Abstract
Progress in mechanobiology allowed us to better understand the important role of mechanical forces in the regulation of biological processes. Space research in the field of life sciences clearly showed that gravity plays a crucial role in biological processes. The space environment offers the unique opportunity to carry out experiments without gravity, helping us not only to understand the effects of gravitational alterations on biological systems but also the mechanisms underlying mechanoperception and cell/tissue response to mechanical and gravitational stresses. Despite the progress made so far, for future space exploration programs it is necessary to increase our knowledge on the mechanotransduction processes as well as on the molecular mechanisms underlying microgravity-induced cell and tissue alterations. This white paper reports the suggestions and recommendations of the SciSpacE Science Community for the elaboration of the section of the European Space Agency roadmap “Biology in Space and Analogue Environments” focusing on “How are cells and tissues influenced by gravity and what are the gravity perception mechanisms?” The knowledge gaps that prevent the Science Community from fully answering this question and the activities proposed to fill them are discussed.
Similar content being viewed by others
Introduction
It is well documented that cell behavior is also regulated by mechanical cues the cells receive from their microenvironment1,2. Cells perceive changes in the mechanical properties of their environment and alterations in gravity. This perception leads to the elaboration of a biological response which can affect processes at the cell and tissue level. Hence, mechanical and gravitational forces can modulate several functions such as proliferation3, differentiation4, apoptosis5,6,7, gene expression8, signaling9, and adhesion/migration properties10,11, thus affecting the organization and functioning of 3D structures formed by cells (constructs, tissues, and organs)12. The resulting effects might underlie the pathophysiological changes observed at the whole organism level during spaceflight. Therefore, understanding the mechanotransduction processes and the resulting changes, not only in cell functions but also in the extracellular environment (e.g. cell-to-cell communication, extracellular matrix (ECM) production, ECM properties, etc.), is a necessary condition to increase our knowledge on the spaceflight adaptation processes, focusing on mechanobiology.
Over the past two decades, many studies have revealed the crucial role that mechanical stressors of the cell microenvironment play in the control of cell morphology and volume, cell cycle and growth. These studies produced significant progress in the field of mechanobiology. For example, it has been revealed that the stiffness of the microenvironment has an important role in regulating cell differentiation/dedifferentiation13, adhesion/migration14, and also the onset and progression of serious diseases, such as cancer15 and fibrosis16,17. Other studies have demonstrated that the process by which cells translate mechanical stimuli into biochemical signals involves cytoskeleton18, integrins19, ECM molecules20, ion channels21,22, guanosine triphosphate hydrolase enzymes (GTPases)23, and mitogen-activated protein kinases (MAPKs)24. However, the exact mechanisms that underlie how cells perceive mechanical stimuli through the diverse mechanosensitive molecules are still not fully understood. Research carried out in altered gravity conditions represents an extraordinary opportunity to investigate the impact of gravity and mechanical forces on biological processes as well as the response of cells and tissues to mechanical and gravitational stimuli.
This white paper reports the suggestions and recommendations of the SciSpacE Science Community (SSC) for answering the question “How are cells and tissues influenced by gravity and what are the gravity perception mechanisms?”, which is one of the major issues of the European Space Agency (ESA) roadmap “Biology in Space and analogue environments”. Key knowledge gaps are indicated and activities to fill them are discussed.
Key knowledge gaps
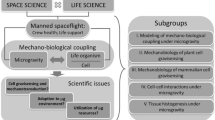
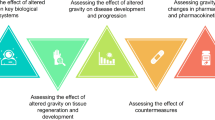
With the avenue of the Artemis program that intends to construct a human-operated Moon base camp in preparation for human space exploration missions of greater distance and duration, five key issues were determined to be of great importance to better understand how gravity affects cell and tissue structure and what are the gravity perception mechanisms (Fig. 1):
-
1.
Identifying changes in the mechanical properties of single cells, tissue models, and organisms in response to gravity alterations.
-
2.
Assessing gravity-induced mechanical and functional alterations in complex 3D cell/tissue models.
-
3.
Evaluating the effects of altered gravity on epigenetics, genetic protection, and repair mechanisms.
-
4.
Investigating the stress response induced by altered gravity conditions and possible countermeasures.
-
5.
Understanding how gravity alterations affect other space-related physiological responses.
In addition, a particular emphasis on the use of complex 3D models was given as these models are crucial to bridge the knowledge of biological processes at the molecular, cellular, and tissue level with human research. In particular, these 3D models are a valuable tool in the efforts to better understand: (i) organ and tissue mechanical properties; (ii) the role of gravity in modulating organ morphology and function; (iii) mechanisms of adaptation to altered gravity, consequent long-term alterations, and possible countermeasures25,26. In parallel, cell-based 2D models can also support the elucidation of this understanding.
Proposed research activities to answer the key knowledge gaps
A fundamental question in space physiology, space medicine, and space biology is how cells adapt to gravity changes. All species that have evolved on Earth possess the structure and function that is adjusted to their living environment, thus to Earth’s gravity. Yet, during spaceflights, astronauts are exposed to microgravity, partial gravity (on the Moon and Mars), and hypergravity. Understanding the mechano-biological coupling mechanisms through which gravity regulates biological responses at molecular, sub-cellular, cellular, tissue, and organism levels is therefore vital. With the aim to reproduce fundamental cell-cell contacts, intra- and inter-cellular signaling, elucidating the underlying mechanisms of how a cell detects, transmits, transduces, and reacts to modified gravitational impulses would aid in identifying changes in the mechanical properties of single cells, tissue models and organisms in response to gravitational alterations.
While, at this stage, it is impossible to recreate the complete physiological complexity of an organ, 3D models (including spheroids, organoids, organ-on-chip and ex-vivo tissue explants from the skin, cornea, retina, blood vessel, muscle tissues, brain, or spinal cord) make it possible to explore tissue-like responses to gravity alterations at the level of morphology, structure, gene/protein expression, cell function, and physiology27,28,29,30. Therefore, implementing 3D model systems enables the assessment of gravity-induced mechanical and functional alterations at a more complex and higher organizational level. In particular, the understanding of the effects of changes in the transport coefficient of culture fluids on the growth and development of tissues and 3D constructs31,32. These 3D models can be used to evaluate or develop new therapeutic treatments. Changes in mitochondrial activity and energy metabolism in response to gravity alterations are also important to investigate. In addition, the comparison between 2D and 3D models would be useful to understand how gravity affects intercellular communication and exchange (proteins, lipids, genetic material, role of extracellular vesicles). Further research is needed about the influence of gravity on the regulation of cell cycle and cell death (in particular cell senescence and apoptosis). Furthermore, studying nanomaterial interactions with biological systems (2D and 3D) in altered gravity conditions at any level (molecular, transcriptional, translational, and phenotypic amongst others) may offer opportunities for developing effective countermeasures (including medications carrying physicochemical cues) against the deleterious effects of the space environment.
Another important aspect to consider when studying the biological effects of microgravity is its interplay with space radiation that poses a constant threat to the DNA integrity of astronauts33,34. Cells have developed sophisticated systems that can find and repair DNA lesions as a defense mechanism against the effects of DNA damage. These mechanisms are comprised of a network of cellular proteins that are active participants in the DNA damage response (DDR) pathways. Some examples of these pathways include cell cycle regulation, DNA repair, and apoptosis. In general, cells are able to compensate for moderate DNA damage through these many repair processes. However, the circumstances of space, particularly gravity alterations, may have a negative impact on the DDR pathways, leading to a loss of genomic integrity. Hence, evaluating the effects of altered gravity in combination with space radiation on epigenetics, genetic protection, and repair mechanisms remains crucial.
Furthermore, cells respond to stress through the activation of processes that support cellular functioning and, as a result, the maintenance of micro-environmental and organismal homeostasis35. Microgravity is a stressor that disrupts cell homeostasis, which was especially observed in the musculoskeletal, cardiopulmonary, and central nervous system36,37,38. Yet, little is known about the signals that emerge from altered gravity-stressed cells in order to provide a coordinated adaptive response across tissues, organs, and the entire organism. Investigating the stress response induced by altered gravity (from microgravity to hypergravity) remains important to establish proper countermeasures. Therefore, well-designed ground-based research programs that investigate the influence of gravity should accompany in-flight research in order to favor a deeper comprehension of the findings from the in-flight research. Finally, the subcellular and cellular mechanisms related to the adaptation responses (intra- and extra-cellular) to gravity alterations from 2D and 3D models should be the basis for designing in-flight studies to understand space-related physiological changes at the tissue and/or whole organism level. These studies should be conducted in order to gain a better understanding of how space affects the body as a whole.
All the available platforms (International Space Station (ISS), parabolic flights, centrifuges, on-ground systems for modeling micro- or hypogravity, etc.) can be profitably used to test responses of in vitro, ex vivo, and in vivo models to gravitational alterations and the mechanisms of gravity perception. The use of on-ground facilities is helpful to increase the number of tests and better prepare the in-flight experiments. Finally, it is very important to have access to facilities that simultaneously model the different space stressors (for example, microgravity and radiation), to study the resulting combination of effects.
Early outcomes of the activities above described can be obtained in a relatively short time (about 3 years), the implementation of most parts of the activities requires from medium to long time (6–10 years).
Priorities for the ESA Space Exploration Program
To prevent the onset of diseases, to prepare effective countermeasures, and to develop tools for diagnostics and health monitoring, it is crucial to understand the molecular and cellular mechanisms underlying the processes of adaptation to spaceflight, the resulting pathophysiological changes, and the associated health risks39,40,41. Therefore, the above-indicated activities can be regarded as fundamental support to the future Programs of Space Exploration which concern the stay of humans in space. Specific recommendations for these activities are presented in Table 1.
Benefit for Earth and industrial relevance
The conditions encountered during spaceflights, especially microgravity, offer a unique opportunity to eliminate the stress caused by mechanical forces from a living system over the long-term, regardless of whether the system in question is a cell, a tissue, or an entire organism. Hence, such an environment provides optimal research conditions to measure the impact of gravity and mechanical forces on biological processes and living systems42. By taking advantage of this one-of-a-kind research environment, we have the chance to advance our fundamental understanding of the cellular mechanisms that are involved in gravity or mechano-sensing, as well as the response that is induced by them. Beside supporting space exploration missions, these findings have the potential to find applications on Earth in many different fields, such as general medicine, rehabilitation, and training, just to mention some examples. Indeed, in the biomedical field, the knowledge gained through space research can provide huge translational value for understanding disease conditions, provide novel therapeutic targets, and, consequently, new therapeutic strategies as well as personalized treatments43. Further information on the role of gravitational/mechanical stress in biological processes can bring progress not only in the biomedicine area, but also in the fields of physiatry, rehabilitation, sports medicine, and training. The advancement of technology is one of the key benefits of space exploration. For example, remote sensing technologies could be useful in the study of the epidemiology of infectious diseases. Data may be used to monitor disease patterns, understand environmental triggers for the spread of diseases, predict risk areas, and define regions that require disease-control planning44,45. This is particularly important in developing countries, where infectious diseases remain among the top causes of death. Space exploration also offers the potential for new discoveries in pharmacology. Drugs widely used on Earth, such as bisphosphonates, have been tested in Space46. Moreover, during spaceflight, humans live in conditions of isolation and confinement, in an environment where air, water, food, and waste are recycled. Hence, the risk of pathogen development may increase. This condition, associated with alterations in astronauts’ microbial flora and possible decrease in drug susceptibility, due to possible changes in physical–chemical properties of drugs as well as to changes in drug pharmacodynamics and pharmacokinetics, makes Space an excellent environmental model to study and develop new drugs47,48.
Finally, the findings obtained from space exploration missions can be directly used in the R&D process for several industries including manufacturing, materials, cosmetics, and many more.
Discussion
From the perspective of future long-duration missions in/beyond the Low Earth Orbit (LEO), it is imperative to expand our knowledge in the fields of space biology and physiology. In this chapter of the SSC white paper, we indicated the activities needed to improve our understanding of how gravity influences biological processes as well as cell and tissue structure and function in biological systems. A very important topic is the investigation of the gravity perception mechanisms, which are still far from clear and whose comprehension is crucial to identify the pathways that transduce the gravitational alterations in biological response. Table 1 summarizes the main questions still unresolved, the activities aimed at answering these scientific problems, the platforms and timelines necessary to conduct the above research, and the repercussions that the results could have both on future space exploration programs and in the biotechnological and biomedical fields on Earth.
Reporting summary
Further information on research design is available in the Nature Research Reporting Summary linked to this article.
References
Bradbury, P. et al. Modeling the impact of microgravity at the cellular level: implications for human disease. Front. Cell Dev. Biol. 8, 96 (2020).
Vandenburgh, H. H. Mechanical forces and their second messengers in stimulating cell growth in vitro. Am. J. Physiol. 262, R350–R355 (1992).
Unsworth, B. R. & Lelkes, P. I. Growing tissues in microgravity. Nat. Med. 4, 901–907 (1998).
Imura, T., Otsuka, T., Kawahara, Y. & Yuge, L. “Microgravity” as a unique and useful stem cell culture environment for cell-based therapy. Regen. Ther. 12, 2–5 (2019).
Morbidelli, L. et al. Simulated hypogravity impairs the angiogenic response of endothelium by up-regulating apoptotic signals. Biochem. Biophys. Res. Commun. 334, 491–499 (2005).
Monici, M. et al. Modeled gravitational unloading triggers differentiation and apoptosis in preosteoclastic cells. J. Cell. Biochem. 98, 65–80 (2006).
Riwaldt, S. et al. Role of apoptosis in wound healing and apoptosis alterations in microgravity. Front. Bioeng. Biotechnol. 9, 679650 (2021).
Panciera, T., Azzolin, L., Cordenonsi, M. & Piccolo, S. Mechanobiology of YAP and TAZ in physiology and disease. Nat. Rev. Mol. Cell Biol. 18, 758–770 (2017).
Siamwala, J. H., Rajendran, S. & Chatterjee, S. Strategies of manipulating BMP signaling in microgravity to prevent bone loss. Vitam. Horm. 99, 249–272 (2015).
Maier, J. A., Cialdai, F., Monici, M. & Morbidelli, L. The impact of microgravity and hypergravity on endothelial cells. Biomed. Res. Int. 2015, 434803 (2015).
Cialdai, F. et al. Modeled microgravity affects fibroblast functions related to wound healing. Microgravity Sci. Technol. 29, 121–132 (2017).
Zhang, C., Li, L., Chen, J. & Wang, J. Behavior of stem cells under outer-space microgravity and ground-based microgravity simulation. Cell Biol. Int. 39, 647–656 (2015).
Humphrey, J. D., Dufresne, E. R. & Schwartz, M. A. Mechanotransduction and extracellular matrix homeostasis. Nat. Rev. Mol. Cell Biol. 15, 802–812 (2014).
Doyle, A. D. & Yamada, K. M. Mechanosensing via cell-matrix adhesions in 3D microenvironments. Exp. Cell Res. 343, 60–66 (2016).
Grimm, D. et al. The fight against cancer by microgravity: the multicellular spheroid as a metastasis model. Int. J. Mol. Sci. 23, 3073 (2022).
Piersma, B., Hayward, M. K. & Weaver, V. M. Fibrosis and cancer: a strained relationship. Biochim. Biophys. Acta Rev. Cancer 1873, 188356 (2020).
Cialdai, F., Risaliti, C. & Monici, M. Role of fibroblasts in wound healing and tissue remodeling on Earth and in space. Front. Bioeng. Biotechnol. 10, 958381 (2022).
Vorselen, D. et al. The role of the cytoskeleton in sensing changes in gravity by nonspecialized cells. FASEB J. 28, 536–547 (2014).
Zhong, J., Yang, Y., Liao, L. & Zhang, C. Matrix stiffness-regulated cellular functions under different dimensionalities. Biomater. Sci. 8, 2734–2755 (2020).
Buravkova, L., Larina, I., Andreeva, E. & Grigoriev, A. Microgravity effects on the matrisome. Cells 10, 2226 (2021).
Nday, C. M., Frantzidis, C., Jackson, G., Bamidis, P. & Kourtidou-Papadeli, C. Neurophysiological changes in simulated microgravity: an animal model. Neurol. India 67, S221–S226 (2019).
Locatelli, L. & Maier, J. A. M. Cytoskeletal remodeling mimics endothelial response to microgravity. Front. Cell Dev. Biol. 9, 733573 (2021).
Louis, F., Deroanne, C., Nusgens, B., Vico, L. & Guignandon, A. RhoGTPases as key players in mammalian cell adaptation to microgravity. Biomed. Res. Int. 2015, 747693 (2015).
Sun, Y., Kuang, Y. & Zuo, Z. The emerging role of macrophages in immune system dysfunction under real and simulated microgravity conditions. Int. J. Mol. Sci. 22, 2333 (2021).
Navran, S. The application of low shear modeled microgravity to 3-D cell biology and tissue engineering. Biotechnol. Annu. Rev. 14, 275–296 (2008).
Aleshcheva, G. et al. Scaffold-free tissue formation under real and simulated microgravity conditions. Basic Clin. Pharm. Toxicol. 119, 26–33 (2016).
Jensen, C. & Teng, Y. Is it time to start transitioning from 2D to 3D cell culture? Front. Mol. Biosci. 7, 33 (2020).
Moroni, L. et al. What can biofabrication do for space and what can space do for biofabrication? Trends Biotechnol. 40, 398–411 (2022).
Edmondson, R., Broglie, J. J., Adcock, A. F. & Yang, L. Three-dimensional cell culture systems and their applications in drug discovery and cell-based biosensors. ASSAY Drug Dev. Technol. 12, 207–218 (2014).
Cubo-Mateo, N. et al. Can 3D bioprinting be a key for exploratory missions and human settlements on the Moon and Mars? Biofabrication 12, 43001 (2020).
Mekala, N. K., Baadhe, R. R. & Potumarthi, R. Mass transfer aspects of 3D cell cultures in tissue engineering. Asia-Pac. J. Chem. Eng. 9, 318–329 (2014).
Nijhuis, J., Schmidt, S., Tran, N. N. & Hessel, V. Microfluidics and macrofluidics in space: ISS-proven fluidic transport and handling concepts. Front. Space Technol. 2, 779696 (2022).
Moreno-Villanueva, M. et al. Interplay of space radiation and microgravity in DNA damage and DNA damage response. NPJ Microgravity 3, 14 (2017).
Barravecchia, I. et al. Microgravity and space radiation inhibit autophagy in human capillary endothelial cells, through either opposite or synergistic effects on specific molecular pathways. Cell. Mol. Life Sci. 79, 28 (2021).
Galluzzi, L., Yamazaki, T. & Kroemer, G. Linking cellular stress responses to systemic homeostasis. Nat. Rev. Mol. Cell Biol. 19, 731–745 (2018).
Juhl, O. J. et al. Update on the effects of microgravity on the musculoskeletal system. NPJ Microgravity 7, 28 (2021).
Baran, R. et al. The cardiovascular system in space: focus on in vivo and in vitro studies. Biomedicines 10, 59 (2021).
Clément, G. R. et al. Challenges to the central nervous system during human spaceflight missions to Mars. J. Neurophysiol. 123, 2037–2063 (2020).
Steller, J. G. et al. Oxidative stress as cause, consequence, or biomarker of altered female reproduction and development in the space environment. Int. J. Mol. Sci. 19, 3729 (2018).
Zhang, Y. et al. Transcriptomics, NF-κB pathway, and their potential spaceflight-related health consequences. Int. J. Mol. Sci. 18, 1166 (2017).
Johnson, I., Nguyen, C. T., Wise, P. & Grimm, D. Implications of altered endosome and lysosome biology in space environments. Int. J. Mol. Sci. 21, 8205 (2020).
Davis, S. A. & Davis, B. L. Exercise equipment used in microgravity: challenges and opportunities. Curr. Sports Med. Rep. 11, 142–147 (2012).
Pavez Loriè, E. et al. The future of personalized medicine in space: from observations to countermeasures. Front. Bioeng. Biotechnol. 9, 739747 (2021).
Beck, L. R., Lobitz, B. M. & Wood, B. L. Remote sensing and human health: new sensors and new opportunities. Emerg. Infect. Dis. 6, 217–227 (2000).
De La Rocque, S., Michel, V., Plazanet, D. & Pin, R. Remote sensing and epidemiology: examples of applications for two vector-borne diseases. Comp. Immunol. Microbiol. Infect. Dis. 27, 331–341 (2004).
Leblanc, A. et al. Bisphosphonates as a supplement to exercise to protect bone during long-duration spaceflight. Osteoporos. Int. 24, 2105–14 32 (2013).
Braddock, M. From target identification to drug development in space: using the microgravity assist. Curr. Drug Discov. Technol. 17, 45–56 (2020).
Dello Russo, C. et al. Physiological adaptations affecting drug pharmacokinetics in space: what do we really know? A critical review of the literature. Br. J. Pharmacol. 179, 2538–2557 (2022).
Acknowledgements
This manuscript has been written in the frame of the activities for the preparation and dissemination of the ESA-ROADMAPS FOR FUTURE RESEARCH which define strategic goals for future space research on the ISS and supporting research platforms.
Author information
Authors and Affiliations
Contributions
Conceptualization, Monica Monici (M.M.) and Nathaniel J. Szewczyk (N.J.S.); writing, Trent Davis (T.D.), Kevin Tabury (K.T.), Shouan Zhu (S.Z.), N.J.S., and M.M.; writing review and editing, T.D., K.T., S.Z., M.M., and N.J.S.; supervision, M.M., N.J.S., Christiane Hahn (C.H.), Jason Hatton (J.H.), Angelique van Ombergen (A.v.O.), and Christopher Puhl (C.P.). The other authors contributed to writing the white paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Davis, T., Tabury, K., Zhu, S. et al. How are cell and tissue structure and function influenced by gravity and what are the gravity perception mechanisms?. npj Microgravity 10, 16 (2024). https://doi.org/10.1038/s41526-024-00357-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41526-024-00357-9
- Springer Nature Limited