Abstract
Following the survival benefit demonstrated in the OlympiA trial, one year of adjuvant olaparib is now recommended for all patients with germline BRCA1/2 pathogenic/likely pathogenic variants (PV) and high-risk, HER2-negative early breast cancer after chemotherapy. However, optimal identification of high-risk patients who may derive benefit from this genomically-directed therapy is debated. In this study, we sought to characterize the real-world proportion of gBRCA1/2 PV carriers eligible for adjuvant olaparib according to the OlympiA criteria, and to compare clinicopathologic characteristics and outcomes between eligible and ineligible patients.
Similar content being viewed by others
Approximately 5% of breast cancers occur in patients who carry a germline BRCA1/2 (gBRCA1/2) pathogenic or likely pathogenic variant (PV)1,2. In the OlympiA trial one year of adjuvant olaparib improved invasive disease-free survival (iDFS) and overall survival (OS) in gBRCA1/2 PV carriers with high-risk, HER2-negative early breast cancer3,4, and olaparib became the first systemic adjuvant therapy specifically approved for these patients. Eligibility criteria of OlympiA differed for triple-negative breast cancer (TNBC) and hormone receptor-positive (HR+) tumors. Patients with TNBC were eligible either when residual disease was present after neoadjuvant chemotherapy, or after upfront surgery and adjuvant chemotherapy for tumors ≥2 cm or with nodal involvement. Patients with HR+ tumors were eligible either if there was residual disease and a clinical and pathologic stage (CPS) and estrogen receptor status and histologic grade (EG) (CPS + EG) score ≥3 after neoadjuvant chemotherapy, or if ≥4 nodes were involved at surgery prior to adjuvant chemotherapy.
Whether these criteria identify all gBRCA PV carriers with high-risk breast tumors who may benefit from olaparib is debated. This is particularly relevant for HR+ breast cancers, as alternative trials used different criteria to select high-risk patients. In monarchE, for instance, patients were eligible either if they had ≥4 positive nodes at surgery, or 1-3 positive nodes and at least one additional high-risk criterion among grade 3 disease, tumor size > 5 cm, or Ki67 ≥ 20%. In this study, we used a prospectively maintained single institution database to characterize the real-world proportion of gBRCA1/2 PV carriers with early breast cancer who meet OlympiA inclusion criteria, and compared clinicopathologic characteristics and outcomes between eligible and ineligible patients. Additionally, we investigated the overlap between criteria in OlympiA and monarchE in an effort to identify additional high-risk patients who might benefit from novel targeted therapies in the adjuvant setting.
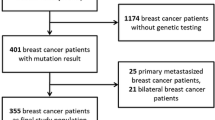
We identified 205 gBRCA1/2 PV carriers with newly diagnosed, HER2-negative early breast cancer, including 113 with HR+ and 92 with TNBC (Fig. 1, Supplementary Table 1). Of them, 15 had synchronous primaries for which only the highest risk tumor was considered in the analysis. Median age at diagnosis was 43 years and most patients (n = 129, 62.9%) were premenopausal. A gBRCA1 PV was identified in 115 (56.1%) patients and a gBRCA2 PV in 90 (43.9%). Overall, 73 (35.6%) patients underwent genetic testing before the diagnosis of breast cancer. A total of 166 (81.0%) patients received chemotherapy; of them, 130 (78.3%) received an anthracycline-containing regimen and 32 (19.3%) received platinum. Neoadjuvant chemotherapy was administered to 107 patients (77 gBRCA1, 30 gBRCA2), of whom 47 (40 gBRCA1, 7 gBRCA2) achieved a pCR (43.9%). Only 8 (3.9%) patients received immunotherapy. Eleven patients received (neo)adjuvant PARP inhibitors, and 4 participated in the OlympiA trial.
Overall, 60 (29.3%) patients were eligible for adjuvant olaparib according to OlympiA criteria (39 gBRCA1 and 21 gBRCA2 PV carriers), including 40 (66.7%) with TNBC and 20 (33.3%) with HR+ breast cancer (Table 1; Fig. 2).
a iDFS for patients eligible and ineligible for adjuvant olaparib; b RFS for patients eligible and ineligible for adjuvant olaparib; c Proportion of eligible and ineligible patients among gBRCA PV carriers with TNBC; d Proportion of eligible and ineligible patients among gBRCA PV carriers with HR+/HER2− breast cancer. iDFS invasive disease-free survival, RFS recurrence-free survival, HER2− human epidermal growth factor receptor 2 negative, HR+ hormone receptor positive, PV pathogenic/likely pathogenic, TNBC triple-negative breast cancer.
Major reasons why most patients identified with gBRCA1/2 PV and breast cancer were deemed ineligible for adjuvant olaparib were lack of prior chemotherapy (n = 9), low anatomic stage at upfront surgery (n = 16 with stage I TNBC; n = 63 with HR+ tumors and <4+ nodes), or evidence of substantial response to neoadjuvant chemotherapy (n = 36 TNBC had pCR; n = 21 with HR+ disease had a pCR or a CPS&EG score <3).
Eligible patients were more likely to have higher grade and higher stage tumors (p < 0.001) compared to ineligible patients. Consistent with the higher stage at presentation, eligible patients were less frequently diagnosed through screening imaging (23.3 vs 42.8%, p = 0.026) and had genetic testing performed more often after diagnosis (80 vs 57.9%, p = 0.004). Chemotherapy regimens administered to eligible patients more frequently included anthracyclines (p = 0.002) and/or platinum salts (p = 0.005). Among patients with HR+ breast cancer, only 2 out of 20 (10%) patients eligible for adjuvant olaparib had recurrence scores (RS) assessed, compared to 48 out of 93 (51.6%) ineligible patients (Table 1).
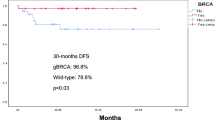
After a median follow up for disease status of 31 months (IQR 16.53 months), 20 iDFS and 13 recurrence-free survival (RFS) events were recorded (Table 2). Three-year iDFS was 84.2% (95% CI, 74.6–95.0%) for eligible and 90.0% (95% CI, 84.1–96.3%) for ineligible patients (hazard ratio 1.54 [95% CI, 0.63–3.78], p = 0.34). Three-year RFS was 86.1% (95% CI, 77.0–96.3%) for eligible and 94.2% (95% CI, 89.3–99.4%) for ineligible patients (hazard ratio 2.38 [95% CI, 0.83–6.80], p = 0.11) (Fig. 2).
Similar results were observed when comparing eligible versus ineligible patients among gBRCA1 and gBRCA2 PVs separately, and among patients with HR+ disease (Table 3). Interestingly, among patients with TNBC, eligible patients had substantially worse outcomes compared to those who were ineligible, with a 12.8% and 12.1% 3-year iDFS and RFS absolute difference, respectively, although with a small number of events and large confidence intervals. Overall, outcomes appeared to be worse for gBRCA2-associated breast tumors compared to gBRCA1, and for HR+ tumors compared to TNBC (Table 3).
Of the 113 patients with HR+ breast cancer, 16 (14%) were eligible for both adjuvant olaparib and abemaciclib (2 received abemaciclib, 1 received both agents, 1 enrolled in OlympiA), 4 (3%) patients were eligible for olaparib only (1 received olaparib and 1 enrolled in OlympiA) and 18 (8.8%) for abemaciclib only (3 received abemaciclib). Using monarchE criteria, we identified 18 (8.8%) additional patients who may be considered for treatment escalation who were not identified by OlympiA criteria.
In this study, we analyzed a large cohort of gBRCA1/2 PV carriers to assess the proportion of patients in a real-world setting deemed eligible for adjuvant olaparib according to OlympiA criteria. Among our patients, approximately 30% met OlympiA criteria. When comparing clinicopathologic characteristics between eligible and ineligible patients, we observed that eligible patients were less frequently diagnosed following imaging-based screening and more often underwent genetic testing after diagnosis. Although this is expected given OlympiA’s requirements in terms of tumor stage at diagnosis, it also underlines that earlier recognition of gBRCA1/2 PV carriers with associated recommended breast imaging monitoring could allow identification of smaller tumors with higher chance of cure following primary therapy.
We observed a large number of RFS events among ineligible patients although, as expected, there was a non-significant trend towards worse outcomes for eligible compared to ineligible patients. Similar iDFS and RFS differences were observed among eligible and ineligible patients when gBRCA1 PV carriers, gBRCA2 PV carriers, and patients with HR+ or TNBC tumors were analyzed separately. Interestingly, the gap between eligible and ineligible patients appeared larger for TNBC, which may suggest that the eligibility criteria employed in OlympiA were better at discriminating between high- and low-risk for TNBC than for HR+ disease. Of note, when comparing monarchE and OlympiA inclusion criteria, we observed that almost half of high-risk patients defined by monarchE eligibility criteria did not meet OlympiA criteria.
This present analysis has limitations inherent to the nature of this study. First, we used real-world data, and survival measures may be biased by how patients were monitored over time. Second, the small sample size and relatively low number of events limits the power of our analyses, in particular for subgroup analyses. Third, we did not consider multifocal disease, which is common among gBRCA1/2 PV carriers and does impact risk of recurrence. Fourth, our median follow up of 31 months is relatively short, especially given the known long-term risk of relapse of HR+ breast tumors. Fifth, 35.6% of patients underwent genetic testing before receiving the diagnosis of breast cancer, which may have favored the detection of lower stage tumors and reduced the proportion of patients eligible for adjuvant olaparib.
To date, olaparib is recommended for all patients with gBRCA1/2 PV and HER2-negative breast cancer considered at high-risk of recurrence, although the definition of “high-risk” remains unclear5. It is worth noticing that both the U.S. Food and Drug Administration6 and the European Medical Agency7 approved adjuvant olaparib for gBRCA PV carriers with high-risk, early-stage, HER2-negative breast cancer, without considering the specifics of OlympiA criteria. Here, in a real world-cohort of patients with gBRCA PV, we described that only 30% met the OlympiA eligibility criteria, and that ineligible patients were still at high risk of recurrence, especially those with HR+ tumors. Therefore, the proportion of gBRCA PV carriers who may benefit from adjuvant olaparib is likely to extend beyond OlympiA criteria. This choice allowed the study to report results sooner, but likely excluded lower risk patients who may benefit from this approach. However, exactly how broad eligibility should be is unknown, and further research is needed to understand whether low risk patients could also benefit from adjuvant PARP inhibitors. In this setting, whether these agents could replace chemotherapy is worth of further investigation.
Methods
Study design
Clinicopathologic and genetic data from all consecutive patients with gBRCA1/2 PV who underwent surgery between 1/4/2016 and 4/7/2022 for a first diagnosis of HER2-negative invasive breast cancer at Dana-Farber Brigham Cancer Center between 2016 and 2022 were extracted from prospectively collected institutional datasets. HR+ disease was defined according to ASCO/CAP guidelines (i.e., estrogen and/or progesterone receptor ≥1%). Patients with prior invasive breast cancer were excluded, whereas patients with prior invasive non-breast cancer were included in the analysis. For patients with synchronous breast tumors, the highest risk tumor was considered. Eligibility for adjuvant olaparib was defined according to the OlympiA study inclusion criteria4. This study was conducted in accordance with the Declaration of Helsinki. The Dana-Farber/Harvard Cancer Center Institutional Review Board classified this study as exempt from IRB approval and included a waiver of informed consent in accordance with the U.S Common Rule.
Statistical analysis
Clinicopathological characteristics and treatment patterns were compared between eligible and ineligible patients using Fisher’s exact test, Chi-squared test, or Wilcoxon rank-sum test, as appropriate. iDFS and RFS were defined according to STEEP 2.0 criteria8 and calculated using the Kaplan-Meier method with the log-rank test to compare between eligible and ineligible patients. As exploratory analyses, the proportion of gBRCA PV carriers with HR+ breast cancer deemed as high-risk by the monarchE criteria (1–3 positive nodes and grade 3 or tumor size >5 cm)9 and the overlap between eligibility to OlympiA and monarchE were assessed. All statistical tests were two-sided with P values ≤ 0.05 considered statistically significant.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
All analyses were performed with R version 4.0.3. R codes are available from the corresponding author on reasonable request.
References
Breast Cancer Association Consortium. et al. Breast cancer risk genes - association analysis in more than 113,000 women. N. Engl. J. Med. 384, 428–439 (2021).
Hu, C. et al. A population-based study of genes previously implicated in breast cancer. N. Engl. J. Med. 384, 440–451 (2021).
Geyer, C. E. Jr et al. Overall survival in the OlympiA phase III trial of adjuvant olaparib in patients with germline pathogenic variants in BRCA1/2 and high risk, early breast cancer. Ann. Oncol. 33, 1250–1268, (2022).
Tutt, A. N. J. et al. Adjuvant Olaparib for Patients with BRCA1- or BRCA2-Mutated Breast Cancer. N. Engl. J. Med. 384, 2394–2405 (2021).
Morganti, S. et al. Adjuvant olaparib for germline BRCA carriers with HER2-negative early breast cancer: Evidence and controversies. Oncologist 28, 565–574, (2023).
Center for Drug Evaluation & Research. FDA approves olaparib for adjuvant treatment of high-risk early breast cancer. U.S. Food and Drug Administration https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-olaparib-adjuvant-treatment-high-risk-early-breast-cancer (2022).
EMA. Lynparza. European Medicines Agency https://www.ema.europa.eu/en/medicines/human/summaries-opinion/lynparza-1 (2022).
Tolaney, S. M. et al. Updated Standardized Definitions for Efficacy End Points (STEEP) in adjuvant breast cancer clinical trials: STEEP version 2.0. J. Clin. Oncol. 39, 2720–2731 (2021).
Johnston, S. R. D. et al. Abemaciclib plus endocrine therapy for hormone receptor-positive, HER2-negative, node-positive, high-risk early breast cancer (monarchE): results from a preplanned interim analysis of a randomised, open-label, phase 3 trial. Lancet Oncol. https://doi.org/10.1016/S1470-2045(22)00694-5 (2022).
Acknowledgements
S.M. acknowledges the support from the Italian Association for Cancer Research (AIRC) and Gianni Bonadonna Foundation. F.L. acknowledges the support of the Friends of Dana-Farber Cancer Institute. The authors acknowledge Kaitlyn T. Bifolck, a full-time employee of Dana-Farber Cancer Institute, for editorial and submission assistance in the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization: S.M., S.M.T., F.L. Data curation: S.M., Q.J., J.V., R.B., S.R., S.S., T.P. Formal Analysis: S.M., Q.J., N.T. Funding acquisition: S.M., J.E.G., T.A.K., E.A.M., F.L. Investigation: S.M., S.M.T., F.L. Methodology: S.M., Q.J., N.T., F.L. Project administration: F.L. Resources: S.M.T., J.E.G., T.A.K., E.A.M., F.L. Supervision: S.M.T., J.E.G., N.U.L., F.L. Validation: S.M., Q.J., N.T., F.L. Visualization: S.M., Q.J., F.L. Writing – original draft: S.M., F.L. Writing – review & editing: All authors.
Corresponding author
Ethics declarations
Competing interests
E.A.M. reports compensated service on scientific advisory boards for Astra Zeneca, BioNTech and Merck; uncompensated service on steering committees for Bristol Myers Squibb and Roche/Genentech; speakers’ honoraria and travel support from Merck Sharp & Dohme; and institutional research support from Roche/Genentech (via SU2C grant) and Gilead. EAM also reports research funding from Susan Komen for the Cure for which she serves as a Scientific Advisor, and uncompensated participation as a member of the American Society of Clinical Oncology Board of Directors. T.A.K. reports speaker honoraria and compensated service on scientific Advisory Board of Exact Sciences. A.W. reports compensated service on scientific advisory boards for Merck and Myriad, and institutional research funding from Myriad. G.C. reports consulting fees from Seagen, Roche, Novartis, Lilly, Daiichi Sankyo, Astra Zeneca, Pfizer, Sanofi, Pierre Fabre, and Gilead and fees for non-CME services (e.g., speakers’ bureaus) from Lilly, Pfizer, and Daiichi Sankyo. N.T. reports research funding and consulting from AstraZeneca. N.U.L. reports institutional research support from Genentech (and Zion Pharmaceutical as part of GNE), Pfizer, Merck, Seattle Genetics (now Pfizer), Olema Pharmaceuticals, and AstraZeneca; consulting honoraria from Puma, Seattle Genetics, Daiichi-Sankyo, AstraZeneca, Olema Pharmaceuticals, Janssen, Blueprint Medicines, Stemline/Menarini, Artera Inc., and Eisai; royalties from UpToDate; and travel support from Olema Pharmaceuticals. J.E.G. reports research funding from Invitae Genetics, Ambry Genetics and Myriad Genetics. S.M.T. reports consulting/advisory role Novartis, Pfizer, Merck, Eli Lilly, AstraZeneca, Genentech/Roche, Eisai, Sanofi, Bristol Myers Squibb, Seattle Genetics, CytomX Therapeutics, Daiichi Sankyo, Gilead, OncXerna, Zymeworks, Zentalis, Blueprint Medicines, Reveal Genomics, ARC Therapeutics, Infinity Therapeutics, Sumitovant Biopharma, Umoja Biopharma, Artios Pharma, Menarini/Stemline, Aadi Bio, Bayer, Incyte Corp, Jazz Pharmaceuticals, Natera, Tango Therapeutics, Systimmune, eFFECTOR, Hengrui USA; research funding from Genentech/Roche, Merck, Exelixis, Pfizer, Lilly, Novartis, Bristol Myers Squibb, Eisai, AstraZeneca, Gilead, NanoString Technologies, Seattle Genetics, and OncoPep; and travel support from Eli Lilly, Sanofi, and Gilead. S.M. reports support from AstraZeneca and Menarini/Stemline. F.L. reports consulting/advisory role for AstraZeneca, Pfizer, Merck and Daiichi Sankyo; and institutional research funding from Eisai, Incyte, Genentech, AstraZeneca, CytomX and Gilead Sciences. All other authors report no conflicts of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Morganti, S., Jin, Q., Vincuilla, J. et al. Clinicopathological characteristics and eligibility for adjuvant olaparib of germline BRCA1/2 mutation carriers with HER2-negative early breast cancer. npj Breast Cancer 10, 28 (2024). https://doi.org/10.1038/s41523-024-00632-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41523-024-00632-8
- Springer Nature Limited