Abstract
Anterior cervical discectomy and fusion (ACDF) and total cervical disc replacement (TDR) are considered effective treatments for patients with cervical degenerative disc disease (CDDD). An indirect meta-analysis including 19 randomized controlled trials (5343 patients) was conducted to compare the clinical outcomes of ACDF with TDR. Primary outcomes including functional indicators (NDI [neck disability index] score, neurological success and patient satisfaction), secondary outcomes including surgical outcomes (operation time, blood loss and length of stay) and secondary surgical procedures (secondary surgery at an adjacent level, secondary surgery at the index level, secondary surgery at both levels, removal, reoperation, revision and supplemental fixation) were included in the study. TDR using the Bryan disc was associated with a greater improvement in NDI score than ACDF (MD = −5.574, 95% CrIs [credible intervals] −11.73–−0.219). For neurological success, the Bryan (odds ratio [OR] = 0.559, 95% CrIs 0.323–0.955) and Prestige (OR = 0.474, 95% CrIs 0.319–0.700) discs were superior to ACDF. However, no differences in the patient satisfaction rate were shown between TDR and ACDF. For patients with CDDD, ACDF using allograft and a plate is most effective for determining the surgical parameters. Moreover, TDR using the ProDisc-C, Mobi-C, Prestige and Bryan discs are good choices for improving functional outcomes and reducing secondary surgeries.
Similar content being viewed by others
Introduction
Anterior cervical discectomy and fusion (ACDF) is considered the traditional standard operation and is widely performed to treat cervical degenerative disc disease (CDDD)1, 2. ACDF, which was described by Smith and Robinson3, may relieve pain significantly by improving nerve function and providing a stable biomechanical environment for the cervical operative segment4. However, approximately one-quarter of ACDF patients underwent reoperation procedures within ten years5.
Total cervical disc replacement (TDR) recently became an alternative to ACDF. TDR may provide normal structure and range of motion to the treated disc level. Moreover, adjacent segment disease is further reduced. However, TDR may lead to complications including heterotopic ossification, erosion of the replacements and increased flexibility of the adjacent cervical disc level.
To date, although several meta-analyses have compared the clinical effectiveness of total TDR and total ACDF, the outcomes were controversial. Whereas some results indicated that TDR was significantly superior to ACDF6,7,8 regarding several clinical outcomes, other results showed no significant difference between the two treatments9,10,11. Additionally, the clinical effectiveness among TDR using different replacements and ACDF using different implants remain unresolved.
The aim of this study was to perform an indirect meta-analysis to estimate the relative effectiveness of each type of TDR or ACDF using different implants on surgical parameters including operative time, blood loss and length of stay, functional indicators including the Neck Disability Index (NDI) score, Short Form 36 (SF-36) Physical Component Score (PCS), SF-36 Mental Component Score (MCS), Visual Analogue Scale (VAS) neck pain score, VAS arm pain score, neurological success, patient satisfaction, return-to-work status and secondary surgical procedures including secondary surgery at an adjacent level, both levels, the index level and removal, reoperation, revision, and supplemental fixation.
Results
Included studies
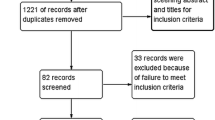
Three thousand six hundred and sixty nine abstracts were retrieved from electronic searches. After duplicates were removed, the titles and abstracts of 2181 studies were screened. Finally, nineteen trials12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30 including twenty-four comparisons between TDRs and ACDF, were included in the review for data extraction and were included in the meta-analysis. The study selection process is shown in the PRISMA flow diagram (Fig. 1).
Basic study characteristics
The characteristics of the included studies are shown in Table 1. In total, eight trials12, 14, 18, 19, 25, 27,28,29 compared the Bryan disc with ACDF using allograft bone and a plate, five trials13, 22, 25, 26, 28 compared the Prestige disc with ACDF using allograft bone and a plate, five trials17, 23, 24, 28, 30 compared the ProDisc-C disc with ACDF using allograft bone and a plate, three trials16, 20, 21 compared the Mobi-C disc with ACDF using allograft and a plate, and two trials14, 15 compared the Kineflex|C disc with ACDF using allograft bone and a plate.
Seven studies13, 17, 23, 25,26,27, 29 reported NDI scores, nine studies13, 17, 19, 22, 23, 25, 26, 28, 30 reported neurological success rates, six studies16, 17, 20, 24,25,26 reported satisfaction rates, nine studies16, 20, 23,24,25,26,27, 29, 30 reported operation times, seven studies16, 20, 23,24,25, 27, 30 reported blood loss, six studies16, 20, 23, 25, 27, 30 reported length of stay, ten studies12,13,14,15, 18, 19, 21, 22, 27, 29 reported secondary surgery at an adjacent level, eight studies12, 14, 15, 18,19,20,21, 27 reported secondary surgery at the index level, seven studies12, 14, 15, 18, 19, 21, 27 reported secondary surgery at both levels, nine studies13, 17,18,19,20,21,22,23, 25 reported removal rates, fourteen studies12,13,14,15, 17,18,19,20,21,22,23, 25, 27, 29 reported reoperation rates, seven studies13, 17, 19, 22, 23, 25, 27 reported revision rates, and ten studies13, 17,18,19,20,21,22,23, 25, 27 reported supplemental fixation rates.
Risk of bias
The risk of bias of the 18 studies is shown in Fig. 2. A total of six included studies had a low risk of bias for random sequence generation. None of the studies described allocation concealment. Four studies described the blinding participants and personnel. However, the participants and personnel were not blinded in one study. None of the included studies described blinding to outcome assessment. Twelve studies were considered at low risk for incomplete outcome data. Five studies were at high risk of incomplete outcome data. Only two studies were considered low risk for selective reporting.
Assessments of heterogeneity
Heterogeneity for each outcome is shown in Table S1. For length of stay, high heterogeneity (I2 = 89.0%) was shown in the comparisons of the Bryan disc vs. ACDF using allograft and a plate. For NDI scores, moderate heterogeneity (I2 = 55.8%) was shown in the comparisons of Bryan disc vs. ACDF using allograft and a plate, indicating random variation between the investigations by chance. The remaining comparisons of TDR vs. ACDF showed minimal to low heterogeneity for all outcomes.
Functional indicators
NDI score
The network plot of comparisons of NDI score is shown in Fig. 3. In total, 520 patients were assigned to ACDF using allograft and a plate, 166 patients to TDR using the ProDisc-C disc, 139 patients to TDR using the Bryan disc, and 280 patients to TDR using the Prestige ST disc.
Regarding NDI scores, the results showed that TDR with the Bryan disc was significantly more effective than ACDF using allograft bone and a plate (MD = −5.574, 95% CrI −11.73–−0.219) (Table 2).
Neurological success
In total, 1406 patients were assigned to ACDF using allograft and a plate, 754 patients to TDR using the Prestige disc, 342 patients to TDR using the ProDisc-C disc, and 507 patients to TDR using the Bryan disc.
The current results indicated that TDR using the Prestige disc significantly improved neurological success than ACDF using allograft and a plate (OR = 0.474, 95% CrI 0.319–0.700). Moreover, TDR using the Bryan disc was significantly more efficacious in terms of neurological success than ACDF using allograft and a plate (OR = 0.559, 95% CrI 0.323–0.955) (Table S2).
Patient satisfaction
In all, 285 patients were assigned to ACDF using allograft and a plate, 58 patients to TDR using the Prestige disc, 109 patients to TDR using the ProDisc-C disc, and 338 patients to TDR using the Mobi-C disc.
The results showed no significant differences in the satisfaction rates between the pairwise comparisons (Table S3).
Surgical Parameters
Operation time
In total, 547 patients were assigned to ACDF using allograft and a plate, 220 patients to TDR using the ProDisc-C disc, 338 patients to TDR using the Mobi-C disc, 68 patients to TDR using the Prestige ST disc, and 139 patients to TDR using the Bryan disc.
The patients who underwent ACDF using allograft and a plate experienced less operation time than those who underwent TDR using the ProDisc-C disc (MD = 8.368, 95% CrI 1.068–15.63), Mobi-C disc (MD = 16.97, 95% CrI 7.379–26.81), Prestige ST disc (MD = 14.52, 95% CrI 2.62–26.32) and Bryan disc (MD = 30.79, 95% CrI 22.79–39.23), respectively. Additionally, TDR using the Bryan disc was associated with significantly increased operation time than TDR using the ProDisc-C disc (MD = 22.42, 95% CrI 11.65–33.72), Mobi-C disc (MD = 13.82, 95% CrI 1.391–26.77) and Prestige ST disc (MD = 16.27, 95% CrI 1.94–31.18), respectively (Table S4).
Blood loss
In total, 435 patients were assigned to ACDF using allograft and a plate, 220 patients to TDR using the ProDisc-C disc, 338 patients to TDR using the Mobi-C disc, and 83 patients to TDR using the Bryan disc.
The meta-analysis results showed that ACDF using allograft and a plate was significantly associated with less blood loss than TDR using the ProDisc-C disc (MD = 21.81, 95% CrI 10.82–32.56) and Bryan disc (MD = 21.11, 95% CrI 4.333–37.29), respectively. Additionally, TDR using the Mobi-C disc was significantly associated with less blood loss than TDR using the ProDisc-C disc (MD = −23.38, 95% CrI −40.71–−5.79) and Bryan disc (MD = 22.69, 95% CrI 1.136–43.56) (Table S5).
Length of stay
In total, 392 patients were assigned to ACDF using allograft and a plate, 176 patients to TDR using the ProDisc-C disc, 338 patients to TDR using the Mobi-C disc, and 83 patients to TDR using the Bryan disc.
For length of stay, the results showed no significant differences between the pairwise comparisons (Table S6).
Secondary surgical procedures
Secondary surgery at an adjacent level
In total, 1257 patients were assigned to ACDF using allograft and a plate, 433 patients to TDR using the Prestige disc, 603 patients to TDR using the Bryan disc, 365 patients to TDR using the Mobi-C disc, and 135 patients to TDR using the Kineflex|C disc.
ACDF using allograft and a plate showed a significantly higher rate of secondary surgery at an adjacent level than TDR with the Prestige disc (OR = 3.527, 95% CrI 1.396–9.439) and TDR with the Mobi-C disc (OR = 3.197, 95% CrI 1.185–8.908), respectively (Table S7).
Secondary surgery at the index level
In all, 886 patients were assigned to ACDF using allograft and a plate, 547 patients to TDR using the Bryan disc, 503 patients to TDR using the Mobi-C disc, and 135 patients to TDR using the Kineflex|C disc.
The results showed no significant difference between the pairwise comparisons in the rate for rate of secondary surgery at the index level (Table S8).
Secondary surgery at both levels
In total, 822 patients were assigned to ACDF using allograft and a plate, 547 patients to TDR using the Bryan disc, 365 patients to TDR using the Mobi-C disc, and 135 patients to TDR using the Kineflex|C disc.
ACDF using allograft and a plate showed a significantly higher rate of secondary surgery at both levels than TDR with the Mobi-C disc (OR = 3.155, 95% CrI 1–10.78) (Table S9).
Removal
In all, 1016 patients were assigned to ACDF using allograft and a plate, 492 patients to TDR using the Prestige disc, 166 patients to TDR using the ProDisc-C disc, 248 patients to TDR using the Bryan disc, and 503 patients to TDR using the Mobi-C disc.
For the removal rate, TDR using the ProDisc-C disc showed a significantly higher rate of removal surgery than when the Prestige disc was used (OR = 16.9, 95% CrI 1.027–803.6) (Table S10).
Reoperation
In sum, 1511 patients were assigned to ACDF using allograft and a plate, 433 patients to TDR using the Prestige disc, 166 patients to TDR the ProDisc-C disc, 650 patients to TDR using the Bryan disc, 503 patients to TDR using the Mobi-C disc, and 135 patients to TDR using the Kineflex|C disc.
Regarding the reoperation rate, TDR using the Mobi-C disc showed a significantly lower rate of reoperation surgery than ACDF using allograft and a plate (OR = 0.275, 95% CrI 0.103–0.740) (Table S11).
Revision
In all, 812 patients were assigned to ACDF using allograft and a plate, 492 patients to TDR using the Prestige disc, 166 patients to TDR using the ProDisc-C disc, and 266 patients to TDR using the Bryan disc.
The results indicated that TDR with the Prestige disc was significantly associated with a reduced rate of revision surgery than ACDF using allograft and a plate (OR = 0.077, 95% CrI 0.003–0.767). Additionally, TDR using the ProDisc-C disc was also significantly associated with a reduced rate of revision than ACDF using allograft and a plate (OR = 0.037, 95% CrI 0.000–0.629) (Table S12).
Supplemental fixation
In all, 1040 patients were assigned to ACDF using allograft and a plate, 433 patients to TDR using the Prestige disc, 166 patients to TDR using the ProDisc-C disc, 331 patients to TDR using the Bryan disc, and 503 patients to TDR using the Mobi-C disc.
TDR with the Prestige disc was significantly associated with a reduced rate of supplemental fixation surgery than ACDF using allograft and a plate (OR = 0.033, 95% CrI 0.001–0.305). Additionally, TDR with the Mobi-C disc was also significantly correlated with a reduced rate of supplemental fixation surgery than ACDF using allograft and a plate (OR = 0.116, 95% CrI 0.013–0.714) (Table S13).
Ranking of treatments
The ranking results of the treatments are shown in Tables 2 and S2 to S13. For reducing blood loss and operation time, ACDF using allograft and a plate may be the best choice. For reducing length of stay, secondary surgery rates at both levels and at the index level and the reoperation rate, TDR using the Mobi-C disc may be the best option. TDR using the Bryan disc for improving the NDI score and the Prestige disc for increasing neurological success, reducing secondary surgery at an adjacent level, removal, and supplemental fixation, and the ProDisc-C disc for improving satisfaction rate and reducing the revision rate were the best choices, respectively. ACDF ranked second in reducing length of stay but was second to last in reducing the removal rate. Whereas it ranked lowest in improving NDI, neurological success, patient satisfaction and reducing secondary surgery at an adjacent level, both levels and the index level, reoperation, revision and supplemental fixation.
Sensitivity analyses
The results of each outcome were not altered by the sensitivity analyses.
Discussion
All relevant studies available concerning treatment of CDDD patients with TDR using the Prestige/ProDisc-C/Bryan/Mobi-C/Kineflex|C discs and ACDF using allograft and a plate were included in the present meta-analysis. In total, thirteen available outcomes were estimated in the indirect comparison using random-effects models. Six to fourteen studies were included for each outcome.
The ranking results showed that TDR with the Mobi-C disc may be the best choice to reduce the length of stay, secondary surgery rate at both levels, secondary surgery rate at the index level and reoperation rate. TDR using the Bryan disc may be the first choice to improve patient NDI scores. TDR using the Prestige disc may be the best choice to increase the neurological success rate and to reduce secondary surgery rate at an adjacent level, removal surgery rate and supplemental fixation rate. TDR with the ProDisc-C disc may optimally increase the satisfaction rate and reduce the revision rate. Interestingly, the ranking results indicated that ACDF using allograft and a plate may be the best choice only for reducing blood loss and the operation time. However, ACDF using allograft and a plate may be the worst choice for improving the NDI score, neurological success and the satisfaction rate, and for reducing the secondary surgery rate at an adjacent level, secondary surgery rate at both levels, secondary surgery at the index level, reoperation, revision and the supplemental fixation rate. For reducing the removal surgery rate, ACDF using allograft and a plate ranked second to last. That is, for patients with CDDD, TDR using replacements including the Mobi-C disc, Bryan disc, Prestige disc and ProDisc-C disc are better choices than ACDF using allograft and a plate.
The basic characteristics of the meta-analyses comparing TDR and ACDF are shown in Table S14. In total, sixteen published meta-analyses6, 7, 9, 10, 31,32,33,34,35,36,37,38,39,40,41,42 compared TDR using different replacements with ACDF. One published meta-analysis11 was a comparison study between TDR using the Bryan disc and ACDF. The last search date of the seventeen studies was from March 2011 to October 2015.
In all, five meta-analyses32, 34, 35, 39, 41 and the short-term results of one meta-analysis11 indicated that no significant difference existed between TDR and ACDF in improving the NDI score, although another five studies7, 31, 38, 40, 42 and the long-term results of one study11 showed that TDR was superior to ACDF, similar to the meta-analyses. The reasons for these findings may be as follows: (1) number of included studies differed significantly; (2) unfitted data was included in published meta-analysis; e.g., the data of Heller JG 200919 included the NDI improvement from baseline but not NDI score at the last follow-up; (3) some outcome data lacked SD values such as those of Mummaneni P 2007 in Xing D 201339, which were excluded from the current study; and (4) comparison of Nabhan A 2011 in Xing D 201339 was between the ProDisc-C and ACDF using the Solis cage and a titanium plate, which was the only ACDF study included in the comparison.
For neurological success rate, twelve meta-analyses6, 9, 31,32,33,34, 36,37,38,39,40, 42 compared TDR and ACDF. Almost all results of the included studies were in accord with the current study except for Ren C 201437, the short-term results of Boselie TF 201331 and the midterm results of Zhang Y 201542, perhaps because the numbers of patients included in those three studies were significantly less than the current study.
In total, two meta-analyses7, 31 compared TDR with ACDF in terms of patient satisfaction. Muheremu A 20157 indicated that TDR was better than ACDF, which differed from the current study. The reasons for this findings may be that Heller JG 2009, Mummaneni P 2007 and Sun 2008 in Muheremu A 20157 were not included in the current study. Data of the three studies were not identified in the articles.
In total, four meta-analyses6, 9, 11, 32 and five studies6, 9, 11, 32, 33 comparing TDR with ACDF investigated operation time reduction and length of stay separately. The results of these studies were in accord with the current study.
For blood loss, four meta-analyses6, 9, 32, 33 comparing TDR with ACDF and one meta-analysis11 comparing TDR using the Bryan disc with ACDF were published. The results of two studies6, 9 indicated no significant difference existed between TDR and ACDF, which differed from the current study. Two reasons may explain this difference: (1) several studies included in the published meta-analyses were excluded from the current study because only one study was included in the individual treatment comparison. For example, the comparisons of Philips FM 2015 and Coric D 2011 in Luo J 20156 were TDR using the PCM disc vs. ACDF using a CSLP or SLIM LOC anterior plate and the Kineflex|C vs. ACDF using allograft and a plate, and (2) some outcome data lacked SD values such as those of Zhang X 2012 included in Rao M 20159, which were excluded from the current study.
A total of thirteen meta-analyses6, 9,10,11, 32,33,34, 37,38,39,40,41,42 investigated the secondary surgery rate of TDR and ACDF. Almost all the meta-analyses results were in accord with the current study except for four studies10, 38, 40, 42. The results of secondary surgery at an adjacent level reported in two studies10, 40 and those of secondary surgery at the index level reported in three studies38, 40, 42 differed from the current study. The reason for these differences may be the significantly different number of patients included.
In all, two meta-analyses33, 35 described reoperation rates of TDR and ACDF. The results of Luo J35 showed that TDRs was associated with a greater reduction in the reoperation rate than ACDF, which was in accord with the current study. However, the results of Gao Y 201333 showed that no significant difference existed between the two treatments, which differed from the current study. The only results on removal and supplemental fixation rates were also described in Gao Y 201333, which also differed from the current meta-analysis. However, the article did not provide detailed outcome data about reoperation, removal and supplemental fixation. Therefore, the reason for the discrepancy between Gao Y 201333 and the current study could not be discerned. Two studies33, 41 that compared TDR with ACDF investigated revision. The results of those studies were in accord with the current study.
The current meta-analysis has several strengths: (1) pairwise comparisons between ACDF using different implants and TDR using different replacements were conducted for the first time to provide comprehensive treatment information for patients with CDDD, and (2) available evidence was applied to the ranking of treatments to provide suggestions about the best choice for patients for each clinical outcomes. However, this article has some limitations: (1) this study lacks direct comparisons between TDR using different replacements and comparisons between different ACDF methods because only one study comparing outcomes between different TDRs was retrieved and less than 3 pairwise comparisons between different TDRs were included in the network plot for the outcome, (2) the numbers of patients included for each treatment were small, e.g., the sample sizes of the Bryan disc for blood loss and length of stay and the Prestige disc for operation time and satisfaction rate were less than 100; thus, the results presented in the current study should be interpreted cautiously, (3) different follow-up durations and races of patients in the included studies might be potential study confounders, (4) the results of outcomes including the SF-36 PCS, SF-36 MCS, VAS neck pain score, VAS arm pain score and return-to-work status were excluded from the current study and (5) the data of several studies were excluded because only one trial was included in each comparison. Therefore, the results of the current meta-analysis could not be assessed comprehensively.
Conclusion
Available randomized controlled study data show that ACDF using allograft and a plate is superior to TDR in terms of surgical parameters, whereas TDR using the Bryan, Prestige, Mobi-C, and ProDisc-C discs are more effective than ACDF using allograft and a plate in regard to functional parameter improvement and secondary surgical procedure reduction. More head-to-head RCTs that directly compare ACDF using different implants or TDR using different replacements in a pairwise fashion are needed to comprehensively confirm the current results.
Materials and Methods
Inclusion criteria
Patients
Adult patients with symptomatic cervical degenerative disc disease including radiculopathy and/or myelopathy.
Interventions and comparisons
We included each pairwise comparison between total disc replacements (TDRs) using different replacements and anterior cervical discectomy and fusions (ACDFs) using different implants.
Outcomes
The primary outcomes included functional outcomes (NDI, SF-36 PCS, SF-36 MCS, VAS neck pain score, VAS arm pain score, neurological success, patient satisfaction and return-to-work status) and secondary outcomes included surgical parameters (operative time, blood loss and length of stay) and secondary surgical procedures (secondary surgery at an adjacent level, secondary surgery at the index level, secondary surgery at both levels, removal, reoperation, revision and supplemental fixation).
Study design
Randomized controlled trials were included in the study.
Exclusion criteria
-
1.
Patients with tumor, metabolic bone disease or trauma.
-
2.
Lack of a detailed description of the surgical implants, e.g., TDR using the Bryan disc, ACDF using allograft and a plate.
-
3.
Results of outcomes were not synthesized in the network meta-analysis in the following situations: (1) less than 3 different pairwise comparisons were included in the network plot for any outcome and (2) only one study was included in any pairwise comparison.
Search and Selection of Studies
We searched CENTRAL, EMBASE and MEDLINE via Ovid SP on May 6, 2016 with no date/time, language, and document type limitations. Keywords were collected through expert opinion, literature review, controlled vocabulary (Medical Subject Headings = MeSH and Excerpta Medica Tree = EMTREE), and by reviewing the primary search results. Additionally, one study was hand searched through reference checking. The search strategies were developed with the assistance of a medical information specialist as reported in Appendix 1. Search results were de-duplicated in EndNote X5 and then sent to two authors for screening.
Data extraction
Extraction of the available data was conducted by two reviewers (Xu B and Ma JX) separately. Any disagreement regarding the eligibility of data generated between the two reviewers was resolved via discussion among the investigators. Information concerning the author names, publication year, country, search duration, comparison of interventions, sample size, age and gender, number of treated levels, length of follow-up period, and clinical outcomes were extracted.
Risk of bias assessment
The quality of the included studies was estimated using the Cochrane Collaboration tool43 for estimating risk of bias. Items for assessment included sequence generation, allocation concealment, blinding of outcome assessor, incomplete outcome data and selective outcome reporting. Each item was assessed using three degrees including high risk of bias, low risk of bias and unclear.
Data synthesis and analysis
Each treatment effect was estimated using the mean difference (MD) with 95% credible intervals (CrIs) or odds ratio (OR) with 95% CrIs for continuous or dichotomous variables, respectively.
Statistical heterogeneity was estimated for each outcome of each pairwise comparison using I2 values. I2 values larger than 25%, 50% and 75% indicated low, moderate and high heterogeneity, respectively44.
A Bayesian indirect meta-analysis with random-effects models was conducted for each outcome using WinBUGS version 1.4 (UK). The model convergence was estimated using trace plots and the Brooks-Gelman-Rubin statistic; a burn-in of 10,000 followed by another 50,000 iterations was considered appropriate for convergence for each outcome45. The surface under the cumulative ranking area (SUCRA) was calculated to rank each treatment. SUCRA values of 100% and 0% indicate the best treatment and the worst treatment46, respectively.
Next, sensitivity analyses were conducted for each outcome by excluding each study included.
References
Cincu, R., Lorente, F. A., Gomez, J., Eiras, J. & Agrawal, A. Long term preservation of motion with artificial cervical disc implants: A comparison between cervical disc replacement and rigid fusion with cage. Asian J Neurosurg 9, 213–217, doi:10.4103/1793-5482.146608 (2014).
Wu, T. K., Liu, H., Wang, B. Y. & Meng, Y. Minimum four-year subsequent surgery rates of cervical disc replacement versus fusion: A meta-analysis of prospective randomized clinical trials. Orthop Traumatol Surg Res 103, 45–51, doi:10.1016/j.otsr.2016.10.008 (2017).
Anakwenze, O. A., Auerbach, J. D., Milby, A. H., Lonner, B. S. & Balderston, R. A. Sagittal cervical alignment after cervical disc arthroplasty and anterior cervical discectomy and fusion: results of a prospective, randomized, controlled trial. Spine 34, 2001–2007, doi:10.1097/BRS.0b013e3181b03fe6 (2009).
Grasso, G. Clinical and radiological features of hybrid surgery in multilevel cervical degenerative disc disease. Eur Spine J 24(Suppl 7), 842–848, doi:10.1007/s00586-015-4281-7 (2015).
Yanbin, Z., Yu, S., Zhongqiang, C. & Zhongjun, L. Sagittal alignment comparison of Bryan disc arthroplasty with ProDisc-C arthroplasty: a prospective, randomized controlled clinical trial. J Spinal Disord Tech 24, 381–385, doi:10.1097/BSD.0b013e318201855f (2011).
Luo, J. et al. Comparison of artificial cervical arthroplasty versus anterior cervical discectomy and fusion for one-level cervical degenerative disc disease: a meta-analysis of randomized controlled trials. Eur J Orthop Surg Traumatol 25(Suppl 1), S115–125, doi:10.1007/s00590-014-1510-4 (2015).
Muheremu, A., Niu, X., Wu, Z., Muhanmode, Y. & Tian, W. Comparison of the short- and long-term treatment effect of cervical disk replacement and anterior cervical disk fusion: a meta-analysis. Eur J Orthop Surg Traumatol 25(Suppl 1), S87–100, doi:10.1007/s00590-014-1469-1 (2015).
Zou, S. et al. Anterior cervical discectomy and fusion (ACDF) versus cervical disc arthroplasty (CDA) for two contiguous levels cervical disc degenerative disease: a meta-analysis of randomized controlled trials. Eur Spine J 26, 985–997, doi:10.1007/s00586-016-4655-5 (2016).
Rao, M. J. et al. Cervical disc arthroplasty versus anterior cervical discectomy and fusion for treatment of symptomatic cervical disc disease: a meta-analysis of randomized controlled trials. Arch Orthop Trauma Surg 135, 19–28, doi:10.1007/s00402-014-2122-5 (2015).
Yang, B., Li, H., Zhang, T., He, X. & Xu, S. The incidence of adjacent segment degeneration after cervical disc arthroplasty (CDA): a meta analysis of randomized controlled trials. PLoS ONE 7, e35032, doi:10.1371/journal.pone.0035032 (2012).
Zhu, Y. et al. Bryan Cervical Disc Arthroplasty Versus Anterior Cervical Discectomy and Fusion for Treatment of Cervical Disc Diseases: A Meta-analysis of Prospective, Randomized Controlled Trials. Spine (Phila Pa 1976) 41, E733–741, doi:10.1097/brs.0000000000001367 (2016).
Anderson, P. A., Sasso, R. C. & Riew, K. D. Comparison of adverse events between the Bryan artificial cervical disc and anterior cervical arthrodesis. Spine 33, 1305–1312, doi:10.1097/BRS.0b013e31817329a1 (2008).
Burkus, J. K., Traynelis, V. C., Haid, R. W. Jr. & Mummaneni, P. V. Clinical and radiographic analysis of an artificial cervical disc: 7-year follow-up from the Prestige prospective randomized controlled clinical trial: Clinical article. J Neurosurg Spine 21, 516–528, doi:10.3171/2014.6.SPINE13996 (2014).
Coric, D., Cassis, J., Carew, J. D. & Boltes, M. O. Prospective study of cervical arthroplasty in 98 patients involved in 1 of 3 separate investigational device exemption studies from a single investigational site with a minimum 2-year follow-up. Clinical article. J Neurosurg Spine 13, 715–721, doi:10.3171/2010.5.SPINE09852 (2010).
Coric, D. et al. Prospective, randomized, multicenter study of cervical arthroplasty: 269 patients from the Kineflex|C artificial disc investigational device exemption study with a minimum 2-year follow-up: clinical article. J Neurosurg Spine 15, 348–358, doi:10.3171/2011.5.SPINE10769 (2011).
Davis, R. J. et al. Two-level total disc replacement with Mobi-C cervical artificial disc versus anterior discectomy and fusion: a prospective, randomized, controlled multicenter clinical trial with 4-year follow-up results. J Neurosurg Spine 22, 15–25, doi:10.3171/2014.7.SPINE13953 (2015).
Delamarter, R. B. et al. Results at 24 months from the prospective, randomized, multicenter Investigational Device Exemption trial of ProDisc-C versus anterior cervical discectomy and fusion with 4-year follow-up and continued access patients. Sas J 4, 122–128, doi:10.1016/j.esas.2010.09.001 (2010).
Garrido, B. J., Taha, T. A. & Sasso, R. C. Clinical outcomes of Bryan cervical disc arthroplasty a prospective, randomized, controlled, single site trial with 48-month follow-up. J Spinal Disord Tech 23, 367–371, doi:10.1097/BSD.0b013e3181bb8568 (2010).
Heller, J. G. et al. Comparison of BRYAN cervical disc arthroplasty with anterior cervical decompression and fusion: clinical and radiographic results of a randomized, controlled, clinical trial. Spine 34, 101–107, doi:10.1097/BRS.0b013e31818ee263 (2009).
Hisey, M. S. et al. Prospective, Randomized Comparison of Cervical Total Disk Replacement Versus Anterior Cervical Fusion: Results at 48 Months Follow-up. J Spinal Disord Tech 28, E237–243, doi:10.1097/BSD.0000000000000185 (2015).
Jackson, R. J. et al. Subsequent surgery rates after cervical total disc replacement using a Mobi-C Cervical Disc Prosthesis versus anterior cervical discectomy and fusion: a prospective randomized clinical trial with 5-year follow-up. J Neurosurg Spine 24, 734–745, doi:10.3171/2015.8.SPINE15219 (2016).
Mummaneni, P. V., Burkus, J. K., Haid, R. W., Traynelis, V. C. & Zdeblick, T. A. Clinical and radiographic analysis of cervical disc arthroplasty compared with allograft fusion: a randomized controlled clinical trial. J Neurosurg Spine 6, 198–209, doi:10.3171/spi.2007.6.3.198 (2007).
Murrey, D. et al. Results of the prospective, randomized, controlled multicenter Food and Drug Administration investigational device exemption study of the ProDisc-C total disc replacement versus anterior discectomy and fusion for the treatment of 1-level symptomatic cervical disc disease. Spine J 9, 275–286, doi:10.1016/j.spinee.2008.05.006 (2009).
Murrey, D. B. et al. Two-Year Results of a Randomized Controlled Clinical Trial Comparing ProDisc-C and Anterior Cervical Discectomy and Fusion. Sas J 2, 76–85, doi:10.1016/SASJ-2007-0124-RR (2008).
Riew, K. D. et al. Cervical disc arthroplasty compared with arthrodesis for the treatment of myelopathy. J Bone Joint Surg Am 90, 2354–2364, doi:10.2106/JBJS.G.01608 (2008).
Riina, J. et al. Comparison of single-level cervical fusion and a metal-on-metal cervical disc replacement device. Am J Orthop 37, E71–77 (2008).
Sasso, R. C., Smucker, J. D., Hacker, R. J. & Heller, J. G. Clinical outcomes of BRYAN cervical disc arthroplasty: a prospective, randomized, controlled, multicenter trial with 24-month follow-up. J Spinal Disord Tech 20, 481–491, doi:10.1097/BSD.0b013e3180310534 (2007).
Upadhyaya, C. D. et al. Analysis of the three United States Food and Drug Administration investigational device exemption cervical arthroplasty trials. J Neurosurg Spine 16, 216–228, doi:10.3171/2011.6.SPINE10623 (2012).
Zhang, X. et al. Randomized, controlled, multicenter, clinical trial comparing BRYAN cervical disc arthroplasty with anterior cervical decompression and fusion in China. Spine 37, 433–438, doi:10.1097/BRS.0b013e31822699fa (2012).
Zigler, J. E., Delamarter, R., Murrey, D., Spivak, J. & Janssen, M. ProDisc-C and anterior cervical discectomy and fusion as surgical treatment for single-level cervical symptomatic degenerative disc disease: five-year results of a Food and Drug Administration study. Spine 38, 203–209, doi:10.1097/BRS.0b013e318278eb38 (2013).
Boselie, T. F. et al. Arthroplasty versus fusion in single-level cervical degenerative disc disease: a Cochrane review. Spine (Phila Pa 1976) 38, E1096–1107, doi:10.1097/BRS.0b013e3182994a32 (2013).
Gao, F. et al. An Updated Meta-Analysis Comparing Artificial Cervical Disc Arthroplasty (CDA) Versus Anterior Cervical Discectomy and Fusion (ACDF) for the Treatment of Cervical Degenerative Disc Disease (CDDD). Spine 40, 1816–1823, doi:10.1097/BRS.0000000000001138 (2015).
Gao, Y. et al. A meta-analysis comparing the results of cervical disc arthroplasty with anterior cervical discectomy and fusion (ACDF) for the treatment of symptomatic cervical disc disease. J Bone Joint Surg Am 95, 555–561, doi:10.2106/JBJS.K.00599 (2013).
Jiang, H. et al. Cervical disc arthroplasty versus fusion for single-level symptomatic cervical disc disease: a meta-analysis of randomized controlled trials. Arch Orthop Trauma Surg 132, 141–151, doi:10.1007/s00402-011-1401-7 (2012).
Luo, J., Gong, M., Huang, S., Yu, T. & Zou, X. Incidence of adjacent segment degeneration in cervical disc arthroplasty versus anterior cervical decompression and fusion meta-analysis of prospective studies. Arch Orthop Trauma Surg 135, 155–160, doi:10.1007/s00402-014-2125-2 (2015).
McAfee, P. C., Reah, C., Gilder, K., Eisermann, L. & Cunningham, B. A meta-analysis of comparative outcomes following cervical arthroplasty or anterior cervical fusion: results from 4 prospective multicenter randomized clinical trials and up to 1226 patients. Spine 37, 943–952, doi:10.1097/BRS.0b013e31823da169 (2012).
Ren, C., Song, Y., Xue, Y. & Yang, X. Mid- to long-term outcomes after cervical disc arthroplasty compared with anterior discectomy and fusion: a systematic review and meta-analysis of randomized controlled trials. Eur Spine J 23, 1115–1123, doi:10.1007/s00586-014-3220-3 (2014).
Wu, A. M. et al. Minimum 4-year outcomes of cervical total disc arthroplasty versus fusion: a meta-analysis based on prospective randomized controlled trials. Medicine (Baltimore) 94, e665, doi:10.1097/MD.0000000000000665 (2015).
Xing, D. et al. A meta-analysis of cervical arthroplasty compared to anterior cervical discectomy and fusion for single-level cervical disc disease. J Clin Neurosci 20, 970–978, doi:10.1016/j.jocn.2012.03.046 (2013).
Yin, S., Yu, X., Zhou, S., Yin, Z. & Qiu, Y. Is cervical disc arthroplasty superior to fusion for treatment of symptomatic cervical disc disease? A meta-analysis. Clin Orthop 471, 1904–1919, doi:10.1007/s11999-013-2830-0 (2013).
Yu, L., Song, Y., Yang, X. & Lv, C. Systematic review and meta-analysis of randomized controlled trials: comparison of total disk replacement with anterior cervical decompression and fusion. Orthopedics 34, e651–658, doi:10.3928/01477447-20110826-09 (2011).
Zhang, Y. et al. Cervical total disc replacement is superior to anterior cervical decompression and fusion: a meta-analysis of prospective randomized controlled trials. PLoS ONE 10, e0117826, doi:10.1371/journal.pone.0117826 (2015).
Higgins, J. P. T. & Green, S. In The Cochrane Collaboration (www.cochrane-handbook.org, 2011).
Higgins, J. P., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ 327, 557–560, doi:10.1136/bmj.327.7414.557 (2003).
Ge, L., Tian, J. H., Li, L., Wang, Q. & Yang, K. H. Mesh fixation methods in open inguinal hernia repair: a protocol for network meta-analysis and trial sequential analysis of randomised controlled trials. BMJ Open 5, e009369, doi:10.1136/bmjopen-2015-009369 (2015).
Salanti, G., Ades, A. E. & Ioannidis, J. P. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol 64, 163–171, doi:10.1016/j.jclinepi.2010.03.016 (2011).
Acknowledgements
We thank Dr. Xiao-yuan Duan and Dr. Hao-bo Jia for their valuable discussion and suggestion.
Author information
Authors and Affiliations
Contributions
B.X. conducted literature search, determined studies for inclusion and exclusion and wrote the main manuscript text. B.X. and J.-X.M. extracted data from the included studies; B.X., J.-X.M. and X.-L.M. prepared Figures 1–3, J.-H.T. and L.G. contributed data analysis. X.-L.M. conceived the idea of the study. All authors reviewed the manuscript and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Xu, B., Ma, Jx., Tian, Jh. et al. Indirect meta-analysis comparing clinical outcomes of total cervical disc replacements with fusions for cervical degenerative disc disease. Sci Rep 7, 1740 (2017). https://doi.org/10.1038/s41598-017-01865-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-01865-3
- Springer Nature Limited