Abstract
Poor insight in schizophrenia spectrum disorders (SSD) is linked with negative outcomes. This single-centre, assessor-blind, parallel-group 1-year follow-up randomised controlled trial (RCT) tested whether metacognitive training (MCT) (compared to psychoeducation) may improve insight and outcomes in outpatients with SSD assessed: at baseline (T0); after treatment (T1) and at 1-year follow-up (T2). Insight (primary outcome) was measured with (i) the Schedule for Assessment of Insight-Expanded version- (SAI-E), including illness recognition (IR), symptom relabelling (SR), treatment compliance (TC) and total insight scores (TIS); and (ii) the Beck Cognitive Insight Scale (BCIS). Between-group comparisons were nonsignificant, while within the MCT group (but not within controls) there was a significant medium effect size for improved TIS at T2 (d = 0.67, P = 0.02). Secondary outcomes included cognitive measures: Jumping to Conclusions (JTC), Theory of Mind (ToM), plus symptom severity and functioning. Compared to psychoeducation, MCT improved the PANSS excitement (d = 1.21, P = 0.01) and depressed (d = 0.76, P = 0.05) factors at T2; and a JTC task both at T1 (P = 0.016) and at T2 (P = 0.031). Participants in this RCT receiving MCT showed improved insight at 1-year follow-up, which was associated with better mood and reduced JTC cognitive bias. In this pilot study, no significant benefits on insight of MCT over psychoeducation were detected, which may have been due to insufficient power.
Similar content being viewed by others
Introduction
Insight (i.e., clinical insight) in schizophrenia spectrum disorders (SSD) has been linked with outcome—greater insight, better outcomes1,2. However, 50–80% of patients with SSDs3, particularly schizophrenia4, lack insight from first presentation5.
Thirty years ago clinical insight was proposed to be a multidimensional phenomenon encompassing (i) illness awareness, i.e., recognition of having a mental illness, (ii) symptom relabelling, defined as the ability to recall unusual mental events (e.g., hallucinations) as abnormal and (iii) treatment compliance6. This multidimensional model of clinical insight has been supported by three decades of research7. More specifically, independent first-episode psychosis (FEP) samples8,9 have replicated the David’s three-dimension model of clinical insight6. Interventions for improving clinical insight, including psychoeducation, psychoanalytically oriented therapies, cognitive-behavioural therapy (CBT), video-recorded self-observation and antipsychotics, have been minimally effective to date10,11, although metacognitive interventions revealed more promising results12.
Metacognition, defined as ‘knowledge and cognition about cognitive phenomena’13 or ‘the ability to think of one’s own and others’ thinking’14, has received much attention from research over the past few years. Specifically, metacognitive deficits have been consistently reported in SSDs15, and have been linked with a lack of clinical insight7,16. Of note, clinical insight, as detailed above, should be distinguished from the broader construct, cognitive insight, a metacognitive domain which includes the ability to evaluate and correct one’s distorted beliefs and misinterpretations (self-reflectiveness) and the tendency to overconfidence in one’s conclusions (self-certainty)15,17. Perhaps surprisingly, the relationship between cognitive and clinical insight has been found to be somewhat weak17.
In 2007, metacognitive training (MCT) was developed in Germany by Steffen Moritz and Todd Woodward. MCT seeks to plant the seeds of doubt by targeting cognitive biases leading to delusional thoughts rather than asking patients directly to talk about of their beliefs. MCT can be delivered individually or in group sessions by psychiatrists, psychologists, social workers, nurses and other therapists. The MCT manual consists of a PowerPoint presentation available at http://www.uke.de/mkt in thirty-seven languages free of charge, which includes ten Modules on different topics: Attributional Style (Module 1), Jumping to Conclusions (Modules 2 and 7), Changing Beliefs (Module 3), Empathy (Modules 4 and 6), Memory (Module 5), Depression and Self-Esteem (Modules 8 and 9) and Stigma (Module 10).
Although not consistently18,19, MCT was demonstrated to reduce positive20,21,22,23,24,25 and negative26 psychotic symptoms severity, cognitive biases, self-esteem and functioning26. However, evidence supporting the effects of specific MCT modules is limited27. Given the heterogeneity of delusional experiences in schizophrenia phenomenology28 and the MCT mechanism of action, namely inducing some doubt about the generation of such delusional ideas29,30, it would be interesting to know the effects of particular modules of MCT on specific delusion types28.
Of relevance, two core treatment targets of MCT, namely Jumping to Conclusions (JTC) cognitive bias and Theory of Mind (ToM) reasoning, have been linked with clinical insight31. Hence, MCT may improve insight via addressing cognitive insight, JTC and/or ToM. JTC, i.e., reaching a conclusion based on incomplete evidence, can be considered as a decision-making style common in psychosis32,33,34 which has been linked with delusions35 and poor clinical insight31. ToM can be defined as ‘the ability to attribute mental states— beliefs, intents, desires, emotions and knowledge—primarily to others’36. ToM deficits have been linked to lack of clinical insight31, consistently reported in patients with psychosis from first presentation28 associated with paranoia in schizophrenia, and finally, likely involve specific brain regions and pathways37. Although MCT targets other cognitive biases leading to delusional ideas, such as Bias Against Disconfirmatory Evidence (BADE), we decided to focus on JTC and ToM based on previous literature linking JTC and ToM (but not BADE) with clinical insight31.
Most importantly, only five previous short-term (over 6 months) randomised controlled trials (RCTs) using early-onset psychosis patients samples investigated the MCT effects on clinical insight38,39,40,41,42, with relatively modest results. To our knowledge, no previous long-term MCT RCT has examined the effects on cognitive and clinical insight changes in non-first-episode schizophrenia patients, including clinical and social outcomes12.
This RCT aimed to investigate whether MCT may improve clinical and cognitive insight (as co-primary outcomes) in outpatients with SSD over a 1-year follow-up. Secondary outcomes included JTC, ToM, symptomatic severity and psychosocial functioning. Compared with controls (an active psychoeducation group) we hypothesised that MCT will result in: (i) greater cognitive and clinical insight levels; (ii) an improvement in JTC cognitive bias and ToM performance, reduced symptom severity and better functioning, and (iii) we sought to explore whether these effects would persist at post treatment and at 1-year follow-up.
Results
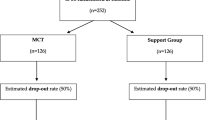
Figure 1 shows the CONSORT flow diagram of participants over the trial period. N = 77 individuals were assessed at T0 and randomised. n = 34 subjects (44.15%), who attended at least four sessions (e.g., ref. 43), were assessed and analysed at T1. At T2, n = 28 subjects were available and analysed. There were no between-group differences at any assessment. Of n = 34, subjects available at T1, 50% of them (median) attended 6 sessions (mean = 5.8 ± 2.4), with no between-group differences.
Sample characteristics
There were no baseline differences in sociodemographic, clinical, premorbid adjustment, neurocognitive, psychopathological, insight, metacognitive and functioning variables between MCT and controls psychoeducation groups (Table 1), except that there was a significantly (P = 0.021) higher proportion of unmarried people in the MCT group (n = 35, 89.7%) than in controls (n = 26, 68.4%).
As noted above, only those individuals who attended four treatment sessions were analysed. In the MCT group no relevant differences between those who attended at least four sessions and those who did not were found (see Supplementary Table S1) except for one insight dimension, namely symptom relabelling (4.8 ± 2.3 vs. 6.9 ± 2.9, t37 = −2.44, P = 0.019). However, in the Psychoeducation control group (Supplementary Table S2), attendees (compared with non-attendees) had higher IQ (109.7 ± 12.0 vs. 100.7 ± 10.1, t36 = 2.50, P = 0.017) and BCIS Composite Index (8.9 ± 5.1 vs. 4.5 ± 5.0, t32 = 2.51, P = 0.017), better functioning in terms of WHODAS (21.3 ± 12.9 vs. 12.1 ± 8.7, t35 = 2.61, P = 0.013) and SLDS (72.9 ± 12.1 vs. 83.6 ± 10.8, t34 = −2.81, P = 0.008) and they were less likely to have JTC as assessed by the 85:15 Beads Task (n = 4, 25.0% vs. n = 15, 68.2%, X21 = 6.91, P = 0.009) (Supplementary Table S2). Most importantly, for those who attended at least 4 treatment sessions (n = 34) no significant baseline differences between MCT (n = 18) and psychoeducation (n = 16) groups were found except for WHODAS (P = 0.047) and SLDS (P = 0.034) total scores, none of which were the primary outcomes of the study (Supplementary Table S2). Analyses were not therefore controlled for baseline data.
Between-group differences in outcomes
Table 2 presents between-group differences in continuous outcome measures after treatment and at follow-up. Of note, the only nominal non-continuous variable was JTC.
Primary outcomes
Although the MCT group SAI-E scores improved from T0 to T1 and from T0 to T2, between-arm comparisons yielded nonsignificant effect sizes (Table 2).
Secondary outcomes
With regard to the ‘easier’ JTC 85:15 Task, in the MCT group, there was a decrease in the number of subjects with JTC from T0 (n = 9, 50%) to T1 (n = 6, 33.3%) (P = 0.25), while at T2 only n = 4 subjects (26.7%) jumped to conclusions (P = 0.22). In the psychoeducation group, there was an increase in the number of individuals who jumped to conclusions from T0 (n = 4, 25%) to T1 (n = 6, 37.5%) (P = 0.62), although at T2 only n = 5 subjects (38.4%) had JTC (P = 0.99).
Regarding the JTC 60:40 Task, in the MCT group there was a significant decrease in the number of subjects who jumped to conclusions from T0 (n = 9, 50%) to T1 (n = 2, 11.1%) (P = 0.016) and from T0 (n = 9, 50%) to T2 (n = 2, 11.1%) (P = 0.031), while in the psychoeducation group differences were nonsignificant: n = 5 (27.8%) both at T0 and at T1 (P = 0.99) and n = 2 (12.5%) (P = 0.37) at T2.
No small or significant effects on ToM tasks were found (Table 2).
With regard to positive and negative psychotic symptoms and disorganisation, effect sizes did not reach significance, although MCT was superior to psychoeducation at an almost significant large effect size in terms of depression (d = 0.76, P = 0.05), which was significant for the PANSS excitement factor (d = 1.21, P = 0.01), at T2.
In terms of functioning, effect sizes were nonsignificant (Table 2).
Between-treatment differences in insight changes over the trial period are also plotted in Fig. 2 (SAI-E total score, i.e., clinical insight) and in Fig. 3 (BCIS Composite Index, i.e., cognitive insight).
Within each treatment group, Student’s t test compared SAI-E total scores changes from baseline to post treatment and from baseline to 1-year follow-up. The blue line indicates values changes for the MCT group, while the orange line indicates values changes for the psychoeducation group. The text boxes report the effect sizes as Cohen’s d coefficient and its P value.
Within each treatment group, Student’s t test compared BCIS Composite Index changes from baseline to post treatment and from baseline to 1-year follow-up. The blue line indicates values changes for the MCT group, while the orange line indicates values changes for the psychoeducation group. The text boxes report the effect sizes as Cohen’s d coefficient and its P value.
Within-group differences in outcome measures
In terms of insight gain in the MCT group at T2 we found a significant medium effect size for TIS (d = 0.67, P = 0.02) and almost significant effect sizes for SR (d = 0.55, P = 0.05) and for TC (d = 0.52, P = 0.06) at T2, which was not replicated at T1. In the psychoeducation group, all effect sizes for insight changes at T1 and at T2 were nonsignificant.
Further within-group differences in outcomes are shown in Table 3 (MCT group) and in Table 4 (Controls). Also, within-group differences in the JTC Beads Tasks over the trial period are plotted in Fig. 4 (85:15) and Fig. 5 (60:40), below.
Discussion
Principal findings
We carried out a pilot study to test whether metacognitive training (MCT) (compared with psychoeducation) may improve clinical and cognitive insight in an unselected sample of outpatients with SSD in order to path the way for a larger trial. As secondary outcomes, we looked at decision-making and mentalizing, that is, JTC cognitive bias and ToM tasks, respectively, symptoms severity and psychosocial functioning.
Results provided some support for our first hypothesis, which predicted that (compared to psychoeducation) MCT would result in greater clinical and cognitive insight gain. In particular, we found MCT to improve both clinical and cognitive insight, although MCT did not show significant benefits compared to psychoeducation, which may have been due to insufficient statistical power. However, within-group comparisons revealed that while the MCT effect on total clinical insight scores changes at 1-year follow-up reached significance, psychoeducation failed to do this, somewhat consistent with our first hypothesis. This said, given the pilot nature of the trial and its limited power, this finding should be taken very cautiously. Regarding secondary outcomes (compared to psychoeducation) MCT showed significant effects sizes on JCT and almost significant effects sizes on the excitement and depressed PANSS factors; however, no such effects were observed on ToM and functioning. These findings partially supported our second hypothesis, which postulated that MCT would result in an improvement in JTC cognitive bias and ToM performance, reduced symptom severity and better functioning. We also noted that most of these effects were larger after treatment than at a 1-year follow-up.
Metacognitive training effects on clinical and cognitive insight
Not much progress has been made in treatments for (clinical) insight in psychosis10,11, although a very recent systematic review and meta-analysis12, which included five RCTs (N = 244) on MCT38,39,41,42,44, showed MCT to increase clinical insight at a larger effect than in controls. In this pilot study, however, between-group comparisons for all SAI-E scores failed to reveal MCT to be superior to psychoeducation, which may have been owing to the relatively small sample size as a result of the COVID-19 outbreak in the middle of the trial. In addition, we tested MCT against an active control intervention, which diminishes effect sizes in comparison with trials using TAU as comparator45.
Taking a multidimensional approach to clinical insight6, the larger effect sizes were observed for the symptom relabelling component, thus replicating some previous trials38,39,44. Therefore, MCT seems to be more useful in improving the ability to reframe the meaning of certain psychotic symptoms rather than in improving illness awareness as a whole or treatment compliance. Moreover, treatment compliance does not appear to have a metacognitive basis, hence being less amenable to metacognitive interventions12, although one trial41 conflicted with this notion.
Interestingly, we found larger effects immediately after treatment than at follow-up. Only three previous RCTs38,42,44 examined the effects of MCT on clinical insight at 6 months, two of which38,42 replicated this pattern. Future trials are warranted to compare whether adding MCT maintenance sessions may alter these results. Regretfully, we could not evaluate the impact of each MCT module on clinical insight changes due to limited statistical power. This said, the more relevant benefits for an individual clinical insight dimension, such as symptom relabelling, are likely to particularly reflect the benefits from two specific modules, namely attributional style (Module 1) and Changing Beliefs (Module 3), which warrants future investigation in a full-scale trial.
Cognitive insight was put forward by Beck and colleagues in 2004, who also validated a 15-item self-rated scale, the Beck Cognitive Insight Scale (BCIS) which yielded two factors, namely self-reflectiveness and self-certainty15. Therefore, interventions targeting cognitive insight aim to increase self-reflectiveness and to decrease self-certainty. In this respect, in contrast with our 2020 meta-analysis12 and recent FEP studies27,46,47, we found nonsignificant effect sizes when comparing MCT to psychoeducation, which may have been due to lack of power. Therefore, future trials with larger sample sizes and more prolonged follow-up periods are warranted to address this clinically relevant issue. More specifically, our results showing (non-significantly) greater effects for self-reflectiveness than for self-certainty appear to suggest that self-reflectiveness may be more amenable to group MCT than self-certainty, although this remains to be confirmed. Future studies may examine whether individual MCT-based interventions, such as a MCT-based smartphone application (https://clinical-neuropsychology.de/app_en)48 and individual face-to-face MCT sessions38,49 may reduce self-certainty levels at a larger effect size than group MCT. On the other hand, cognitive insight, especially self-certainty, may behave as a trait (rather than a state), which cannot be modified through intervention.
Metacognitive training effects on JTC and ToM
We replicated the positive effect of MCT on JTC46,50. Indeed, MCT specifically addresses JTC in two modules23, although the potential influence of JTC cognitive bias on lack of insight in psychosis remains unknown.
Contrary to our expectations (hypothesis ii), MCT was not shown to improve ToM deficits when compared with psychoeducation. It could be argued, however, that the two ToM measures of the trial, namely the Emotions Recognition Test Faces51,52 activity and the Hinting Task53, may have failed to capture those ToM elements targeted by MCT54. Also, ToM deficits in early psychosis may be more prone to modification28 than in later stages of the illness. Certainly, mentalizing impairment appears to be a trait marker of schizophrenia55.
Positive effects of metacognitive training on mood
MCT improved mood by reducing excitement and depressive symptoms severity, particularly at follow-up. In particular, it should be noted that both excitement and depressive symptoms were based on the PANSS factors56 which included the following PANSS items: excitement—excitement (P4), Hostility (P7), Uncooperativeness (G8) and Poor impulse control (G14)—and depression—Anxiety (G2), Guilt Feelings (G3) and Depression (G6). In particular, a recent meta-analysis of 63 studies across 22 countries demonstrated self-stigma to positively correlate with depressive symptoms in schizophrenia spectrum disorders—greater stigma, more severe depressive symptoms—while the correlation between self-stigma and self-esteem was negative—greater stigma, worse self-esteem-57. Hence, there are grounds to speculate that the above MCT-induced mood improvement could be explained, in part, by two MCT additional modules, namely Self-Esteem (Module 9) and Stigma (Module 10), which were later added to the original 8-module MCT package58. However, these results were not replicated with the Calgary Depression Scale for Schizophrenia (CDSS)59 total score, which could be attributable to the limited power of the trial. Future studies using different scales are therefore warranted.
Although two previous meta-analyses replicated the relationship between mood and clinical insight—lower mood, greater insight60,61—the causality direction remains far from clear. On the one hand, becoming aware of having a psychotic illness could be thought to lead to more severe depressive symptoms, which is known as the demoralisation syndrome62. On the other hand, depressed patients are subject to cognitive distortions which makes them more pessimistic about themselves, including illnesses—the depressive realism model63—hence scoring higher on insight scales at assessment. The so-called (clinical) Insight Paradox64 has also been replicated for cognitive insight, particularly for self-reflectiveness65,66 in SSD, which was found to mediate the impact of depression on general psychological distress67. However, the relationship between insight and increased suicide risk has not been confirmed68,69. Nonetheless, more theoretical debate and empirical research is needed to better understand the clinical meaning of depression in schizophrenia, thus improving patient outcomes70.
Of note, no baseline differences were found between those who attended four treatment sessions and those who did not except for the MCT group (see Supplementary Tables S1, S2 and S3 in the Supplementary online material). In particular, within the MCT group those who dropped out of the trial before the fourth session had greater baseline insight into mental symptoms than those who attended four or more sessions. Certainly, recalling mental events as pathological must be very distressful. Hence, those with greater ability to recall mental experiences as abnormal may be at a higher risk of disengagement from an intervention such as MCT, which seeks to encourage self-reflection on these phenomena.
Strengths and limitations
This is the first 1-year follow-up RCT examining the effects of MCT on multiple dimensions of clinical and cognitive insight (as co-primary outcomes) measured with validated instruments (SAI-E and BCIS, respectively) in a sample of SSD outpatients. Participants were randomised to either MCT or psychoeducation and the same assessor (JDLM) blinded to the treatment allocation evaluated outcomes across assessments. MCT founders were uninvolved in the trial and participants did not receive a financial compensation. We also controlled for the effect of attending a weekly therapeutic group since controls received an active intervention (psychoeducation), which probably improved recruitment to the study and conferred some ethical benefits in comparison to studies with TAU comparators, although this may have diminished effect sizes favouring MCT45.
However, some limitations should be borne in mind when interpreting our pilot RCT results. First, recruitment and intervention groups had to be stopped in March 2020 owing to the COVID-19 outbreak in Spain. Not only did this reduce the study power, but also measures for combating COVID-19, such as prolonged confinement periods, may have had a negative impact on functioning-related outcomes at follow-up. Although unlikely given the consistency of results from this pilot study, a potential ‘regression to the mean’ phenomenon cannot be fully ruled out. Future large-scale trials are therefore required. Hence, the true effect size of MCT on insight changes remains to be established. In addition, given the aforementioned power issues we did not examine potential MCT effects on individual PANSS items, such as delusions, or the module-specific effects on insight. Also, we only analysed those who attended at least four treatment sessions, in line with previous studies38,43. Second, other variables such as antipsychotics71, which were not evaluated in this trial, may have affected our results. Third, although not evaluated in this study, the potential improvement in other cognitive processes targeted by MCT, such as Bias Against Disconfirmatory Evidence, may have contributed to MCT-related insight gain, which requires further investigation. Fourth, regretfully we did not conduct a satisfaction survey or feedback questionnaire72, although the high attendance rates and the lack of issues raised by attendees suggested high levels of satisfaction. We cannot rule out that between-arm differences in insight levels over the trial period may have contributed to attrition issues which may have affected the results, although this seems unlikely. Finally, these findings may not apply to other settings such as primary care and those living in rural areas. In addition, participants consented to a lengthy protocol, including three face-to-face assessments over one year, which may have excluded those individuals with poorer insight.
Conclusions
This RCT was designed to compare MCT with psychoeducation in an unselected sample of outpatients with SSD with the aim of investigating effects on insight and some clinical and social outcomes. Regretfully, the COVID-19 outbreak in Spain in March 2020 prevented the trial from recruiting the required sample size, thus requiring us to reconsider the original RCT as a pilot study. Although much caution is therefore needed when interpreting the results, MCT proved useful in improving insight and some outcomes, such as JTC and mood, in this sample of SSD patients. Of note, conducting such a trial remains feasible since no adverse effects were observed and most participants remained clinically stable. These promising results therefore strongly justify a larger scale RCT and future research in this area.
Methods
Study design and randomisation process
Single-centre, assessor-blind, parallel-group, two-armed, 1-year follow-up RCT. After baseline (T0) assessment, participants were randomised to either group MCT (experimental intervention) or a psychoeducation group (controls) through a computerised algorithm independent of the investigators (no stratification factors) in blocks of 10 subjects (maximum number of each group) and assessor (JDLM)-patient blind. Participants were aware of the intervention so the RCT was not double-blind, as in most non-pharmacological trials. Reassessments took place after treatment (T1) and at 1-year follow-up (T2), which were carried out by the same assessor (JDLM) blind to the group allocation (assessor-blind).
Sample and eligibility criteria
Participants came from the publicly-funded Hospital Universitario Fundación Jiménez Díaz (Madrid, Spain). Inclusion criteria were: (i) outpatient status; (ii) age: 18-64 years, both inclusive; and iii) diagnosis, namely SSD based on the Mini International Neuropsychiatric Interview, 5th Edition, (MINI)73, which included schizophrenia, schizoaffective disorder, delusional disorder and psychotic disorder Not Otherwise Specified, according to either International Statistical Classification of Diseases (ICD), 10th Revision74.
Recruitment began on the 06/17/2019 and had to be stopped on the 03/11/2020 due to the COVID-19 outbreak in Spain. Exclusion criteria were: (i) IQ ≤ 70, which was assessed with the short form of the Wechsler Adults Intelligence Scale (WAIS)-IV75, (ii) a history of head injury and/or a neurological condition; (iii) having received a metacognitive intervention within the previous year; (iv) low level of Spanish; (v) lack of cooperativeness for participating in the intervention groups detailed below, as judged by the treating consultant psychiatrist or psychologist. Participants provided written informed consent as approved by the Local Research Ethics Committee (EC044-19_FJD-HRJC). The RCT is registered at ClinicalTrials.gov (NCT04104347). Participants were not financially compensated for completing the assessments and/or receiving the interventions. The full study protocol of this RCT was published elsewhere76.
Variables
Co-primary outcomes
Clinical insight was assessed with the Spanish version77 of the Schedule for Assessment of Insight, expanded version (SAI-E)78, which provides scores on three insight dimensions based on David’s model of insight6—illness recognition (IR), symptom relabelling (SR), treatment compliance (TC)—and a total insight score (TIS). The scale was found to be easily applicable in routine clinical practice79 and good to excellent inter-rater reliability was reported, with total insight scores intra-class correlations coefficients ranging from 0.92 to 0.98 (P < 0.001)8. JDLM was trained by the author scale (ASD) and they both co-led the validation study of the SAI-E Spanish version77, which was used in this RCT.
Cognitive insight was evaluated by the Spanish version80 of the Beck Cognitive Insight Scale (BCIS)15. The BCIS is a 15-item self-administered scale which includes 9 items assessing self-reflectiveness and 6 items enquiring about self-certainty. A composite index can thus be calculated by subtracting self-certainty from self-reflectiveness. Internal consistency was found to be acceptable, with Cronbach’s α ranging from 0.60 to 0.68 across individual BCIS items80.
Secondary outcomes
Secondary outcomes included Jumping to Conclusions (JTC) and Theory of Mind (ToM), symptomatic severity and functioning.
Jumping to Conclusions (JTC) was measured with a computerised version of the Beads Task81. Participants are shown two jars containing coloured beads in different, although reciprocal, proportions. On the basis of probability (in task 1 the probability is 85:15, while in task 2 the probability is 60:40), the individual must decide the jar to which the extracted bead belongs. JTC was rated as present/absent based on the ‘two or less draws to decision threshold’, which was found to be most reliably associated with delusions34 and widely used in previous studies32,46,82. However, concerns have been raised about the Beads Task as a measurement of JTC cognitive bias since patients’ tendency to ‘over-adjustment’ may be explained by miscomprehension of the test83. In keeping with this, we did not consider the Beads Task as a continuous variable due to power-related issues detailed below.
In order to assess mentalizing or Theory of Mind (ToM), two instruments were administered. First, two different stories from the Hinting Task53 Spanish version84, which was found to have acceptable internal consistency (α = 0.64)84, were used in each assessment to avoid learning. Scores therefore ranged from 0 to 4. Second, the Emotions Recognition Test Faces activity (ERTF)51,52, which is composed of 20 different photographs showing people’s facial expressions, evaluated patients’ ability to recognise people’ emotions between two given options. Each right answer is given a score of 1, which can be summed up to create total scores ranging from 0 to 20; higher scores indicated better ToM performance.
Although the Spanish version85 of the Positive and Negative Syndrome Scale (PANSS)86 which revealed three psychopathological dimensions—positive, negative and disorganised85, was used to assess symptoms severity, five symptomatic dimensions, namely positive, negative, disorganisation, excitement and depression, were taken based on a more updated review of previous PANSS factor analysis studies56 as follows: positive (P1, P3, G5, G9), negative (N1, N2, N3, N4, N6, G7), disorganisation (P2, N5, G11), excitement (P4, P7, G8, G14) and depression (G2, G3, G6). Specifically, Depressive symptoms severity was also measured with the Spanish version87 of the Calgary Depression Scale for Schizophrenia (CDSS)59, which is a 9-item structured interview enquiring about symptoms of depression, each of which is scored within a 4-point Likert scale ranging from 0 (absent) to 3 (severe). Total CDSS scores therefore range from 0 to 27. Based on the first Kraepelinian classification of endogenous psychoses88 schizophrenia has long been considered as a ‘non-affective’ psychotic illness. However, not only recent research has supported the dimensional model of psychoses89, but also mania and depression symptoms have been shown to be intrinsic to schizophrenia70,90,91.
Functioning was recorded through the Global Assessment of Functioning (GAF)92 and the 12-item version of the World Health Organization Disability Schedule (WHODAS)93, while the Spanish adaptation94 of the Satisfaction Life Domains Scale (SLDS)95 measured quality of life.
Additional variables
We collected baseline data on age, gender, education level, marital status, employment status, living status, ICD-10 diagnosis, previous suicidal behaviour (present/absent), illness duration and number of previous admissions, number of antipsychotics (one or more than one), being on long-acting injections (present/absent), taking clozapine (present/absent) and chlorpromazine equivalents (mg), premorbid adjustment assessed with the Premorbid Adjustment Scale (PAS)96 and neurocognition. The Wechsler Adult Intelligence Scale (WAIS)-IV -vocabulary subtest-75 estimated participants’ IQ and the Trail Making Test (TMT)97 assessed executive function, particularly ‘time to complete Task A (in seconds) minus time to complete Task B’, which provides a brief measure of executive function (set maintenance/shifting), whilst controlling for processing speed9. We did not report on medication changes over the trial, which were marginal (data available on request).
Interventions
In addition to treatment as usual (TAU), which consisted of regular face-to-face appointments with the treating consultant psychiatrist, consultant psychologist and registered mental health nurse as appropriate and taking antipsychotic medication, participants were randomised either to receive MCT or to attend a psychoeducation group. Hence, all participants were meant to receive one weekly 45–60-min group session lasting over 8 weeks.
Metacognitive training (MCT)
Metacognitive Training (MCT)58 addresses positive symptoms of schizophrenia from an indirect approach which seeks to plant the seeds of doubt regarding cognitive biases leading to delusional thoughts. MCT focuses on different topics (Modules) shown by structured powerpoint presentations: Attributional Style (Module 1), Jumping to Conclusions (Modules 2 and 7), Changing Beliefs (Module 3), Empathy (Modules 4 and 6), Memory (Modules 5), Depression and Self-Esteem (Module 8) and two additional modules, namely Self-Esteem (Module 9) and Stigma (Module 10), although Modules 8, 9 and 10 were delivered together as one session. Although subject to future investigation, MCT was found to be efficacious for those who attended (at least) four sessions38,43. As a result, only those who attended four or more sessions were analysed.
Psychoeducation control group
Controls attended eight weekly psychoeducation group sessions on: (1) basic and (2) instrumental activities of daily living, (3) physical health, (4) newspapers-based work, (5) emotions, (6) illness, (7) social and family relationships. One additional session allowed participants to raise further concerns.
Both groups were conducted by a higher-trainee clinical psychologist (ASEM), who received direct training from one co-author of the Spanish version of the MCT manual (MLB). Treatment fidelity was looked at by this researcher (MLB) against the MCT manual criteria (http://www.uke.de/mkt), while a significant exposure of controls to MCT elements was ruled out during two random sessions over a month.
Statistical analysis
First, we explored baseline between-group differences in sociodemographic, premorbid adjustment, clinical, neurocognitive variables and outcome measures, including insight levels (Table 1). Second, after confirmation of the normal distribution of the co-primary outcomes of the RCT by means of the Kolmogorov–Smirnov test, we conducted Student’s t tests to examine between-group differences in the SAI-E and BCIS total and subtotal scores changes from T0 to T1 and from T0 to T2 (as the dependent variable) (Table 2). Third, within-group SAI-E and BCIS score changes from T0 to T1 and from T0 to T2 were also investigated (Tables 3 and 4). Effect sizes (Cohen’s d) were calculated for between- and within-group comparisons, which were classified as small (d < 0.2), medium (d = 0.5) or large (d > 0.8)98. JTC was the only binary outcome measure so McNemar’s test investigated between-assessment changes in each treatment group.
It is true that General Linear Mixed Models are particularly useful in longitudinally analysing between-group differences by modelling fixed and random effects. However, our small sample size and limited power, as detailed below, and the ‘normal’ distribution of the dependent variable, namely, SAI-E and BCIS scores ‘changes’ (i.e., whilst controlling for baseline data) led us to use Student’s t test for the analyses, which provided a unique P value of significance for between-group comparisons at post treatment and at follow-up99.
Analyses were performed for those participants who completed at least 4 treatment sessions regardless of the group38,43 using the Statistical Package for Social Science version 25.0 (SPSS, IBM Corp.; Armonk, NY, USA). Power calculations indicated that a total sample size of N = 102 subjects (n = 51 in each treatment arm) at the end of the trial would be needed to detect a medium effect size (d = 0.50, α = 5%, 1-β = 80%) for the primary outcome measure (SAI-E total score). As detailed above, recruitment and assessments had to be stopped due to unforeseen circumstances related to the COVID-19 outbreak in Spain in March 2020, which prevented us from reaching the required sample size. On the other hand, given the final underpowered sample size we did not apply corrections for multiple testing since Type I error was very unlikely.
Data availability
Data supporting these results are available upon reasonable request to the corresponding author, provided the dataset access policy is complied with.
References
Escobedo-Aedo, P. J. et al. Investigating the role of insight, decision-making and mentalizing in functional outcome in schizophrenia: a cross-sectional study. Behav. Sci. 12, 28 (2022).
Lincoln, T. M., Lüllmann, E. & Rief, W. Correlates and long-term consequences of poor insight in patients with schizophrenia. A systematic review. Schizophr. Bull. 33, 1324–1342 (2007).
Carpenter, W. T., Strauss, J. S. & Bartko, J. J. Flexible system for the diagnosis of schizophrenia: report from the WHO International Pilot Study of Schizophrenia. Science 182, 1275–1278 (1973).
Amador, X. F. et al. Awareness of illness in schizophrenia and schizoaffective and mood disorders. Arch Gen. Psychiatry 51, 826–836 (1994).
Ayesa-Arriola, R. et al. Lack of insight 3 years after first-episode psychosis: an unchangeable illness trait determined from first presentation? Schizophr. Res. 157, 271–277 (2014).
David, A. S. Insight and psychosis. Br. J. Psychiatry 156, 798–808 (1990).
David, A. S. Insight and psychosis: the next 30 years. Br. J. Psychiatry 217, 521–523 (2019).
Morgan, K. D. et al. Insight, grey matter and cognitive function in first-onset psychosis. Br. J. Psychiatry 197, 141–148 (2010).
Wiffen, B. D. R. et al. Are there specific neuropsychological deficits underlying poor insight in first episode psychosis? Schizophr. Res. 135, 46–50 (2012).
Henry, C. & Ghaemi, S. N. Insight in psychosis: a systematic review of treatment interventions. Psychopathology 37, 194–199 (2004).
Pijnenborg, G. H. M., van Donkersgoed, R. J. M., David, A. S. & Aleman, A. Changes in insight during treatment for psychotic disorders: a meta-analysis. Schizophr. Res. 144, 109–117 (2013).
Lopez-Morinigo, J.-D. et al. Can metacognitive interventions improve insight in schizophrenia spectrum disorders? A systematic review and meta-analysis. Psychol. Med. 50, 2289–2301 (2020).
Flavell, J. H. Metacognition and cognitive monitoring: a new area of cognitive-developmental inquiry. Am. Psychologist 34, 906–911 (1979).
Wells, Adrian Metacognition and cognitive-behaviour therapy: a special issue. Clin. Psychol. Psychother. 6, 71–72 (1999).
Beck, A. T., Baruch, E., Balter, J. M., Steer, R. A. & Warman, D. M. A new instrument for measuring insight: the Beck Cognitive Insight Scale. Schizophr. Res. 68, 319–329 (2004).
Lysaker, P. H., Pattison, M. L., Leonhardt, B. L., Phelps, S. & Vohs, J. L. Insight in schizophrenia spectrum disorders: relationship with behavior, mood and perceived quality of life, underlying causes and emerging treatments: world psychiatry. World Psychiatry 17, 12–23 (2018).
Van Camp, L. S. C., Sabbe, B. G. C. & Oldenburg, J. F. E. Cognitive insight: a systematic review. Clin. Psychol. Rev. 55, 12–24 (2017).
van Oosterhout, B. et al. Metacognitive training for schizophrenia spectrum patients: a meta-analysis on outcome studies. Psychol. Med. 46, 47–57 (2016).
van Oosterhout, B. et al. Metacognitive group training for schizophrenia spectrum patients with delusions: a randomized controlled trial. Psychol. Med. 44, 3025–3035 (2014).
Eichner, C. & Berna, F. Acceptance and efficacy of metacognitive training (MCT) on positive symptoms and delusions in patients with schizophrenia: a meta-analysis taking into account important moderators. Schizophr. Bull. 42, 952–962 (2016).
Jiang, J., Zhang, L., Zhu, Z., Li, W. & Li, C. Metacognitive training for schizophrenia: a systematic review. Shanghai Arch Psychiatry 27, 149–157 (2015).
Liu, Y.-C., Tang, C.-C., Hung, T.-T., Tsai, P.-C. & Lin, M.-F. The efficacy of metacognitive training for delusions in patients with schizophrenia: a meta-analysis of randomized controlled trials informs evidence-based practice. Worldviews on Evidence-Based Nursing 15, 130–139 (2018).
Moritz, S. et al. Sowing the seeds of doubt: a narrative review on metacognitive training in schizophrenia. Clin. Psychol. Rev. 34, 358–366 (2014).
Philipp, R. et al. Effectiveness of metacognitive interventions for mental disorders in adults—a systematic review and meta-analysis (METACOG). Clin. Psychol. Psychother. 26, 227–240 (2019).
van Oosterhout, B. et al. Letter to the editor: should we focus on quality or quantity in meta-analyses? Psychol. Med. 46, 2003–2005 (2016).
Penney, D. et al. Immediate and sustained outcomes and moderators associated with metacognitive training for psychosis: a systematic review and meta-analysis. JAMA Psychiatry 79, 417–429 (2022).
Birulés, I. et al. Cognitive insight in first-episode psychosis: changes during metacognitive training. J. Pers. Med. 10. https://doi.org/10.3390/jpm10040253 (2020).
Bora, E. & Pantelis, C. Theory of mind impairments in first-episode psychosis, individuals at ultra-high risk for psychosis and in first-degree relatives of schizophrenia: systematic review and meta-analysis. Schizophr. Res. 144, 31–36 (2013).
Moritz, S. et al. A two-stage cognitive theory of the positive symptoms of psychosis. Highlighting the role of lowered decision thresholds. J. Behav. Ther. Exp. Psychiatry 56, 12–20 (2017).
Brar, P. S., Sass, L., Beck, D. & Kalarchian, M. A. Metacognitive training for schizophrenia: a scoping review and phenomenological evaluation. Psychosis 14, 1–21 (2022).
Lopez-Morinigo, J.-D. et al. Investigating the contribution of decision-making, cognitive insight, and theory of mind in insight in schizophrenia: a cross-sectional study. Psychopathology 55, 104–115 (2022).
Falcone, M. A. et al. Jumping to conclusions, neuropsychological functioning, and delusional beliefs in first episode psychosis. Schizophr Bull 41, 411–418 (2015).
Garety, P. Reasoning and delusions. Br. J. Psychiatry Suppl. 159, 14–18 (1991).
Garety, P. A. et al. Reasoning, emotions, and delusional conviction in psychosis. J. Abnorm. Psychol. 114, 373–384 (2005).
Falcone, M. A. et al. Jumping to conclusions and the persistence of delusional beliefs in first episode psychosis. Schizophr. Res. 165, 243–246 (2015).
Baron-Cohen, S., Leslie, A. M. & Frith, U. Does the autistic child have a ‘theory of mind’? Cognition 21, 37–46 (1985).
Weng, Y., Lin, J., Ahorsu, D. K. & Tsang, H. W. H. Neuropathways of theory of mind in schizophrenia: a systematic review and meta-analysis. Neurosci. Biobehav. Rev. 137, 104625 (2022).
Balzan, R. P., Mattiske, J. K., Delfabbro, P., Liu, D. & Galletly, C. Individualized metacognitive training (MCT+) reduces delusional symptoms in psychosis: a randomized clinical trial. Schizophr. Bull. 45, 27–36 (2019).
Briki, M. et al. Metacognitive training for schizophrenia: a multicentre randomised controlled trial. Schizophr. Res. 157, 99–106 (2014).
Favrod, J. et al. Sustained antipsychotic effect of metacognitive training in psychosis: a randomized-controlled study. Eur. Psychiatry 29, 275–281 (2014).
Gawęda, Ł., Krężołek, M., Olbryś, J., Turska, A. & Kokoszka, A. Decreasing self-reported cognitive biases and increasing clinical insight through meta-cognitive training in patients with chronic schizophrenia. J. Behav. Ther. Exp. Psychiatry 48, 98–104 (2015).
Kuokkanen, R., Lappalainen, R., Repo-Tiihonen, E. & Tiihonen, J. Metacognitive group training for forensic and dangerous non-forensic patients with schizophrenia: a randomised controlled feasibility trial. Crim. Behav. Mental Health 24, 345–357 (2014).
Ahuir, M. et al. Improvement in cognitive biases after group psychoeducation and metacognitive training in recent-onset psychosis: a randomized crossover clinical trial. Psychiatry Res. 270, 720–723 (2018).
Favrod, J. et al. Sustained antipsychotic effect of metacognitive training in psychosis: a randomized-controlled study. Eur. Psychiatry 29, 275–281 (2014).
Mehl, S., Werner, D. & Lincoln, T. M. Does Cognitive Behavior Therapy for psychosis (CBTp) show a sustainable effect on delusions? A meta-analysis. Front. Psychol. 6, 1450 (2015).
Ochoa, S. et al. Randomized control trial to assess the efficacy of metacognitive training compared with a psycho-educational group in people with a recent-onset psychosis. Psychol. Med. 47, 1573–1584 (2017).
Salas-Sender, M. et al. Gender differences in response to metacognitive training in people with first-episode psychosis. J. Consult. Clin. Psychol. 88, 516–525 (2020).
Lüdtke, T., Pult, L. K., Schröder, J., Moritz, S. & Bücker, L. A randomized controlled trial on a smartphone self-help application (Be Good to Yourself) to reduce depressive symptoms. Psychiatry Res. 269, 753–762 (2018).
Andreou, C. et al. Individualized metacognitive therapy for delusions: a randomized controlled rater-blind study. J. Behav. Ther. Exp. Psychiatry 56, 144–151 (2017).
Aghotor, J., Pfueller, U., Moritz, S., Weisbrod, M. & Roesch-Ely, D. Metacognitive training for patients with schizophrenia (MCT): feasibility and preliminary evidence for its efficacy. J. Behav. Ther. Exp. Psychiatry 41, 207–211 (2010).
Baron-Cohen, S., Wheelwright, S. & Jolliffe, T. Is there a ‘language of the eyes’? Evidence from normal adults, and adults with autism or Asperger syndrome. Visual Cognition 4, 311–331 (1997).
Huerta-Ramos, E. et al. Translation and validation of Baron Cohen’s face test in a general population from Spain. Actas Esp Psiquiatr 49, 106–113 (2021).
Corcoran, R., Mercer, G. & Frith, C. D. Schizophrenia, symptomatology and social inference: Investigating “theory of mind” in people with schizophrenia. Schizophr. Res. 17, 5–13 (1995).
Langdon, R., Still, M., Connors, M. H., Ward, P. B. & Catts, S. V. Theory of mind in early psychosis. Early Interv. Psychiatry 8, 286–290 (2014).
Sprong, M., Schothorst, P., Vos, E., Hox, J. & van Engeland, H. Theory of mind in schizophrenia: meta-analysis. Br. J. Psychiatry 191, 5–13 (2007).
Wallwork, R. S., Fortgang, R., Hashimoto, R., Weinberger, D. R. & Dickinson, D. Searching for a consensus five-factor model of the positive and negative syndrome Scale for schizophrenia. Schizophr. Res. 137, 246–250 (2012).
Sarraf, L., Lepage, M. & Sauvé, G. The clinical and psychosocial correlates of self-stigma among people with schizophrenia spectrum disorders across cultures: a systematic review and meta-analysis. Schizophr. Res. 248, 64–78 (2022).
Moritz, S. & Woodward, T. S. Metacognitive training for schizophrenia patients (MCT): a pilot study on feasibility, treatment adherence, and subjective efficacy. German J. Psychiatry 10, 69–78 (2007).
Addington, D., Addington, J., Maticka-Tyndale, E. & Joyce, J. Reliability and validity of a depression rating scale for schizophrenics. Schizophr. Res. 6, 201–208 (1992).
Belvederi Murri, M. et al. Is good insight associated with depression among patients with schizophrenia? Systematic review and meta-analysis. Schizophr. Res. 162, 234–247 (2015).
Mintz, A. R., Dobson, K. S. & Romney, D. M. Insight in schizophrenia: a meta-analysis. Schizophr. Res. 61, 75–88 (2003).
Drake, R. E. & Cotton, P. G. Depression, hopelessness and suicide in chronic schizophrenia. Br. J. Psychiatry 148, 554–559 (1986).
Ghaemi, S. N. Feeling and time: the phenomenology of mood disorders, depressive realism, and existential psychotherapy. Schizophr. Bull. 33, 122–130 (2007).
Lysaker, P. H., Roe, D. & Yanos, P. T. Toward understanding the insight paradox: internalized stigma moderates the association between insight and social functioning, hope, and self-esteem among people with schizophrenia spectrum disorders. Schizophr. Bull. 33, 192–199 (2007).
Lysaker, P. H., Pattison, M. L., Leonhardt, B. L., Phelps, S. & Vohs, J. L. Insight in schizophrenia spectrum disorders: relationship with behavior, mood and perceived quality of life, underlying causes and emerging treatments. World Psychiatry 17, 12–23 (2018).
Palmer, E. C., Gilleen, J. & David, A. S. The relationship between cognitive insight and depression in psychosis and schizophrenia: a review and meta-analysis. Schizophr. Res. 166, 261–268 (2015).
García-Mieres, H., De, Jesús-Romero, R., IDENTITY group, Ochoa, S. & Feixas, G. Beyond the cognitive insight paradox: Self-reflectivity moderates the relationship between depressive symptoms and general psychological distress in psychosis. Schizophr. Res. 222, 297–303 (2020).
Ayesa-Arriola, R. et al. The dynamic relationship between insight and suicidal behavior in first episode psychosis patients over 3-year follow-up. Eur. Neuropsychopharmacol. 28, 1161–1172 (2018).
Lopez-Morinigo, J.-D. et al. Insight and risk of suicidal behaviour in two first-episode psychosis cohorts: Effects of previous suicide attempts and depression. Schizophr. Res. 204, 80–89 (2019).
Upthegrove, R., Marwaha, S. & Birchwood, M. Depression and schizophrenia: cause, consequence, or trans-diagnostic issue? Schizophr. Bull. 43, 240–244 (2017).
Pijnenborg, G. H. M. et al. Differential effects of antipsychotic drugs on insight in first episode schizophrenia: data from the European First-Episode Schizophrenia Trial (EUFEST). Eur. Neuropsychopharmacol. 25, 808–816 (2015).
Lam, K. C. K. et al. Metacognitive training (MCT) for schizophrenia improves cognitive insight: a randomized controlled trial in a Chinese sample with schizophrenia spectrum disorders. Behav. Res. Ther. 64, 38–42 (2015).
Sheehan, D. V. et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J. Clin. Psychiatry 59(Suppl 20), 22–33 (1998).
World Health Organization. The ICD-10 Classification of Mental and Behavioural Disorders: Diagnostic Criteria for Research (World Health Organization, 1993).
Wechsler, D. The psychometric tradition: developing the wechsler adult intelligence scale. Contemporary Educ. Psychol. 6, 82–85 (1981).
Lopez-Morinigo, J.-D. et al. Study protocol of a randomised clinical trial testing whether metacognitive training can improve insight and clinical outcomes in schizophrenia. BMC Psychiatry 20, 30 (2020).
Soriano-Barceló, J. Insight assessment in psychosis and psychopathological correlates: validation of the Spanish version of the Schedule for Assessment of Insight - Expanded Version. Eur. J. Psychiatry 30, 55–65 (2016).
Kemp, R. & David, A. S. Insight and compliance. Chronic mental illness. in Treatment Compliance and the Therapeutic Alliance (ed. Blackwell, B.) Vol. 5, 61–84 (Gordon and Breach, 1997).
Sanz, M., Constable, G., Lopez-Ibor, I., Kemp, R. & David, A. S. A comparative study of insight scales and their relationship to psychopathological and clinical variables. Psychol. Med. 28, 437–446 (1998).
Gutiérrez-Zotes, J. A. et al. Spanish adaptation of the Beck Cognitive Insight Scale (BCIS) for schizophrenia. Actas Esp. Psiquiatr. 40, 2–9 (2012).
Brett-Jones, J., Garety, P. & Hemsley, D. Measuring delusional experiences: a method and its application. Br. J. Clin. Psychol. 26, 257–265 (1987).
O’Connor, J. A. et al. Can cognitive insight predict symptom remission in a first episode psychosis cohort? BMC Psychiatry 17, 54 (2017).
Balzan, R. P., Delfabbro, P. H., Galletly, C. A. & Woodward, T. S. Over-adjustment or miscomprehension? A re-examination of the jumping to conclusions bias. Aust N Z J Psychiatry 46, 532–540 (2012).
Gil, D., Fernández-Modamio, M., Bengochea, R. & Arrieta, M. Adaptation of the Hinting Task theory of the mind test to Spanish. Revista de Psiquiatría y. Salud Mental 5, 79–88 (2012).
Peralta, V. & Cuesta, M. Validation of positive and negative symptom scale (PANSS) in a sample of Spanish schizophrenic patients. Psychiatry Res. 53, 31–40 (1994).
Kay, S. R., Fiszbein, A. & Opler, L. A. The positive and negative syndrome scale (PANSS) for Schizophrenia. Schizophr. Bull. 13, 261–276 (1987).
Sarró, S. et al. Cross-cultural adaptation and validation of the Spanish version of the Calgary Depression Scale for Schizophrenia. Schizophr. Res. 68, 349–356 (2004).
Jablensky, A. Living in a Kraepelinian world: Kraepelin’s impact on modern psychiatry. Hist Psychiatry 18, 381–388 (2007).
Craddock, N. & Owen, M. J. The Kraepelinian dichotomy—going, going… but still not gone. Br. J. Psychiatry 196, 92–95 (2010).
Peralta, V. & Cuesta, M. J. How many and which are the psychopathological dimensions in schizophrenia? Issues influencing their ascertainment. Schizophr. Res. 49, 269–285 (2001).
van Os, J. & Kapur, S. Schizophrenia. Lancet 374, 635–645 (2009).
Endicott, J., Spitzer, R. L., Fleiss, J. L. & Cohen, J. The global assessment scale. A procedure for measuring overall severity of psychiatric disturbance. Arch Gen. Psychiatry 33, 766–771 (1976).
Üstün, T. B. (ed.). Measuring Health and Disability: Manual for WHO Disability Assessment Schedule WHODAS 2.0 (World Health Organization, 2010).
Carlson, J. et al. Adaptation and validation of the quality-of-life scale: satisfaction with life domains scale by Baker and Intagliata. Compr. Psychiatry 50, 76–80 (2009).
Baker, F. & Intagliata, J. Quality of life in the evaluation of community support systems. Eval Program Plann. 5, 69–79 (1982).
Cannon-Spoor, H. E., Potkin, S. G. & Wyatt, R. J. Measurement of premorbid adjustment in chronic schizophrenia. Schizophr. Bull. 8, 470–484 (1982).
Reitan, R. M. Validity of the trail making test as an indicator of organic brain damage. Percept. Mot. Skills 8, 271–276 (1958).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences. 2nd edn. (L. Erlbaum Associates, 1988).
Mishra, P., Singh, U., Pandey, C. M., Mishra, P. & Pandey, G. Application of student’s t-test, analysis of variance, and covariance. Ann. Card. Anaesth. 22, 407–411 (2019).
Acknowledgements
We are very grateful for all participants. We acknowledge departmental support from the Department of Psychiatry, Universidad Autónoma de Madrid (Madrid, Spain) and Instituto de Investigaciones Sanitarias-Fundación Jiménez Díaz (Madrid, Spain). This study was co-funded by the Universidad Autónoma de Madrid and European Union via the Intertalentum Project Grant-Marie Skłodowska Curie Actions (GA 713366), which was awarded to the Principal Investigator (JDLM). The funders had no role in hypothesis generation, study design, decision to publish or manuscript writing. The funders had no conflict of interest in relation to the study subject.
Author information
Authors and Affiliations
Contributions
J.D.L.M. was the Principal Investigator of this randomised controlled trial. J.D.L.M., A.S.E.M., M.L.B., P.J.E.A., V.G.R.R., S.S.A., L.M.I., L.M.L., D.C., S.O., E.B.G. and A.S.D. contributed in the process of protocol design, hypothesis generation, manuscript preparation and fulfilled the criteria for authorship. J.D.L.M. wrote the first draft. M.L.B., S.O., E.B.G. and A.S.D. conceived of the study, participated in its design, implemented the project and contributed to the interpretation of results. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
41537_2022_316_MOESM2_ESM.doc
Table S2. Psychoeducation group: Baseline differences between those who attended 4 sessions (n=16) and those who did not (n=22)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lopez-Morinigo, JD., Martínez, A.SE., Barrigón, M.L. et al. A pilot 1-year follow-up randomised controlled trial comparing metacognitive training to psychoeducation in schizophrenia: effects on insight. Schizophr 9, 7 (2023). https://doi.org/10.1038/s41537-022-00316-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41537-022-00316-x
- Springer Nature Limited