Abstract
Bronchopulmonary dysplasia (BPD), the main consequence of prematurity, has a significant heritability, but little is known about predisposing genes. The aim of this study was to identify gene loci predisposing infants to BPD. The initial genome-wide association study (GWAS) included 174 Finnish preterm infants of gestational age 24–30 weeks. Thereafter, the most promising single-nucleotide polymorphisms (SNPs) associated with BPD were genotyped in both Finnish (n = 555) and non-Finnish (n = 388) replication cohorts. Finally, plasma CRP levels from the first week of life and the risk of BPD were assessed. SNP rs11265269, flanking the CRP gene, showed the strongest signal in GWAS (odds ratio [OR] 3.2, p = 3.4 × 10−6). This association was nominally replicated in Finnish and French African populations. A number of other SNPs in the CRP region, including rs3093059, had nominal associations with BPD. During the first week of life the elevated plasma levels of CRP predicted the risk of BPD (OR 3.4, p = 2.9 × 10–4) and the SNP rs3093059 associated nominally with plasma CRP levels. Finally, SNP rs11265269 was identified as a risk factor of BPD (OR 1.8, p = 5.3 × 10−5), independently of the robust antenatal risk factors. As such, in BPD, a potential role for variants near CRP gene is proposed.
Similar content being viewed by others
Introduction
Bronchopulmonary dysplasia (BPD) is a major complication of prematurity. In the USA alone, about 10,000–15,000 new cases are diagnosed each year1. The potential adverse consequences of BPD include asthma and chronic obstructive pulmonary disease (COPD)2,3,4. Effective prevention of BPD does not exist, and even with modern state-of-the-art therapies the incidence has not fallen1.
In BPD, poor alveolarization and disrupted pulmonary vascularization lead to impaired gas exchange, clinically seen as prolonged need for respiratory support5. Inflammation plays a key role in the pathogenesis of BPD, but the molecular mechanisms remain unknown6, 7. According to studies in twins, genetic factors account for 53–82% of the variance in susceptibility to BPD8, 9. However, identification of predisposing genes has been challenging. Although a suggestive association between SPOCK2 gene and BPD was discovered in the first published genome-wide association study (GWAS) on BPD10, two subsequent GWASs did not reveal any significant associations at the genome-wide level11, 12. Moreover, candidate gene studies have been largely unsuccessful at producing replicable results13, 14. One probable reason for this “missing heritability” is the heterogeneity of study populations. In Hadchouel et al.’s study, the infants were of either Caucasian or African origin. Wang et al.’s population consisted of four identified ethnic groups, and Ambalavanan et al.’s study included both Caucasians and African-Americans. Different ethnic groups differ in their allele frequencies, thus complicating genetic analyses.
The population of Finland is genetically relatively homogeneous and has therefore been used extensively in genetic studies15, 16. Furthermore, neonatal practices vary little in Finland due to standardized treatment protocols. This is of importance when studying a trait such as BPD, the incidence of which is affected by neonatal treatment practices.
In the present investigation, we conducted a GWAS on BPD in a Finnish population. To make the results more generalizable, we studied two replication populations from Finland and two populations from Canada and France. We identified a single nucleotide polymorphism (SNP), rs11265269, as an independent risk factor of BPD.
Methods
Study design and study populations
A flow chart of the study is shown in Fig. 1. The total numbers of BPD cases and controls in the entire study were 319 and 798, respectively.
Inclusion criteria for all infants included in the study were (1) gestational age (GA) <31 wk and (2) no major congenital malformations. The discovery population used for the GWAS (step 1 of the study, n = 174) and the two internal replication populations (first internal replication, step 2 of the study, n = 326 and second internal replication, step 4 of the study, n = 229) consisted of Finnish infants collected prospectively at the five university hospitals in Finland. The external replication populations (step 3 of the study, n = 388) comprised infants from Canada and France. To augment the power of the GWAS study, two control infants were chosen for each BPD case. Only one infant from each sibling pair was selected. For details of patient selection, see Supplementary Information. Clinical characteristics of the population in the GWAS are shown in Table 1, and those of the replication populations are shown in Supplementary Tables S1 and S2. The study was approved by the Ethics Committee of Oulu University Hospital, the University of British Columbia Clinical Research Ethics Board, the University of Alberta Ethics Board, and the Comités de Protection des Personnes (CPP) Ile de France VI and IX. Written informed consent was obtained from the infants’ parents. All experiments were performed in accordance with relevant guidelines and regulations.
Diagnostic criteria for BPD
BPD was defined as a continuing need for supplemental oxygen or positive pressure support (including high-flow 5–6 litre per minute via nasal probes) at 36 wks. postmenstrual age (PMA); i.e., moderate-to-severe BPD according to National Institute of Child Health and Human Development (NICHD) criteria17. This criterion was based on twin studies, in which moderate-to-severe BPD is significantly affected by genetic factors whereas mild BPD is not9. The controls were infants without BPD or with mild BPD. According to NICHD criteria, mild BPD is defined as a need for supplementary oxygen or positive pressure support at the age of 28 days but no longer at the age of 36 wks. PMA. No BPD is defined as no need for supplementary oxygen or positive pressure support at the age of 28 days.
Statistical analyses and outline of other methods
Specific details of statistical analyses, genotyping, data processing, imputation and selection of SNPs for replication analyses, and analyses of CRP values, are shown in Supplementary Information. Information about DNA sample preparation is shown in Supplementary Table S3.
Genome-wide SNP genotyping was performed with the Infinium HumanCoreExome BeadChip (Illumina, San Diego, CA, USA). GWAS was used to identify SNPs associated with moderate-to-severe BPD; data processing and statistical analyses were performed with PLINK, v. 1.07 or 1.0918. After quality control, 276,306 SNPs with minor allele frequency >0.01 remained for GWAS. We used a filtering strategy to narrow down the SNPs analyzed for association with BPD in each step (Fig. 1): the most promising GWAS SNPs were analyzed in the internal Finnish replicate, and the most consistent SNPs were further analyzed in two external populations (Caucasian and French African). At the final step, four SNPs were analyzed in the second internal Finnish replicate. Logistic regression under the additive model with GA as a covariate was used to assess associations with BPD in these populations. The SNP selection criteria are listed in the Supplementary Information. At the final stage, we also included small-for-gestational age (SGA, Z-score ≤−2 SD) as a covariate in a logistic regression model to assess how the significant antenatal factors together (GA and SGA) with the SNP with most consistent signal affect the risk of BPD.
Because the SNP with most consistent signal was located near the CRP gene, we analyzed plasma CRP levels for association with BPD and SNP genotypes. Daily CRP levels were available because of the current practice in screening for infection during the first week after very premature birth. The infants included in the plasma CRP studies were a subset of the populations analyzed in the GWAS and the first internal replicate; infants for whom CRP levels could be recorded were included (n = 275; 112 infants from GWAS, 163 infants from first internal replicate). Logistic regression was used to evaluate whether SNP genotypes were associated with maximum and mean CRP level (CRP values above/below median) during the first week of life with PLINK, v. 1.0918. The number of surfactant doses was included as a covariate, because this variable associated with CRP levels (p < 0.001). Association of the BPD incidence or SNP genotypes with CRP levels was assessed by the χ 2 or Kruskal–Wallis test with SPSS Statistics 20.0, IBM Corporation. At the final phase, logistic regression with SPSS was used to evaluate whether CRP levels are significant predictors of BPD using GA and SGA as covariates.
Results
Associations in GWAS
The GWAS was performed in a discovery cohort of 60 cases with moderate-to-severe BPD and 114 controls. GA, gender, proportion of singletons, exposure to antenatal or postnatal steroids or to clinical chorioamnionitis did not differ between the cases and controls. As expected19, 20, both birth weight and birth-weight Z-score were lower in BPD cases compared with controls, suggesting a lower foetal growth rate among BPD infants. Pre-eclampsia was more common in the BPD group (Table 1).
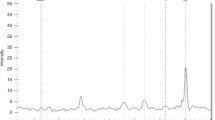
In the GWAS, none of the SNPs reached our stringent genome-wide significance level (p < 1.8 × 10−7), but there were many suggestive associations (Supplementary Figure S1). Results for the SNPs with the strongest suggestive signals (p < 1 × 10−4) are shown in Table 2. The most promising SNP was rs11265269 (odds ratio [OR] 3.22, p = 3.43 × 10−6), which is located between the CRP and DUSP23 genes (approximately 44 and 23 kb upstream of each gene, respectively). Population stratification was minimal (Supplementary Figure S2). The imputed SNPs did not show any association signals that were clearly more significant than those of the genotyped SNPs (Supplementary Figure S3). The SNPs with p < 5 × 10−4 in the GWAS were selected for further genotyping, and are listed in Supplementary Table S4.
Association of rs11265269 with BPD in replication populations
First, we chose 155 SNPs for analysis in the 1st internal replication population (105 cases and 221 controls) (Supplementary Table S1). These SNPs were selected on the basis of the strongest signals in GWAS; in addition, previously BPD-associated SNPs with consistent signals were included. The selection criteria are given in detail in Supplementary Information. Thereafter, the top 41 SNPs, based on GWAS results and 1st internal replication, were further analysed in the external replication populations (Caucasian, n = 312, and French African, n = 76) (Supplementary Table S2 and Supplementary Information). Finally, to increase power and to validate the most promising associations, SNPs rs11265269 (the top SNP identified by GWAS, near CRP/DUSP23), rs1889268 (COL15A1) and rs5999125 (LARGE) were genotyped in a second internal replication population (Finnish, n = 229). Of all the SNPs analysed in the external replication populations, these three SNPs showed the most consistent results and were thus selected for this final genotyping step. Because GA was significantly different between cases and controls in the replication populations, the analyses were performed using logistic regression (LR) analysis with GA as a covariate.
In the first internal replicate (Supplementary Table S4), only one SNP, rs6690148, showed nominal association with BPD (p < 0.05). The top SNP in GWAS, rs11265269 showed an effect similar to that detected in GWAS (OR 1.42, p = 0.097). Of the SNPs analysed further, two showed nominal association in the external replication populations: rs11265269 in the French African population and COL15A1 SNP (rs1889268) in the Caucasian population (Supplementary Table S5). Overall, SNP rs11265269 (the SNP with the strongest signal in GWAS) was the most consistent SNP. Table 3 summarizes the results for this SNP in all the replication populations. In addition to the nominal replication in the French African population, this SNP associated with BPD when the internal replication populations were combined (p = 0.029). With all of the Finnish populations combined, the OR for the minor allele of SNP rs11265269 was 1.84 (p = 2.39 × 10−5) (Table 3); i.e., the effect was smaller than in the initial GWAS, which may be due to overestimated effect size in GWAS. The case–control allele frequency differences for the COL15A1 SNP (rs1889268) were also consistent in all Caucasian populations, although the difference was not significant in the internal replication populations (Supplementary Tables S5 and S6). The results for the LARGE SNP (rs5999125) were more inconsistent (Supplementary Tables S5 and S6). The results for the other SNPs analysed in the replication populations are listed in Supplementary Tables S4 and S5.
We reanalysed the three SNPs with the strongest signals (rs11265269, rs1889268 and rs5999125) in the Finnish populations by excluding the mild BPD infants from the controls. This exclusion did not notably alter the results (Supplementary Table S7). For rs11265269 the OR was 3.28 in GWAS (p = 5.94 × 10−5) and 1.79 in the joint analysis of all Finnish populations (p = 1.41 × 10−3). Altered treatment practices or changes in the diagnostics of BPD (Supplementary Information) could also influence the results. Therefore, the genetic data was reanalysed separately for infants born during 1998–2010 and after 2010. During both time periods the association of rs11265269 with the risk of BPD was maintained in GWAS (OR 4.78 and 2.37, respectively) and in joint analysis of all Finnish populations (OR 1.87 and 1.71, respectively).
SNPs previously associated with BPD: no clear association signals
Next, we analysed, first in our GWAS data, and thereafter the most promising SNPs in the internal replication population, the SNPs in proximity to genes that were previously found to be associated with BPD10, 11. Some of these SNPs showed a nominal association in the GWAS, but none were associated with BPD in the first internal replication population (Supplementary Tables S8 and S9). Consistently, none of the SNPs in previously-associated genes that were analysed further associated with BPD in the Caucasian and French African external replication populations (Supplementary Table S10). Overall, SNP rs2536512 in SOD3 was the most promising. This SNP associated nominally with BPD in GWAS (OR = 0.53, p = 0.017), and showed a similar effect in the first internal replicate (OR = 0.75, p = 0.173) and external Caucasian population (OR = 0.70, p = 0.072). SNP rs2536512 reached p < 0.05 when the Caucasian population was combined with the internal replication population. However, there was no association in the French African replicate or in the second internal replicate (Supplementary Table S10). With all the Finnish populations combined, this SNP had an OR of 0.76 (p = 0.060).
Evaluation of BPD risk and SNPs in the CRP region and in other regions previously associated with plasma CRP levels
Because SNP rs11265269 showed the most promising association with BPD and is located between DUSP23 and CRP genes, we investigated this genomic region in more detail. We determined that rs11265269 displays linkage disequilibrium (LD) with some of the SNPs within or flanking the CRP gene rather than those flanking the DUSP23 gene (Supplementary Figure S4, Fig. 2). This is consistent with 1000 Genomes data (http://www.1000genomes.org). Furthermore, some of the other SNPs in the vicinity of only CRP gene also showed association signals in GWAS. Because many SNPs in this region are known to be associated with serum CRP levels, we further analysed 13 SNPs in the region extending from 69 kb 5′ to 3 kb 3′ of CRP to capture all common variations within this region in the first internal replication population (Supplementary Table S11). In addition to rs11265269, two SNPs (rs3093059 and rs12091403) in this region showed p < 0.05 for associations with BPD when the first internal replication population was combined with the population analysed in the GWAS. Haplotype analysis in this region revealed that three haplotypes were associated with BPD (p < 0.05); these could be discriminated by the three SNPs with p < 0.05 in the single SNP analyses (Table 4, Fig. 2). SNP rs11265269 was the only one of these three SNPs nominally associated with BPD in the Finnish and French African replication populations (Supplementary Table S12).
Linkage disequilibrium plot for single-nucleotide polymorphisms (SNPs) within the region flanking rs11265269, the top SNP in genome-wide association study (GWAS) of bronchopulmonary dysplasia. Pairwise D′ values for each SNP pair in the combined Finnish population (discovery GWAS and first internal replicate) are shown. Darker colors indicate stronger linkage disequilibrium. Of the SNPs analysed, rs2794520 and rs3093059 (D′ 0.86 with rs11265269) are located down- and upstream of the CRP gene, respectively; these SNPs are known to be associated with plasma levels of CRP. SNP rs1129923 is located within the DUSP23 (dual specificity phosphatase 23) gene; rs7519478 and rs4233356 are up- and downstream of DUSP23.
Using our GWAS data, we also analysed 108 SNPs (23 SNPs near CRP and 85 SNPs in other genes) that were previously reported to be associated with CRP levels21,22,23. Some of the SNPs had nominal associations with BPD (p < 0.05). However, in addition to those in the CRP gene region (Supplementary Table S11), none of them fulfilled our criteria to be included for genotyping in the replication sets.
SNPs in the CRP gene region and plasma CRP levels
Next, we analysed whether SNPs near the CRP gene are associated with plasma CRP levels during the first week of life. Altogether, 275 Finnish infants were included. CRP values were obtained from clinical laboratory reports. Infants carrying the BPD-predisposing alleles of SNPs rs11265269 and rs3093059 tended to have higher CRP levels than non-carriers. In LR analysis with the number of surfactant doses as the established significant covariate, SNP rs3093059 was associated with mean plasma CRP concentrations (p = 0.046) but not with maximum plasma CRP concentrations (p = 0.074) (Supplementary Table S13) during the first week of life. SNP rs11265269 did not show association.
Plasma CRP levels during the first week of life associated with BPD
Because the SNP with the strongest association signal in GWAS was located upstream of the CRP gene and other SNPs near CRP (including rs3093059) associated with plasma CRP levels in adult populations22, 23, and also showed nominal associations with BPD, we analysed whether maximum or mean CRP levels during the first week of life could predict development of BPD. Available laboratory data from 275 Finnish preterm infants were studied.
Plasma levels of CRP before 12 hours’ age were mostly low; only 8.4% of the infants had a CRP concentration of more than 1 mg/L; the maximum was 53.4 mg/L. These plasma levels were not associated with the risk of BPD (p = 0.66). We next studied whether maximum and mean plasma CRP concentrations during the 1st week were associated with the risk of BPD. The median values of the maximum and mean CRP levels during the first week were 4.0 mg/L and 1.6 mg/L, respectively. The maximum plasma CRP concentration was above the median in 72.6% of the BPD cases and in 40.6% of the controls (p = 3.0 × 10−6). Mean CRP levels during the 1st week of life exceeded the median in 74.0% of the BPD cases and 41.6% of the controls (p = 2.0 × 10−6) (Table 5). The number of surfactant doses given was associated with these CRP levels (p = 8.86 × 10−8 for the maximum and p = 3.25 × 10−8 for the mean level). In contrast, the length of gestation, prolonged rupture of membranes, chorioamnionitis, cord blood pH or blood culture-proven sepsis (either early- or late-onset during the first week) showed no detectable association.
In LR analysis with GA and SGA as covariates, both the maximum CRP level during the first week and the mean CRP level during the first week were significant predictors of BPD, with ORs of 3.4 and 3.6, respectively (Table 5).
SNP rs11265269 in the CRP gene region is an independent risk factor of BPD
To consolidate the evidence on the role of rs11265269 as a risk factor of BPD, this SNP was analysed together with the significant antenatal risk factors, GA and SGA. Table 6 shows the results of LR analysis that included rs11265269 in the model. This SNP predicted the risk of BPD in each population, independently of the degree of prematurity and SGA status.
Discussion
BPD is a multifactorial disease with high heritability. We conducted a GWAS on a relatively genetically homogenous Finnish population and observed a suggestive association between moderate-to-severe BPD and SNP rs11265269 near the CRP gene. This association was replicated in an independent Finnish population and in population of infants of African descent from France. Furthermore, plasma levels of CRP during the first week of life predicted the development of BPD. Additionally, the known antenatal risk factors and rs11265269 additively predisposed to BPD. Finally, another SNP from the CRP-gene region, SNP rs3093059, associated with plasma CRP levels. No significant association was noted with the SNP rs11265269 and plasma CRP, however.
CRP is a prominent clinical marker of infections and inflammatory disease. Persisting inflammation is a major finding in infants developing BPD. Mechanical ventilation and hyperoxia are known postnatal risk factors for BPD and have been shown to induce inflammation24, 25. CRP is mainly produced in the liver. One of the main ligands of CRP is phosphocholine, a constituent of many bacterial and fungal polysaccharides. Phosphocholine is also present in dead or dying cells26. IL-6 and IL-1 have a major synergistic effect on the induction of CRP expression. High level of CRP activates phagocytic cells, stimulates production of inflammatory cytokines, regulates the classical complement pathway and may cause adverse vascular events when present in excess27.
The plasma levels of CRP are generally low at birth28, and a mechanism of deficient CRP synthesis in fetal liver has been proposed29. In present study, plasma levels of CRP before age 12 hours were mostly low and predicted neither BPD nor sepsis. By contrast, it was the increase in plasma CRP after birth and the maintenance of variably elevated CRP levels during the first week of life that discriminated between infants who will develop BPD and those who will not. This is in line with what earlier has been reported about CRP and BPD, namely that CRP on day 28 and BPD associated30, and that there was a qualitative association between CRP levels from days 0 to 21 and BPD31. In present study, the association between CRP values and BPD was demonstrated already during the first week of life, which is of clinical importance. Our novel finding, that elevated plasma CRP values from the first week of life can be used as a biomarker for an increased risk of BPD, needs to be confirmed in independent studies. The clinical feasibility of CRP makes it superior to the other markers identified with an increased risk of BPD32. Noteworthy the CRP values did not correlate with either early- or late-onset sepsis. This is in consonance with what is known about CRP in the preterm i.e. that both the baseline level of CRP and the response to infection are lower in premature infants compared to the term new-borns. Also the sensitivity and specificity of CRP in diagnosing sepsis are lower at preterm neonates compared to their term peers33. As all other laboratory values, the interpretation of CRP requires the context of other findings and clinical signs of the patient.
Besides inflammation, infection and other environmental factors, genetic polymorphisms influence the expression of CRP. The heritability of plasma CRP is estimated to be 25–40%, and levels of CRP vary among different ethnic groups34, 35. Moreover, the SNPs reported to affect CRP levels vary among different populations23, 36. As such, the lack of an association between rs11265269 and BPD in one of the external replication populations is not surprising. CRP response varies also between species and for instance in mice Crp has generally low expression levels37, thus complicating further confirmation of the findings of this study.
A strength of this study is the relative homogeneity of the population in the GWAS and in the internal replication sets. Because of the Finnish population history with a restricted number of founders, the allelic diversity in Finland is lower than in most other populations38. This compensates for the main limitation of the study, namely the small sample size in the GWAS. However, it is possible that the small sample size limits the overall generalization of our GWAS findings to other study populations. Another limitation is the lack of association between rs11265269 and plasma CRP levels. This SNP, however, is located in the same LD region with rs3093059 and other CRP-associated SNPs, and also haplotypes within the region associated with BPD. We propose that rs11265269 may show milder association with CRP plasma levels that remains undetected in our set of study subjects, or it may associate with the CRP synthesis in lung cells39. Unfortunately, potentially valuable data on expression of CRP in lung compartments was not available. Further credence to association between rs11265269 and moderate-to-severe BPD is given by the finding that the association remained similar when infants with mild BPD were excluded from the controls. Furthermore, as altered treatment practices and changes in diagnostics may have influenced the results, we repeated the analyses in subgroups of infants born in two time periods (before and after 2010) and the association remained similar. It is also possible that the potential predisposing role of rs11265269 in BPD is linked to a gene other than CRP, such as DUSP23, the adjacent gene, encoding dual specificity phosphatase 23. DUSP23 is a phosphatase involved in regulation of cell-cell adhesion40. Its potential link to BPD remains to be investigated.
Although none of the initial GWAS signals were consistently replicated in our replication populations, some of these may represent real association signals. As an example, of the SNPs suggestively associated with BPD in GWAS, those within RASGFR1 (encoding Ras protein specific guanine nucleotide releasing factor 1) may represent real associations, since other SNPs located near this gene showed association signals in a previous GWAS of BPD11. Furthermore, some of the other genes with suggestively associated SNPs could be biologically relevant for the BPD phenotype. As an example, variants near the SGCD gene (encoding sarcoglycan delta) are known to be associated with airway responsiveness, a process that is linked to decline in lung function in subjects with COPD41. Larger studies are required to assess the potential role of these genes in BPD.
Although the heritability of BPD is estimated to be high, consistent and statistically significant associations have not been detected in genomic studies. Current evidence suggests a multiplicity of genes and pathways involved in the pathogenesis of BPD. Besides GWAS, these pathways have been suggested in other large-scale studies, including exome sequencing and transcriptomic studies. It is possible that rare predisposing variants detected by exome sequencing can explain part of the missing heritability of BPD; these rare variants would be missed with the GWAS strategy. In line with our present finding, CRP was one of the top five genes characterizing severe BPD compared to non-BPD infants in a recent exome sequencing study42. In another exome sequencing study, Li et al. tentatively identified 258 genes with rare nonsynonymous variants in patients with moderate-to-severe BPD. These genes represented pulmonary structure and function, morphogenesis of embryonic epithelium, and regulation of the Wnt signalling pathway43. On the other hand, Ambalavanan and co-workers combined a GWAS with pathway analysis and found evidence suggesting involvement of both known (phosphorus oxygen lyase activity) and new (targets of miR-219) pathways in the pathophysiology of BPD12. Bhattacharya et al. performed a genome-wide gene-expression study and reported evidence on accumulation of connective tissue inflammatory mast cells in the lungs of infants who died of BPD44. In addition to further exome and whole-genome sequencing studies to detect rare predisposing variants, additional approaches, such as meta-analyses, pharmacogenomics and experimental studies are required. Identification of BPD-predisposing genes would bring information about the pathogenesis of BPD, ultimately enabling discovery of new individualized strategies against this complex syndrome.
We discovered a suggestive association between rs11265269 polymorphism near CRP gene and the risk of BPD. Plasma CRP shortly after birth predicts the risk of BPD. Together with the known antenatal risk factors rs11265269 influences susceptibility to BPD. As such, we propose that genetic and environmental factors influence the expression of CRP that may stimulate pathways predisposing to BPD.
References
Stoll, B. J. et al. Trends in Care Practices, Morbidity, and Mortality of Extremely Preterm Neonates, 1993–2012. JAMA 314, 1039–1051 (2015).
Baraldi, E. & Filippone, M. Chronic lung disease after premature birth. N. Engl. J. Med. 357, 1946–1955 (2007).
Islam, J. Y., Keller, R. L., Aschner, J. L., Hartert, T. V. & Moore, P. E. Understanding the Short- and Long-Term Respiratory Outcomes of Prematurity and Bronchopulmonary Dysplasia. Am. J. Respir. Crit. Care Med. 192, 134–156 (2015).
Gough, A., Spence, D., Linden, M., Halliday, H. L. & McGarvey, L. P. General and respiratory health outcomes in adult survivors of bronchopulmonary dysplasia: a systematic review. Chest 141, 1554–1567 (2012).
Abman, S. H. Bronchopulmonary dysplasia: “a vascular hypothesis”. Am. J. Respir. Crit. Care Med. 164, 1755–1756 (2001).
Balany, J. & Bhandari, V. Understanding the Impact of Infection, Inflammation, and Their Persistence in the Pathogenesis of Bronchopulmonary Dysplasia. Front. Med. (Lausanne) 2, 90 (2015).
Jobe, A. H. Mechanisms of Lung Injury and Bronchopulmonary Dysplasia. Am. J. Perinatol. 33, 1076–1078 (2016).
Bhandari, V. et al. Familial and genetic susceptibility to major neonatal morbidities in preterm twins. Pediatrics 117, 1901–1906 (2006).
Lavoie, P. M., Pham, C. & Jang, K. L. Heritability of bronchopulmonary dysplasia, defined according to the consensus statement of the national institutes of health. Pediatrics 122, 479–485 (2008).
Hadchouel, A. et al. Identification of SPOCK2 as a susceptibility gene for bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 184, 1164–1170 (2011).
Wang, H. et al. A genome-wide association study (GWAS) for bronchopulmonary dysplasia. Pediatrics 132, 290–297 (2013).
Ambalavanan, N. et al. Integrated Genomic Analyses in Bronchopulmonary Dysplasia. J. Pediatr. (2014).
Shaw, G. M. & O’Brodovich, H. M. Progress in understanding the genetics of bronchopulmonary dysplasia. Semin. Perinatol. 37, 85–93 (2013).
Lavoie, P. In Bronchopulmonary Dysplasia (ed Bhandari, V.) 109–27 (Springer International, 2016).
Peltomaki, P. et al. Genetic mapping of a locus predisposing to human colorectal cancer. Science 260, 810–812 (1993).
Sabatti, C. et al. Genome-wide association analysis of metabolic traits in a birth cohort from a founder population. Nat. Genet. 41, 35–46 (2009).
Jobe, A. H. & Bancalari, E. Bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 163, 1723–1729 (2001).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Klinger, G. et al. Perinatal risk factors for bronchopulmonary dysplasia in a national cohort of very-low-birthweight infants. Am. J. Obstet. Gynecol. 208, 115.e1–115.e9 (2013).
Bose, C. et al. Fetal growth restriction and chronic lung disease among infants born before the 28th week of gestation. Pediatrics 124, e450–8 (2009).
Schick, U. M. et al. Association of exome sequences with plasma C-reactive protein levels in >9000 participants. Hum. Mol. Genet. 24, 559–571 (2015).
Ridker, P. M. et al. Loci related to metabolic-syndrome pathways including LEPR,HNF1A, IL6R, and GCKR associate with plasma C-reactive protein: the Women’s Genome Health Study. Am. J. Hum. Genet. 82, 1185–1192 (2008).
Okada, Y. et al. Genome-wide association study for C-reactive protein levels identified pleiotropic associations in the IL6 locus. Hum. Mol. Genet. 20, 1224–1231 (2011).
Bhandari, V. & Elias, J. A. Cytokines in tolerance to hyperoxia-induced injury in the developing and adult lung. Free Radic. Biol. Med. 41, 4–18 (2006).
Allison, B. J. et al. Ventilation of the very immature lung in utero induces injury and BPD-like changes in lung structure in fetal sheep. Pediatr. Res. 64, 387–392 (2008).
Weismann, D. & Binder, C. J. The innate immune response to products of phospholipid peroxidation. Biochim. Biophys. Acta 1818, 2465–2475 (2012).
Volanakis, J. E. Human C-reactive protein: expression, structure, and function. Mol. Immunol. 38, 189–197 (2001).
Ishibashi, M., Takemura, Y., Ishida, H., Watanabe, K. & Kawai, T. C-reactive protein kinetics in newborns: application of a high-sensitivity analytic method in its determination. Clin. Chem. 48, 1103–1106 (2002).
Nishikawa, T. et al. Transcriptional complex formation of c-Fos, STAT3, and hepatocyte NF-1 alpha is essential for cytokine-driven C-reactive protein gene expression. J. Immunol. 180, 3492–3501 (2008).
Ambalavanan, N., Ross, A. C. & Carlo, W. A. Retinol-binding protein, transthyretin, and C-reactive protein in extremely low birth weight (ELBW) infants. J. Perinatol. 25, 714–719 (2005).
D’Angio, C. T. et al. Blood Cytokine Profiles Associated with Distinct Patterns of Bronchopulmonary Dysplasia among Extremely Low Birth Weight Infants. J. Pediatr. (2016).
Lal, C. V. & Ambalavanan, N. Cellular and humoral biomarkers of Bronchopulmonary Dysplasia. Early Hum. Dev. (2016).
Hofer, N., Zacharias, E., Muller, W. & Resch, B. An update on the use of C-reactive protein in early-onset neonatal sepsis: current insights and new tasks. Neonatology 102, 25–36 (2012).
Pankow, J. S. et al. Familial and genetic determinants of systemic markers of inflammation: the NHLBI family heart study. Atherosclerosis 154, 681–689 (2001).
Anand, S. S. et al. C-reactive protein as a screening test for cardiovascular risk in a multiethnic population. Arterioscler. Thromb. Vasc. Biol. 24, 1509–1515 (2004).
Reiner, A. P. et al. Genome-wide association and population genetic analysis of C-reactive protein in African American and Hispanic American women. Am. J. Hum. Genet. 91, 502–512 (2012).
Pepys, M. B. & Hirschfield, G. M. C-reactive protein: a critical update. J. Clin. Invest. 111, 1805–1812 (2003).
Peltonen, L., Jalanko, A. & Varilo, T. Molecular genetics of the Finnish disease heritage. Hum. Mol. Genet. 8, 1913–1923 (1999).
Dong, Q. & Wright, J. R. Expression of C-reactive protein by alveolar macrophages. J. Immunol. 156, 4815–4820 (1996).
Gallegos, L. L. et al. A protein interaction map for cell-cell adhesion regulators identifies DUSP23 as a novel phosphatase for beta-catenin. Sci. Rep. 6, 27114 (2016).
Hansel, N. N. et al. Genome-Wide Association Study Identification of Novel Loci Associated with Airway Responsiveness in Chronic Obstructive Pulmonary Disease. Am. J. Respir. Cell Mol. Biol. 53, 226–234 (2015).
Carrera, P. et al. Exome sequencing and pathway analysis for identification of genetic variability relevant for bronchopulmonary dysplasia (BPD) in preterm newborns: A pilot study. Clin. Chim. Acta 451, 39–45 (2015).
Li, J. et al. Exome Sequencing of Neonatal Blood Spots and the Identification of Genes Implicated in Bronchopulmonary Dysplasia. Am. J. Respir. Crit. Care Med. 192, 589–596 (2015).
Bhattacharya, S. et al. Genome-wide transcriptional profiling reveals connective tissue mast cell accumulation in bronchopulmonary dysplasia. Am. J. Respir. Crit. Care Med. 186, 349–358 (2012).
Acknowledgements
The authors wish to thank Maarit Haarala for laboratory assistance; Riitta Vikeväinen, Marita Suni, Kaisa Jaakkonen, Leena Kivinen, Jaana Vuollet-Puurunen, and Satu Ekblad for sample and data collection; Leena Haataja and Helena Lapinleimu for subject enrollment and sample collection; Mihoko Ladd and Barb Kamstra for coordinating sample collection and enrollment in Vancouver and Edmonton (Canada), respectively; Claude Danan, Pierre-Henri Jarreau, Richard Lenclen and the EPIPAGEADO study group for coordinating sample collection and subject enrolment in France, and the Centre de Ressources Biologiques (CRB, CHI Créteil) for French DNA storing; and Kira O’Day Heller for language editing of the manuscript. Genotyping of SNP markers was performed by the Technology Centre, Institute for Molecular Medicine Finland (FIMM), University of Helsinki. The study was supported by Jane and Aatos Erkko Foundation (M.H., M.R.), The European Union Grant (Health H-FS-2009-223062; M.H., D.B., C.F.P.), The Academy of Finland (M.H.), The Sigrid Juselius Foundation (M.H.), Alma and K.A. Snellman Foundation (M.M., M.K.K., M.R.), Emil Aaltonen Foundation (M.K.K.), Foundation of Pediatric Research in Finland (M.K.K.), The Research Foundation of the Pulmonary Diseases (HES) (M.K.K.), the Competitive State Research Financing of the Tampere University Hospital (M.R.), the Competitive State Research Financing of the Oulu University Hospital (M.R.), The Programmes Hospitaliers de Recherche Clinique AOR 07 018 and AOM P100117, Assistance Publique–Hôpitaux de Paris, and Agence Nationale de la Recherche ANR-09-GENO-037 (AH), The British Columbia Lung Association (P.M.L.).
Author information
Authors and Affiliations
Contributions
Acquisition of data: M.M., M.K.K., J.M.H., S.A., M.A.K., O.K.T.T., U.S., L.L., R.H.M., T.L.-M., C.D., Ch.D., P.M.L., A.H., M.R., M.H; conception and design: M.M., M.K.K., D.B., C.F.P., A.P., L.M., M.R., M.H.; analysis and interpretation: M.M., M.K.K., J.M.H., M.R., M.H.; writing of the manuscript: M.M., M.K.K., M.R., M.H.; editing of the manuscript: J.M.H., S.A., M.A.K., O.K.T.T., U.S., L.L., D.B., C.F.P., A.P., L.J.M., P.M.L., A.H., M.R., M.H.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mahlman, M., Karjalainen, M.K., Huusko, J.M. et al. Genome-wide association study of bronchopulmonary dysplasia: a potential role for variants near the CRP gene. Sci Rep 7, 9271 (2017). https://doi.org/10.1038/s41598-017-08977-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-08977-w
- Springer Nature Limited
This article is cited by
-
Prospective epigenome and transcriptome analyses of cord and peripheral blood from preterm infants at risk of bronchopulmonary dysplasia
Scientific Reports (2023)
-
Epigenome-wide association study of bronchopulmonary dysplasia in preterm infants: results from the discovery-BPD program
Clinical Epigenetics (2022)
-
Verification of immunology-related genetic associations in BPD supports ABCA3 and five other genes
Pediatric Research (2022)
-
Potential role of vitamin D receptor-related polymorphisms in bronchopulmonary dysplasia
Egyptian Journal of Medical Human Genetics (2021)
-
Exome sequencing of extreme phenotypes in bronchopulmonary dysplasia
European Journal of Pediatrics (2020)