Abstract
New World primates feature a complex colour vision system. Most species have polymorphic colour vision where males have a dichromatic colour perception and females can be either dichromatic or trichromatic. The adaptive value of high allelic diversity of opsins, a light sensitive protein, found in primates’ eyes remains unknown. Studies revealing the allelic diversity are important as they shed light on our understanding of the adaptive value of differences in the colouration of species and their ecologies. Here we investigate the allelic types found in Pitheciidae, an understudied New World primate family, revealing the diversity of medium/long wavelength sensitive opsins both in cryptic and conspicuous species of this primate family. We found five alleles in Cacajao, six in Callicebinae (i.e. Plecturocebus, Cheracebus, and Callicebus), four in Chiropotes, and three in Pithecia, some of them reported for the first time. Both cryptic and conspicuous species in this group presented high allelic diversity.
Similar content being viewed by others
Introduction
While most terrestrial mammals possess dichromatic colour perception, similar to a red-green colour blind human, primates may exhibit: (1) monochromatic colour vision, which occurs in several nocturnal species; (2) routine dichromatic colour vision in tarsiers and some lemur species; (3) routine trichromatic colour vision, mostly in Old World primates; and (4) polymorphic colour vision in most New World primates where males are obligatory dichromats, whereas females can have either dichromatic or trichromatic colour vision systems1,2,3,4,5. Strepsirrhine primate species have a potential polymorphic colour vision, however, this primate suborder lacks behavioural studies confirming a polymorphic colour vision as possessed by New World primates6, 7. This variation in colour vision in New World primates arises from a single locus for the gene coding the medium-long (M/L) wavelength sensitive opsin on the X chromosome8, 9; that is, males are obligatory dichromats as they are hemizygotes, and females can be either dichromats (i.e. homozygotes) or trichromats (i.e. heterozygotes)10. In New World primates, allelic diversity of the Medium-Long wavelength sensitive opsin results in intraspecific variation in colour vision perception11.
The functional role of opsin polymorphism is still not completely understood; however, both trichromatic and dichromatic colour vision phenotypes are proven to have different behavioural advantages. Dichromatic individuals are better at detecting camouflaged stimuli and seeing in low light levels, whereas ripe food and new leaves are better detected by trichromats12,13,14,15,16. Social signals, observed on bare skin, such as emotional states, sexual signals, threat displays are cited as factors leading to the evolution of routine trichromatic colour vision17. Undoubtedly, bare skin is a relevant source of information, which might not be perceived by individuals with dichromatic colour vision. Even species that possess trichromatic colour vision might have difficulty to differentiate colours if the M/L opsin alleles in a heterozygotic female are separated by less than 10 nm18. Anomalous trichromacy (i.e., impaired colour vision but not complete loss) is frequent in howler monkeys and might be common in highly polymorphic colour vision species19.
Uakaries, genus Cacajao, are Neotropical primate species occurring in the upper Amazonian region and they are represented by two contrasting colour phenotypes, one red-faced (i.e. Cacajao calvus) and the other black-faced (Cacajao ayresi, Cacajao hosomi, Cacajao melanocephalus)20. Uakaris are seed-eating specialist species living in groups with up to 200 individuals in a fission-fusion society21,22,23,24,25. Cacajao was, until recently, a major lineage not investigated regarding its colour vision. It is now known that Cacajao calvus has high allelic diversity of opsins26. The bare face of the bald red-uakari is thought to be related to communication among group members. Its simple one coloured face could allow for efficient communication through facial movements and expressions27. Importantly, the red colourful display on the bald uakari’s face may present variations in haemoglobin pigmentation, which might indicate emotional states or health status17, 28. In addition, the bald uakari’s vision was recently reported as being highly polymorphic with a six functional alleles for the medium-long wavelength sensitive opsin. This makes the species an interesting subject to test the importance of reddish displays on the evolution of colour vision. Interestingly, the cryptic black-faced congeneric (i.e. Cacajao melanocephalus, Cacajao ayresi and Cacajao hosomi) lack a bare face. To date, nothing is known about the colour vision in this species and this is one of the objectives of the present study.
The genus Chiropotes, also possesses species with red skin exposed on the face (Chiropotes albinasus), whereas other species in the genus do not possess this characteristic29. These medium sized primates are secretive and difficult to observe in the wild; this is why there are few studies on their behaviour30. However, it is known that Chiropotes form fission-fusion groups with highly affiliative behaviour, and males are more gregarious and tolerant towards juveniles than females30, 31. One Chiropotes species (Chiropotes utahickae) was investigated with regards to its colour vision and three M/L opsin alleles were found, also possessing a colour vision similar to other New World primates32.
Pithecia is a cryptic species and lives in small group sizes (i.e. 4 to 6 individuals); however, their diet is similar to Cacajao and Chiropotes 33. In the Pitheciidae, the genus Pithecia presents the highest degree of sexual dichromatism30. This is a relevant factor when studying the polymorphic colour vision system in New World primates, especially considering the importance of this characteristic in relation to visual communication. This genus also possesses a polymorphic colour vision system as observed in other New World primates with three different known alleles for the M/L opsin gene32.
In Callicebinae, the genus Plecturocebus was also found to possess a relatively high number of alleles for M/L opsins. While most New World primates have three types of photopigments for the medium or long wavelength, Plecturocebus moloch have five cone types in a range from 530 to 562 nm11. Despite similarities in the higher allelic diversity, Cacajao and Plecturocebus are contrasting in many ways. Callicebinae is the New World primate subfamily with the greatest number of species (33 recognised species)34, 35. Despite the number of species, it is one of the least studied primate subfamilies36. Titi monkeys are small (1.5 kg) with timid behaviour spending most of the time under umbrella-like canopy, forming family groups commonly of five individuals with a diet consisting of leaves and fruits36,37,38. Some species show countershading coloration with a bright coppery colour. It is assumed that this colouration is not visible to dichromatic colour vision individuals and the role of this conspicuous colouration is still unknown39. Interestingly, only two species have been investigated so far regarding their colour vision: Plecturocebus brunneus and Plecturocebus moloch.
There are three main methods employed to determine colour vision perception; behavioural studies, direct physiological measurements and molecular analysis of opsin genes40, 41. In the behavioural approach, animals are trained to select colour referenced stimuli in discrimination experiments, which evaluate the degree of difficulty to detect the colour stimuli41. In the physiological approach, an electroretinogram where the spectral sensitivity of photoreceptors in the retina is measured or spectrophotometry of in vitro reconstituted photopigments from cDNA are measured8. Molecular analysis is the most widely employed method. From molecular analysis, it is possible to infer the peak of sensitivity of the expressed opsin gene by verifying amino acid changes at specific sites42. The combination of both molecular and ecological data promises to provide new insights on the role of colour vision evolution in primates43.
The maintenance of such a high number of alleles in this family, strongly, suggests that it has an adaptive function. Here we evaluate, qualitatively, the allelic diversity in the family Pitheciidae (Cacajao, Plecturocebus, Cheracebus, Callicebus, Chiropotes, and Pithecia).
Results
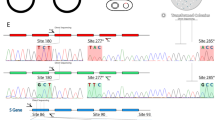
All individuals had their sex confirmed by molecular analysis. By analysing the sites 180 at the exon 3 and sites 277, 285, and 294 at the exon 5 we were able to find six allelic variants of the M/L opsin gene (Tables 1 and 2). We found five alleles in the Cacajao genus (532, 545, 550, 555, 560 λmaxnm). We found C. ayresi with the alleles AFA (532 λmaxnm), AYT (555 λmaxnm) and SYT (560 λmaxnm); C. hosomi with the alleles AFA, AFT (545 λmaxnm) and SYT; C. melanocephalus with AFT, AYT and SYT. Thus, in total, they had a total of five different alleles for the M/L opsin gene. These three Cacajao species have highly pigmented skin. C. calvus, which has exposed red facial skin, had four of these alleles, namely, AFA, AFT, SFT and SYT.
We found 6 alleles in Callicebinae (532, 534, 545, 550, 555, 560 λmaxnm). P. bernhardi with AFT, AYT, and SYT; C. cinerascens with SYT, AYT, and SFT (550 λmaxnm); C. lugens with AFA, SFA, AFT, SFT, AYT and SYT; P. miltoni with SFT and AFT; P. moloch with AFT and SFT; and C. nigrifrons with SFT, AYT, AFT, and SYT. It was not possible to identify the allele in one individual of C. lugens due to double heterozygous sites at 277 and 285.
We found four alleles in Chiropotes (532, 545, 555, 560 λmaxnm). C. albinasus with allele SYT; C. israelita with the alleles AFA, AFT, AYT and SYT; and C. satanas with allele AFA. In Pithecia, three alleles (532, 545, 555 λmaxnm): P. irrorata with AFA, AFT and AYT and P. pithecia with AFT.
All sequences were identified from repeated sequencings from both strands and independent PCR reactions. Sequences are available in GenBank (Accession numbers KY345056-KY345113).
Discussion
The results reported here show that the diversity of M/L opsins found in Cacajao, Callicebus, Plecturocebus, Cheracebus, Chiropotes, and Pithecia is greater than previously reported. Although the samples’ origins and the sample sizes were not appropriate to accurately estimate the opsin frequency, our results contribute to the knowledge of colour vision polymorphism in New World primates. Some allelic variants were found in only one individual per species requiring further studies for confirmation. For instance, the allele SFA was found in one specimen of Cheracebus lugens, and we suggest further confirmation.
We found three alleles in C. ayresi, three in C. hosomi, and three in C. melanocephalus. The different alleles found in this species group indicates that the black-headed uakaris have similar high allelic diversity as found in the polymorphic red-faced uakari26. Further studies of allelic frequencies with black-headed morphs might confirm a highly polymorphic colour vision, suggesting that the red colourful display found in C. calvus is not the ultimate cause for the high number of allelic variants of the Medium/Long wavelength sensitive opsin in New World primates. Thus, challenging the importance of socio-sexual displays in the evolution of routine trichromatic colour vision17. To the best of our knowledge, this is the first report on the opsin diversity of black-headed uakaris (i.e. Cacajao ayresi, Cacajao hosomi, Cacajao melanocephalus).
From physiological studies using electroretinogram flicker photometry, Plecturocebus was reported to have the highest number of opsin alleles within a species (P. moloch), showing five different M/L photopigments in captive individuals11. In a study with wild populations of P. brunneus using a molecular approach, three alleles were found representing the most common types from the aforementioned research (i.e. AFA, AFT, and SYT)44. These three alleles were found for the Callicebinae subfamily in our analysis. In addition, from the two individuals of P. moloch investigated here, the allelic variant SFT was found representing an absorbance peak of 550 λmaxnm. Considering the subfamily Callicebinae only, we have evidence of six different alleles, which increases the known opsin diversity found.
In the genus Chiropotes, in addition to the alleles AFA, AFT, and SYT reported in the literature32, we found an additional variant AYT with a peak spectral sensitivity of 555 λmaxnm in Chiropotes israelita, increasing the number of functional alleles from three to four. Furthermore, the genus Pithecia was reported with the alleles AFA, AFT, and SYT45. We found a variant AYT allele with the sensitivity peak of 555 λmaxnm, increasing the number of known opsin alleles from three to four.
Spectral shifts in opsin sensitivity could also result from mutations in other sites of the M/L opsin gene46. However, the M/L opsin sensitivity peaks are best explained by the “three-site-rule”47. Now that this variation in Pitheciidae is known, one possible approach is to confirm the sensitivity of this protein in vitro. cDNA can be used to produce functioning opsins in vitro by cloning in cultured cells to measure the photopigment sensitivity by spectrophotometric measurements48. Alternatively, electroretinography from Pitheciidae species in captivity would help to confirm the high allelic variation found.
Similarly to Corso et al.26, we found no evidence of routine trichromatic colour vision in the red-faced uakari. Additionally, we also found no evidence of routine trichromatic colour vision in the black-headed uakaris. If routine trichromatic colour vision was found in the red-faced uakari, as in Alouatta 49, this would support the importance of socio-sexual signals in the evolution of colour vision in primates17, which was not the case. However, both cryptic and conspicuous Cacajao morphs share high opsin diversity. This results in an increased number of heterozygotes and potentially more trichromatic females in a group. Similarly in Callicebinae (a subfamily in which most of its skin is covered with fur) shows high allelic diversity again resulting in a high proportion of trichromatic females.
Despite the fact that trichromatic colour vision is best suited to distinguish colour modulations on the skin17, there is evidence that the ability to discriminate red colours in primate vision evolved after red visual traits in primate species50. For example, primate species that are able to discriminate red-green hues have less red fur than dichromate species51. Other species, such as Chiropotes albinasus, have strong red facial marking, but do not have routine trichromatic colour vision. Geographical and ecological factors may also affect the morphology of primate species. Group size and incidence of UV light may lead to more complex faces and dark facial masks27. Thus, variations in facial colouration are expected to be generated from both social and biogeographical pressures.
The exaggerated reddish displays in uakaris and the coppery coloration in Callicebinae could be an evolutionary adaptation to allow dichromatic colour vision individuals to identify social signals. Further studies measuring skin and fur colouration would be useful to understand the role of exaggerated reddish displays in New World primates, for instance, if variants in the red-faced uakari are detectable by dichromatic colour vision individuals. This would indicate why these species show this exaggerated colouration pattern. Future research should focus on the benefits of primate groups possessing both dichromatic and trichromatic individuals rather than focusing only on the consequences of different colour vision on an individual primate.
Methods
DNA extraction
Genomic DNA was extracted from muscle tissue, deposited in museum collections (Table 3), using DNeasy blood and tissue kit (QIAGEN) following the manufacture’s protocol without modifications. The extracted DNA was quantified using a NanoDrop 2000 (Thermo Scientific) to rule out allelic dropouts (i.e. sequencing one chromosome only in heterozygous individuals) by using samples with a DNA concentration higher than 200 picograms/microliter52, 53. We used 1 μL of extracted DNA in buffer AE to obtain 260 nm readings providing the DNA concentration, which were, on average 20000 pg/μl.
Sex Assignment
To confirm the primates’ sex from all samples, Polymerase Chain Reactions (PCR) were conducted using the primers for the Amelogenin in the X chromosome (Forward: 5′–ACCACCAGCTTCCCAGTTTA–3′; and Reverse: 5′–GCTGGGWTAGAACCAAGCTG–3′) for a ~200 bp fragment, and the Y-linked sex-determining region (SRY) (Forward: 5′–AGTGAAGCGACCCATGAACG–3′; and Reverse: 5′–TGTGCCTCCTGGAAGAATGG–3′) for a ~165 bp fragment54. A 25 μL PCR was performed 0.25 μL of TaKaRa Ex Taq® Hot Start Version (1.25 units), with 2.5 μL of × 10 Ex Taq buffer, 4 μL of 2.5 mM dNTP mixture, 0.8 μL of each primer at 100 pmol/μL, 3 μL of template DNA, and 14 μL of pure PCR water to complete the final volume. All reactions were performed with a negative control using pure PCR water instead of DNA template.
The thermal cycling profile followed one cycle of initial denaturation at 94 °C for two minutes; then 40 cycles of denaturation at 94 °C for 30 seconds, annealing at 58 °C for 30 seconds, and elongation at 72 °C for 30 seconds; lastly, a final elongation cycle of 72 °C for 5 minutes. Amplifications were confirmed by electrophoresis in a 1.3% agarose gel, using HyperLadder I™ (Bioline) as reference. The amplification of the Amelogenin, gene present in all samples at the X chromosome, was used as a positive control for all reactions and confirmed through molecular weight. The presence of two bands (i.e. one for the X and one for the Y chromosome) in the agarose gel allowed the assignment of males, while one band determinate females (i.e. one band for the X chromosome) through a benchtop verification54.
Amplification and sequencing
Primers for the Exon 3 were: Forward 5′–CTCCAACCAAAGATGGGCGG–3′; Reverse 5′–ATCACAGGTCTCTGGTCTCTG–3′. Primers for the Exon 5 were: Forward 5′–GAATCCACCCAGAAGGCAGAG–3′; Reverse 5′–ACGGGGTTGTAGATAGTGGCA–3′. PCRs were carried out using 0.25 μL of TaKaRa Ex Taq® Hot Start Version (1.25 units), with 2.5 μL of × 10 Ex Taq buffer, 4 μL of 2.5 mM dNTP mixture, 0.25 μL of each primer at 100 pmol/μL, 0.5 μL of BSA (New England BioLabs) 3 μL of template DNA, and 14.25 μL of pure PCR water to a final 25 μL reaction. Negative controls were employed in all reactions using pure PCR water instead of DNA template. The Exon 3 thermocycling profile consisted of one initial cycle of 98 °C for five minutes; 40 cycles of 98 °C for 30 seconds, 62 °C for 30 seconds decreasing 0.1 °C per cycle, and 72 °C for 30 seconds; followed by a final elongation cycle of 72 °C for 5 minutes. The Exon 5 used the same thermocycling profile, but the annealing temperature was 60 °C decreasing 0.1 °C per cycle. PCR clean-up and sequencing were performed independently by Source Bioscience Sequencing commercial service (Rochdale, UK) using Applied Biosystems 3730 series DNA Analysers.
Genotype determination
Amino acid changes at site 180 in Exon 3, 277 and 285 in Exon 5 are responsible for major shifts in the peak absorbance of the M/L photopigment and are known as the “three-site rule”47, 55. For instance, a change at the site 180 from an Alanine for a Serine shift the absorbance peak in +7 nanometres; at the site 277, a phenylalanine for a Tyrosine shift +8 nanometres, and at the site 285 a change of an Alanine for a Threonine shift the peak of absorbance in +15 nanometres. In the case of an opposite substitution, it is possible to subtract the constant for the site and obtain an approximate sensitivity peak. The site 294 is also known to shift the predicted peak in spectral sensitivity in Atelids and was also verified56. Examining these sites it is possible to identify five major types of photopigments found in New World monkeys. Sequencing from both forward and reverse strands from, at least, two independent PCRs were used to determinate the type of M/L photopigment. Fragments were edited and mapped to the reference opsin gene (GenBank NM000513)57 analysed using Geneious58. Double peaks in the chromatogram were used to assign the individual as a homozygote, heterozygote, or hemizygote employing a specific plugin of Geneious software (Heterozygotes)58. Individuals assigned as double heterozygotes (i.e. double peaks at 180, 277, or 285) were not considered.
References
Jacobs, G. H. Evolution of colour vision in mammals. Philos. Trans. R. Soc. B Biol. Sci. 364, 2957–2967 (2009).
Bowmaker, J. K. Evolution of colour vision in vertebrates. Eye 12, 541–547 (1998).
Moritz, G. L., Ong, P. S., Perry, G. H. & Dominy, N. J. Functional preservation and variation in the cone opsin genes of nocturnal tarsiers. Philos. Trans. R. Soc. London B Biol. Sci. 372 (2017).
Jacobs, G. H., Deegan, J. F., Neitz, J., Crognale, M. A. & Neitz, M. Photopigments and color vision in the nocturnal monkey, Aotus. Vision Res. 33, 1773–1783 (1993).
Jacobs, G. H. & Deegan, J. F. Photopigments underlying color vision in ringtail lemurs (Lemur catta) and brown lemurs (Eulemur fulvus). Am. J. Primatol. 30, 243–256 (1993).
Leonhardt, S. D., Tung, J., Camden, J. B., Leal, M. & Drea, C. M. Seeing red: behavioral evidence of trichromatic color vision in strepsirrhine primates. Behav. Ecol. 20, 1–12 (2009).
Surridge, A. K., Osorio, D. & Mundy, N. I. Evolution and selection of trichromatic vision in primates. Trends Ecol. Evol. 18, 198–205 (2003).
Jacobs, G. H. New world monkeys and color. Int. J. Primatol. 28, 729–759 (2007).
Jacobs, G. H. & Rowe, M. P. Evolution of vertebrate colour vision. Clin. Exp. Optom. 87, 206–216 (2004).
Jacobs, G. H., Neitz, J. & Neitz, M. Genetic basis of polymorphism in the color vision of platyrrhine monkeys. Vision Res. 33, 269–274 (1993).
Jacobs, G. H. & Deegan, J. F. Polymorphic New World monkeys with more than three M/L cone types. J. Opt. Soc. Am. A 22, 2072 (2005).
Morgan, M. J., Adam, A. & Mollon, J. D. Dichromats detect colour-camouflaged objects that are not detected by trichromats. Proceedings. Biol. Sci. 248, 291–5 (1992).
Regan, B. C. et al. Fruits, foliage and the evolution of primate colour vision. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 356, 229–83 (2001).
Verhulst, S. & Maes, F. W. Scotopic vision in colour-blinds. Vision Res. 38, 3387–3390 (1998).
Dominy, N. J. & Lucas, P. W. Ecological importance of trichromatic vision to primates. Nature 410, 363–6 (2001).
Osorio, D. & Vorobyev, M. Colour vision as an adaptation to frugivory in primates. Proc. Biol. Sci. 263, 593–9 (1996).
Changizi, M. A., Zhang, Q. & Shimojo, S. Bare skin, blood and the evolution of primate colour vision. Biol. Lett. 2, 217–21 (2006).
DeMarco, P., Pokorny, J. & Smith, V. C. Full-spectrum cone sensitivity functions for X-chromosome-linked anomalous trichromats. J. Opt. Soc. Am. A 9, 1465 (1992).
Matsushita, Y., Oota, H., Welker, B. J., Pavelka, M. S. & Kawamura, S. Color Vision Variation as Evidenced by Hybrid L/M Opsin Genes in Wild Populations of Trichromatic Alouatta New World Monkeys. 35, 71–87 (2014).
Hershkovitz, P. Uacaries, new world monkeys of the genusCacajao(Cebidae, Platyrrhini): A preliminary taxonomic review with the description of a new subspecies. Am. J. Primatol. 12, 1–53 (1987).
Bowler, M., Knogge, C., Heymann, E. W. & Zinner, D. Multilevel Societies in New World Primates? Flexibility May Characterize the Organization of Peruvian Red Uakaris (Cacajao calvus ucayalii). Int. J. Primatol. 33, 1110–1124 (2012).
Boubli, J. P. et al. A Taxonomic Reassessment of Cacajao melanocephalus Humboldt (1811), with the Description of Two New Species. Int. J. Primatol. 29, 723–741 (2008).
Bowler, M. & Bodmer, R. Social behavior in fission-fusion groups of red uakari monkeys (Cacajao calvus ucayalii). Am. J. Primatol. 71, 976–87 (2009).
Bowler, M. & Bodmer, R. E. Diet and Food Choice in Peruvian Red Uakaris (Cacajao calvus ucayalii): Selective or Opportunistic Seed Predation? Int. J. Primatol. 32, 1109–1122 (2011).
Barnett, A. A. & Brandon-Jones, D. The Ecology, Biogeography and Conservation of the Uakaris, Cacajao (Pitheciinae). Folia Primatol. 68, 223–235 (1997).
Corso, J. et al. Highly polymorphic colour vision in a New World monkey with red facial skin, the bald uakari (Cacajao calvus). Proceedings. Biol. Sci. 283, 620–626 (2016).
Santana, S. E., Lynch Alfaro, J. & Alfaro, M. E. Adaptive evolution of facial colour patterns in Neotropical primates. Proc. Biol. Sci. 279, 2204–11 (2012).
Mayor, P. et al. Proximate causes of the red face of the bald uakari monkey (Cacajao calvus). Open Sci. 2 (2015).
Hershkovitz, P. A preliminary taxonomic review of the South American bearded saki monkeys genus Chiropotes (Cebidae, Platyrrhini), with the description of a new supspecies/Philip Hershkovitz. Fieldiana 27, 1–64 (1985).
Bicca-Marques, J. C. & Heymann, E. W. Evolutionary Biology and Conservation of Titis, Sakis and Uacaris. (Cambridge University Press, doi:10.1017/CBO9781139034210 2013).
Veiga, L. M., Silva, S. S. B. & Ferrari, S. F. Relatives or just good friends? Affiliative relationships among male southern bearded sakis (Chiropotes satanas). In Livro de Resumos XI Congresso Brasileiro de Primatologia 174 (2005).
de Lima, E. M. et al. Polymorphic color vision in captive Uta Hick’s cuxiús, or bearded sakis (Chiropotes utahickae). Am. J. Primatol. 77, 66–75 (2015).
Barnett, A. A., Boyle, S. A. & Thompson, C. L. Pitheciid research comes of age: Past puzzles, current progress, and future priorities. Am. J. Primatol. 78, 487–492 (2016).
Mittermeier, R. a., Rylands, A. B. & Wilson, D. E. Handbook of the Mammals of the World. Vol. 3. Primates (2013).
Byrne, H. et al. Phylogenetic relationships of the New World titi monkeys (Callicebus): first appraisal of taxonomy based on molecular evidence. Front. Zool. 13, 10 (2016).
Bicca-Marques, J. C. & Heymann, E. Ecology and behavior of titi monkeys (genus Callicebus). Evol. Biol. Conserv. Titis, Sakis Uacaris 196–207 (2013).
Veiga, L. M. et al. Evolutionary Biology and Conservation of Titis, Sakis and Uacaris. Evolutionary Biology and Conservation of Titis, Sakis and Uacaris (Cambridge University Press, doi:10.1017/CBO9781139034210 2013).
Mason, W. Social organization of the south american monkey, Callicebus moloch: a preliminary report. Tulane Stud. Zool. 13, 23–28 (1966).
Sumner, P. & Mollon, J. D. Colors of primate pelage and skin: objective assessment of conspicuousness. Am. J. Primatol. 59, 67–91 (2003).
Jacobs, G. H., Neitz, J. & Krogh, K. Electroretinogram flicker photometry and its applications. J. Opt. Soc. Am. A 13, 641 (1996).
Gomes, Ú. R., Pessoa, D. M., Tomaz, C. & Pessoa, V. F. Color vision perception in the capuchin monkey (Cebus apella): a re-evaluation of procedures using Munsell papers. Behav. Brain Res. 129, 153–157 (2002).
Yokoyama, S., Yang, H. & Starmer, W. T. Molecular basis of spectral tuning in the red- and green-sensitive (M/LWS) pigments in vertebrates. Genetics 179, 2037–43 (2008).
Buchanan-Smith, H. M. Recent advances in color vision research. Am. J. Primatol. 67, 393–398 (2005).
Bunce, J. A. et al. Characterization of opsin gene alleles affecting color vision in a wild population of titi monkeys (Callicebus brunneus). Am. J. Primatol. 73, 189–96 (2011).
Boissinot, S. et al. Origins and antiquity of X-linked triallelic color vision systems in New World monkeys. Proc. Natl. Acad. Sci. USA. 95, 13749–54 (1998).
Shyue, S.-K. et al. Molecular Genetics of Spectral Tuning in New World Monkey Color Vision. J. Mol. Evol. 46, 697–702 (1998).
Yokoyama, S. & Radlwimmer, F. B. The Molecular Genetics and Evolution of Red and Green Color Vision in Vertebrates. Genetics 158, 1697–1710 (2001).
Hiramatsu, C., Radlwimmer, F. B., Yokoyama, S. & Kawamura, S. Mutagenesis and reconstitution of middle-to-long-wave-sensitive visual pigments of New World monkeys for testing the tuning effect of residues at sites 229 and 233. Vision Res. 44, 2225–2231 (2004).
Araújo, A. C. et al. Color vision in the black howler monkey (Alouatta caraya). Vis. Neurosci. 25, 243–8 (2008).
Fernandez, A. A. & Morris, M. R. Sexual selection and trichromatic color vision in primates: statistical support for the preexisting-bias hypothesis. Am. Nat. 170, 10–20 (2007).
Kamilar, J. M., Heesy, C. P. & Bradley, B. J. Did trichromatic color vision and red hair color coevolve in primates? Am. J. Primatol. 75, 740–51 (2013).
Beja-Pereira, A., Oliveira, R., Alves, P. C., Schwartz, M. K. & Luikart, G. Advancing ecological understandings through technological transformations in noninvasive genetics. Mol. Ecol. Resour. 9, 1279–301 (2009).
Morin, P. A., Chambers, K. E., Boesch, C. & Vigilant, L. Quantitative polymerase chain reaction analysis of DNA from noninvasive samples for accurate microsatellite genotyping of wild chimpanzees (Pan troglodytes verus). Mol. Ecol. 10, 1835–44 (2001).
Di Fiore, A. A rapid genetic method for sex assignment in non-human primates. Conserv. Genet. 6, 1053–1058 (2005).
Hiramatsu, C. et al. Color-vision polymorphism in wild capuchins (Cebus capucinus) and spider monkeys (Ateles geoffroyi) in Costa Rica. Am. J. Primatol. 67, 447–61 (2005).
Matsumoto, Y. et al. Evolutionary renovation of L/M opsin polymorphism confers a fruit discrimination advantage to ateline New World monkeys. Mol. Ecol. 23, 1799–812 (2014).
Nathans, J., Thomas, D. & Hogness, D. Molecular genetics of human color vision: the genes encoding blue, green, and red pigments. Science (80−.). 232, 193–202 (1986).
Kearse, M. et al. Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28, 1647–9 (2012).
Acknowledgements
Surveys and collections were funded by the Sustainable Development unit of the Brazilian Biodiversity Program – PROBIO/MMA/BIRD/GEF/CNPq, the Zoological Society of San Diego, University of Auckland and National Geographic Waitts grants and CAPES/FAPEAM grant to JPB. Additional support came from FAPEAM, FAPESP, CAPES, CNPq (307951/2012-0) and NSF (Dimensions US-Biota-São Paulo grant). Molecular analyses and four field expeditions were funded by CNPq/FAPEAM SISBIOTA Program (No. 563348/2010-0) and CAPES (AUX n 3261/2013). Permission to conduct fieldwork and to collect tissue samples was granted by IBAMA (License N 005/2005 – CGFAU/LIC) and ICMBio. Felipe Ennes, Malu Messias, Aryanne Clyvia are thanked for providing samples. VDLRG was in receipt of a CAPES postgraduate scholarship Proc. 1213-13-0.
Author information
Authors and Affiliations
Contributions
V.D.L.R.G., J.P.B., and R.J.Y. conceived the study, analysed the data, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Goulart, V.D.L.R., Boubli, J.P. & Young, R.J. Medium/Long wavelength sensitive opsin diversity in Pitheciidae. Sci Rep 7, 7737 (2017). https://doi.org/10.1038/s41598-017-08143-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-08143-2
- Springer Nature Limited