as either an increase or decrease and whether statistically significant. Statistical significance was deemed where the p-value was <0.05.
Results
Fifteen articles [22,23,24,25,26,27,28,29,30,31,32,33,34,35,36] reporting fourteen interventions were included in this systematic review (Fig. 1). One intervention was reported in two articles [22, 23], and subsequently data from both articles were pooled to report the one intervention. In addition, Harvie et al. [28] evaluated two separate Mediterranean diet groups (delivered as home versus community group consultations) compared to a usual care group, and for the purpose of this systematic review these were treated as two separate intervention groups. The sample sizes ranged from 23 [22] to 1542 [25] participants; the average age of participants ranged from 41 [29] to 66 [22] years (Table 1).
Based on the Cochran risk-of-bias assessment, 12 of the 14 interventions scored positive [22, 24, 27,28,29, 33, 34, 36] (Supplementary Material 2). Jaleli [29] and Zuniga [36] had incomplete outcome data, which resulted in an unclear rating. Allocation concealment was the least positively (42%) scored outcome in the risk of bias tool.
Study characteristics
Ten interventions included women with breast cancer [24,25,26, 28, 31,32,33,34,35,36] and one intervention included a mix of cancers however were mostly women with breast cancer [30]. Other interventions included people with each of the following cancer types: prostate cancer [22, 23], acute myeloid leukaemia [29], and lung cancer [27]. Five interventions included participants undergoing active treatment [22, 23, 29, 30, 32, 35] and five interventions commenced after cancer treatment [24, 26, 31, 33, 36]; that included two months [31, 36], three months [24], or twelve months [33] after treatment. Three interventions included participants either during or post-treatment [27, 28, 34], and one intervention commenced within 5 years post-diagnosis [25]. The intervention length ranged from one month [29] to twelve months [24, 25, 31, 33,34,35,36].
Mediterranean diet characteristics
Details on the MED-diet intervention design was reported in all studies. Nine interventions were administered through a trained nutrition professional [22, 24,25,26,27,28, 30, 32, 34], six used individualised consultation to deliver the MED-diet [22, 27,28,29, 32, 34], five used education webinars/seminars [25, 31, 33, 35, 36], and two provided the MED-diet ingredients/meals to participants with consultation support [26, 30]. Variations in the MED-diet prescription were seen (Table 1), with nutrient or food group targets prescribed in seven of 14 interventions [22, 26,27,28,29, 32, 36]. Collectively, nutrient targets ranged from 20 to 40% of total energy from fat, 4 to 10% from polyunsaturated fat, 10 to 24% from monounsaturated fat, 45 to 65% from carbohydrates, and 15 to 25% from protein. Five interventions recommended food groups [22, 26, 28, 36]: 1.5 to 3 servings/day of fruit, 2 to 7 servings/day of vegetables, 2 to 4 servings/week of fish or seafood, 3 to 5 servings/week of nuts, and 15 to 30 ml/day of olive oil. Others indicated to increase or decreased food groups without specific targets. In addition, some interventions added olive leaf extract [24], emphasised fermented macrobiotics [25, 35], or flaxseed and green tea [34] to the MED-diet prescription.
Most interventions aimed to achieve weight loss [22, 24,25,26, 28, 32, 34, 35], whilst two aimed to prevent weight gain or maintain weight [27, 29], and others focused on reducing cancer-related fatigue [30] or pro-inflammatory cytokines [31], or improving diet quality [36], and did not report desired changes in body weight. Variations were seen in energy restriction applied to the MED-diet prescription across interventions. For participants classified as overweight or obese (BMI ≥ 25 kg/m2) energy restrictions ranged from −2000 to −4000 kJ/day [22], −250 kcal/day [35], −500 kcal/day [32], −25% estimated energy requirements [28], and total energy intake restricted to 1500 kcal/day [26]. Others aimed to achieve energy deficits through portion control [24], increasing satiating foods [25], or restricting energy by an unknown deficit [34]. MED-diet interventions that aimed to maintain body weight were prescribed to match estimated energy requirements to prevent malnutrition during treatment [27, 29].
MED-diet adherence and dietary change
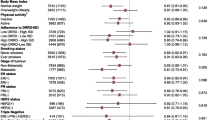
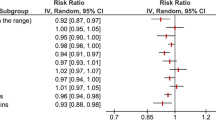
Eight of the 14 interventions reported adherence to the MED-diet [24, 26, 27, 30,31,32,33, 36], of which five used the Mediterranean Diet Adherence Score (MEDAS) tool [22, 23, 26, 30, 31, 33]. Of the eight interventions, six showed a significant improvement in adherence compared to the control group [24, 26, 27, 31, 34, 36]; whilst seven showed a significant increase in adherence within the MED-diet group only [23, 26, 27, 30,31,32,33]. No adverse events were attributed to the MED-diet in any of the 14 interventions.
Change in energy and nutrients from the MED-diet is represented in Table 2. Food Frequency Questionnaires and 3-day or 7-day food diaries were the predominant tool used to quantify energy and nutrient intake to compare to the MED-diet targets within each study. Eight interventions reported energy intake [22, 26, 28,29,30,31, 33, 36], only two reported a significant between-group reduction [31, 36]. Nine reported total fat and saturated fat (SFA) intake [22, 27, 29, 31,32,33,34], with two [26, 29] and three studies [22, 23, 27, 32], respectively, showing a between-group reduction following a MED-diet intervention. Five reported fibre intake [22, 27, 30,31,32] with two showing a between-group increase in fibre intake from the MED-diet intervention [22, 27].
Anthropometric data
Thirteen interventions reported body weight at baseline and endpoint (Table 2). Six of the thirteen interventions reported a significant between-group decrease in body weight in favour of the MED-diet [22, 25, 28, 32, 33, 35]. Body mass index (BMI) was recorded in 11 interventions [22, 24, 26, 27, 29, 32,33,34, 36], seven reported a significant between-group decrease in BMI [24, 25, 32,33,34,35]. Additionally, body composition was (fat mass or lean mass) was reported in seven interventions [22, 26,27,28, 32, 34, 35]; five reported a significant between-group decrease [22, 28, 32, 34, 35], and three reported a significant between-group decrease in lean mass [22, 28, 35].
Biomarkers
Twelve of the 14 interventions reported the effects of the MED-diet on biomarkers [22, 24,25,26,27,28,29,30, 32,33,34,35] including glucose metabolism, cardiovascular risk factors, protein stores, and inflammatory markers (Table 2). Three interventions reported a significant decrease in blood glucose levels [25,26,27, 34], four showed a significant decrease in triglycerides [25, 26, 28, 33], three increased HDL [32,33,34], and Harvie et al’s community-group showed a decrease in LDL and total cholesterol [28]. Individual trials reported a significant decrease interleukin-8 [22], and a significant increase in albumin [29].
Quality of life
Quality of life measurements were assessed using either the Functional Assessment of Chronic Illness Therapy (FACT) [37] or the European Organisation for Research and Treatment of Cancer – Quality of Life Questionnaire (EORTC-QLQ-C30) [38]. Seven interventions reported the effects of the MED-diet on quality of life [22, 24, 28, 30,31,32,33]. Four interventions reported significant between-group improvements in quality of life in favour of the MED-diet [22, 28, 30, 32], whilst two showed significant improvements in fatigue [22, 30]. Other domains of quality of life that include role [32, 33], physical [30, 33], emotional [32], and cognitive health composite [23] were inconsistently improved across interventions (Table 2).
Discussion
This review showed that the MED-diet is safe (zero adverse events), feasible, and well adhered to, however heterogeneity in the prescription of the MED-diet and intervention design precludes identifying the optimal approach to supporting individual health outcomes for adult cancer survivors. Hypocaloric MED-diets show particularly promising results for reducing body weight (range: −3.9 kg to −0.7 kg) in overweight or obese adults who have finished cancer treatment or currently treated with hormone therapy for breast or prostate cancer. Interventions with the goal to maintain weight showed promising findings in preventing weight loss during chemotherapy, however, both studies were small in sample size and individualised to manage nutritional impact symptoms (i.e., nausea and vomiting). Whilst evidence for weight loss from the MED-diet is in accordance with several guidelines advocating for healthy body weight post-treatment to reduce the risk of mortality and morbidity [5, 6], the current evidence for the MED-diet in patients with cancer is limited to mostly women who have finished breast cancer treatment. Despite the evidence to support the MED-diet for preventing and managing chronic diseases, translation of this evidence to cancer survivors, where chronic disease risk is high, requires further investigation.
Feasibility of Mediterranean diet interventions
The MED-diet interventions in this review were safe (zero adverse events), feasible with a high completion rate (range: 64% to 98%), and showed high attendance to consultations or educational workshops (>75%). These factors likely contributed to the high adherence to the MED-diet food groups across interventions in this review. Most studies provided dietary goals for food groups that participants could follow and all studies incorporated guidance or accountability check-ins with trained staff, though the frequency of informative workshops or interaction with a trained nutrition professional. More intense intervention delivery may account for the improvement in MED-diet adherence scores amongst these studies [22, 23, 27, 28, 36], as behaviour change is more likely when dietary advice is paired with improved nutritional literacy [39]. Interventions delivered by a nutrition professional, with accompanied education material (i.e., recipes and cooking demonstrations), offers the chance to build rapport and iteratively change dietary behaviours across consultations, and is likely a contributing factor to the high adherence seen in these studies.
Body weight and composition
This review indicates that the MED-diet when prescribed with an energy reduction has weight loss benefits (range: −3.9 kg to −0.7 kg). Similar reductions in weight were reported in a meta-analysis that evaluated the effects of MED-diet interventions compared to low fat diets (−4.1 to 10.1 kg vs. 2.9 to −5.0 kg) in overweight or obese adults [40]. Whilst MED-diet interventions have shown long term weight loss (≥12 months) in overweight or obese adults at risk of chronic disease [40, 41], the long term effect in cancer survivors are limited. Two 12-month MED-diet interventions prescribed by a nutrition specialist has demonstrated significant weight loss in women with breast cancer that are treated with hormone therapy (−2.4 kg vs. −0.9 kg) [25] or that are post-surgery and treated with adjuvant hormone and/or chemotherapy (home-based: −1.5 kg vs. 0.8 kg and community-based: −1.6 kg vs. 0.8 kg) [28]. The present review also suggests the MED-diet can prevent weight gain from hormone therapy in prostate cancer [22], however this study was underpowered in sample size. Whilst most studies in the present review were designed to promote weight loss, the methodological differences in intervention prescription, behaviour change support techniques, and reporting of dietary adherence varied. As such it is plausible that improvements in diet quality from the MED-diet, characterised by an increased consumption of vegetables, fruits, and whole grains led to weight loss.
Interventions that measured body composition showed reductions in fat mass but also lean mass. Whilst reductions in weight and fat mass is beneficial for optimising body composition, the reductions in lean mass seen in some studies in concerning for maintaining strength, physical function, and quality of life following or during treatment. Nonetheless, reductions in lean mass from weight loss interventions are expected [42], and our findings are in line with a previous meta-analysis evaluating nutrition and exercise interventions on body composition in adults with cancer reporting muscle loss (mean difference −0.58 kg) from weight-loss focused interventions [18]. Efforts to attenuate muscle loss in cancer survivorship is a research priority given most cancer treatments are associated with reduced lean mass. Increased protein intake (1.0–1.5 g/kg/day), and/or supplemental approaches (i.e., β-hydroxy β-methylbutyrate at 3 g/day) may protect muscle or increase muscle when paired with exercise [43]. Yet whether these approaches protect muscle mass within a MED-diet that intends to reduce body weight and fat mass requires investigation.
Cardiometabolic health
The MED-diet promotes improvements in cardiometabolic health by encouraging foods high in monounsaturated and polyunsaturated fats and fibre, which play an important role in lowering low-density lipoprotein (LDL) and triglycerides and increasing HDL cholesterols [8, 9, 44]. A limited number of studies showed improvements in cardiovascular biomarkers, however results were inconsistent and varying intervention components preclude identifying whether the benefits in cardiovascular health were primarily attributable to the MED-diet. For example, studies varied in the MED-diet prescription, with some promoting a specified amount of olive leaf extract [24], flaxseed, and green tea consumption per day [34], or including macrobiotic foods like soy and miso [25], whilst others included an exercise program with the MED-diet [28, 33]. Secondly, most interventions ranged between 12 and 26 weeks, which may not be a sufficient duration needed for the MED-diet to see changes to cardiometabolic biomarkers. Whilst a larger body of evidence supports the use of the MED-diet in reducing cardiovascular biomarkers (LDL, triglycerides and cholesterol) in adults who already have chronic disease(s) [39, 45, 46], the benefits of the MED-diet in adults with cancer who are at a higher risk of chronic diseases however may not have clinically elevated biomarkers at baseline may be less sensitive to change in a short timeframe and therefore requires further investigation.
Chronic inflammation and oxidative stress are known predictive risk factors of cardiovascular disease [47]. The MED-diet effects on cardiometabolic health are theorised to be associated with dietary properties high in bioactive nutrients and phenolic compounds, which combat oxidation and lower circulating inflammatory markers [48,49,50]. Several biomarkers are used to identify endothelial cell damage and systemic inflammation, for example, interleukins (IL-6, IL-8, IL-1β), however, the types of inflammatory markers measured from the MED-diet in adults with cancer in this review varied and were often secondary or tertiary outcome measures. Inflammatory pathways and networks are complex and can be influenced by cancer treatment. As such, studies that are powered to investigate the effects of the MED-diet on inflammatory markers, considering covariates (i.e., medications and treatment), are needed before definitive conclusions can be made.
Quality of life
This review provides preliminary evidence for quality-of-life benefits from the MED-diet in adults with cancer and corroborates previous reports of mixed benefits to quality of life from lifestyle interventions in cancer [51,52,53]. The studies that demonstrate high adherence to the MED-diet and improved global and domains of quality of life (i.e., fatigue, physical and cognitive functioning) also had improvements to body weight and composition (free fat mass, weight, BMI) [22, 23, 28, 33]. Our results suggest that weight loss from the MED-diet might have additional benefits in improving mental domains of quality of life and body image, which is a research priority highlighted elsewhere [54]. Most MED-diet interventions that were delivered by a dietitian showed improvements in quality of life or domains of quality of life (i.e., role, emotional). These findings may be attributable to the benefits of interaction with a health care professional, which offers individualisation of dietary preferences, social and environmental influences on dietary intake. The utilisation of behaviour change strategies within a consultation, where participants are empowerment to be actively involved in their cancer health, may contribute to the improvements seen in quality of life [55]. Whilst the MED-diet is likely to have improved quality of life, weight loss and interactions with a health professional may be mediators, and further research is needed to optimize the components of a MED-diet intervention for quality-of-life outcomes.
Future directions and clinical implications
The results of this systematic review should be considered in light of its strengths and limitations. This review followed the PRISMA guidelines in reporting a systematic review and provides a high level of evidence for the benefits of the MED-diet compared to usual care across health outcomes. Whilst this review demonstrates that hypocaloric MED-diets may reduce weight for people with cancer that are overweight or obese, the effects on body composition (i.e., fat mass and muscle mass) are limited. The heterogeneity in MED-diet prescription, the intent of each intervention on body weight, and the use of co-interventions (i.e., exercise) precluded pooling the mean difference in a meta-analysis. This highlights the importance for future interventions to report MED-diet prescriptions using calculating estimated energy requirements, nutrient targets along with food groups to identify components of the MED-diet to health outcomes. Translation of the MED-diet in patients with cancer are limited to reducing or maintaining body weight. However, whether the benefits in weight control are attributable to the energy restriction or the MED-diet itself is unknown. Future longer term RCTs should focus on reducing the risk of, or managing, cardiovascular or metabolic disease after cancer treatment to improve the potential clinical implications of the MED-diet.
Conclusion
This systematic review showed that the MED-diet is safe, feasible, and well adhered to among adult cancer survivors during and after treatment. Collectively, there was considerable variation in the MED-diet prescription (i.e., macronutrients, micronutrients, and food groups), health outcomes measured, resulting in mixed findings from the heterogeneity in intervention design. Interventions that prescribed an energy restriction in addition to the MED-diet showed reductions in body weight, however, whether the benefits in weight loss can be attributable to the MED-diet over and above the energy restriction requires further investigation. Whilst the MED-diet is consistent with dietary recommendations for cancer survivors, there is limited evidence to indicate that the MED-diet offers benefits to managing side effects, chronic disease prevention, or improving quality of life. Attention to reporting the MED-diet prescription and adherence on other important outcomes, such as cardiometabolic function, is important for evidence-based recommendations across multiple health outcomes for cancer survivors.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49. https://doi.org/10.3322/caac.21660.
Hurria A, Jones L, Muss HB. Cancer treatment as an accelerated aging process: assessment, biomarkers, and interventions. Am Soc Clin Oncol. 2016;36. https://doi.org/10.1200/EDBK_156160.
Curigliano G, Lenihan D, Fradley M, Ganatra S, Barac A, Blaes A, et al. Management of cardiac disease in cancer patients throughout oncological treatment: ESMO consensus recommendations. Ann Oncol. 2020;31:171–90. https://doi.org/10.1016/j.annonc.2019.10.023.
Dent SF, Kikuchi R, Kondapalli L, Ismail-Khan R, Brezden-Masley C, Barac A, et al. Optimizing cardiovascular health in patients with cancer: a practical review of risk assessment, monitoring, and prevention of cancer treatment–related cardiovascular toxicity. American Society of Clinical Oncology Educational Book. 2020:501–15. https://doi.org/10.1200/edbk_286019.
Rock CL, Thomson CA, Sullivan KR, Howe CL, Kushi LH, Caan BJ, et al. American Cancer Society nutrition and physical activity guideline for cancer survivors. CA Cancer J Clin. 2022;72:230–62. https://doi.org/10.3322/caac.21719.
Ligibel JA, Bohlke K, May AM, Clinton SK, Demark-Wahnefried W, Gilchrist SC, et al. Exercise, diet, and weight management during cancer treatment: ASCO guideline. J Clin Oncol. 2022;40:2491–507. https://doi.org/10.1200/JCO.22.00687.
Arends J, Bachmann P, Baracos V, Barthelemy N, Bertz H, Bozzetti F, et al. ESPEN guidelines on nutrition in cancer patients. Clin Nutr. 2017;36:11–48. https://doi.org/10.1016/j.clnu.2016.07.015.
Ahmad S, Moorthy MV, Demler OV, Hu FB, Ridker PM, Chasman DI, et al. Assessment of risk factors and biomarkers associated with risk of cardiovascular disease among women consuming a Mediterranean diet. JAMA Netw Open. 2018;1:e185708. https://doi.org/10.1001/jamanetworkopen.2018.5708.
Buckland G, Gonzalez CA, Agudo A, Vilardell M, Berenguer A, Amiano P, et al. Adherence to the Mediterranean diet and risk of coronary heart disease in the Spanish EPIC cohort study. Am J Epidemiol. 2009;170:1518–29. https://doi.org/10.1093/aje/kwp282.
Trichopoulou A, Martínez-González MA, Tong TYN, Forouhi NG, Khandelwal S, Prabhakaran D, et al. Definitions and potential health benefits of the Mediterranean diet: views from experts around the world. BMC Med. 2014;12:112. https://doi.org/10.1186/1741-7015-12-112.
Bach-Faig A, Berry EM, Lairon D, Reguant J, Trichopoulou A, Dernini S, et al. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011;14:2274–84. https://doi.org/10.1017/s1368980011002515.
Dinu M, Pagliai G, Casini A, Sofi F. Mediterranean diet and multiple health outcomes: an umbrella review of meta-analyses of observational studies and randomised trials. Eur J Clin Nutr. 2018;72:30–43. https://doi.org/10.1038/ejcn.2017.58.
Mentella MC, Scaldaferri F, Ricci C, Gasbarrini A, Miggiano GAD. Cancer and Mediterranean diet: a review. Nutrients. 2019;11:2059 https://doi.org/10.3390/nu11092059.
Kenfield SA, DuPre N, Richman EL, Stampfer MJ, Chan JM, Giovannucci EL. Mediterranean diet and prostate cancer risk and mortality in the Health Professionals Follow-up Study. Eur Urol. 2014;65:887–94. https://doi.org/10.1016/j.eururo.2013.08.009.
Di Maso M, Dal Maso L, Augustin LSA, Puppo A, Falcini F, et al. Adherence to the Mediterranean diet and mortality after breast cancer. Nutrients. 2020;12:3649 https://doi.org/10.3390/nu12123649.
Haseen F, Murray LJ, Cardwell CR, O’Sullivan JM, Cantwell MM. The effect of androgen deprivation therapy on body composition in men with prostate cancer: systematic review and meta-analysis. J Cancer Surviv. 2010;4:128–39. https://doi.org/10.1007/s11764-009-0114-1.
Sheean PM, Hoskins K, Stolley M. Body composition changes in females treated for breast cancer: a review of the evidence. Breast Cancer Res Treat. 2012;135:663–80. https://doi.org/10.1007/s10549-012-2200-8.
Baguley BJ, Dalla Via J, Fraser SF, Daly RM, Kiss N. Effectiveness of combined nutrition and exercise interventions on body weight, lean mass, and fat mass in adults diagnosed with cancer: a systematic review and meta-analysis. Nutr Rev. 2022. https://doi.org/10.1093/nutrit/nuac079.
Castro-Espin C, Agudo A. The role of diet in prognosis among cancer survivors: a systematic review and meta-analysis of dietary patterns and diet interventions. Nutrients. 2022;14:348. https://doi.org/10.3390/nu14020348.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement:an updated guideline for reporting systematic reviews. BMJ. 2021; 372. https://doi.org/10.1136/bmj.n71.
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al. Cochrane Handbook for Systematic Reviews of Interventions, vol. 6.4. (updated August 2023). Cochrane, 2023. Available from www.training.cochrane.org/handbook.
Baguley BJ, Skinner TL, Jenkins DG, Wright ORL. Mediterranean-style dietary pattern improves cancer-related fatigue and quality of life in men with prostate cancer treated with androgen deprivation therapy: a pilot randomised control trial. Clin Nutr. 2021;40:245–54. https://doi.org/10.1016/j.clnu.2020.05.016.
Baguley BJ, Adlard K, Jenkins D, Wright ORL, Skinner TL. Mediterranean style dietary pattern with high intensity interval training in men with prostate cancer treated with androgen deprivation therapy: a pilot randomised control trial. Int J Environ Res Public Health. 2022;19:5709. https://doi.org/10.3390/ijerph19095709.
Braakhuis A, Campion P, Bishop K. The effects of dietary nutrition education on weight and health biomarkers in breast cancer survivors. Med Sci. 2017;5. https://doi.org/10.3390/medsci5020012.
Bruno E, Krogh V, Gargano G, Grioni S, Bellegotti M, Venturelli E, et al. Adherence to dietary recommendations after one year of intervention in breast cancer women: the DIANA-5 trial. Nutrients. 2021;13:2990. https://doi.org/10.3390/nu13092990.
Cho AR, Choi WJ, Kwon YJ, Lee HS, Ahn SG, Lee JW. Mediterranean diet and naltrexone/bupropion treatment for weight loss in overweight and obese breast cancer survivors and non-cancer participants: a pilot randomized controlled trial. Diabetes Metab Syndr Obes. 2020;13:3325–35. https://doi.org/10.2147/DMSO.S269237.
Gioxari A, Tzanos D, Kostara C, Papandreou P, Mountzios G, Skouroliakou M. Mediterranean diet implementation to protect against advanced lung cancer index (ALI) rise: study design and preliminary results of a randomised controlled trial. Int J Environ Res Public Health. 2021;18:3700. https://doi.org/10.3390/ijerph18073700.
Harvie M, Pegington M, McMullan D, Bundred N, Livingstone K, Campbell A, et al. The effectiveness of home versus community-based weight control programmes initiated soon after breast cancer diagnosis: a randomised controlled trial. Br J Cancer. 2019;121:443–54. https://doi.org/10.1038/s41416-019-0522-6.
Jalali, Mostafa, Abdollahi S, Hosseini M, Bozorg A, Ajami DK, et al. The positive effects of mediterranean-neutropenic diet on nutritional status of acute myeloid leukemia patients under chemotherapy. Front Biol. 2018;13:475–80. https://doi.org/10.1007/s11515-018-1519-x.
Kleckner AS, Reschke JE, Kleckner IR, Magnuson A, Amitrano AM, Culakova E, et al. The effects of a Mediterranean diet intervention on cancer-related fatigue for patients undergoing chemotherapy: a pilot randomized controlled trial. Cancers (Basel). 2022;14:4202. https://doi.org/10.3390/cancers14174202.
Long Parma DA, Reynolds GL, Munoz E, Ramirez AG. Effect of an anti-inflammatory dietary intervention on quality of life among breast cancer survivors. Support Care Cancer. 2022;30:5903–10. https://doi.org/10.1007/s00520-022-07023-4.
Papandreou P, Gioxari A, Nimee F, Skouroliakou M. Application of clinical decision support system to assist breast cancer patients with lifestyle modifications during the COVID-19 pandemic: a randomised controlled trial. Nutrients. 2021;13:2115. https://doi.org/10.3390/nu13062115.
Ruiz-Vozmediano J, Löhnchen S, Jurado L, Recio R, Rodríguez-Carrillo A, López M, et al. Influence of a multidisciplinary program of diet, exercise, and mindfulness on the quality of life of stage iia-iib breast cancer survivors. Integr Cancer Ther. 2020;19:1–11. https://doi.org/10.1177/1534735420924757.
Skouroliakou M, Grosomanidis D, Massara P, Kostara C, Papandreou P, Ntountaniotis D, et al. Serum antioxidant capacity, biochemical profile and body composition of breast cancer survivors in a randomized Mediterranean dietary intervention study. Eur J Nutr. 2018;57:2133–45. https://doi.org/10.1007/s00394-017-1489-9.
Villarini A, Pasanisi P, Raimondi M, Gargano G, Bruno E, Morelli D, et al. Preventing weight gain during adjuvant chemotherapy for breast cancer: a dietary intervention study. Breast Cancer Res Treat. 2012;135:581–9. https://doi.org/10.1007/s10549-012-2184-4.
Zuniga KE, Parma DL, Munoz E, Spaniol M, Wargovich M, Ramirez AG. Dietary intervention among breast cancer survivors increased adherence to a Mediterranean-style, anti-inflammatory dietary pattern: the Rx for better breast health randomized controlled trial. Breast Cancer Res Treat. 2019;173:145–54. https://doi.org/10.1007/s10549-018-4982-9.
Cella D, Tulsky D, Gray G, Sarafian B, Linn E, Bonomi A, et al. The functional assessment of Cancer therapy scale: development and validation of the general measure. J Clinical Oncology. 1993;11:570–9. https://doi.org/10.1200/JCO.1993.11.3.570.
Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, et al. The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. JNCI: Journal of the National Cancer Institute. 1993;85:365–376. https://doi.org/10.1093/jnci/85.5.365.
Aridi YS, Walker JL, Roura E, Wright ORL. Adherence to the Mediterranean diet and chronic disease in Australia: national nutrition and physical activity survey analysis. Nutrients. 2020;12:1251. https://doi.org/10.3390/nu12051251.
Mancini JG, Filion KB, Atallah R, Eisenberg MJ. Systematic review of the Mediterranean diet for long-term weight loss. Am J Med. 2016;129:407–15.e404. https://doi.org/10.1016/j.amjmed.2015.11.028.
Sanchez-Sanchez ML, Garcia-Vigara A, Hidalgo-Mora JJ, Garcia-Perez MA, Tarin J, Cano A. Mediterranean diet and health: a systematic review of epidemiological studies and intervention trials. Maturitas. 2020;136:25–37. https://doi.org/10.1016/j.maturitas.2020.03.008.
Willoughby D, Hewlings S, Kalman D. Body composition changes in weight loss: strategies and supplementation for maintaining lean body mass, a brief review. Nutrients. 2018;10:1876. https://doi.org/10.3390/nu10121876.
Prado CM, Purcell SA, Laviano A. Nutrition interventions to treat low muscle mass in cancer. J Cachexia Sarcopenia Muscle. 2020;11:366–80. https://doi.org/10.1002/jcsm.12525.
Sofi F, Macchi C, Abbate R, Gensini GF, Casini A. Mediterranean diet and health status: an updated meta-analysis and a proposal for a literature-based adherence score. Public Health Nutr. 2014;17:2769–82. https://doi.org/10.1017/s1368980013003169.
Muscogiuri G, Verde L, Sulu C, Katsiki N, Hassapidou M, Frias-Toral E, et al. Mediterranean diet and obesity-related disorders: what is the evidence? Curr Obes Rep. https://doi.org/10.1007/s13679-022-00481-1.
Tuttolomondo A, Simonetta I, Daidone M, Mogavero A, Ortello A, Pinto A. Metabolic and vascular effect of the Mediterranean diet. Int J Mol Sci. 2019;20:4716. https://doi.org/10.3390/ijms20194716.
Wongwarawipat T, Papageorgiou N, Bertsias D, Siasos G, Tousoulis D. Olive oil-related anti-inflammatory effects on atherosclerosis: potential clinical implications. Endocr Metab Immune Disord Drug Targets. 2018. https://doi.org/10.2174/1871530317666171116103618.
Wu PY, Chen KM, Tsai WC. The Mediterranean dietary pattern and inflammation in older adults: a systematic review and meta-analysis. Adv Nutr. 2021;12:363–73. https://doi.org/10.1093/advances/nmaa116.
Schwingshackl L, Hoffmann G. Mediterranean dietary pattern, inflammation and endothelial function: a systematic review and meta-analysis of intervention trials. Nutr Metab Cardiovasc Dis. 2014;24:929–39. https://doi.org/10.1016/j.numecd.2014.03.003.
Koelman L, Egea Rodrigues C, Aleksandrova K. Effects of dietary patterns on biomarkers of inflammation and immune responses: a systematic review and meta-analysis of randomized controlled trials. Adv Nutr. 2022;13:101–15. https://doi.org/10.1093/advances/nmab086.
Kim NH, Song S, Jung SY, Lee E, Kim Z, Moon HG, et al. Dietary pattern and health-related quality of life among breast cancer survivors. BMC Women’s Health. 2018;18:65. https://doi.org/10.1186/s12905-018-0555-7.
Baguley BJ, Bolam KA, Wright ORL, Skinner TL. The effect of nutrition therapy and exercise on cancer-related fatigue and quality of life in men with prostate cancer: a systematic review. Nutrients. 2017;9. https://doi.org/10.3390/nu9091003.
Du S, Hu L, Dong J, Xu G, Jin S, Zhang H, et al. Patient education programs for cancer-related fatigue: a systematic review. Patient Educ Couns. 2015;98:1308–19. https://doi.org/10.1016/j.pec.2015.05.003.
Niedzwiedz CL, Knifton L, Robb KA, Katikireddi SV, Smith DJ. Depression and anxiety among people living with and beyond cancer: a growing clinical and research priority. BMC Cancer. 2019;19:943. https://doi.org/10.1186/s12885-019-6181-4.
Harvey BI, Youngblood SM, Kleckner AS. Barriers and facilitators to adherence to a Mediterranean diet intervention during chemotherapy treatment: a qualitative analysis. Nutr Cancer. 2023:1–12. https://doi.org/10.1080/01635581.2023.2192891.
Acknowledgements
We would like to thank Emily Hummel, Emily Moroney, Jana Labiris, and Tess Henderson for their contributions in screening and data extraction for this manuscript.
Funding
Brenton Baguley is the recipient of a Victorian Government Early Career Fellowship through the Victorian Cancer Agency (ECRF22019). Elena George is the recipient of a Deakin University Faculty of Health Dean’s Fellowship. This publication was supported by funds through the Maryland Department of Health’s Cigarette Restitution Fund Program (CH-649-CRF to AK). Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
BB, EG, and AK were responsible for the conceptualisation and protocol development for this systematic review. AM, BG, EOC, and PR completed title and abstract screening and full text screening. All discrepancies were discussed with BB for full text review. Quality assessment and data extract was completed by all authors. PR, BG, and EOC were responsible for preparing the original draft. BB, AK, and EG were responsible for reviewing and editing the draft manuscript. BB was responsible for study supervision and project administration. All authors agreed to the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
McHugh, A., O’Connell, E., Gurd, B. et al. Mediterranean-style dietary interventions in adults with cancer: a systematic review of the methodological approaches, feasibility, and preliminary efficacy. Eur J Clin Nutr 78, 463–476 (2024). https://doi.org/10.1038/s41430-024-01426-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41430-024-01426-8
- Springer Nature Limited