Abstract
Early Palaeozoic sites with soft-tissue preservation are predominantly found in Cambrian rocks and tend to capture past tropical and temperate ecosystems. In this study, we describe the diversity and preservation of the Cabrières Biota, a newly discovered Early Ordovician Lagerstätte from Montagne Noire, southern France. The Cabrières Biota showcases a diverse polar assemblage of both biomineralized and soft-bodied organisms predominantly preserved in iron oxides. Echinoderms are extremely scarce, while sponges and algae are abundantly represented. Non-biomineralized arthropod fragments are also preserved, along with faunal elements reminiscent of Cambrian Burgess Shale-type ecosystems, such as armoured lobopodians. The taxonomic diversity observed in the Cabrières Biota mixes Early Ordovician Lagerstätten taxa with Cambrian forms. By potentially being the closest Lagerstätte to the South Pole, the Cabrières Biota probably served as a biotic refuge amid the high-water temperatures of the Early Ordovician, and shows comparable ecological structuring to modern polar communities.
Similar content being viewed by others
Main
Early Palaeozoic sites with soft-tissue preservation1 provide a wealth of information on the evolution of past life and enhance our understanding of previous ecosystems2,3, but are unequally distributed in time and space. While approximately 100 assemblages with soft-tissue preservation4 have been described from the Cambrian, around 30 are known from the Ordovician5,6,7,8,9,10,11,12,13,14,15,16,17, and only a few Lagerstätten are discovered in Early Ordovician rocks4.
The distribution of Early Palaeozoic Lagerstätten is also palaeogeographically skewed, as approximately 97% of discovered biotas represent tropical and temperate ecosystems within 65° north and south of the palaeoequator4. This pattern is particularly true for the Ordovician, where very few Lagerstätten are known from polar environments4. Among the most famous Ordovician Lagerstätten, the Soom Shale (Upper Ordovician, South Africa), Big Hill (Late Ordovician, United States) and Winneshiek (Middle Ordovician, United States) biotas are indicative of tropical ecosystems11,12,13 (Extended Data Fig. 1). The Liexi Fauna, along with the Fenxiang and Tonggao biotas from the Early Ordovician of China, represent tropical to warm temperate ecosystems5,6,7 (Extended Data Fig. 1). The Afon Gam (Early Ordovician, United Kingdom), Castle Bank (Middle Ordovician, United Kingdom) and Llanfawr (Middle Ordovician, United Kingdom) biotas provide valuable information on cold to temperate Ordovician communities near the polar circle8,9,10 (Extended Data Fig. 1). The Early Ordovician Fezouata (Morocco) and Klabava (Czech Republic) biotas are the rare exceptions to this pattern, providing insights into strictly polar ecosystems15,16 (Extended Data Fig. 1). Taken together, all these sites exhibit a mix of typical Cambrian and later Palaeozoic taxa, and suggest that marine assemblages were in transition between two early biodiversification events, the Cambrian Explosion and the Great Ordovician Biodiversification Event18,19.
Considering the rarity of Ordovician Lagerstätten (Extended Data Fig. 1) and their skewed palaeogeographic distribution (Extended Data Fig. 1), the discovery of new biotas with soft-tissue preservation beyond the aforementioned palaeogeographic zones and environments is crucial for expanding our understanding of this time period and gaining better insights into the factors driving the rise of animal diversity on Earth. In this study, we describe a new fossil assemblage with soft-tissue preservation, the Cabrières Biota, from the Early Ordovician of southern Montagne Noire, France. The taxonomic diversity of this fossil biota is described, and the preservation of the fossils is investigated. The recent findings are then discussed in light of other Early Ordovician Lagerstätten. This newly discovered biota is of particular importance as it is a close Ordovician Lagerstätte to the contemporaneous South Pole (Extended Data Fig. 1), constituting a cornerstone for understanding ancient polar ecosystems and their evolution.
Results and discussion
Stratigraphy and environmental context
The Early Ordovician Cabrières Biota is a newly discovered assemblage from the southern Montagne Noire, France (Extended Data Fig. 2). During the Early Ordovician, the Montagne Noire was an open marine environment located in the Southern Hemisphere at high polar latitudes on the margin of the supercontinent Gondwana20 (Extended Data Fig. 1). The biota is preserved in stratigraphically equivalent layers to the Landeyran Formation, but more to the east than traditional localities, specifically within the Apatokephalus incisus trilobite biozone21,22,23,24, which dates it to an upper Floian age25 (Fl3; Extended Data Fig. 2). The Landeyran Formation corresponds to an offshore environment deposited in a transgressive phase26,27, succeeding the sandy shoreface to upper offshore Foulon Formation24,27. However, a proper investigation based on recent knowledge of mud deposition28 is needed to properly frame the sedimentary context. Soft-tissue preservation occurs within an interval of 1 m thickness (Extended Data Fig. 2), located 15 m above the base of the Landeyran Formation (Extended Data Fig. 2).
Faunal content
The biota contains numerous taxa that exhibit biomineralization (Fig. 1 and Extended Data Fig. 3). These include animals such as molluscs (14%), trilobites (12%), brachiopods (9%), hyoliths (7%) and cnidarians (6%) (Extended Data Fig. 3). Trilobites are primarily represented by the genera Ampyx (Fig. 1a), Asaphellus and calymenine trilobites (possibly Colpocoryphe) (Fig. 1e), which is in accordance with deposition in an open marine offshore environment29. Gastropods can be found in association with elongated tubes probably representing the enigmatic cnidarian (possible conulariid) Sphenothallus (Fig. 1b). Biomineralized conulariid cnidarians showing a quadrilateral aperture and phosphatized body are also preserved (Fig. 1c). Articulated brachiopods, mostly orthids, are abundant in the Cabrières Biota and can be observed either attached to possible leptomitid sponges with long monaxons30 (Fig. 1d, and Extended Data Figs. 4 and 5) or randomly positioned near trilobites and possible non-biomineralized bivalved arthropods (Fig. 1e). Hyoliths are also present, although they are often poorly preserved. In one instance, a hyolith preserves possible traces of internal organs, probably representing the gut (Fig. 1f). An interesting feature of the Cabrières Biota is the rarity of echinoderms, which are represented by three specimens only (Extended Data Fig. 3).
a, Trilobite of the genus Ampyx (UCBL-FSL713598). b, Gastropods associated with a tube-like structure, probably the conulariid Sphenothallus (UCBL-FSL713599). c, Biomineralized conulariid cnidarian (UCBL-FSL713600). d, Articulated brachiopods attached to a possible leptomitid sponge (UCBL-FSL713601). e, Assemblage formed of articulated brachiopods (centre), flattened carapaces probably of bivalved arthropods (centre left and right) and a calymenine trilobite cranidium (left; UCBL-FSL713602). f, A hyolith with possible internal organs (UCBL-FSL713603). Scale bars represent 4 mm in a and e, 1 cm in b and d, 5 mm in c, and 2 mm in f.
In addition to trilobites, brachiopods, cnidarians, gastropods and hyoliths, the Cabrières Biota is characterized by a prevalence of sponges and branching algae constituting 26% of all identified fossils (Extended Data Fig. 3). Probable cylindrical demosponges can reach large sizes in excess of 10 cm. Specimen UCBL-FSL713604 is large with well-preserved subelliptical ostia within a thin dermal layer formed of fine fibres that are occasionally visibly spiculate (Fig. 2a and Extended Data Fig. 6). The termination of this fossil is unclear owing to incomplete preservation, obscuring whether it is a branched individual or two individuals close to each other (Fig. 2a). At the distal ends of the specimens, the oscula are not clearly defined (Fig. 2a). Other sponge specimens show detailed preservation (Fig. 2b), and the use of multispectral imaging allows for the differentiation of their soft tissues and skeleton (Fig. 2c). Algae of the Cabrières Biota vary in shape and size (Extended Data Fig. 7), including forms with a thick branching structure (Fig. 2d), delicate branching forms (Fig. 2e) and more intricate morphologies consisting of multiple compact branches and nodes (Fig. 2f). This site also preserves specimens (Fig. 2g) similar to Margaretia from the Burgess Shale, an organism previously attributed to green algae31 but recently reinterpreted as organic tubes of the enteropneust hemichordate Oesia32.
a, A large sponge from the Cabrières Biota, possibly a demosponge (UCBL-FSL713604). b, Sponge (UCBL-FSL713605). c, Same specimen showing clear differentiation between the soft tissues (pink) and the mineralized skeleton (green) under multispectral imaging. d, Thick branching algae (UCBL-FSL713606). e, Thin branching algae (UCBL-FSL713607). f, More complex algae (UCBL-FSL713608). g, Organic tube of an Oesia-like enteropneust hemichordate (=Margaretia; UCBL-FSL713609). Scale bars represent 1 cm in a and f, 5 mm in e, and 3 mm in b–d and g.
The Cabrières Biota also showcases a variety of bivalved arthropod carapaces forming 16% of identified fossils (Figs. 1e and 3a,b, and Extended Data Fig. 8). Most notable are the elongate suboval valves ornamented with very closely spaced, longitudinal striations (Fig. 3a,b), on rare occasions associated with abdominal segments not covered by the carapace (Fig. 3a), which represent a new taxon of phyllocarid crustacean. In addition, numerous fragments of non-biomineralized arthropods are present, including structures resembling chelicerate gnathobases (Fig. 3c) and a spiny appendage that could belong to either Radiodonta or Chelicerata (Fig. 3d). Some fossils of non-biomineralized arthropods exhibit segmented bodies adorned with ornamentation resembling that seen in chelicerates (Fig. 3e,f), sometimes with possible segmented appendages (Fig. 3f and Extended Data Fig. 9). One specimen also preserves what probably is a lunar-shaped eye and a rectangular prosoma (Fig. 3e), both of which are consistent with features seen in eurypterids, synziphosurids or even chasmataspids33,34. The post-prosomal anatomy of this specimen reveals a possible opisthosoma divided into a pre-abdomen and an abdomen (Fig. 3e and Extended Data Fig. 9).
a, Phyllocarid carapace valves ornamented with very closely spaced, longitudinal striations and associated with abdominal segments (UCBL-FSL713609). b, Phyllocarid carapace valve with longitudinal striations preserved near a graptolite (UCBL-FSL713610). c, Possible chelicerate gnathobase (UCBL-FSL713611). d, Spiny arthropod appendage (UCBL-FSL713612). e, Segmented arthropod with chelicerate-like ornamentation and an eye (UCBL-FSL713613). f, Part of a segmented arthropod with chelicerate-like ornamentation and an appendage (UCBL-FSL713614). ap, appendage; co, chelicerate ornamentation; ey, eye; ls, longitudinal striations; sb, segmented body. Scale bars represent 2 mm in a; 8 mm in b, c and f; 5 mm in d and e; and 4 mm in j.
Some vermiform organisms are also present in the Cabrières Biota (∼1% of identified fossils), one of which exhibits external ornamentation consisting of many tiny nodes and preserves gut remains (Fig. 4a). Two other specimens consist of a partially preserved elongated and annulated soft body bearing two thick oval plates (Fig. 4b,c). These plates are approximately 2 mm and 6 mm long in the first and second specimens, respectively, and present a complex internal morphology (Fig. 4b,c) with an outer surface showing some reticulate ornamentation in places where thickness is preserved (Fig. 4d,e). A lateral extension at the base of one of the plates in the first specimen probably represents the remains of the proximal part of an appendage (?lo; Fig. 4b). At a similar position in the second specimen, a strong annulated area ends laterally into a series of lateral outgrowths (Fig. 4d,e) that probably represent spines or appendicules. The combination of a soft annulated body (and potentially appendages) and sclerite plates is characteristic of armoured lobopodians.
a, Unidentified vermiform organism UCBL-FSL713615, with possible remains of the gut and external ornamentation of tiny nodes. b,c, Incomplete armoured lobopodians UCBL-FSL713616 (b) and UCBL-FSL713617 (c) exhibiting two sclerite plates along an elongated soft body with annulations. A lateral extension in b possibly represents remains of the proximal part of a lobopod (?lo). d,e Close-up three-dimensional lateral (d) and top (e) views of part of UCBL-FSL713617, from the dotted box in c. Arrowheads point to lateral outgrowths associated with annulations that could represent spines or lobopod appendicules. an, annulations; gu, gut; lo, lobopod; otn, ornamentation of tiny nodes; ro, reticulate ornamentation; sp, sclerite plates. Scale bars represent 5 mm in a and c, and 1 mm in b and e; note that due to the three-dimensional rotation, no scale bar is given for d and the reader is instead invited to refer to scale bars in c and e.
Preservation mode
Fossils from the Cabrières Biota commonly exhibit brown, red or orange hues and are embedded within a siliciclastic matrix composed of mudstone and siltstone, which can range in colour from blue to green and yellow (Figs. 1–4). Scanning electron microscopy (SEM) backscattered electron and energy-dispersive X-ray (EDX) analyses indicate that the fossils are made of dense, shapeless iron oxide crystals lacking distinct framboids or euhedral minerals (Fig. 5a–c), surrounded by a matrix of aluminium-rich phyllosilicates (Fig. 5c). Synchrotron-based investigations of the chemical speciation of iron using Fe K-edge X-ray absorption near edge structure (XANES) spectroscopy show that iron is present as Fe(III) oxides and hydroxides (Fig. 5d; see Extended Data Fig. 10 for the position of the spectrum). In addition, black films, probably representing carbonaceous material, can be observed on some fossils (Fig. 5a).
a, Backscattered electron microscopy image revealing white iron oxide minerals and some limited black carbonaceous material within the fossils of the Cabrières Biota. b, The iron oxides appear shapeless, lacking distinct framboids or euhedral minerals. c, Semi-quantitative elemental proportions from SEM–EDX analyses indicate that the fossils exhibit a higher iron content compared with their surrounding aluminosilicate matrices. d, Fe K-edge XANES spectroscopy shows that the iron present in the Montagne Noire fossils exists in the form of oxides and hydroxides. e,f, Synchrotron µXRF major-to-trace elemental mapping shows that modern weathering elements such as manganese and arsenic are deposited on the surface of the samples. Scale bars represent 100 µm in a, 50 µm in b and 5 mm in e and f.
The preservation of the Cabrières Biota exhibits similarities with the preservation seen in the Fezouata Biota, including comparable fossil colours and chemical signatures35,36. At least part of the iron oxides and hydroxides in the Cabrières Biota (Fig. 5a–d), such as the Fezouata Shale36, may result from alteration by modern precipitation events because weathering products, such as manganese and arsenic, are deposited on the surface of the samples (Fig. 5e,f and Extended Data Fig. 10). The future collection of an expanded range of fossils will enable a more comprehensive taphonomic analysis of the modes and mechanisms of preservation within the Cabrières Biota and will facilitate comparisons with other Lagerstätten37,38,39,40,41.
Taxonomic and taphonomic importance
Many organisms of the Cabrières Biota are not fully mineralized and exhibit preservation of sclerotized, that is, toughened cuticle, in addition to cuticularized and cellular structures. As such, the Cabrières Biota is considered a Lagerstätte. Although the Montagne Noire region contains numerous fossil sites24 with a wide range of temporal and palaeogeographical distributions, its Ordovician outcrops were previously recognized mainly for their biomineralized elements, such as trilobites20,21,22,23,24,25,26,27,42,43,44, echinoderms44,45,46, molluscs, brachiopods, hyoliths21 and conulariids47 as well as heavily sclerotized organisms such as graptolites48. The discovery of the Cabrières Biota expands the range of preserved tissue types found in the Ordovician of Montagne Noire, revealing entirely soft taxonomic groups such as algae and non-biomineralized animals (Figs. 2–4 and Extended Data Fig. 3).
Preliminary quantifications of the overall diversity within this biota reveal that organisms with biomineralized body walls (for example, brachiopods, echinoderms, trilobites) that do not preserve soft tissues make up approximately 41% of the total diversity. By contrast, over half of the total diversity comprises non-biomineralized organisms, such as bivalved arthropods, chelicerates, lobopodians and hemichordates, or biomineralized animal groups that do preserve soft tissues, such as the figured sponges (Fig. 2a,b). It is worth noting that these percentages are comparable to those of other well-known Lagerstätten from the Early Ordovician, such as the Fezouata Biota, which has approximately 44% of its taxa37,38, preserving only biomineralized remains. Moreover, many organisms in the Cabrières Biota can be fragmentary, which may indicate that they were either exposed to decay for relatively long periods of time or transported by sedimentary flows. Regardless of the processes responsible for such fragmentation, which will require further investigations, similar preservation is also observed in some localities from the Fezouata Biota, in which animals are dominantly fragmentary, with fully articulated organisms being the exception rather than the norm49. Despite the difference in collection efforts between the Fezouata Biota and the Cabrières Biota, which was only recently discovered, the latter still yielded some complete organisms. Many animals are preserved in high detail as well, as exemplified by the longitudinal striations observed on the bivalved arthropod carapaces and the ornamentations on the chelicerates and the worms (Figs. 3a,b,e,f and 4, and Extended Data Figs. 8 and 9).
All animal groups in the Cabrières Biota are known from other Cambrian and Ordovician Lagerstätten, yet the taxonomic composition of the Cabrières Biota is particularly unique for the Early Ordovician. The newly described biota is almost as diverse as the range of clades seen in the Liexi Fauna5 and Fezouata Biota15, yet echinoderms, which are otherwise abundant in the Ordovician, are extremely rare in this biota. This scarcity of echinoderms in the Cabrières Biota is similar to that in the Fenxiang Biota7 and the Klabava Biota16 but differs from that of other Early Ordovician Lagerstätten such as the Leixi Fauna and particularly the Fezouata Shale50,51,52,53,54,55,56 (Extended Data Fig. 3). The Cabrières Biota yields a higher diversity of arthropods compared with the Fenxiang Biota and lacks evidence of nematodes, scalidophorans and corals. Furthermore, there are no bryozoans present in the Cabrières Biota in contrast to the Klabava Biota. The Cabrières Biota preserves an abundance of algae and sponges (Fig. 2a–f and Extended Data Fig. 3), similar to the Afon Gam Biota9, but with a greater number of non-biomineralized arthropods (Fig. 3a–f). The Cabrières Biota provides further evidence that armoured lobopodians (Fig. 4b,c) persisted until at least the Ordovician. Armoured lobopodians were important components of Cambrian ecosystems and are highly abundant in Cambrian Lagerstätten, such as the Chengjiang Biota57, and can be present in Ordovician ecosystems58. With the discovery of the Cabrières Biota, it is becoming clearer that many elements of the classic Cambrian fauna persisted into the Ordovician. Findings such as these are increasingly connecting the Cambrian Explosion and the Great Ordovician Biodiversification Event59 albeit existing differences between them60.
Ecological and evolutionary implications
The Cabrières Biota represents a close Lagerstätte to the Ordovician South Pole (Extended Data Fig. 1). High-latitude marine habitats can offer a range of favourable spatiotemporal conditions supporting high species richness61, and play a crucial role as taxonomic refugia during periods of environmental stress62,63,64. Given the extremely warm waters of the Early Ordovician65, the high-latitude Cabrières Biota would have experienced less extreme temperatures compared with lower-latitude regions, fostering the development of a unique diversity of taxa that had shifted southwards into cooler climatic bands. For instance, trilobite fauna from Montagne Noire shares taxa with Iran such as Taihungshania and Damghanampyx. The latter genus is only known in these two regions, while Taihunghsania also occurs in Turkey, United Arab Emirates and South China66,67,68,69. The Montagne Noire also shares numerous taxa with the Anti-Atlas in Morocco70,71,72,73. This melting pot in the Montagne Noire is restricted to the Lower Ordovician, and faunal affinities become strictly Gondwanan during the Upper Ordovician74,75. The unique taxonomic diversity of the Montagne Noire during the Early Ordovician might have been facilitated by the oceanic circulation permitting taxa to migrate towards the pole from warmer, more stressful, lower latitudes66, and might have been made easy by a possible position of the Montagne Noire in a slightly more eastern location within the polar circle76,77,78.
Some modern polar biotas, similar to the Cabrières Biota, can be dominated by algae and sponges79,80,81,82. Macroalgae possess specific characteristics and adaptations in polar settings, which explain their ecological success in these environments83. Sponges play a key role in the community dynamics of polar settings84 and can attain large sizes85, as was observed in the Cabrières Biota. The prevalence of sponges in the Cabrières Biota cannot be ascribed to environmental factors such as oxygen depletion, even though sponges typically thrive in hypoxic environments. This is because hypoxic environments are characterized by low diversity, which is clearly not the case for the Cabrières Biota preserving a diverse array of organisms, including brachiopods, trilobites, bivalved arthropods, lobopodians, worms, cnidarians, hyoliths and molluscs. The diversity of arthropods in polar ecosystems is comparable to that of less harsh environments in the sub-Arctic and low-Arctic regions86, which could explain the similarities in general arthropod diversity between the Cabrières Biota and nearby Lagerstätten, such as the Fezouata Biota (that is, the presence of trilobites, bivalved arthropods, chelicerates and possibly radiodonts)87,88,89. Cnidarians are also present in modern polar ecosystems90, as is the case in the Cabrières Biota. Their success is related to their wide range of diets and opportunistic behaviour, enabling them to take advantage of the available food sources in these extreme ecosystems90.
The patterns for echinoderms are more complex. Echinoderms can be found in the Arctic and on the Antarctic margins91,92. However, their diversity is lower in these regions compared with other areas such as the Atlantic or Indo-Pacific oceans93. Within a certain polar setting, echinoderms can be locally abundant91,92. The scarcity of echinoderms in the Cabrières Biota could be attributed to its polar settings, among other local factors, especially that other sites from the Early Ordovician of Montagne Noire45 have yielded diverse assemblages of echinoderms. Thus, similarly to modern polar ecosystems, echinoderms did not colonize the entire seafloor in the Montagne Noire. In fact, echinoderms thrive when the diversity of other animal groups is limited52. By adapting to oligotrophic conditions, echinoderms are often found in low-diversity, low-competition assemblages, where the conditions of the water column did not allow for the colonization of other animal groups, a pattern that is also respected in the Fezouata Biota. For instance, when examined at high resolution, echinoderms are abundantly found in levels of specific bathymetry where other animals are not diverse, constituting the building blocks for the high echinoderm diversity in the Fezouata Biota49. It is likely that echinoderms are rarely found in the Cabrières Biota owing to its high diversity and therefore increased competition, unlike nearby environments from the Montagne Noire that were favourable for echinoderm colonization. This pattern would have been accentuated if the bathymetric conditions in the Cabrières Biota were unfavourable for echinoderm colonization. This would explain echinoderm abundance in traditional Montagne Noire localities and their absence in the specific level yielding the Cabrières Biota.
The position of the Cabrières Biota in the polar zone, the preservation of a diverse assemblage, the dominance of sponges and algae, and the habitat selectivity of echinoderms, among other observed patterns such as the absence of bryozoans that are dominantly found in tropical and temperate waters, all support the notion that this assemblage represents a polar ecosystem characteristic of the Early Ordovician. The resemblance between the Cabrières Biota (Fig. 6) and modern polar ecosystems raises questions on how warmer Early Ordovician polar regions65 supported similar niche partitioning and structuring to colder modern-world polar ecosystems. Moreover, considering the global cooling happening after the Early Ordovician, similar ecosystems to the high-latitude Cabrières Biota can be anticipated in low-latitude environments during Middle to Late Ordovician. Further global-scale investigations are required to confirm this trend, despite the presence of local palaeontological evidence suggesting the existence of low-latitude sponge-dominated Lagerstätten in the Late Ordovician of China94. Yet, it is possible to suggest that refugial zones found at high latitudes in the Early Ordovician migrated to lower latitudes during the subsequent Ordovician cooling.
In the foreground, a row of Ampyx (trilobites) and various shelly organisms, including brachiopods and a hyolith (bottom left corner). Behind the trilobites, a lobopodian, a chelicerate, cnidarians (blue), sponges (green), thin branching algae (red and green) and hemichordate tubes (purple), along with some molluscs. Bivalved arthropods inhabit the water column along with graptolites. Credit: Christian McCall (Prehistorica Art).
Methods
The search for Lagerstätten in the Early Ordovician of Montagne Noire (France) has gained momentum over the past decade. In 2018, the first potential soft tissues were discovered, and new discoveries by two authors (E.M. and S.M.), from the ‘écailles de Cabrières’, followed since. The fossiliferous sites are found in outcrops within a 1 km radius of the Cabrières village (Extended Data Figs. 1 and 2). Over 400 fossils have been collected so far, and all are registered and housed under the collection ‘Monceret’ at the University of Lyon (Université Lyon 1, Villeurbanne, France), under the acronym UCBL-FSL.
Collected fossils were examined with a WILD type 308700 (×6.4, ×16 and ×40) binocular microscope. Optical photos were taken with a Canon 800D camera coupled to a Canon MP-E 65 mm macro lens equipped with a polarizing filter. Various lighting conditions, including normal and polarized light, as well as dry and alcohol-submerged photography, were used. Z-stacks were processed using Helicon Focus software and the pyramid function. Three-dimensional surface images were produced for one specimen through an automatic vertical stacking process using a Keyence VHX-7000 digital microscope equipped with a VH-ZOOT Macro lens (×0–50 magnification) connected to a VXH-7020 high-performance 3.19-megapixel complementary metal-oxide semiconductor (CMOS) camera.
Some specimens were further documented using multispectral imaging at the Institute of Earth Science of the University of Lausanne (Switzerland) to see whether certain anatomies are better seen under different light combinations. Reflection and luminescence images in various spectral ranges were collected using a set-up consisting of a low-noise 2.58-megapixel back-illuminated sCMOS camera with high sensitivity from 200 to 1,000 nm, fitted with a UV–VIS–IR 60 mm 1:4 Apo Macro lens (CoastalOptics) in front of which is positioned a filter wheel holding eight interference band-pass filters (Semrock) to collect images in eight spectral ranges from 435 to 935 nm. Illumination was provided by 16 light-emitting diodes, with wavelengths ranging from 365 nm to 700 nm (CoolLED pE-4000), coupled to a liquid light guide fitted with a fibre-optic ring light guide. As such, more than 90 different illumination and detection couples are available, and the resulting greyscale images can be combined into false-colour RGB images to enhance morphological contrasts or reveal details invisible in traditional photography using only visible light. Stacking, image registration of the different couples (excitation/emission of 385/935, 660/775 and 365/571) and production of false-colour RGB composites were performed using ImageJ.
Four specimens were analysed using an FEI Quanta 250 SEM at the Electron Microscopy Facility of the Faculty of Biology and Medicine of the University of Lausanne to investigate the mode of preservation of the Cabrières Biota fossils and their mineralogical composition. The SEM was equipped with backscattered and secondary electron detectors in addition to an EDX analyser. To detect heavy elements such as iron, specimens were analysed uncoated in environmental mode using a 10 keV beam. Elemental percentages (semi-quantifications) were obtained using the associated Bruker Nano Analytics software.
The trace elemental composition of two fossils was further investigated using synchrotron micro-X-ray fluorescence (µXRF) mapping at the PUMA beamline of the SOLEIL synchrotron source (France) to better constrain the differences between matrices and fossils. The incoming monochromatic X-ray beam was focused using Kirkpatrick–Baez mirrors down to a spot size of ∼7 × 5 μm2 (Horizontal × Vertical, full width at half maximum) and set to an energy of 18,500 eV optimized for the excitation of elements from phosphorus to zirconium (K-lines) and from cadmium to uranium (L-lines). The sample was mounted on a scanner stage allowing 150 mm and 100 mm movements (in horizontal and vertical directions, respectively) with micrometre accuracy, and oriented at 60° to the incident beam, producing an effective beam size of ∼10 × 5 μm2 (Horizontal × Vertical) on the sample. XRF data were collected using a SiriusSD silicon drift detector (SGX Sensortech, 100 mm2 active area) oriented at 90° to the incident beam, in the horizontal plane. Two-dimensional spectral images, that is, images for which each pixel is characterized by a full XRF spectrum, were collected with a 60–80 ms dwell time at a 100–200 µm lateral resolution depending on the samples (see figure captions for the precise scanning parameters). The results are shown herein as false-colour RGB overlays of three elemental distributions reconstructed from full spectral decomposition using the batch-fitting procedure of the PyMCA data-analysis software95, with polynomial baseline subtraction, and assuming a Hypermet peak shape. XANES spectroscopy at the Fe K-edge was performed to determine iron speciation. Fe XANES spectra were collected in fluorescence mode in the 7,050–7,550 eV range with energy steps of 5 eV between 7,050 and 7,100 eV, 0.5 eV between 7,100 and 7,200 eV, and 2 eV between 7,200 and 7,250 eV. The count time was set to 2 s per energy step. Energy was calibrated using a reference metallic Fe foil and setting the first inflection point of the Fe K-edge at 7,111 eV.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All the data needed to reproduce this paper are available in the main text and the extended data figures. All material can be accessed at the public collections of the University of Lyon (France).
References
Seilacher, A., Reif, W. E. & Westphal, F. Sedimentological, ecological and temporal patterns of fossil Lagerstätten. Philos. Trans. R. Soc. Lond. B 311, 5–24 (1985).
Allison, P. A. & Briggs, D. E. Exceptional fossil record: distribution of soft-tissue preservation through the Phanerozoic. Geology 21, 527–530 (1993).
Brasier, M. D., Antcliffe, J. B. & Callow, R. H. T. in Taphonomy: Process and Bias Through Time 2nd edn, Vol. 32 (eds P. A. Allison & D. J. Bottjer) Ch. 15, 519–567 (Springer Science, 2011).
Muscente, A. D. et al. Exceptionally preserved fossil assemblages through geologic time and space. Gondwana Res. 48, 164–188 (2017).
Van Roy, P. et al. Ordovician faunas of Burgess Shale type. Nature 465, 215–218 (2010).
Muir, L. A., Ng, T. W., Li, X. F., Zhang, Y. D. & Lin, J. P. Palaeoscolecidan worms and a possible nematode from the Early Ordovician of South China. Palaeoworld 23, 15–24 (2014).
Balinski, A. & Sun, Y. Fenxiang biota: a new Early Ordovician shallow-water fauna with soft-part preservation from China. Sci. Bull. 60, 812–818 (2015).
Botting, J. P., Muir, L. A., Sutton, M. D. & Barnie, T. Welsh gold: a new exceptionally preserved pyritized Ordovician biota. Geology 39, 879–882 (2011).
Botting, J. P., Muir, L. A., Jordan, N. & Upton, C. An Ordovician variation on Burgess Shale-type biotas. Sci. Rep. 5, 9947 (2015).
Botting, J. P. et al. A Middle Ordovician Burgess Shale-type fauna from Castle Bank, Wales (UK). Nat. Ecol. Evol. 7, 666–674 (2023).
Gabbott, S. E., Browning, C., Theron, J. N. & Whittle, R. J. The late Ordovician Soom Shale Lagerstätte: an extraordinary post-glacial fossil and sedimentary record. J. Geol. Soc. 174, 1–9 (2017).
Lamsdell, J. C., LoDuca, S. T., Gunderson, G. O., Meyer, R. C. & Briggs, D. E. A new Lagerstätte from the Late Ordovician Big Hill Formation, Upper Peninsula, Michigan. J. Geol. Soc. 174, 18–22 (2017).
Briggs, D. E., Liu, H. P., McKay, R. M. & Witzke, B. J. The Winneshiek biota: exceptionally well-preserved fossils in a Middle Ordovician impact crater. J. Geol. Soc. 175, 865–874 (2018).
Kimmig, J., Couto, H., Leibach, W. W. & Lieberman, B. S. Soft-bodied fossils from the upper Valongo Formation (Middle Ordovician: Dapingian–Darriwilian) of northern Portugal. Sci. Nat. 106, 1–13 (2019).
Fang, X. et al. The Liexi fauna: a new Lagerstätte from the Lower Ordovician of South China. Proc. R. Soc. B 289, 20221027 (2022).
Kraft, P. et al. Ordovician of the Bohemian Massif. Geol. Soc. Lond. Spec. Publ. 532, 433–464 (2023).
Moysiuk, J., Izquierdo-López, A., Kampouris, G. E. & Caron, J. B. A new marrellomorph arthropod from southern Ontario: a rare case of soft-tissue preservation on a Late Ordovician open marine shelf. J. Paleontol. 96, 859–874 (2022).
Daley, A. C., Antcliffe, J. B., Drage, H. B. & Pates, S. Early fossil record of Euarthropoda and the Cambrian Explosion. Proc. Natl Acad. Sci. USA 115, 5323–5331 (2018).
Stigall, A. L., Edwards, C. T., Freeman, R. L. & Rasmussen, C. M. Coordinated biotic and abiotic change during the Great Ordovician Biodiversification Event: Darriwilian assembly of early Paleozoic building blocks. Palaeogeogr. Palaeoclimatol. Palaeoecol. 530, 249–270 (2019).
Álvaro, J. J., González-Gómez, C. & Vizcaïno, D. Paleogeographic patterns of the Cambrian–Ordovician transition in the southern Montagne Noire (France): preliminary results. Bull. Soc. Geol. Fr. 174, 217–225 (2003).
Courtessole, R., Marek, L., Pillet, J., Ubaghs, G. & Vizcaïno, D. Calymenina, Echinodermata et Hyolitha de l’Ordovicien inférieur de la Montagne Noire (France meridionale) (Société d’Etudes Scientifiques de l’Aude, Carcassonne 1983).
Courtessole, R., Pillet, J. & Vizcaïno, D. Nouvelles données sur la biostratigraphie de l’Ordovicien inférieur de la Montagne Noire. Revision des Taihungshaniidae, de Megistaspis (Ekeraspis) et d’Asaphopsoides (Trilobites) (Société d’Etudes Scientifiques de l’Aude, Carcassonne, 1981).
Courtessole R., Pillet J., Vizcaïno D. & Eschard R. Etude biostratigraphique et sédimentologique des formations arénacées de l’Arenigien du Saint-Chinianais oriental (Hérault) versant sud de la Montagne Noire (France méridionale) (Société d’Etudes Scientifiques de l’Aude, Carcassonne, 1985).
Vizcaïno, D., Alvaro, J. J. & Lefebvre, B. The Lower Ordovician of the southern Montagne Noire. Ann. Soc. Géol. Nord. 8, 213–220 (2001).
Fortey, R. A. Trilobites of the genus Dikelokephalina from Ordovician Gondwana and Avalonia. Geol. J. 46, 405–415 (2011).
Noffke, N. & Nitsch, E. Sedimentology of Lower Ordovician clastic shelf deposits, Montagne Noire (France). Géol. de la France 4, 3–19 (1994).
Vizcaïno, D. & Álvaro, J. J. Adequacy of the Early Ordovician trilobite record in the southern Montagne Noire (France): biases for biodiversity documentation. Earth Environ. Sci. Trans. R. Soc. Edinb. 93, 393–401 (2003).
Saleh, F. et al. The Chengjiang Biota inhabited a deltaic environment. Nat. Commun. 13, 1569 (2022).
Vidal, M. Le modèle des biofaciès à trilobites: un test dans l’Ordovicien inférieur de l’Anti-Atlas, Maroc. C.R. Acad. Sci., Ser. IIa: Sci. Terre Planets 327, 327–333 (1998).
Brayard, A. et al. Unexpected Early Triassic marine ecosystem and the rise of the Modern evolutionary fauna. Sci. Adv. 3, e1602159 (2017).
Conway Morris, S. & Robison, R. A. More soft-bodied animals and algae from the Middle Cambrian of Utah and British Columbia. Univ. Kansas. Paleo. Contrib. 122, 1–48 (1988).
Nanglu, K., Caron, J. B., Conway Morris, S. & Cameron, C. B. Cambrian suspension-feeding tubicolous hemichordates. BMC Biol. 14, 56 (2016).
Schoenemann, B., Poschmann, M. & Clarkson, E. N. Insights into the 400 million-year-old eyes of giant sea scorpions (Eurypterida) suggest the structure of Palaeozoic compound eyes. Sci. Rep. 9, 17797 (2019).
Bicknell, R. D., Amati, L. & Ortega-Hernández, J. New insights into the evolution of lateral compound eyes in Palaeozoic horseshoe crabs. Zool. J. Linn. Soc. 187, 1061–1077 (2019).
Saleh, F. et al. Taphonomic pathway of exceptionally preserved fossils in the Lower Ordovician of Morocco. Geobios 60, 99–115 (2020).
Saleh, F. et al. Insights into soft-part preservation from the Early Ordovician Fezouata Biota. Earth Sci. Rev. 213, 103464 (2021).
Saleh, F. et al. Taphonomic bias in exceptionally preserved biotas. Earth Planet. Sci. Lett. 529, 115873 (2020).
Saleh, F. et al. A novel tool to untangle the ecology and fossil preservation knot in exceptionally preserved biotas. Earth Planet. Sci. Lett. 569, 117061 (2021).
Saleh, F. et al. Probability-based preservational variations within the early Cambrian Chengjiang biota (China). PeerJ 10, e13869 (2022).
Saleh, F., Clements, T., Perrier, V., Daley, A. C. & Antcliffe, J. B. Variations in preservation of exceptional fossils within concretions. Swiss J. Palaeontol. 142, 20 (2023).
Whitaker, A. F., Schiffbauer, J. D., Briggs, D. E., Leibach, W. W. & Kimmig, J. Preservation and diagenesis of soft-bodied fossils and the occurrence of phosphate-associated rare earth elements in the Cambrian (Wuliuan) Spence Shale Lagerstätte. Palaeogeogr. Palaeoclimatol. Palaeoecol. 592, 110909 (2022).
Vidal, M. Quelques Asaphidae (Trilobita) de la Formation de Saint-Chinian, Ordovicien inférieur, Montagne Noire (France): systématique et paléoenvironnements. Geobios 29, 725–744 (1996).
Tortello, M. F., Vizcaïno, D. & Álvaro, J. J. Early Ordovician agnostoid trilobites from the southern Montagne Noire, France. J. Paleontol. 80, 477–495 (2006).
Saleh, F. et al. Contrasting Early Ordovician assembly patterns highlight the complex initial stages of the Ordovician Radiation. Sci. Rep. 12, 3852 (2022).
Vizcaïno, D. & Lefebvre, B. Les échinodermes du Paléozoïque inférieur de Montagne Noire: Biostratigraphie et paléodiversité. Geobios 32, 353–364 (1999).
Nardin, E.New specimens of Lingulocystis Thoral, 1935 (Eocrinoidea, Blastozoa) from the Arenig (Lower Ordovician) of Montagne Noire (southern France): intraspecific morphological variability, stratigraphic, and palaeoecological implications. Ann. Paléontol. 93, 199–214 (2007).
Van Iten, H. & Lefebvre, B. Conulariids from the Lower Ordovician of the southern Montagne Noire, France. Acta Palaeontol. Pol. 65, 629–639 (2020).
Fortey, R. A. A critical graptolite correlation into the Lower Ordovician of Gondwana. Proc. Yorks. Geol. Soc. 58, 223–226 (2011).
Saleh, F., Lefebvre, B., Martin, E. L., Nohejlová, M. & Spaccesi, L. Skeletal elements controlled soft-tissue preservation in echinoderms from the Early Ordovician Fezouata Biota. Geobios 81, 51–66 (2023).
Allaire, N., Lefebvre, B., Martin, E., Nardin, E. & Vaucher, R. in Progress in Echinoderm Palaeobiology (eds Zamora, S. & Rábano, I.) 21–26 (Instituto Geológico y Minero De España, 2015).
Allaire, N. et al. Morphological disparity and systematic revision of the eocrinoid genus Rhopalocystis (Echinodermata, Blastozoa) from the Lower Ordovician of the central Anti-Atlas (Morocco). J. Paleontol. 91, 685–714 (2017).
Lefebvre, B. et al. Palaeoecological aspects of the diversification of echinoderms in the Lower Ordovician of central Anti-Atlas, Morocco. Palaeogeogr. Palaeoclimatol. Palaeoecol. 460, 97–121 (2016).
Lefebvre, B. et al. Exceptionally preserved soft parts in fossils from the Lower Ordovician of Morocco clarify stylophoran affinities within basal deuterostomes. Geobios 52, 27–36 (2019).
Martin, E., Lefebvre, B. & Vaucher, R. in Progress in Echinoderm Palaeobiology (eds Zamora, S. & Rábano, I.) 95–100 (Instituto Geológico y Minero De España, 2015).
Martin, E. L. et al. The Lower Ordovician Fezouata Konservat-Lagerstätte from Morocco: age, environment and evolutionary perspectives. Gondwana Res. 34, 274–283 (2016).
Saleh, F., Lefebvre, B., Hunter, A. W. & Nohejlová, M. Fossil weathering and preparation mimic soft tissues in eocrinoid and somasteroid echinoderms from the Lower Ordovician of Morocco. Microsc. Today 28, 24–28 (2020).
Hou, X. G., et al. The Cambrian Fossils of Chengjiang, China: the Flowering of Early Animal Life (John Wiley & Sons, 2017).
Van Roy, P., Briggs, D. E. & Gaines, R. R. The Fezouata fossils of Morocco; an extraordinary record of marine life in the Early Ordovician. J. Geol. Soc. 172, 541–549 (2015).
Servais, T. et al. No (Cambrian) explosion and no (Ordovician) event: a single long-term radiation in the early Palaeozoic. Palaeogeogr. Palaeoclimatol. Palaeoecol. 623, 111592 (2023).
Saleh, F., Antcliffe, J. B., Lustri, L., Daley, A. C. & Gibert, C. Contrasting Cambrian and Ordovician diversity fluctuations could be resolved through a single ecological hypothesis. Lethaia 56, 1–13 (2023).
Alabia, I. D. et al. Marine biodiversity refugia in a climate‐sensitive subarctic shelf. Glob. Change Biol. 27, 3299–3311 (2021).
Ashcroft, M. B. Identifying refugia from climate change. J. Biogeogr. 37, 1407–1413 (2010).
Stewart, J. R., Lister, A. M., Barnes, I. & Dalén, L. Refugia revisited: individualistic responses of species in space and time. Proc. R. Soc. B 277, 661–671 (2010).
Morelli, T. L. et al. Climate Change refugia and habitat connectivity promote species persistence. Clim. Change Responses 4, 8 (2017).
Trotter, J. A., Williams, I. S., Barnes, C. R., Lécuyer, C. & Nicoll, R. S. Did cooling oceans trigger Ordovician biodiversification? Evidence from conodont thermometry. Science 321, 550–554 (2008).
Fortey, R. A., Heward, A. P. & Miller, C. G. Sedimentary facies and trilobite and conodont faunas of the Ordovician Rann Formation, Ras al Khaimah, United Arab Emirates. GeoArabia 16, 127–152 (2011).
Ghobaldi Pour, M. G., Vidal, M. & Hosseini-Nezhad, M. An Early Ordovician trilobite assemblage from the Lashkarak Formation, Damghan area, northern Iran. Geobios 40, 489–500 (2007).
Ghobadi Pour, M. & Popov, L. E. The Ordovician of the Middle East (Iran, Afghanistan, Pakistan). Geol. Soc. Lond. Spec. Publ. 533, 279–312 (2023).
Pillola, G. L. & Vidal, M. Lower Ordovician Trilobites from SE Sardinia (Italy): a new record of the “Taihungshania bioprovince”. Geobios 81, 67–84 (2023).
Saleh, F. et al. Large trilobites in a stress-free Early Ordovician environment. Geol. Mag. 158, 261–270 (2021).
Laibl, L., Saleh, F. & Pérez-Peris, F. Drifting with trilobites: the invasion of early post-embryonic trilobite stages to the pelagic realm. Palaeogeogr. Palaeoclimatol. Palaeoecol. 613, 111403 (2023).
Laibl, L. et al. Babies from the Fezouata Biota: early developmental trilobite stages and their adaptation to high latitudes. Geobios 81, 31–50 (2023).
Martin, E. L. et al. Biostratigraphic and palaeoenvironmental controls on the trilobite associations from the Lower Ordovician Fezouata Shale of the central Anti-Atlas, Morocco. Palaeogeogr. Palaeoclimatol. Palaeoecol. 460, 142–154 (2016).
Colmenar, J., Villas, E. & Vizcaino, D. Upper Ordovician brachiopods from the Montagne Noire (France): endemic Gondwanan predecessors of Prehirnantian low-latitude immigrants. Bull. Geosci. 88, 153–174 (2013).
Álvaro, J. J., Colmenar, J., Monceret, E., Pouclet, A. & Vizcaïno, D. Late Ordovician (post-Sardic) rifting branches in the North Gondwanan Montagne Noire and Mouthoumet massifs of southern France. Tectonophysics 681, 111–123 (2016).
Ballèvre, M., Bosse, V., Ducassou, C. & Pitra, P. Palaeozoic history of the Armorican Massif: models for the tectonic evolution of the suture zones. C.R. Geosci. 341, 174–201 (2009).
Catalan, J. R. M., Schulmann, K. & Ghienne, J. F. The Mid-Variscan Allochthon: keys from correlation, partial retrodeformation and plate-tectonic reconstruction to unlock the geometry of a non-cylindrical belt. Earth Sci. Rev. 220, 103700 (2021).
Caroff, M. et al. Relations between basalts and adakitic–felsic intrusive bodies in a soft-substrate environment: the South Ouessant Visean basin in the Variscan belt, Armorican Massif, France. Can. J. Earch Sci. 53, 441–456 (2016).
Smetacek, V. & Nicol, S. Polar ocean ecosystems in a changing world. Nature 437, 362–368 (2005).
Downey, R. V., Griffiths, H. J., Linse, K. & Janussen, D. Diversity and distribution patterns in high southern latitude sponges. PLoS ONE 7, e41672 (2012).
Michel, C. in Arctic Biodiversity Assessment: Status and Trends in Biodiversity (eds Meltofte, H., Josefson, A.B. & Payer, D.) Ch. 14, 487–527 (Arctic Council, 2013).
Stratmann, T. et al. Habitat types and megabenthos composition from three sponge-dominated high-Arctic seamounts. Sci. Rep. 12, 20610 (2022).
Karsten, U. et al. Physiological responses of polar benthic algae to ultraviolet radiation. Botanica Marina 52, 639–654 (2009).
Dayton, P. K., Robilliard, G. A., Paine, R. T. & Dayton, L. B. Biological accommodation in the benthic community at McMurdo Sound, Antarctica. Ecol. Monogr. 44, 105–128 (1974).
McClintock, J. B., Amsler, C. D., Baker, B. J. & Van Soest, R. W. Ecology of Antarctic marine sponges: an overview. Integr. Comp. Biol. 45, 359–368 (2005).
Matheson, P. & McGaughran, A. How might climate change affect adaptive responses of polar arthropods? Diversity 15, 47 (2022).
Saleh, F. et al. New fossil assemblages from the Early Ordovician Fezouata biota. Sci. Rep. 12, 20773 (2022).
Laibl, L. et al. Early developmental stages of a Lower Ordovician marrellid from Morocco suggest simple ontogenetic niche differentiation in early euarthropods. Front. Ecol. Evol. 11, 1232612 (2023).
Potin, G. J. M., Gueriau, P. & Daley, A. C. Radiodont frontal appendages from the Fezouata Biota (Morocco) reveal high diversity and ecological adaptations to suspension-feeding during the Early Ordovician. Front. Ecol. Evol. 11, 1214109 (2023).
Orejas, C., Gili, J., López-González, P. J. & Arntz, W. Feeding strategies and diet composition of four Antarctic cnidarian species. Polar Biol. 24, 620–627 (2021).
O’Loughlin, P. M., Paulay, G., Davey, N. & Michonneau, F. The Antarctic region as a marine biodiversity hotspot for echinoderms: diversity and diversification of sea cucumbers. Deep Sea Res. Part II Top. Stud. Oceanogr. 58, 264–275 (2011).
Smirnov, A. V. in Echinoderms Through Time (eds David, B., Guille, A., Feral, J. P. & Roux, M.) 135–143 (CRC Press, 1994).
Stöhr, S., O’Hara, T. D. & Thuy, B. Global diversity of brittle stars (Echinodermata: Ophiuroidea). PLoS ONE 7, e31940 (2012).
Botting, J. P. et al. Flourishing sponge-based ecosystems after the end-Ordovician mass extinction. Curr. Biol. 27, 556–562 (2017).
Solé, V. A., Papillon, E., Cotte, M., Walter, P. & Susini, J. A multiplatform code for the analysis of energy-dispersive X-ray fluorescence spectra. Spectrochim. Acta Part B 62, 63–68 (2007).
Scotese, C. R. Ordovician plate tectonic and palaeogeographical maps. Geol. Soc. Lond. Spec. Publ. 532, 91–109 (2023).
Cocks, L. R. M. & Torsvik, T. H. Ordovician palaeogeography and climate change. Gondwana Res. 100, 53–72 (2021).
Acknowledgements
We thank A. Mucciolo and R. Abi Habib for their assistance during SEM analyses. We also thank D. Vizcaïno, C. Goujon and M. Goujon for their assistance during fieldwork. We are grateful to SOLEIL Synchrotron for the provision of beamtime and to A. Logghe and S. Sanchez for help at the PUMA beamline. We also thank L. Parry for participating in discussions during his visit to the University of Lausanne. This paper is a contribution to the International Geoscience Programme (IGCP) Project 735—Rocks and the Rise of Ordovician Life (Rocks n’ROL). F.S. thanks the Faculty of Geoscience and Environment of the University of Lausanne and the Swiss National Science Foundation (SNF; Ambizione grant, number PZ00P2_209102) for funding. L. Laibl was supported by the programme Dynamic Planet Earth of the Czech Academy of Sciences (StrategieAV21/30) and by the institutional support RVO 67985831 of the Institute of Geology of the Czech Academy of Sciences. M.N. was supported by an internal grant from the Czech Geological Survey (number 311410), part of the Strategic Research Plan of the Czech Geological Survey (DKRVO/CGS 2023–2027). H.B.D., S.L. and J.B.A. are funded by an SNF Sinergia grant (number CRSII5_198691) awarded to A.C.D. and three other principal investigators. L. Lustri and F.P.-P. were funded during the early stages of this project through an SNF project grant (number 205321_179084) awarded to A.C.D. V.J. is funded by an SNF Ambizione grant (PZ00P2_193520). G.J.-M.P. is supported by the Canton de Vaud. B.L. is funded by the project ECO-BOOST of the French National Research Agency (ANR-22-CE01-0003).
Funding
Open access funding provided by University of Lausanne.
Author information
Authors and Affiliations
Contributions
F.S., B.L. and M.N. did fieldwork in 2018 upon invitation from E.M., and E.M. and S.M. found the fossils after extensive fieldwork over 3 years, did the initial fossil identification and donated the samples to the University of Lyon. F.P.-P., G.J.-M.P., L. Laibl and V.J. helped with the inventory of the collection. Fossil examination included all authors, particularly L. Lustri and P.G. F.P.-P., L. Lustri, S.L., V.J. and G.J.-M.P. took photographs of the fossils. F.S. examined specimens using electron microscopy, A.V. applied multispectral imaging on selected samples, and P.G., S.S. and E.B. carried out synchrotron analyses. All authors interpreted and discussed the results. F.S. wrote the paper and made the figures with the help of all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Ecology & Evolution thanks Khadija El Hariri, Dongjing Fu and Julien Kimmig for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
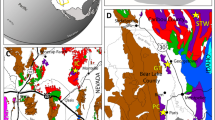
Extended Data Fig. 1 Distribution of Ordovician Lagerstätten.
(a) Early Ordovician palaeomap showing some of the significant Ordovician Lagerstätten, in addition to the newly discovered Cabrières Biota. (b) Stratigraphic formations yielding soft-tissue preservation during the Ordovician. The Cabrières Biota from the Landeyran Formation is the close to the Ordovician South Pole. It should be noted that some of the plotted sites in (a) date from the Middle or Upper Ordovician and not from the Early Ordovician. The paleomap is modified from Scotese96 but different reconstructions97 show rather comparable paleogeographic distributions. Dapingien (Dapin), Hirnantian (Hirn).
Extended Data Fig. 2 Geographic and stratigraphic context.
(a) Geographic position of the Cabrières Biota in the Montagne Noire in southern France, (b) within a 1 km radius around the Cabrières village. Map data ©2022 Google. The Cabrières Biota is discovered in the late Floian (Fl3) Landeyran Formation (c). (d) Photograph of the outcrop that yielded most soft-tissue preservation. (e) Geological map of the Montagne Noire showing the position of the new Lagerstätte in the southern part of the complex.
Extended Data Fig. 3 Taxonomic abundances in the Cabrières Biota and their comparison with the Early Ordovician Fezouata Biota.
The Cabrières Biota showcases a notably higher abundance of algae and sponges, while echinoderms are remarkably scarcer in comparison to the Fezouata Biota. Other animal taxa display relatively comparable representation between the two Lagerstätten. It is important to note that this data is highly probable to evolve with forthcoming fossil discoveries. Hence, it should be used only as an initial comparative reference. The Fezouata Biota dataset derives from the Marrakesh collections.
Extended Data Fig. 4 Additional views of the possible leptomitid sponge UCBL-FSL713601 shown in Fig. 1d.
(a) Optical photograph under polarised light of the entire specimen. (b) Close-up view under polarised light from the box in (a) showing projected longitudinal spicules. Scale bars represent 1 cm in (a), and 5 mm in (b).
Extended Data Fig. 5 Another possible leptomitid sponge, associated to the thin branching algae UCBL-FSL713607.
(a) Optical photograph under polarised light of the entire specimen. (b) Close-up view under polarised light from the box in (a) showing longitudinal skeletal elements. Scale bars represent 1 cm in (a), and 1 mm in (b).
Extended Data Fig. 6 Additional views of the large sponge UCBL-FSL713604 shown in Fig. 2a.
(a) Optical photograph of the entire organism. (b) Close-up view of one side of the organism, from the box in (a). (c) Close-up view of the osculum from the box in (b). (d, e) Close-up views under polarised light showing the skeletal framework, from the corresponding boxes in (c). Scale bars represent 1 cm in (a) and (b), 5 mm in (c), and 1 mm in (d) and (e).
Extended Data Fig. 7 Additional views of the algae shown in Fig. 2.
(a) Optical photograph of the thick branching algae UCBL-FSL713606 shown in Fig. 2d. (b) Close-up view under polarised light of basal branches, from the box in (a). (c) Optical photograph under polarised light of the thin branching algae UCBL-FSL713607 shown in Fig. 2e. (d) Close-up view under polarised light from the box in (c) highlighting textural differences between algae (right) and sponges (left). (e) Optical photograph under low angle and polarised light of the more complex algae UCBL-FSL713608 shown in Fig. 2f. (f) Close-up view under polarised light from the box in (e) highlighting branched clumps of flat kidney-shaped segments. Scale bars represent 5 cm in (a), (b), (c) and (f), 1 mm in (d), and 1 cm in (e).
Extended Data Fig. 8 Phyllocarid crustaceans from the Cabrières Biota.
(a, b) Optical photograph (a) and interpretative line drawing (b) of the counterpart of UCBL-FSL713609 shown in Fig. 3a. The specimen contains two left valves ornamented with very closely spaced, longitudinal striations. The bottom valve (white) is associated with a few abdominal segments. Fossil remains located anteriorly to the top valve (light blue) may represent a rostral plate. (c, d) Optical photograph (c) and interpretative line drawing (d) of the left valve UCBL-FSL713610 shown in Fig. 3b. Abbreviations: al, anterodorsal lobe; as, abdominal segments; cc, cephalic carina; co, carapace ornamentation; mdp, median dorsal plate; rp, rostral plate. Scale bars represent 5 mm.
Extended Data Fig. 9 Additional images and interpretative drawings of the chelicerates in Fig. 3e, f.
(a, b) Optical photograph (a) and interpretative line drawing (b) of the chelicerate UCBL-FSL713613. The presence of a prosoma lacking enlarged axial nodes on opisthosomal tergites, reduced genal spines, and an opisthosoma that is largest at the third and fourth tergite, likely suggest chasmataspid affinities. (c) Close up of the eye and the ornamentation. (d–g) Optical photographs under polarised light of the part (d) and the counterpart (f), and corresponding interpretative line drawings (e, g) of the chelicerate UCBL-FSL713614. Green highlights ventral features. Pale purple shows dorsal features. The sample displays a carapace, three tergites, and a possible appendage. The presence of a possible metastoma suggest non-arachnid dekatriatan affinities. (h, i) Close-up photographs showing the ornamentation. Abbreviations: appendage (ap); (ax) axis; cardiac lobe (cdl); doublure (db); ornamentation (or); eye (ey); metastoma (m); and tergite (t). Scale bars represent 2 mm in (c)s and (i), 3 mm in (h), 5 mm in (a) and (b), and 8 mm in (d), (e), (f), and (g).
Extended Data Fig. 10 Synchrotron-based X-ray fluorescence major-to-elemental mapping of the Cabrières Biota.
(a) Optical photograph under polarised light of the assemblage UCBL-FSL713602 shown in Fig. 1e. (b, c) False-colour overlays of manganese (red), iron (green) and potassium (blue) (b), and arsenic (red), iron (green) and potassium (blue) (c) distributions from the box in (a). Acquisition parameters: 200 μm steps, 80 ms dwell time, 51,100 pixels. (d) Optical photograph under polarised light of sponge UCBL-FSL713618. (e) Close-up view from the box in (d). (f, g) False-colour overlays of manganese (red), iron (green) and potassium (blue) distributions from the corresponding boxes in (e) and (f). Acquisition parameters: 200 μm steps, 60 ms dwell time, 6,150 pixels in (f); 100 μm steps, 60 ms dwell time, 4,800 pixels in (g). The white circle in (g) locates the XANES analysis shown in Fig. 4d. (h) Average µXRF spectra and main elemental contributions from the corresponding numbered boxes in c (156 pixels) and f (208 pixels). Scale bars represent 1 cm in (a), (b), (c) and (d), 5 mm in (e) and (f), and 1 mm in (g).
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Saleh, F., Lustri, L., Gueriau, P. et al. The Cabrières Biota (France) provides insights into Ordovician polar ecosystems. Nat Ecol Evol 8, 651–662 (2024). https://doi.org/10.1038/s41559-024-02331-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-024-02331-w
- Springer Nature Limited
This article is cited by
-
Highly resolved taphonomic variations within the Early Ordovician Fezouata Biota
Scientific Reports (2024)
-
Inherited challenges in the pursuit of an academic career abroad
Nature Ecology & Evolution (2024)