Abstract
Since the origin of life, temperatures on earth have fluctuated both on short and long time scales. How such changes affect the rate at which Darwinian evolution can bring forth new phenotypes remains unclear. On the one hand, high temperature may accelerate phenotypic evolution because it accelerates most biological processes. On the other hand, it may slow phenotypic evolution, because proteins are usually less stable at high temperatures and therefore less evolvable. Here, to test these hypotheses experimentally, we evolved a green fluorescent protein in E. coli towards the new phenotype of yellow fluorescence at different temperatures. Yellow fluorescence evolved most slowly at high temperature and most rapidly at low temperature, in contradiction to the first hypothesis. Using high-throughput population sequencing, protein engineering, and biochemical assays, we determined that this is due to the protein-destabilizing effect of neofunctionalizing mutations. Destabilization is highly detrimental at high temperature, where neofunctionalizing mutations cannot be tolerated. Their detrimental effects can be mitigated through excess stability at low temperature, leading to accelerated adaptive evolution. By modifying protein folding stability, temperature alters the accessibility of mutational paths towards high-fitness genotypes. Our observations have broad implications for our understanding of how temperature changes affect evolutionary adaptations and innovations.
Similar content being viewed by others
Introduction
Temperature influences all processes in the biosphere. On the smallest scale of biomolecules, it affects processes such as protein folding and catalytic efficiency1,2,3,4,5,6. On the intermediate scale of organisms, it affects the activity and metabolic rate of individuals7,8,9. On the largest scale of ecosystems, it affects biodiversity and productivity1,10,11,12,13. These large-scale effects are ultimately also caused by temperature’s effects on the molecular scale1,14,15,16,17,18.
Most modern proteins are derived from the last universal common ancestor of today’s organisms, which existed more than 3.5 billion years ago19,20. Since this time, temperature on Earth has changed dramatically on both long- and short-time scales. We know that organisms can adapt to such changes by acquiring adaptive DNA mutations5,14,15,21,22. However, we still lack direct experimental evidence on how temperature affects the rate at which new phenotypic traits evolve. On the one hand, increasing temperature can accelerate both enzyme-catalyzed and spontaneous chemical reactions. It can also accelerate protein folding by increasing the rate at which molecules collide23,24. On the other hand, the very same collisions can cause increasing temperatures to destabilize, denature, and inactivate proteins. These two opposing factors also entail conflicting selection pressures during the adaptive evolution of proteins. At elevated temperatures, proteins must evolve greater stability to reduce heat denaturation and maintain structural integrity2,3,23. At lower temperatures, proteins need to evolve greater flexibility to combat the slowing down of molecular motion2,25,26,27. These opposing demands make it difficult to predict whether temperature changes would facilitate or hinder the evolution of a new protein phenotype.
Comparative and empirical studies have focused on the effects of temperature on the evolution of physiological performance, such as biomass growth15,28,29, but how temperature affects the evolution of new phenotypes has not been studied. Because increasing temperature accelerates most processes in the biosphere1,13,30, it may also accelerate the evolution of new phenotypes. Alternatively, increasing temperature may impede phenotypic evolution, because high temperature can destabilize proteins and thus reduce protein evolvability31,32,33. We validated these conflicting hypotheses by experimentally evolving green fluorescent protein (GFP, a derivative of the green fluorescent protein of the jellyfish Aequorea Victoria (avGFP); see Fig. S1)34 in E. coli. Because GFP is neither native to nor essential for E.coli, it interferes minimally with the E.coli proteome and physiology, and is thus especially well-suited for this purpose. We asked how a change in temperature affects the ability of GFP to evolve the new color phenotype of yellow fluorescence (Fig. 1a). During each of five rounds (“generations”) of mutation and selection, we mutagenized the gfp gene and selected cells for survival based on single-cell fluorescence phenotypes that are quantifiable through fluorescence-activated cell sorting (FACS) – another key benefit of using this protein. The starting (“ancestral”) GFP we used is well adapted to the native 37 °C growth temperature of E.coli34. We evolved it both at this temperature, as well as at a higher and lower temperature. We also monitored its evolutionary dynamics via single-molecule real-time sequencing (SMRT), and identified the effect of key genetic changes through mutant engineering and biochemical assays. In this work, we show that low temperature rather than high temperature can promote phenotypic adaptation through neofunctionalizing mutations – mutations that bring forth a new protein phenotype.
a Experimental design. We subjected four replicate populations of GFP to five generations of directed evolution at 25 °C (blue; populations L), 37 °C (black; populations M) or 44 °C (red; populations H) under strong directional selection for yellow fluorescence (λex = 488 nm and λem = 530 ± 15 nm, see ‘Methods’), allowing only the top 0.5% of cells to survive. We extracted plasmids from the surviving cells in each generation, and used the GFP inserts of these plasmids as templates for the next mutation-selection cycle and for SMRT sequencing. b, c H populations evolved new excitation and emission peaks most slowly and L populations did so most rapidly. b Relative change in peak excitation during evolution quantified by measuring the fluorescence intensity of evolving populations excited at their optimal excitation peak relative to excitation at 402 nm (note: the optimal excitation peak is 508 nm for both L and M populations and 512 nm for H populations, whereas it is 402 nm for ancestral GFP; see also Fig. S2a); (c) emission peaks of evolving populations in each generation (horizontal axes) (see also Fig. S2b). d Specific yellow fluorescence of evolving populations in each generation. The vertical axis indicates specific yellow fluorescence (arbitrary units) for evolving populations in each generation (horizontal axis). We calculated specific yellow fluorescence for each population and time point, dividing yellow fluorescence intensity by the amount of soluble fluorescent protein, as quantified by an ELISA (enzyme-linked immunosorbent assay; see ‘Methods’). We performed One-way ANOVAs with Dunnett’s post hoc test to ask whether a significant difference existed between L and M, as well as between M and H populations in panels b–d. Error bars represent one standard error of the mean (SEM) from four replicate populations (colored symbols, see color legend) in panels b–d. *P < 0.05; **P < 0.01; ***P < 0.001.
Results
Elevated temperature delays and lowered temperature accelerates evolution of a new phenotype
We evolved GFP in populations of at least 2 × 106 E. coli cells towards yellow fluorescence at 25 °C (L for low temperature), 37 °C (M for medium or unchanged temperature), and 44 °C (H for high temperature). Specifically, we evolved four replicate E. coli populations at each temperature, and allowed only the top 0.5% of yellow fluorescing cells in each generation to survive (see ‘Methods’). Because we selected directly for yellow fluorescence, we also refer to yellow fluorescence as ‘fitness’. In each generation, we generated ~ 106 GFP variants by applying mutagenic PCR to randomly introduce 1.07-1.75 amino acid changing mutations on average into the coding region of GFP (Table S1–S3).
Selection of the new yellow fluorescence phenotype involves a change in both the excitation and emission spectra (λex = ~402 nm and λem = 512 nm for wild-type GFP vs. λex = ~510 nm and λem = ~526 nm for evolved YFP). Temperature affected the evolution of both aspects of yellow fluorescence in ways that contradict our initial hypothesis. Specifically, in populations evolved at high temperature the excitation peak shifted most slowly, whereas in populations evolved at low temperature it did so most rapidly (Figs. 1b and S2a). The same holds for the emission peak. In low-temperature populations this peak shifted towards yellow fluorescence one generation earlier than in medium-temperature populations, and two generations earlier than in high-temperature populations (Figs. 1c and S2b).
An improvement in fitness (total yellow fluorescence per cell) can be achieved by either increasing the specific yellow fluorescence (yellow fluorescence intensity per protein molecule) or the number of fluorescing protein molecules in a cell. We consider only the first increase to qualify as phenotypic evolution, because it represents intrinsic single-molecule properties. Our study system allows us to accurately quantify such phenotypic changes during evolution. Specifically, we normalized the phenotypic evolution rate of each evolving population, dividing its yellow fluorescence (fitness) by the amount of soluble fluorescent protein in a cell (‘Methods’). Because misfolded proteins are often insoluble, protein solubility can also serve as a proxy for the foldability and stability of proteins at physiological temperatures. We performed this normalization because it excludes the effects of temperature on transcription, translation, and protein folding. Again, H populations achieved significantly lower specific yellow fluorescence (yellow fluorescence intensity per protein molecule) than M populations during the first four generations of evolution (One-way ANOVA with Dunnett’s post hoc test, P < 0.05; Fig. 1d). In contrast, L populations showed significantly higher specific yellow fluorescence than M populations in the first three generations (One-way ANOVA with Dunnett’s post hoc test, P < 0.01; Fig. 1d). Specific yellow fluorescence leveled off in L populations after the third generation, but only reached its maximum in M and H populations at the last generation (Fig. 1d). In sum, high temperature delays and low temperature accelerates the evolution of yellow fluorescence.
Elevated temperature slows and lowered temperature accelerates selective sweeps of neofunctionalizing mutations
To understand the genetic causes of phenotypic evolution at different temperatures, we used SMRT sequencing to genotype an average of 2391 fluorescent proteins for each replicate population in every generation (Table S3). We found that thirteen mutations achieved a frequency higher than 20% in at least one replicate L, M, or H population during evolution, and seven of these mutations (F65L, S66G, V164A, I168T, I168V, I172V, and C204Y; Fig. S3) did so in multiple replicate populations (Fig. S4a). Four of the seven high-frequency mutations (S66G, I168T, I168V, and C204Y) achieved a higher frequency in M populations than in H populations after one round of evolution and three of them reached significantly higher frequency (One-way ANOVA with Dunnett’s post hoc test, P < 0.001; Figs. 2a and S4b). In addition, the frequency for one of these four mutations remained significantly higher at the end of evolution in M populations (I168T; One-way ANOVA with Dunnett’s post hoc test, P < 0.01; Fig. 2a). In contrast, the frequencies for two of these four mutations (C204Y and I168V) were significantly lower in M populations than in L populations (Figs. 2a and S4b). One mutation in particular (C204Y) stood out, because it achieved a significantly higher frequency in L populations than in M and H populations in each generation (One-way ANOVA with Dunnett’s post hoc test, P < 0.01; Fig. 2a). Moreover, its frequency approached 100% at the end of evolution in all replicate populations, and was much higher than the frequency of any other mutation (Figs. 2a and S4b).
a Evolutionary dynamics of selected high-frequency mutations in evolving populations during evolution. b Four high-frequency mutations (dubbed neofunctionalizing) lead to a change in peak excitation. To estimate the magnitude of the shift in the excitation spectrum, we determined the ratio of fluorescence intensity excited at the mutants’ maximum excitation wavelength relative to fluorescence when excited at the ancestral maximum excitation wavelength (vertical axis). c Two neofunctionalizing mutations shift the emission peak towards greener fluorescence (see also Fig. S5). d Relative specific yellow fluorescence of ancestral GFP (WT) and mutants at low (L), medium (M) and high (H) temperatures. We calculated specific yellow fluorescence by dividing yellow fluorescence intensity by the amount of soluble fluorescent proteins quantified by an ELISA, and did so for ancestral GFP and for each mutant. We then normalized the specific fluorescence for each mutant by dividing it by that of ancestral GFP at the corresponding temperature (see ‘Methods’). e Melting temperatures of ancestral GFP (WT) and high-frequency mutants. We estimated the Tm for each mutant by heating cells that expressed the mutant proteins at varying temperatures for 5 min, and then measuring the residual fluorescence (see ‘Methods’). This procedure can estimate Tm, because the loss of fluorescence after denaturation is mainly caused by exposure of the buried chromophore to an aqueous environment65. We performed One-way ANOVAs with Dunnett’s post-hoc test to ask whether a significant difference existed between L and M, as well as between M and H populations in panel a, and to ask whether each mutant was significantly different from ancestral GFP (WT) in panels b-e. Colored symbols (see color legend) and error bars represent one standard error of the mean (SEM) from four replicate populations (colored symbols, see color legend) in panel a. Bar height and error bars represent mean ± SD (one standard deviation) based on three biological replicate fluorescence measurements (open circles) in panels b–e. *P < 0.05; **P < 0.01; ***P < 0.001.
We hypothesized that these four mutations are responsible for the shift to the new phenotype, i.e., they are neofunctionalizing mutations35. To test this hypothesis, we engineered each of these four mutations into ancestral GFP and determined their excitation and emission spectra. Indeed, all four mutations shifted the excitation peak towards that of yellow fluorescence protein, and two of them (S66G and C204Y) also shifted the emission peak towards yellow fluorescence (Figs. 2b, c, and S5). These data are also supported by previous observations, in which the same four mutations changed excitation and/or emission spectra in the genetic background s of avGFP (mutations 65G, 167T, 167V, and 203Y in avGFP36,37,38; see Fig. S1 for the differences in the coordinate system between our GFP and avGFP). In further support, we found that these four mutations also greatly improved specific yellow fluorescence of our ancestral GFP at all three temperatures (Fig. 2d). Specifically, each of them caused a 3.0–12.1-fold increase in specific yellow fluorescence. C204Y caused an even higher increase than the other three mutations. These observations indicate that the slower evolution of a yellow phenotype in H populations resulted from the slower sweep of neofunctionalizing mutations, and especially of C204Y, in H populations. Conversely, faster evolution in L populations resulted from the faster sweep of these mutations.
Neofunctionalizing mutations often destabilize proteins39,40,41. Our four mutations are no exception: All four neofunctionalizing mutations reduce the melting temperature of GFP significantly (One-way ANOVA with Dunnett’s post hoc test, P < 0.001; Figs. 2e and S6). This observation raises an important question. How can these mutations be beneficial while reducing protein stability? To find out, we next examined another class of mutations that often occur in experimental and natural evolution. These are mutations that increase the thermodynamic stability or foldability of proteins3,40,42.
Stabilizing mutations are most beneficial at high temperature
Among the seven mutations that we had identified as rising to high frequency, three (F65L, V164A, and I172V) did not change the emission and excitation spectra of fluorescent protein, nor did they improve specific yellow fluorescence (Fig. 2b–d). Previous studies have revealed that these three mutations can improve the folding stability of YFP42 and of avGFP (mutations 64L, 163A and 171V in the coordinate system of avGFP, see also Fig. S1)38,43. Absent other phenotypes, we hypothesized that these mutations are beneficial, because they can also increase protein folding stability in the genetic background of our ancestral GFP. To test this hypothesis, we engineered each of the three mutations into ancestral GFP, and measured the melting temperature Tm of the engineered fluorescent protein. Indeed, all three mutations significantly increased this melting temperature (One-way ANOVA with Dunnett’s post hoc test, P < 0.001; Figs. 2e and S6).
We next examined the evolutionary dynamics of these mutations and found that they reached the highest frequencies in H populations, intermediate frequencies in M populations, and the lowest frequencies in L populations (Figs. 2a and S4b). For example, at the end of evolution, the frequency of variant F65L was 2.4-fold higher in H populations than in M populations, and 24.2-fold higher in M populations than in L populations. The second mutation V164A also achieved the highest frequency in H populations, lower frequency in M populations, and the lowest frequency in L populations in each generation of evolution (Fig. 2a). These observations suggest that the stabilizing mutations are most beneficial at high temperature and least beneficial at low temperature.
One possible explanation is that these stabilizing mutations increase expression yields by enhancing the transcription and stability of mRNA, or by reducing protein misfolding and insolubilization through slowing translation33,44. To test this hypothesis, we engineered all possible synonymous mutations into the codon encoding each of the three stabilizing mutations. The resulting data shows that none of these synonymous mutations substantially changes green fluorescence at either low, medium or high temperature for any of these variants (Fig. S7). Thus, the greater benefit of these mutations at higher temperatures does probably not just result from their effects on transcription and translation.
Another possible explanation is that proteins expressed at a lower temperature are more stable than they need to be – they have excess folding stability – because the slower and less disruptive molecular motions at that temperature are less likely to disrupt a protein fold45. This hypothesis creates several predictions. First, our stabilizing mutations should preferentially increase protein solubility at high temperature, but less so at medium or low temperature. This is indeed the case (Fig. 3a). They increase solubility on average 66.4-fold at high temperature, 3.0-fold at medium temperature, but do not significantly affect solubility at low temperature (Fig. 3a). We observed a similar solubility increase in a cell-free expression system that only contained the components necessary for in vitro transcription and translation. Specifically, in this cell-free system all three stabilizing mutations increase solubility on average 10.3-fold at high temperature, but only 2.4-fold at medium temperature and 1.9-fold at low temperature (Fig. S8). The difference in magnitude between the in vivo and in vitro solubility improvement might be due to the fact that the in vivo environment contains chaperones and proteases, which can affect the folding and degradation of different variants to a different extent40. Even though the proteins we study are thermally highly stable (Tm > 70 °C), a stabilizing mutation that increases the melting temperature by a few degrees can increase the amount of soluble fluorescent proteins at 44 °C more than 60-fold (Fig. 3a). The reason is that elevated temperature does not just cause inactivation of the mature protein (quantified by Tm) but it can also interfere with the post-translational maturation of GFP46.
a, b Relative solubility (a) and relative yellow fluorescence (b) of single mutants at low (L), medium (M) and high (H) temperatures. The vertical axes indicate the amount of soluble protein (a) or the fluorescence intensity (b) for each mutant (horizontal axes) relative to ancestral GFP. c Neofunctionalizing mutations and stabilizing mutations are more beneficial at low and high temperatures, respectively. The horizontal (x) and vertical (y) axes indicate yellow fluorescence of single and double mutants relative to ancestral GFP at low (L) and high (H) temperatures based on three biological replicate measurements (single small symbols). The dashed line indicates equality (y = x), and values above (below) this line indicate the mutants are more (less) beneficial at low temperature. Colors indicate ancestral GFP (WT, gray), mutants that carry neofunctionalizing mutations (purple), stabilizing mutations (yellow), or both neofunctionalizing and stabilizing mutations (green). d Evolutionary dynamics of neofunctionalizing mutations and stabilizing mutations. The vertical axes indicate the mean number of neofunctionalizing (solid line) or stabilizing mutations (dashed line) per sequence in each generation of evolution (horizontal axes). Bar height and error bars represent mean ± SD based on three biological replicate measurements (single small symbols) in panels a and b, and mean ± SEM based on four replicate populations (circles, squares, or triangles) in panel d. Note the logarithmic vertical scales in panels a and b. We performed One-way ANOVAs with Dunnett’s post hoc test to ask whether fluorescence or solubility of mutants was significantly different from that of ancestral GFP (WT) in panels a and b. We used two-sided t-tests with ‘Holm’ adjustment to test whether the average number for neofunctionalizing mutations per sequence was significantly different from that for stabilizing mutations in L, M, and H populations in panel d. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
Another prediction of the excess-stability hypothesis is that the three stabilizing mutations should be most beneficial at high temperature and least beneficial at low temperature. We tested this by measuring yellow fluorescence at different temperatures. All three stabilizing mutations greatly improved total yellow fluorescence at high temperature, but only one of them (V164A) slightly did so at low temperature (Fig. 3b; note that stabilizing mutations can improve total yellow fluorescence by increasing protein copy number, because ancestral GFP has weak yellow fluorescence). For example, the mutation F65L, which increases the melting temperature by 5.8 °C (Fig. S6b), caused a ~ 24-fold increase in yellow fluorescence at high temperature (One-sided t-test, P = 6.92 × 10−6), but led to a 30.9% decrease of yellow fluorescence at low temperature (One-sided t-test, P = 1.15 × 10−3; Fig. 3b). Similarly, both of the stabilizing mutations V164A and I172V greatly increased yellow fluorescence at high temperature (by 73.8-fold and 11.7-fold, respectively) but neither of them did so at low temperature. All of the stabilizing mutations only moderately increased yellow fluorescence at medium temperature (Fig. 3b). In sum, high temperature enhances and low temperature weakens the fitness benefits of stabilizing mutations (Fig. 3c).
Neofunctionalizing mutations destabilize proteins the most at high temperature but the least at low temperature
The excess stability hypothesis also predicts that fluorescent proteins are most affected at high temperature but least affected at low temperature by the destabilizing effects of neofunctionalizing mutations45. We tested this prediction by examining how neofunctionalizing mutations affect protein solubility. Two of the four mutations reduced protein solubility at high temperature (Fig. 3a), a reduction that was greatest (75.5%) for the neofunctionalizing mutation C204Y (One-sided t-test, P = 0.03; Fig. 3a). In contrast, only one of the four mutations reduced protein solubility (and to a lesser extent) at medium temperature (60.6%). None of the four mutations reduced solubility significantly at low temperature (Fig. 3a).
A further prediction is that neofunctionalizing mutations preferentially increase fitness at low temperature. Indeed, while all four neofunctionalizing mutations enhanced yellow fluorescence greatly, they did so to different extents across the three temperatures (Fig. 3b). Three of them increased yellow fluorescence to a greater extent at low temperature than at high temperature (Fig. 3c). Mutation C204Y led to the greatest increase in yellow fluorescence at low temperature (13.4-fold). Importantly, it improved yellow fluorescence to a greater extent at low temperature than at medium and high temperatures (One-way ANOVA with Dunnett’s post hoc test, P < 0.001; Figs. 3b and S9a). Another neofunctionalizing mutation, I168T, which achieved a 2.9-fold higher frequency in L populations than in H populations at the end of evolution (Fig. 2a), was also more beneficial at low temperature than at medium and high temperatures (Figs. 3b and S9a). These observations confirm that elevated temperature weakens and lowered temperature enhances the fitness benefits of neofunctionalizing mutations.
The excess stability hypothesis also suggests that stabilizing mutations should outcompete neofunctionalizing mutations during evolution in H population, and that the opposite should be the case in L populations. Indeed, we found that the mean number of stabilizing mutations per GFP coding sequence was 1.1–2.4-fold higher than that for neofunctionalizing mutations in H populations during the first three generations, but it was 9.8–41.9-fold lower in L populations during each generation of evolution (Fig. 3d). Taken together, these observations indicate that elevated temperature exacerbates the destabilizing effects and reduces the fitness benefits of neofunctionalizing mutations, whereas lowered temperature reduces their destabilizing effects and thus magnifies their benefits.
Epistatic interactions promote the spreading of stabilizing mutations at high temperature
Neofunctionalizing and stabilizing mutations may interact non-additively when they occur on the same molecule. Such non-additive (epistatic) interactions can be positive or negative, depending on whether two adaptive mutations together lead to a fitter or less fit genotype than expected. To understand how epistasis may affect phenotypic evolution, we first identified genotypes with multiple amino acid mutations in our populations. Only three double mutants (and no higher order mutants) achieved a frequency exceeding 50% in L, M or H populations at the end of evolution (C204Y + I168T, C204Y + F65L, and C204Y + V164A; Fig. 4a).
a Frequency dynamics of high-fitness double mutants during evolution at low (L), medium (M) and high (H) temperatures. b Relative yellow fluorescence of C204Y + I168T at different temperatures. c Negative epistasis between the constituent mutations of C204Y + I168T at different temperatures. d Relative solubility of C204Y + I168T and its constituent mutations at different temperatures. e Relative specific yellow fluorescence of three high-fitness double mutants at low temperature. The vertical axes in panels b and d, respectively, indicate the yellow fluorescence intensity and solubility of each mutant (horizontal axes) relative to ancestral GFP at the corresponding temperature. The vertical axis in panel c indicates the degree of epistasis (see text for details) between the constituent mutations of C204Y + I168T. The vertical axis in panel e indicates the specific yellow fluorescence of each double mutant (horizontal axis) relative to ancestral GFP at low temperature. We calculated specific yellow fluorescence by dividing yellow fluorescence intensity by the amount of soluble fluorescent proteins, and then normalized the specific yellow fluorescence for each mutant by dividing it by that of ancestral GFP at low temperature (see ‘Methods’). Bar height and error bars represent mean ± SEM based on four replicate populations for panel a, and mean ± SD based on three biological replicate measurements (single small symbols) for panels b–e. We performed One-way ANOVAs with Dunnett’s post hoc test in panels a-c and e. In panel d, solid lines between ancestral GFP (WT) and a mutant or between mutants indicate a significant difference in relative solubility between them (P < 0.05, One-sided t-test) and dashed lines indicate no significant difference. *P < 0.05; **P < 0.01; ***P < 0.001.
The first double mutant (C204Y + I168T) consists of two destabilizing neofunctionalizing mutations (Fig. 2d, e). It caused a much smaller increase in yellow fluorescence at high temperature (3.9-fold increase) than at medium and low temperatures (8.1-fold and 35.2-fold increase, respectively; Fig. 4b). Though part of this difference is caused by a lower improvement in yellow fluorescence at high temperature for each of its constituent mutations (Fig. S9a), we wondered whether high temperature can also alter interactions between these mutations and thus further contributes to the double mutant’s lower fitness increase. To find out, we calculated the extent of epistasis between mutation pairs based on the expression e = (FA+B - FWT) – (FA-FWT) - (FB - FWT), where FA+B, FA, FB, and FWT, respectively, represent the logarithmically transformed yellow fluorescence of the double mutant A + B, of its constituent mutants A and B, and of ancestral GFP47. The extent of epistasis e represents the relative difference between the fitness effect of the double mutant and the sum of the effects of both constituent mutations. A negative (positive) value of e indicates negative (positive) epistasis, i.e., the combined effects of the mutations are smaller than (greater than) expected from additivity. A value of e = 0 indicates no epistasis, i.e., additivity. At all temperatures, the extent of epistasis was far below zero, indicating that the double mutant C204Y + I168T increases fitness less than the sum of its constituent single mutants (Fig. 4c). However, at high temperature epistasis was less negative than at medium and low temperatures (Fig. 4c). Consistent with this observation, I168T did not significantly reduce the solubility of C204Y at all temperatures (Fig. 4d). This demonstrates that the lower fitness improvement for the double mutant C204Y + I168T at high temperature mainly resulted from a smaller increase in yellow fluorescence for each of its constituent mutations rather than from negative epistasis between them. The resulting lower fitness improvement caused a much slower sweep of the genotype C204Y + I168T through all H populations than through M or L populations (Fig. 4a). This slower spreading contributed to the slower phenotypic evolution in H populations, because the genotype conveyed much higher specific yellow fluorescence (>1.6-fold) than the other two double mutants C204Y + F65L and C204Y + V164A that swept most rapidly through H populations (One-way ANOVA with Dunnett’s post hoc test, P < 0.001; Fig. 4a, e).
The remaining double mutants (C204Y + F65L and C204Y + V164A) combine the neofunctionalizing (but destabilizing) mutation C204Y with one of the two stabilizing mutations F65L and V164A (Fig. 2e). For the double mutant C204Y + F65L, high and medium temperatures caused a positive interaction between its single constituent mutations, resulting in a higher fitness effect than expected from the fitness effects of two single mutants (Fig. 5a). This positive epistasis was significantly greater at high temperature than at medium temperature (One-way ANOVA with Dunnett’s post hoc test, P < 0.001; Fig. 5a). In contrast, low temperature almost completely suppressed this positive epistasis and caused the interaction to become additive (Fig. 5a). In addition, the stabilizing mutation F65L greatly increased yellow fluorescence of C204Y at high temperature (88.4-fold) and medium temperature (5.9-fold) but significantly reduced it at low temperature (Two-sided t-test, P < 0.001; Fig. 5b). This switch from being beneficial at high and medium temperatures to deleterious at low temperature originates from the extent to which F65L can compensate for the destabilizing effect of C204Y. Because F65L reduces specific yellow fluorescence by 16.7–43.4 % at all temperatures when introduced into the C204Y mutant (Fig. 5c), it can achieve a net fitness benefit only by increasing solubility. This is the case at high temperature, where F65L increases solubility by 116.4-fold (Fig. 5d). At medium temperature, it still increases solubility by 6.9-fold. In contrast, at low temperature, it no longer significantly increases solubility (Fig. 5d). In consequence, increased solubility cannot compensate for F65L’s reduction in specific fluorescence at low temperature. These temperature-specific effects are also underscored by the evolutionary dynamics of the constituent mutations. At high temperature, F65L rose to high frequency before C204Y did (Fig. 5e), which is consistent with its stronger fitness benefit at this temperature (Fig. 3a). At medium temperature, F65L is less beneficial than C204Y, and it thus also rose to a high frequency more slowly than C204Y. Finally, at low temperature, the frequency of F65L stayed below 1.2% during evolution (Fig. 5e). Similarly, high temperature led to greater positive epistasis between the constituent mutations of the double mutant C204Y + V164A than medium temperature but low temperature resulted in slightly negative epistasis (Fig. 5f), consistent with the observations that the stabilizing mutation V164A causes a higher increase in yellow fluorescence on top of C204Y at high temperature than at medium and low temperatures (Fig. 5g), and that it can greatly compensate for the destabilizing effect of C204Y at high temperature (Fig. 5h). By enhancing positive epistasis between C204Y and F65L or V164A, high temperature made both double mutants more beneficial than C204Y + I168T (Fig. S10) which only harbors neofunctionalizing mutations and has higher specific yellow fluorescence (Fig. 4e). As a result, high temperature helped these two genotypes outcompete C204Y + I168T (Fig. 4a), and thus delayed the evolution of the new yellow fluorescence phenotype (Fig. 1).
a High temperature enhances and low temperature neutralizes positive epistasis between the constituent mutations of C204Y + F65L. b–d Effects of F65L on yellow fluorescence (b), specific yellow fluorescence (c) and solubility (d) at the genetic background of C204Y at low (L), medium (M) and high (H) temperatures. e Frequency dynamics of C204Y and F65L during evolution at different temperatures. f Increasing temperature changes epistasis between the constituent mutations of C204Y + V164A from negative to positive. g, h Effects of V164A on yellow fluorescence (g) and solubility (h) at the genetic background of C204Y at different temperatures. The vertical axes in panels a and f indicate the degree of epistasis (see text for details) between the constituent mutations of C204Y + F65L and of C204Y + V164A, respectively. The vertical axes in panels b–d indicate the yellow fluorescence intensity, specific yellow fluorescence, and solubility of C204Y and C204Y + F65L relative to ancestral GFP at the corresponding temperature. The vertical axis of panel e indicates the frequency of each mutant at the corresponding temperature. We calculated specific yellow fluorescence by dividing yellow fluorescence intensity by the amount of soluble fluorescent proteins and then normalized the specific yellow fluorescence for each mutant by dividing it by that of ancestral GFP at the corresponding temperature (see ‘Methods’). The vertical axes in panels g and h indicate the yellow fluorescence intensity and solubility of C204Y and C204Y + V164A relative to ancestral GFP at the corresponding temperature. Bar height and error bars represent mean ± SD based on three biological replicate measurements (single small symbols) for panels a–d and f–h. We performed one-way ANOVAs with Dunnett’s post hoc test in panels a and f, two-sided t-tests in panels b–d and g, h, and two-sided t-tests with ‘Holm’ adjustment in panel e. *P < 0.05; **P < 0.01; ***P < 0.001; ****P < 0.0001.
In sum, high temperature slows phenotypic evolution by favoring stabilizing mutations and enhancing their positive epistasis with neofunctionalizing mutations. In contrast, low temperature accelerates phenotypic evolution by favoring destabilizing neofunctionalizing mutations, and by reducing or eliminating their positive epistasis with stabilizing mutations.
Discussion
Our observations demonstrate that increasing the temperature at which a protein evolves can hinder phenotypic adaptation. In contrast, reducing temperature can accelerate such adaptation (Fig.1). Increasing the temperature worsens the destabilizing effects of neofunctionalizing mutations and thus slows their sweeps (Figs. 2a and 3c). Conversely, lowering the temperature creates excess protein folding stability (Figs. 2e and 3a), which reduces the destabilizing cost of neofunctionalizing mutations, increases their fitness benefit, and thus helps them spread faster through a population (Figs. 2a, 3c, d, and 4a).
Protein stability is crucial for protein evolution31,48,49 and is a major goal in protein engineering50,51. Increasing temperature destabilizes proteins by accelerating molecular motions, whereas decreasing temperature stabilizes proteins by slowing such motions45. Everything else being equal, temperature thus changes the equilibrium between folded and unfolded protein states. In other words, a temperature increase can reduce the amount of correctly folded and thus soluble fluorescent proteins. For example, we found that increasing the temperature from 37 °C to 44 °C led to a 17.2-fold decrease of fluorescent protein solubility – a measurable proxy for protein stability (Fig. S9b). This stability loss can be rescued by stabilizing mutations (Fig. 3a). Such mutations become especially beneficial during evolution at high temperature, where they benefit fitness more than neofunctionalizing mutations (Fig. 3b). In consequence, they spread more rapidly than neofunctionalizing mutations at high temperature (Fig. 3d), which delays phenotypic adaptation (Fig. 1).
At any one location on Earth, temperature may rapidly fluctuate by many degrees due to diurnal cycles and seasonal changes. Such temperature changes can alter the strength of selection to modify protein stability during the evolution of new protein functions. Our experiments show that an increase in temperature strengthens and a decrease weakens selection for stability (Fig. 3d). Because adaptive mutations that bring forth new protein functions usually destabilize proteins39,40,41,52, stronger selection on protein stability at high temperatures can delay the spreading of neofunctionalizing mutations through an evolving population. Consequently, increasing temperature can slow down and lowering temperature can speed up the evolution of a new phenotype (Fig. 1). We expect that our observations will apply whenever neofunctionalizing mutations reduce the stability of adaptively evolving proteins. Whether they also apply when neofunctionalizing mutations do not reduce stability35 is an exciting question for future work.
Populations of organisms cope with elevated temperatures through evolutionary adaptation both in the laboratory and in the wild13,14,15,53,54,55,56. However, once they have exceeded their upper thermal limits they often go extinct15,54,57. Our observations may help explain such extinction. First, evolutionary rescue becomes impossible if high temperature destabilizes important proteins so much that stability-enhancing mutations become exceedingly rare. Second, if the emergence or improvement of an adaptive phenotype necessary for survival requires one or more strongly destabilizing mutations, a temperature increase may render the mutant proteins unable to fold, and thus prevent the rescue of the population through adaptive evolution15,22.
Our work has several practical implications. For example, it suggests that lowering temperature substantially below that required for soluble protein expression can help evolve or engineer proteins with new functions, because it helps function-altering mutations to manifest their beneficial effects. This strategy may be especially helpful when conventional approaches fail, because neofunctionalizing mutations are too strongly destabilizing41,58,59. Another implication relates to ongoing global climate change, which can exert severe thermal stress on organisms and force them to undergo adaptive evolution to higher temperatures60,61. Although global climate change itself alters the average global environment only very gradually, one of its consequences is an increase of extreme climatic events, such as extreme heat waves with rapid temperature increase48,61,62,63. Such a rapid temperature increase can purge neofunctionalizing mutations that destabilize proteins. Consequently, it may impede phenotypic evolution, and thus result in decreased genetic diversity and biodiversity. Most generally, our work suggests that a temperature change can profoundly affect how life finds a way towards phenotypic innovation.
Methods
Strains and plasmids
We used the plasmid pBAD202/D-TOPO (K4202-01, Invitrogen) as our expression vector. This plasmid harbors a Kanamycin-resistance gene and an arabinose-inducible araBAD promoter that controls the expression of a GFP gene, which is integrated into the vector between XhoI and HindIII restriction sites, as described previously34. We refer to the encoded GFP protein as our ‘ancestral’ GFP. It carries a methionine insertion at the second residue and six amino acid substitutions compared to the Aequorea victoria green-fluorescent protein (avGFP) (Fig. S1). It has an excitation maximum at 405 nm, and an emission peak at 512 nm. We used the E. coli host BW27783 (CGSC 12119) throughout to ensure homogeneity of GFP expression.
Preparation of electrocompetent cells
We inoculated a single colony of E. coli strain BW27783 into 5 mL SOB medium in a 50 mL flask, and grew the resulting culture at 37 °C with shaking at 220-250 rpm for 12–16 h. After overnight incubation, we transferred 3 mL of the culture into 300 mL SOB medium in a 2 L flask, and continued the incubation until the OD600 value equaled 0.4–0.6 (1 cm optical path length). We then placed the culture on ice for at least 15 min, and pelleted cells at 4 °C by centrifugation at 1500 g for 15 min. We resuspended the cell pellets using 60 mL ice-cold ddH2O and distributed them equally into three 50 mL tubes. We then used a 10 mL pipette to slowly deliver 10 mL of ice-cold glycerol/mannitol solution (20% glycerol (w/v) and 1.5% mannitol (w/v)) to the bottom of each tube. We set the centrifuge’s acceleration/deceleration to zero and used it to pellet cells by centrifugation at 1500 g and 4 °C for 15 min by a concentrator (Eppendorf 5810/5810R). We removed the supernatant by aspiration and resuspended cell pellets in 3.0–4.0 mL ice-cold glycerol/mannitol solution. We used a dry ice-ethanol bath to freeze the resulting cell suspensions for ~1 min, and then stored at −80 °C for the following electroporation experiments.
Preparation of mutant libraries and expression of fluorescent proteins
In every generation, we inserted mutated GFP coding regions into fresh backbones and transformed into fresh competent cells to avoid the incidence of mutations in other region of plasmid or the genome of E. coli. We conducted mutagenic PCR to introduce random mutations into the coding region of GFP, as previously described64, but with the following modifications. In brief, we prepared 50 µL of a PCR reaction mix that consists of 35.25 µL ddH2O, 2.5 µL template (1 ng/µL), 5 µL of 10×ThermoPol buffer (M0267L, NEB), 0.25 µL of Taq DNA polymerase (5 U/µL, M0267L, NEB), 2 µL of dNTPs (10 mM, R0192, Thermo Scientific), 1.5 µL of 8-oxo-GTP/dPTP (100 µM, Trilink Biotechnologies), and 1 µL of each primer (Mutafp511F - GAAGGAGATATACCTCGAG/Mutafp511R - AGACCGTTTAAACAAGCT T).
We executed the following thermocycling program to perform the PCR: 95 °C/30 s; 20 cycles of 95 °C/20 s, 46 °C/30 s and 68 °C/ 6 min. After PCR amplification, we used a QIAquick PCR purification kit (QIAGEN) to purify PCR products, added 1 µL of DpnI (R0176S, NEB), and incubated at 37 °C for ~1 h to remove the template plasmid. Then we added 2 µL XhoI and HindIII (R0146L/R3104S, NEB), and continued the incubation at 37 °C to digest the purified PCR products. After overnight digestion, we purified the mutated GFP pools as inserts for ligation by using the QIAquick PCR purification kit.
We performed the ligation reaction in a 20 µL solution, which contained ~60 ng of purified inserts, ~100 ng of purified vector backbone, 10 U of T4 DNA ligase and 1×Ligation buffer (M0202L, NEB). We performed the ligation reaction at 20–22 °C for ~16 h, and subsequently purified the ligation product by precipitation. To this end, we mixed the ligation product with 80 µL of ddH2O, 50 µL of 7.5 M ammonium acetate (A2706-100 mL, Sigma), 1 µL of glycogen (R0551, Thermo Scientific), and 375 µL of ice-cold absolute ethanol. We kept the mixture in a −80 °C freezer for ~20 min, and then pelleted the ligated plasmids by centrifugation at 18,000 g for 20 min. We washed the pellet once using 800 µL of cold ethanol (70%), and dried it using a concentrator (Eppendorf 5301). We then used 10 µL of ddH2O to dissolve the pellet as the purified ligation product for electroporation.
We mixed 5 μL of the resulting ligation product with 100 μL of electrocompetent BW27783 cells, and transferred the mixture into a 0.2 cm pre-cooled cuvette (EP202, Cell Projects, UK). We used a Micropulser electroporator (Bio-Rad) to perform electroporation at 15 kV/cm. Then we immediately added 1 mL of pre-warmed SOC medium and transferred the resulting culture into a 50 mL tube and incubated it for 1.5 h at 37 °C with shaking at 220 rpm. We added 10 mL of LB containing 50 µg/mL kanamycin. We serially diluted the resulting transformants using saline, and plated 50 or 100 µL of the serially diluted aliquot on LB agar supplemented with 25 μg/mL of kanamycin to estimate the mutant library size, which equaled ~106 colonies per transformation. We continued to incubate the remaining culture for 12 ~ 14 h. We then sampled 2 mL of this culture and pelleted cells by centrifugation at 9000 g and 4 °C for 5 min. We resuspended cells with 2 mL LB containing 50 µg/mL kanamycin and 0.2% arabinose in a 14 mL tube. We incubated the resulting culture using a microplate shaker with a shaking speed of 480 rpm (VWR) at 25 or 37 °C for ~12 h, or 44 °C for ~8 h. After induction, we put the culture on ice until we performed cell sorting.
Sorting cells
We selected high-fluorescence cells by using a cell sorter (BD Aria III) at ~25 °C for all evolving populations, because this instrument does not allow us to change the temperature of droplets harboring cells when quantifying fluorescence. However, the fluorescence detected at ~25 °C for a given variant is likely to reflect its fluorescence relative to ancestral GFP or to other variants at 37 °C for M populations and at 44 °C for H populations. This is because GFP and its variants are highly thermostable (usually Tm > 60 °C) once they have folded correctly and the chromophore has maturated. In other words, protein folding and the maturation of the chromophore at different temperatures are the key factors that determine the fluorescence of variants for our L, M, and H populations. This is evident from our observation that the fluorescence intensities for all seven high-frequency mutants relative to ancestral GFP did not change substantially when measured at 26, 36, and 42 °C (Fig. S11). In sum, our study system enables us to minimize the interference of potential factors other than temperature on a protein’s phenotype.
After induction of GFP expression at different temperatures, we mixed 20–40 µL of culture with 800 µL of PBS buffer, and used the resulting cell suspension for sorting. Specifically, we selected the top 0.5% of yellow fluorescing cells (Fig. 1a) with an Aria III cell sorter (BD Biosciences) in the FITC channel (λex = 488 nm, λem = 530 ± 15 nm). We sorted cells at 4 °C and collected 104 cells in ~1 mL LB medium in a 1.5 mL tube for every population grown at 25 and 37 °C, as well as 2 × 104 cells for populations grown at 44 °C. We selected two-fold more cells for every population that grew at 44 °C because cell viability for populations grown at 44 °C is roughly half that of populations grown at 25 and 37 °C. We put the selected cells on ice until we had sorted all samples to avoid cell death or proliferation. After sorting all samples, we incubated the selected cells at 37 °C with a shaking speed of 220 rpm for 3 ~ 5 h. We then mixed the culture with 2 mL LB supplemented with 75 µg/mL kanamycin and continued the incubation for ~12 h. We sampled 1 mL of overnight culture for isolating plasmids by using a QIAprep Spin Miniprep Kit (Qiagen). We used the isolated plasmids as templates for the next-round of PCR mutagenesis, as well as for SMRT sequencing. In addition, we mixed 0.9 mL of overnight culture with 0.6 mL 50% glycerol to make a glycerol stock stored at – 80 °C for the further experiments described below.
Engineering GFP mutants
We engineered single mutants and double mutants by using whole plasmid PCR. Specifically, we designed a pair of primers for each single or double mutant that carried the corresponding mutation. To generate all single mutants, we used ancestral GFP as the template for whole plasmid PCR. To engineer double mutants that all harbored the mutation C204Y, we used the mutant C204Y as the template for a whole plasmid PCR. We performed the whole plasmid PCR in a 50 µL reaction solution which contains 1 ng of template plasmid, 10 µL of 5×Q5 reaction buffer, 10 µL of 5×Q5 high GC enhancer, 1.0 µL of 10 nM dNTPs, 1.0 µL of each primer (10 μM), 0.5 μL of Q5 High-Fidelity DNA Polymerase, and 25.5 µL of ddH2O. We executed the PCR with the following program: 98 °C/ 30 s; 8 cycles of 98 °C/15 s, 64 or 72 °C/10 s and 72 °C/2 min; 20 cycles of 98 °C/15 s and 72 °C/130 s; 72 °C/5 min. We purified the PCR products using the QIAquick PCR purification kit and used DpnI to remove the template plasmid by digesting the PCR product at 37 °C for 1.5 ~ 2 h. We sampled 1 μL of the digested PCR product, and mixed it with 30 μL electrocompetent cells for electroporation. After allowing the transformants to recover at 37 °C for ~1 h, we plated 50 μL of recovered transformants on LB agar supplemented with 25 μg/mL kanamycin. We picked three to six colonies from each transformation and Sanger-sequenced the plasmid DNA. We chose a correct construct for each mutant, confirmed by Sanger sequencing, and grew it in 2 mL LB (50 μg/mL kanamycin) at 37 °C with shaking at 220 rpm. Then we mixed 600 μL 50% glycerol with 900 μL overnight culture and stored the mixture at −80 °C for the further experiments. We isolated the plasmids and used a XhoI and HindIII digestion for further confirmation. To add a 6 × His tag at the C-terminal of ancestral GFP and each mutant, we used the correctly constructed plasmids as templates to perform whole plasmid PCR using the following pair of primers (HindHISF- TACAAGAAGCTTCATCATCACCATCACCATtgaGTTTAAACGGTCTCCAGCTTGGCT/ HindHISR- AACTCAATGGTGATGGTGATGATGAAGCTTCTTGTACAGCTCGTCCATGCCGAGAG. The PCR followed the same procedure we have just described. We used the resulting His-tagged ancestral GFP and its mutants for further experiments.
We followed the same procedure as described above to engineer synonymous mutations for the mutants F65L, V164A, and I172V by whole plasmid PCR. We confirmed the correct construction for each mutant by Sanger sequencing and prepared glycerol stocks for further experiments, as described above.
Fluorescence assay, excitation/emission spectrum scan, and quantification of soluble proteins
To perform a fluorescence assay for each replicate evolving population, we transferred 20 μL of glycerol stock into 2 mL LB medium supplemented with 50 μg/mL kanamycin, and grew the resulting culture at 37 °C in a VWR microplate shaker (VWR International, USA) with a shaking speed of 480 rpm or in a WIGGENS microplate shaker with a shaking speed of 480 rpm (WIGGENS GmbH, Germany) for ~12 h. We sampled 1 mL of culture and pelleted cells by centrifugation at 9000 g and 4 °C for 5 min. We resuspended cells in 2 mL LB medium containing 50 μg/mL of kanamycin as well as 0.2% arabinose, and continued the incubation for ~12 h at 25 and 37 °C, as well as for ~10 h at 44 °C. We then transferred 20 μL of the culture into 190 μL PBS, and mixed thoroughly in a 96-well plate. Subsequently, we added 10 μL of the resulting cell suspension to another 190 μL PBS for quantifying fluorescence intensity at the single-cell level (Fig. S12), using a Fortessa (BD Biosciences) or CytoFLEX LX cell analyzer (Beckman Coulter). We used the remainder of the cell suspension for measuring fluorescence intensity, and scanned the excitation/emission spectrum using a Tecan plate reader (Spark). We also sampled 1 mL of culture to extract soluble proteins by using a CelLytic™ B Cell Lysis Reagent (B7435-500 mL, Sigma) or a One-Step Bacterial Active Protein Extraction Kit (Sangon Biotech). We followed the manufacture’s protocol to quantify the amount of soluble fluorescent proteins by using a GFP ELISA Kit (AKR-121, Cell Biolabs Inc.), which can detect GFP, BFP, CFP, and YFP from Aequorea Victoria. We followed the same procedure to perform fluorescence assays, excitation/emission scans, and quantification of soluble proteins for each of the His-tagged engineered mutants, except that we used a His-tag ELISA Detection Kit (L00436, Genscript) rather than a GFP ELISA Kit to quantify soluble protein.
Protein thermal stability assay
To estimate the thermal stability of each mutant and ancestral GFP, we thoroughly mixed 1 μL of crude lysate with 99 μL of TNG (100 mM Tris, 100 mM NaCl, 10% glycerol, 10 mM DTT, 1×cOmplete™ (EDTA-free Protease Inhibitor Cocktail, Roche 11873580001), pH 7.5) buffer by pipetting. We then used a thermocycler to heat the resulting mixture for 5 min in a temperature range of 60–80 °C (60.0, 60.6, 62.0, 64.1, 66.6, 69.0, 71.6, 74.0, 76.5, 78.6, and 80.0 °C), followed by a 30-second incubation at 4 °C. Immediately after the incubation, we transferred 90 μL of each mixture to a 96-well microplate, and used a Tecan Spark plate reader (λex = 400 nm, λem = 512 nm) to measure its fluorescence intensity. We used the unheated lysate-buffer mixture and the GFP-free lysate (from cells without a GFP gene) as positive and blank controls, respectively. We calculated the residual fluorescence as fluorescence relative to the positive control. We then used the resulting data to fit a sigmoidal model, and derived the midpoint temperature of the transition curve (Tm, the temperature at which 50% of proteins unfold) for each mutant, as well as for ancestral GFP (WT) (Fig. S6a). This procedure can estimate the denaturation midpoint of each mutant and of ancestral GFP by directly measuring the green fluorescence change, because the chromophore of fluorescent proteins still stays chemically intact even in the denatured state, and the loss of fluorescence after denaturation is mainly caused by exposing the buried chromophore to an aqueous environment65.
In addition, we also measured the in vivo Tm for ancestral GFP and its variants. Specifically, to quantify Tm for any one of these proteins, we first grew cells for inducible protein expression at 37 °C, as described above. We then sampled ~300 μL of cell culture and pelleted the cells by centrifugation at 4000 g and 4 °C for 15 min. We washed the pelleted cells using 1 mL of cold PBS and resuspended them in ~1.8 mL of cold PBS. Subsequently, we transferred 90 μL of the suspended cells into a 96-well PCR plate,and used a thermocycler to heat the resulting cell suspension for 5 min in a temperature range of 55-95 °C (55.0, 58.6, 62.6, 67.6, 72.5, 77.5, 82.4, 87.4, 91.4, 93.9, and 95.0 °C), followed by a 30-second incubation at 4 °C. Immediately after the incubation, we transferred 80 μL of each cell suspension to a 96-well microplate, and used a Tecan Spark plate reader (λex = 400 nm, λem = 512 nm) to measure its fluorescence intensity. We used the unheated cell suspension and the GFP-free cell suspension (cells without a GFP-coding gene) as positive and blank controls, respectively. We followed the same procedure as described above to calculate the residual fluorescence, to fit the resulting data to a sigmoidal model, and to derive the midpoint temperature of the transition curve (Tm) for each mutant, as well as for ancestral GFP (WT) (see Fig. 2e).
Protein purification and melting temperature (T m) determination by circular dichroism
We purified fluorescent proteins for ancestral GFP and its variants and measured their melting temperatures (Tm) using circular dichroism. To purify the proteins, we inoculated 7.5 μL of a glycerol stock of ancestral GFP or its variants into a 250 mL flask containing 25 mL of LB supplemented with 50 μg/mL Kanamycin. We incubated the culture at 37 °C with shaking at 220 rpm for 12 h. We pelleted the cells by centrifuging the overnight culture at 3000 g and 4 °C for 10 min. We resuspended the cells in 50 mL of LB (containing 50 μg/mL Kanamycin and 0.2% (w/v) arabinose) and continued the incubation at 37 °C with shaking at 250 rpm for another 12 h. We collected the cells by centrifuging the overnight culture at 3,000 g and 4 °C for 15 min, and washed the pelleted cells once with cold 2 × PBS. Then, we pelleted the cells again at 3,000 g and 4 °C for 15 min and resuspended them in 50 mL of 2 × PBS. We broke the cells to release the fluorescent proteins using a pressure cell cracker (UH-06, Union-biotech) and pelleted the insoluble fractions by centrifugation twice at 18,000×g for 10 min. We filtered the resulting supernatant with a 0.45 μM filter and used the high-affinity Ni-charged resin FF (L00666, GenScript) for purification, following the manufacturer’s protocol. We used PD-10 desalting columns (52-1308-00 BB, GE Healthcare) to desalt the purified proteins according to the manufacturer’s instructions, and concentrated the desalted fluorescent proteins using an Amicon Ultra-15 filter (UFC901008, Merck). We assessed the purity of the concentrated proteins using SDS-PAGE and quantified the concentration using a Bradford Protein Assay Kit (P0006, Beyotime).
We diluted the purified proteins to a concentration of 0.1–0.5 mg/mL using MiliQ water. We then transferred 200 μL of diluted proteins into a 1 mm thick cuvette and placed it in a Circular Dichroism spectropolarimeter (Chirascan V100, Applied Photophysics Ltd). We heated these samples from 50 to 90 °C at a rate of 1 °C per minute and measured their absorbance from 195 nm to 255 nm. We used the Global 3 V1.2 (Applied Photophysics Ltd) to calculate the melting temperature for each sample.
Cell-free expression of stabilizing mutants at different temperatures and determination of solubility by ELISA
To exclude the possibility that our observations at high temperature are caused by heat shock proteins, we performed new experiments, in which we expressed our proteins in a cell-free expression system (PURExpress In Vitro Protein Synthesis Kit, E6800S, NEB) at low, medium and high temperatures. This system contains only the components necessary for in vitro transcription and translation, all purified from E. coli, and completely avoids interference by heat shock proteins. Specifically, we replaced the DHFR gene in the PURExpress control DHFR plasmid with each of the genes of the full-length stabilizing mutants using a seamless cloning Kit (D7010M, Beyotime). We transformed the resulting plasmids into E. coli BW27783 by heat shock transformation and isolated the correctly constructed plasmids as templates for performing the in vitro protein synthesis reaction. The protein synthesis reaction contained 2 µL solution A, 1.5 µL solution B, 0.5 µL RNase inhibitor (8 U/µL; M0314S, NEB) and 1.0 µL template (50 ng/µL). The mixed solution was incubated in a PCR thermocycler tube at 25 °C and 44 °C for 8 h and 37 °C for 4 h. We then followed the manufacture’s protocol to quantify the amount of soluble fluorescent proteins in each solution by using a GFP ELISA kit (ab171581, Abcam), which can detect GFP from Aequorea Victoria and its enhanced and superfold variants.
Single-molecule real-time sequencing
As described in a previous study64, we barcoded GFP variants of each replicate population for SMRT (Pacific Biosciences, PacBio) sequencing by two-step PCRs. We used Q5 High-Fidelity DNA Polymerase (M0491L, NEB) to minimize the incidence of mutations during the PCR amplifications. Specifically, we first performed a 12-cycle PCR using primers tsmrtF/tsmrtR (Table S4) to amplify GFP variants for each replicate population. The PCR reaction contained 15.7 µL ddH2O, 6 µL 5×Q5 reaction buffer, 6 µL 5×Q5 high GC enhancer, 0.6 µL dNTPs (10 nM), 0.2 µL each primer (10 μM), 0.3 μL Q5 High-Fidelity DNA Polymerase, and 1 μL template (1 ng/μL). We performed the PCR with the following thermocycling program: 98 °C/30 s; 14 cycles of 98 °C/10 s, 60 °C/10 s, and 72 °C/25 s; 72 °C/2 min. We added 0.5 µL DpnI, 0.5 µL Exonuclease I (EN0581, Fermentas), as well as 0.5 µL ddH2O to the PCR product, and incubated the mixture at 37 °C for 1 h to digest the template plasmid and the primers. We then incubated the mixture at 80 °C for 20 min to inactivate the enzymes. Subsequently, we used 1 µL of PCR product as a template for a barcoding PCR in a 50 μL volume, which utilized a unique combination of a forward and a reverse barcode-tagged primers (Table S4) to barcode each replicate population. Each barcode-tagged primer has a unique 16-bp DNA sequence. The barcoding PCR mix contained 25.5 µL ddH2O, 10 µL 5×Q5 reaction buffer, 10 µL 5×Q5 high GC enhancer, 1 µL dNTPs (10 mM), 1 µL each primer (10 μM), 0.5 μL Q5 High-Fidelity DNA Polymerase, and 1 μL template. We executed the PCR reaction with the following program: 98 °C/30 s; 25 cycles of 98 °C/15 s, 68 °C/10 s and 72 °C/25 s; 72 °C/5 min. We purified the barcode-tagged PCR products by using a QIAquick PCR purification kit, and estimated their quality and concentration with agarose gel electrophoresis, a UV-Vis spectrophotometer (NanoDrop, Thermo Fisher Scientific), and a Qubit 4 Fluorometer (Invitrogen). We also used the ancestral GFP gene as a template for preparing barcode-tagged PCR products by following the same procedure to estimate the incidence of any errors that might have occurred during library preparation and sequencing. We pooled 100 ng DNA for each replicate population and for every generation in a single tube, and then gel-purified the mixture by using a QIAquick Gel Extraction Kit (Qiagen). We sent ~1 µg of the resulting amplicons to the Functional Genomics Center Zurich for sequencing with the PacBio Sequel II platform.
Primary data analysis
We used the SMRT Link V9.0.0.92188 software package (PacBio) to perform the primary sequencing data analysis. Specifically, we assembled consensus reads from single-stranded subreads by using the protocol “Circular Consensus Sequences (CCS)” and set the insert GFP length to 720–1200 bp, the full-pass subread number to ≥3, and the predicted consensus accuracy to ≥0.99. To demultiplex the resulting sequencing data by using the “Demultiplex Barcodes” application, we set the “Minimum Barcode Score” to 80 and the “Filter Minimum Barcode Quality” to 26. We used the ‘Mapping’ application to map the demultiplexed reads to the ancestral GFP sequence by setting the minimum mapped length to 700 bp and the minimum mapped concordance to 70%. We then selected the mapped reads that had an average Phred quality above 20, covered the entire GFP coding region, and didn’t have indels, which yielded an average of 2347 reads for each replicate population at each generation (Table S3). We used the resulting reads for all further analyses.
Identifying mutations and mutation combinations
Our analysis focused on SNPs (point mutations) because > 90% of sequencing errors during SMRT sequencing come from indels66, and because most indels render fluorescent proteins non-fluorescent. We treated a SNP as a true-positive only if the Phred quality score of a mismatch of a variant sequence to the GFP reference sequence was above 20. We wrote Python scripts (Python 3.10.4) to search point mutations, and their combinations and calculated their frequencies in each replicate population.
Statistical analyses
Unless specified otherwise, we performed one-way ANOVA with Dunett’s post hoc test for statistical data analysis by using R version 4.2.1.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data are available in the manuscript or the supplementary materials. All sequencing data is available at GenBank under accession number KIDR00000000. Source data are provided as a Source Data file. Source data are provided with this paper.
Code availability
Custom code used in this study is available at Zenodo under the accession number https://doi.org/10.5281/zenodo.10146677 [https://zenodo.org/records/10146677].
References
Arroyo, J. I., Díezc, B., Kempes, C. P., West, G. B. & Marquet, P. A. A general theory for temperature dependence in biology. Proc. Natl Acad. Sci. USA 119, e2119872119 (2022).
Nguyen, V. et al. Evolutionary drivers of thermoadaptation in enzyme catalysis. Science 355, 289–294 (2017).
Pinney, M. M. et al. Parallel molecular mechanisms for enzyme temperature adaptation. Science 371, eaay2784 (2021).
Scheffers, B. R. et al. The broad footprint of climate change from genes to biomes to people. Science 354, aaf7671 (2016).
Deatherage, D. E., Kepner, J. L., Bennett, A. F., Lenski, R. E. & Barrick, J. E. Specificity of genome evolution in experimental populations of Escherichia coli evolved at different temperatures. Proc. Natl Acad. Sci. USA 114, E1904–E1912 (2017).
Chen, J. Z., Fowler, D. M. & Tokuriki, N. Comprehensive exploration of the translocation, stability and substrate recognition requirements in vim-2 lactamase. Elife 9, 1–31 (2020).
Gillooly, J. F., Brown, J. H., West, G. B., Savage, V. M. & Charnov, E. L. Effects of size and temperature on metabolic rate. Science 293, 2248–2251 (2001).
Deutsch, C., Penn, J. L. & Seibel, B. Metabolic trait diversity shapes marine biogeography. Nature 585, 557–562 (2020).
Rubalcaba, J. G., Verberk, W. C. E. P., Jan Hendriks, A., Saris, B. & Arthur Woods, H. Oxygen limitation may affect the temperature and size dependence of metabolism in aquatic ectotherms. Proc. Natl Acad. Sci. USA 117, 31963–31968 (2020).
Allen, A. P., Brown, J. H. & Gillooly, J. F. Global biodiversity, biochemical kinetics, and the energetic-equivalence rule. Science 297, 1545–1548 (2002).
Antão, L. H. et al. Temperature-related biodiversity change across temperate marine and terrestrial systems. Nat. Ecol. Evol. 4, 927–933 (2020).
García, F. C., Bestion, E., Warfield, R. & Yvon-Durochera, G. Changes in temperature alter the relationship between biodiversity and ecosystem functioning. Proc. Natl Acad. Sci. USA 115, 10989–10994 (2018).
Yvon-Durocher, G. et al. Five years of experimental warming increases the biodiversity and productivity of phytoplankton. PLoS Biol. 13, e1002324 (2015).
Bennett, A. F., Dao, K. M. & Lenski, R. E. Rapid evolution in response to high-temperature selection. Nature 346, 79–81 (1990).
Toll-Riera, M., Olombrada, M., Castro-Giner, F. & Wagner, A. A limit on the evolutionary rescue of an Antarctic bacterium from rising temperatures. Sci. Adv. 8, 3511 (2022).
Leuenberger, P. et al. Cell-wide analysis of protein thermal unfolding reveals determinants of thermostability. Science 355, eaai7825 (2017).
Prentice, E. J. et al. The inflection point hypothesis: the relationship between the temperature dependence of enzyme-catalyzed reaction rates and microbial growth rates. Biochemistry 59, 3562–3569 (2020).
Li, G. et al. Bayesian genome scale modelling identifies thermal determinants of yeast metabolism. Nat. Commun. 12, 1–12 (2021).
Weiss, M. C. et al. The physiology and habitat of the last universal common ancestor. Nat. Microbiol. 1, 16116 (2016).
Povolotskaya, I. S. & Kondrashov, F. A. Sequence space and the ongoing expansion of the protein universe. Nature 465, 922–926 (2010).
Dallinger, W. H. The president’s address. J. R. Microsc. Soc. 7, 185–199 (1887).
Lindsey, H. A., Gallie, J., Taylor, S. & Kerr, B. Evolutionary rescue from extinction is contingent on a lower rate of environmental change. Nature 494, 463–467 (2013).
Arcus, V. L. & Mulholland, A. J. Temperature, dynamics, and enzyme-catalyzed reaction rates. Annu. Rev. Biophys. 49, 163–180 (2020).
MacDonald, P. J., Chen, Y. & Mueller, J. D. Chromophore maturation and fluorescence fluctuation spectroscopy of fluorescent proteins in a cell-free expression system. Anal. Biochem. 421, 291–298 (2012).
Åqvist, J., Isaksen, G. V. & Brandsdal, B. O. Computation of enzyme cold adaptation. Nat. Rev. Chem. 1, 1–14 (2017).
Saavedra, H. G., Wrabl, J. O., Anderson, J. A., Li, J. & Hilser, V. J. Dynamic allostery can drive cold adaptation in enzymes. Nature 558, 324–328 (2018).
Feller, G. & Gerday, C. Psychrophilic enzymes: hot topics in cold adaptation. Nat. Rev. Microbiol. 1, 200–208 (2003).
Angilletta, M. J., Huey, R. B. & Frazier, M. R. Thermodynamic effects on organismal performance: Is hotter better? Physiol. Biochem. Zool. 83, 197–206 (2010).
Tarkington, J. & Zufall, R. A. Temperature affects the repeatability of evolution in the microbial eukaryote Tetrahymena thermophila. Ecol. Evol. 11, 13139–13152 (2021).
Agozzino, L. & Dill, K. A. Protein evolution speed depends on its stability and abundance and on chaperone concentrations. Proc. Natl Acad. Sci. USA 115, 9092–9097 (2018).
Bloom, J. D., Wilke, C. O., Arnold, F. H. & Adami, C. Protein stability promotes evolvability. Proc. Natl Acad. Sci. USA 103, 5869–5874 (2006).
Gong, L. I., Suchard, M. A. & Bloom, J. D. Stability-mediated epistasis constrains the evolution of an influenza protein. Elife 2, 631 (2013).
Strobel, H. M., Horwitz, E. K. & Meyer, J. R. Viral protein instability enhances host-range evolvability. PLoS Genet. 18, e1010030 (2022).
Zheng, J., Payne, J. L. & Wagner, A. Cryptic genetic variation accelerates adaptive evolution by opening access to diverse adaptive peaks. Science 365, 347–353 (2019).
Conant, G. C. & Wolfe, K. H. Turning a hobby into a job: how duplicated genes find new functions. Nat. Rev. Genet. 9, 938–950 (2008).
Heim, R., Prasher, D. C. & Tsien, R. Y. Wavelength mutations and posttranslational autoxidation of green fluorescent protein. Proc. Natl Acad. Sci. USA 91, 12501–12504 (1994).
Ormö, M. et al. Crystal structure of the aequorea victoria green fluorescent. Protein Sci. 273, 1392–1395 (1996).
Delagrave, S., Hawtin, R. E., Silva, C. M., Youvan, D. C. & Yang, M. M. Red-shifted excitation mutants of the green fluorescent protein. Biotechnology 13, 151–154 (1995).
Petrie, K. L. et al. Destabilizing mutations encode nongenetic variation that drives evolutionary innovation. Science 359, 1542–1545 (2018).
Tokuriki, N. & Tawfik, D. S. Chaperonin overexpression promotes genetic variation and enzyme evolution. Nature 459, 668–673 (2009).
Trudeau, D. L. & Tawfik, D. S. Protein engineers turned evolutionists—the quest for the optimal starting point. Curr. Opin. Biotechnol. 60, 46–52 (2019).
Zheng, J., Guo, N. & Wagner, A. Selection enhances protein evolvability by increasing mutational robustness and foldability. Science 370, eabb5962 (2020).
Pédelacq, J. D., Cabantous, S., Tran, T., Terwilliger, T. C. & Waldo, G. S. Engineering and characterization of a superfolder green fluorescent protein. Nat. Biotechnol. 24, 79–88 (2005).
Rosano, G. L. & Ceccarelli, E. A. Rare codon content affects the solubility of recombinant proteins in a codon bias-adjusted Escherichia coli strain. Microb. Cell Fact. 8, 1–9 (2009).
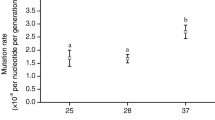
Kontopoulos, D.-G., Patmanidis, I., Barraclough, T. G. & Pawar, S. Higher temperatures worsen the effects of mutations on protein stability. bioRxiv https://doi.org/10.1101/2020.10.13.337972 (2020).
Siemering, K. R., Golbik, R., Sever, R. & Haseloff, J. Mutations that suppress the thermosensitivity of green fluorescent protein. Curr. Biol. 6, 1653–1663 (1996).
Sarkisyan, K. S. et al. Local fitness landscape of the green fluorescent protein. Nature 533, 397–401 (2016).
Geerts, A. N. et al. Rapid evolution of thermal tolerance in the water flea Daphnia. Nat. Clim. Chang. 5, 665–668 (2015).
Tokuriki, N. & Tawfik, D. S. Stability effects of mutations and protein evolvability. Curr. Opin. Struct. Biol. 19, 596–604 (2009).
Nisthal, A., Wang, C. Y., Ary, M. L. & Mayo, S. L. Protein stability engineering insights revealed by domain-wide comprehensive mutagenesis. Proc. Natl Acad. Sci. USA 116, 16367–16377 (2019).
Norrild, R. K. et al. Increasing protein stability by inferring substitution effects from high-throughput experiments. Cell Rep. Methods 2, 100333 (2022).
Tokuriki, N., Stricher, F., Serrano, L. & Tawfik, D. S. How protein stability and new functions trade off. PLoS Comput. Biol. 4, e1000002 (2008).
Lenski, R. E. & Bennett, A. F. Evolutionary response of Escherichia coli to thermal stress. Am. Nat. 142, S47–S64 (1993).
Rudolph, B., Gebendorfer, K. M., Buchner, J. & Winter, J. Evolution of Escherichia coli for growth at high temperatures. J. Biol. Chem. 285, 19029 (2010).
Lai, W.-Y. & Schlötterer, C. Evolution of gene expression variance during adaptation to high temperature in Drosophila. bioRxiv https://doi.org/10.1101/2021.01.19.427270 (2021).
Schou, M. F. et al. Evolutionary trade-offs between heat and cold tolerance limit responses to fluctuating climates. Sci. Adv. 8, 9580 (2022).
Morgan, R., Finnøen, M. H., Jensen, H., Pélabon, C. & Jutfelt, F. Low potential for evolutionary rescue from climate change in a tropical fish. Proc. Natl Acad. Sci. USA 117, 33365–33372 (2020).
Packer, M. S. & Liu, D. R. Methods for the directed evolution of proteins. Nat. Rev. Genet. 16, 379–394 (2015).
Molina, R. S. et al. In vivo hypermutation and continuous evolution. Nat. Rev. Methods Prim. 2, 1–22 (2022).
Bradshaw, W. E. & Holzapfel, C. M. Evolutionary response to rapid climate change. Science 312, 1477–1478 (2006).
Smale, D. A. et al. Marine heatwaves threaten global biodiversity and the provision of ecosystem services. Nat. Clim. Chang. 9, 306–312 (2019).
Laufkötter, C., Zscheischler, J. & Frölicher, T. L. High-impact marine heatwaves attributable to human-induced global warming. Science 369, 1621–1625 (2020).
Frölicher, T. L., Fischer, E. M. & Gruber, N. Marine heatwaves under global warming. Nature 560, 360–364 (2018).
Bratulic, S., Gerber, F. & Wagner, A. Mistranslation drives the evolution of robustness in TEM-1 β-lactamase. Proc. Natl Acad. Sci. USA 112, 12758–12763 (2015).
Reid, B. G. & Flynn, G. C. Chromophore formation in green fluorescent protein. Biochemistry 36, 6786–6791 (1997).
Kim, K. E. et al. Long-read, whole-genome shotgun sequence data for five model organisms. Sci. Data https://doi.org/10.1038/sdata.2014.45 (2014).
Acknowledgements
We acknowledge the experimental supports of the flow cytometry core facility and the instrumentation and service center for molecular sciences at Westlake University, and the flow cytometry facility and the functional genomics center at the University of Zurich. We thank Prof. Ren Sun and Dr. Boxiao Wang for helpful discussions and suggestions. We would like to acknowledge support by Westlake Education Foundation (J.Z.), the URPP Evolution in Action (J.Z.), the National Natural Science Foundation of China grant 32270669 (J.Z.), the European Research Council under grant agreement 739874 (A.W.), and the Swiss National Science Foundation grant 31003A_172887 (A.W.).
Author information
Authors and Affiliations
Contributions
J.Z. designed the experiments. J.Z., N.G., Y.H. and X.G. performed the experiments. N.G., J.Z. and A.W. contributed to data analysis. J.Z. and A.W. wrote the paper. All authors read and edited the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zheng, J., Guo, N., Huang, Y. et al. High temperature delays and low temperature accelerates evolution of a new protein phenotype. Nat Commun 15, 2495 (2024). https://doi.org/10.1038/s41467-024-46332-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-024-46332-6
- Springer Nature Limited