Abstract
Autophagy is known to be implicated in the pathogenesis of Sjögren syndrome (SS), but evidences are limited. We aimed to examine the levels of autophagy markers in tear film and conjunctival epithelial cells from SS dry eye patients, and analyze their correlations with clinical features. Patients with SS dry eye exhibited lower Schirmer values, lower tear breakup time, and higher ocular staining scores. In tears, ATG5 and LC3B-II/I levels were significantly higher in SS dry eye. ATG5 and LC3B-II mRNA in the conjunctiva were also elevated in SS dry eye compared with non-SS dry eye. The immunostaining of conjunctival epithelium showed a punctate pattern of ATG5 and LC3B-II in SS dry eye. These staining patterns were also observed in the lacrimal gland of SS animal models. ATG5 levels in tears and the conjunctival epithelium strongly correlated with ocular staining scores, and one month of topical corticosteroid treatment reduced both ATG5 and LC3B-II/I levels in tear film and the conjunctival epithelium of patients with SS dry eye. Our results suggest that autophagy is enhanced or dysregulated in SS and autophagy markers may be serve as both diagnostic and therapeutic biomarkers in SS dry eye.
Similar content being viewed by others
Introduction
Dry eye (DE) syndrome is a multifactorial disease characterized by tear film instability and ocular surface damage, which cause foreign body sensation, discomfort and visual disturbances1. Among various etiologies, Sjögren syndrome (SS) is a major cause of aqueous deficient type DE1. SS is a systemic autoimmune disease that affects the entire body, mainly the exocrine glands2. The characteristics of SS is the lymphocytic infiltration of the exocrine glands and mucosal epithelia, causing dry eye and dry mouth3,4. The pathogenic mechanisms of SS is assumed to lead to more severe dry eye manifestations compared to non-SS DE5,6,7.
Numerous studies over the past decade extended our understanding of the pathologic mechanisms of SS. Autophagy has been suggested to have a role in the pathogenesis of SS, although data to support this role are limited8,9,10. Recent data demonstrated that enhanced autophagy and apoptosis are involved in the Ro/SAA and La/SSB redistribution in secretory epithelial cells of the salivary gland9,11,12. At the cellular level, the local secretion of autoantibodies follows the re-localization of the autoantigens to the cell surface13,14. Autophagy is a self-eating cellular process to maintain cellular homeostasis via lysosome-mediated degradation and recycling of cytoplasmic components and organelles. In addition to its classic role in response to cellular stress, autophagy has been implicated in the pathogenesis of autoimmune diseases15,16,17. Autophagy regulates various immune processes, such as antigen presentation, pathogen removal, the survival immune cells, and inflammation17. Moreover, recent studies showed that polymorphisms in autophagy-related genes might attribute to susceptibility of systemic lupus erythematosus and Crohn’s disease18,19.
During autophagy, a portion of the cytoplasm and several proteins are incorporated in the autophagosome, a key structure with double layer membranes for intracellular degradation. Among these proteins, ATG5 and the LC3B-II/I ratio are used as typical markers of autophagy. ATG5 (autophagy related gene 5), which conjugates with ATG12, is specifically required for the maturation of the autophagic membrane, and ATG5 deletion plays a role in autophagy-mediated salivary homeostatic control in SS8. A cytosolic form of LC3 (LC3-I) is conjugated to phosphatidylethanolamine to form LC3-phosphatidylethanolamine conjugate (LC3-II), which is recruited to the autophagosomal membranes20. The aim of this study was to investigate these autophagy markers in tear film and conjunctival epithelial cells from SS DE and non-SS DE patients as well as to analyze their correlations with clinical features for determining the implications of autophagy dysregulation in SS DE.
Results
Patient Demographics
The demographics and ocular surface parameters of subjects are listed in Table 1. Schirmer I value and TBUT were significantly lower and the conjunctival staining scores were significantly higher in SS DE than non-SS DE. Age, OSDI, and corneal staining scores did not significantly differ between SS DE and non-SS DE.
Autophagy Marker Expression in Tear and Conjunctival Epithelial Cells
Western blot analysis of tears revealed significant increases in ATG5 protein and the LC3B-II/I conversion ratio in SS DE compared to non-SS DE and controls. The relative densitometry results showed that the levels of ATG5 (vs β-actin) and the LC3B-II/I ratio in tears were higher in patients with SS DE (5.06 ± 1.31 and 3.37 ± 1.08) than in patients with non-SS DE (1.44 ± 0.46 and 1.67 ± 0.40) and healthy controls (1.00 ± 0.35 and 1.00 ± 0.35) (P = 0.0001) (Fig. 1A). The mRNA levels of ATG5 and LC3B-II in the conjunctiva were also upregulated in patients with SS DE (8.15 ± 1.38- and 6.14 ± 1.51-fold change), but not in patients with non-SS DE (0.80 ± 0.17- and 2.21 ± 0.61-fold change), compared to healthy controls (Fig. 1B, all P < 0.0001). The expression levels of autophagy markers in tears and conjunctival impression cytology specimens did not significantly differ between non-SS DE and controls. The immunofluorescent staining of conjunctival impression cytology specimens identified a punctate cytoplasmic staining pattern of ATG5, indicative of autophagy, in SS DE, whereas ATG5 exhibited diffuse staining in samples from non-SS DE and controls (Fig. 1C). Furthermore, the number of punctate staining foci of LC3B-II was also notably increased in SS DE but not in non-SS DE and controls. Overall, autophagy marker expression was upregulated in tears and the conjunctiva of SS DE, suggesting that the dysregulation of autophagy may be implicated in the pathogenesis of SS DE.
Expression of Autophagy Markers ATG5 and LC3B in Tears and Conjunctival Impression Cytology Specimens from Controls, Patients with non-Sjögren Syndrome Dry Eye, and Patients with Sjögren Syndrome Dry Eye. (A). Representative Western blot images and relative densitometry data on ATG5 protein expression and the LC3B-II/I conversion ratio in tears demonstrating increases in patients with Sjögren Syndrome (SS) dry eye (DE) compared to patients with non-SS DE and controls. The protein expressions of ATG5, LC3B-I, LC3B-II and β-actin were developed from the same gel in western blot. (B). The ATG5 and LC3B-II mRNA expression levels in the conjunctiva as measured by real time PCR demonstrating increases in patients with SS DE compared to patients with non-SS DE and controls. (C). Representative confocal images of immunostained conjunctiva from impression cytology specimens showing that the punctate cytoplasmic staining patterns of ATG5 and LC3B-II indicative of autophagy were enhanced in patients with SS DE. *P < 0.05. Scale bar = 20 μm.
Autophagy Marker Expression in the Lacrimal Glands of Animal Models
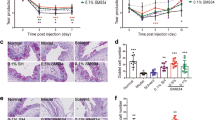
In the lacrimal glands of non-obese diabetic (NOD)/LtJ mice, punctate cytoplasmic staining patterns of ATG5 and LC3B-II were observed at 8 weeks of gestational age and developed prominently in lacrimal gland at 16 weeks of gestational age, compared to 16-week-old BALB/c normal mice (Fig. 2A). Correspondingly, western blot analysis from lysates of lacrimal gland showed higher levels of ATG5 and LC3B-II/I in 16-week-old NOD/LtJ mice (3.36 ± 0.04 and 4.07 ± 0.43 versus BALB/c normal mice) than in 8-week-old NOD/LtJ (2.30 ± 0.02 and 2.94 ± 0.22 versus BALB/c normal mice), respectively (Fig. 2B). Based on these findings in the SS animal model, we speculate that autophagy might be induced in the lacrimal glands of patients with SS DE.
Immunofluorescent Staining of Autophagy Markers (ATG5 and LC3B-II) in the Lacrimal Glands of Sjögren Syndrome Animal Models (NOD/LtJ mice). (A) Representative immunofluorescent photos demonstrating cytoplasmic punctate staining patterns of ATG5 and LC3B-II on the lacrimal gland developed prominently at 16 weeks of gestational age in NOD/LtJ mice. Those markers were not prominent at 8-week-old NOD/LtJ mice. Lacrimal glands of 16-week-old BALB/c mice were used as a control. DAPI (blue) was used for nuclear counterstaining. Scale bar = 20 μm. (n = 6). (B) Western blotting and relative densitometry graph demonstrates ATG5 protein expression and the LC3B-II/I conversion ratio from lysates of 8− and 16-week-old NOD/LtJ, and 16-week-old BALB/c mice. Both markers were significantly higher at 16 weeks than 8 weeks of gestational age in NOD/LtJ mice (n = 6). The protein expressions of ATG5, LC3B-I, LC3B-II and β-actin were developed from the same gel in western blot. *P < 0.0001.
Correlations between Autophagy Markers and Clinical Parameters in SS DE
Because autophagy markers were significantly upregulated in SS DE but not in non-SS DE, we next investigated the correlation between autophagy markers and clinical parameters in patients with SS DE (Table 2). ATG5 expression strongly correlated with corneal and conjunctival staining scores in patients with SS DE, which indicates that ATG5 might be used as a biomarker in SS DE. Additionally, LC3B-II/I conversion ratio was not significantly correlated with any clinical parameters in this study.
The Outcome of Topical Corticosteroid Treatment on Ocular Surface Parameters and the Level of Autophagy Markers in SS DE
After administering topical corticosteroid to SS DE, we evaluated the changes in autophagy marker expression and clinical parameters. One month of topical corticosteroid treatment with artificial tears significantly improved the OSDI score, Schirmer I value, TBUT, corneal and conjunctival staining scores in SS DE (Table 3 and Fig. 3). Accordingly, representative Western blot images demonstrated reductions in the ATG5 and LC3B-II/I ratio in tears after topical corticosteroid treatment (Fig. 4A). The relative densitometry results showed that ATG5 expression (vs β-actin) and the LC3B-II/I ratio in tears significantly decreased after corticosteroid treatment (0.66 ± 0.05 and 0.84 ± 0.08) compared to the respective values before treatment (5.24 ± 0.32 and 1.29 ± 0.07) in SS DE (P < 0.0001 and P = 0.001, respectively). The mRNA levels of ATG5 and LC3B-II (fold) were significantly decreased from 13.16 ± 4.36 and 12.93 ± 4.44 to 1.23 ± 0.27 and 2.57 ± 0.48 (respectively, P = 0.00573 and P = 0.01586) (Fig. 4B). In addition, representative immunostaining pictures demonstrated that the punctate staining patterns of autophagy markers in the conjunctiva were also normalized in SS DE after corticosteroid treatment (Fig. 4C).
The Representative Images of Ocular Surface Staining Before and After Topical Corticosteroid Treatment in Sjögren Syndrome Dry Eye Patient. In this patient, the ocular surface staining scores, which were 5, 3, and 2 in cornea, nasal, and temporal conjunctiva before treatment, reduced to 3, 1, and 0 after 1-month topical corticosteroid treatment, respectively. Corneal and conjunctival staining was performed using fluorescein and lissamine green dye.
Changes in Autophagy Markers after Topical Corticosteroid Treatment. The expression levels of the autophagy markers ATG5 and LC3B-II/I in tears and conjunctival impression cytology specimens from 16 eyes of 16 patients with Sjögren syndrome (SS) Dry Eye (DE) before and after corticosteroid treatment. (A) Representative Western blot image of ATG5 and LC3B-II/I conversion in tears showing reductions in patients with SS DE after corticosteroid treatment. Relative densitometry of ATG5 (vs β- actin) and the LC3B-II/I ratio in tears showing significant decrease in patients with SS DE after corticosteroid treatment. The protein expressions of ATG5, LC3B-I, LC3B-II and β-actin were developed from the same gel in western blot. (B) ATG5 and LC3B-II mRNA expression level in the conjunctiva measured by real time PCR showing significant reductions in patients with SS DE. (C) The cytoplasmic punctate staining patterns of ATG5 and LC3B-II indicative of autophagy were markedly attenuated in the immune-stained conjunctiva impression cytology specimens of patients with SS DE after corticosteroid. *P < 0.05. Scale bar = 20 μm.
DISCUSSION
In the current study, we revealed that the ATG5 level and LC3B-II/I ratio in tears and mRNA expression levels in conjunctival epithelial cells significantly increased in SS DE. Our findings constitute first evidence to suggest that the autophagy markers were upregulated in SS DE. To date, limited studies have examined the role of autophagy in SS and the exact mechanisms linking autophagy to SS pathogenesis are not established. Previous studies reported that autophagy plays important roles in redistributing the auto-antigens Ro/SSA and La/SSB in salivary gland epithelial cells9 or maintaining the homeostasis of salivary acinar cells8. Autophagy dysregulation has been reported in various autoimmune and auto-inflammatory diseases. The promotion of the presentation of cytosolic antigens by major histocompatibility complex (MHC) class II and the control of T lymphocyte homeostasis enhance autophagic reaction, and type 1 T helper cells cytokines and specific serum autoantibodies may also play a role in this reaction21,22,23. ATG5 has been observed to be upregulated in patients with SLE, and complement-inactivated serum from patients significantly activated autophagy24. A more recent study directly investigating autophagy in T cells indicated that autophagy was increased in lupus, which promoted the survival of autoimmune T cells25. Furthermore, ATG5 can reportedly interfere with the maintenance of autoreactive T and B cell clones and their imbalanced crosstalk26,27,28,29,30,31, and ATG5 is also implicated in the pathogenesis of RA32,33. These studies strongly implicate autophagy as a core mechanism of autoimmunity rather than as a bystander.
One study in dry eye murine model indicated enhanced autophagy in the lacrimal glands and assumed that DE stress activates the autophagy process via hypoxia caused by the loss of lacrimal gland vasculature10. However, in our data from non-SS DE patients, autophagy markers from tears and conjunctiva did not significantly increase compared to healthy controls even though we could not directly examine the patient’s lacrimal gland. Our data of NOD/LtJ mice SS animal model showed that protein abundance of ATG5 and LC3B-II/I and their stained feature in lacrimal glands were at 8 weeks of age and became more prominent at 16 weeks of age. Because it has been reported that the salivary and lacrimal gland infiltration occurs at approximately 12 to 16 weeks of age, followed by secretory dysfunction by 16 weeks of age34, the increase of autophagy markers coincides timely to the pathogenic course of SS. This temporal coincidence suggests that enhanced autophagy is closely related to autoimmune inflammation and gland dysfunction of SS, not just as a predecessor or by consequence of DE stress as suggested in the experimental DE model.
When we observed that elevated ATG in SS DE was associated with DE clinical parameters, ATG5 expressions in tear and conjunctival epithelium proved to have positive correlations with corneal and conjunctival staining scores. As ocular staining scores showed higher multi-criteria decision analysis weights and higher importance of variable than the Schirmer test, which is a classic ocular criteria in the new diagnostic criteria for primary SS35, positive correlations of ATG5 with ocular staining scores are very meaningful. In regards to associations with the Schirmer test, ATG5 in the conjunctival epithelium did not correlate with this value but ATG5 in tears showed weak negative correlations, even though it was not statistically significant. We think very low Schirmer numeric value (2.72 ± 1.88 mm) in SS DE could not reach statistical significant correlation results.
Our data suggests the possibility of ATG5 as a diagnostic and therapeutic marker in SS DE. ATG5 in tear and conjunctival epithelium were upregulated in SS DE but not in non-SS DE compared to healthy controls. Previous studies demonstrated elevated tear cytokines such as IL-6, IL-17 or TNF-α in SS DE but these cytokines are not specific to SS DE because they are also elevated in non-SS DE36,37. Although data from a larger clinical study are warranted, ATG5 is expected to serve as a disease-specific diagnostic marker of SS DE. Of the interest, the elevated autophagy markers in tear and conjunctival epithelium were reduced after topical corticosteroid treatment, in conjunction with improvement of clinical parameters in SS DE. The effects of topical corticosteroids on improvement of symptoms and signs in SS DE were very well established38,39. Previous studies exhibited immune activation of the conjunctival epithelium and accompanying elevations of cytokines in SS DE40,41. Topical corticosteroids may exert its therapeutic effects by inhibiting the immune response and the production of cytokines on the conjunctival epithelium resulting in reduction of autophagy markers in conjunctival epithelium after corticosteroid treatment. Elevated autophagy markers in tears were also reduced after corticosteroid treatment in our study. There is no evidence that the topically applied anti-inflammatory treatment influences lacrimal glands by direct drug delivery. Since lacrimal secretion is diluted with secretions from meibomian glands and ocular surface (cornea and conjunctiva) epithelial cells while constituting the tear film at the ocular surface42, elevated autophagy markers in tears in our study might originate from lacrimal glands, as well as ocular surface epithelial cells. Therefore, reduction of autophagy markers in tear after corticosteroid treatment might reflect the therapeutic effects of corticosteroid on conjunctiva epithelium. Also, it should be considered that normalization of neural feedback mechanism to stimulate lacrimal secretion, which is suppressed by ocular surface inflammation43, has led to tear recovery and increase of Schirmer values after corticosteroid treatment.
In conclusion, this cross-sectional case-control study demonstrated that autophagy markers in tears and the conjunctival epithelium were upregulated in patients with SS DE, and ATG5 expression positively correlated with the corneal and conjunctival staining scores in SS DE. Additionally, anti-inflammatory treatment with a topical corticosteroid attenuated these increases in autophagy markers, which was accompanied by improved clinical parameters in patients with SS DE. Further studies using SS animal models and large numbers of human samples are required to reveal the role of autophagy on the pathogenesis of SS DE and validate the sensitivity and specificity of ATG5 as a potential biomarker of SS DE.
Methods
Study Design
The current study was a cross-sectional case-control research performed at Seoul St. Mary’s Hospital, the Catholic University of Korea in the Department of Ophthalmology. Informed consent was attained from all subjects in this research. The study protocols in this research was designed referencing the Declaration of Helsinki guidelines and was affirmed by the Institutional Review Board of Seoul St. Mary’s Hospital.
Subjects
Eighty-two female subjects were included in the present study: 40 SS DE patients (78 eyes), 24 non-SS DE patients (48 eyes), and 16 normal healthy subjects (25 eyes) as control. Sixteen of 40 SS DE patients received topical corticosteroid treatment (4 times a day for 1 month, fluorometholone 0.1%, Sam-Il Pharmaceutics Co. Seoul, Korea). All DE patients had disease severity above grade 2, a classification that was used by referencing the severity grading scheme in the International Dry Eye WorkShop (DEWS)1. All SS DE patients were confirmed with primary SS from a diagnosis by a rheumatologist according to the 2012 Sjögren International Collaborative Clinical Alliance (SICCA) classification criteria44. Patients with secondary SS or subjects taking any disease-modifying medications were excluded from this study. Only females were included in this current study, reflecting on the higher prevalence of SS in females(F:M = 9:1)45. The patients with the conditions that could affect dry eye parameters were excluded from this study. They included those with acute or chronic allergic conjunctivitis, lid abnormalities, any ocular surgeries within a span of the last 6 months, contact lens wearers, punctual plug, meibomian gland dysfunction above grade 2, and subjects taking systemic medications and using any topical treatments other than artificial tears within the last 3 months46.
Clinical Assessments
The same investigator (S.-H.C.) assessed the ocular surface and measured the clinical parameters including the Schirmer I test, ocular surface disease index (OSDI) questionnaire ocular surface staining (OSS) scores, and tear film breakup time (TBUT). Standardized Schirmer strips (Eagle Vision, Memphis, TN, USA) were situated in the lateral 1/3 of the lower lid without anesthesia, and after 5 minutes, the length of strips that were wetted was measured. The strips were then frozen and stored for future analysis by cutting them lengthwise in cryotubes. A fluorescein dye (Haag-Streit, Koeniz, Switzerland) was applied to patients and the TBUT was measured by a slit lamp under a cobalt blue light. The average score of three TBUT measurements were used47. The ocular staining scores (OSS) including corneal staining scores and conjunctival staining scores were determined according to the SICCA registry ocular examination protocol48. To calculate the corneal staining scores, corneal surface images were taken as a screenshot immediately after measuring TBUT. Corneal punctate epithelial erosions (PEEs) were counted and scored that stain with fluorescein. The corneal score was 0 if PEEs were absent, and the score was 1 when 1–5 PEEs could be observed. Additionally, a score of 2 was given to 6–30 PEEs observed, and a 3 if more than 30 PEEs were visible. In the case of more than one patches that could be seen of confluent staining, pupillary area of the cornea, or filament anywhere on the cornea that was stained, an additional point was calculated. The maximum obtainable score for each cornea was 6. To measure the conjunctival staining scores, 1% Lissamine green dye (Leiter’s Pharmacy San Jose, CA, USA) was administered in the inferior conjunctival fornix one drop each in both eyes after fluorescein was washed out with nonpreserved saline solution. After several blinks, the conjunctivae staining scores were examined separately in the temporal and nasal bulbar conjunctivae. A score of 0 was represented 0 to 9 dots for Lissamine green staining; a score of 1 was appointed for 10 to 32 dots, a score of 2 with 33 to 100 dots, and a score 3 indicated >100 dots. Therefore, the maximum possible score was 6 for the conjunctiva (temporal plus nasal).
Tear Collection and Western Blot Analysis
Tears were collected from patients by Schirmer strips that were tear-soaked, in accordance with the Posa’s protocol49. In order for the extraction of the tear fluid, extraction buffer (NaCl 0.5 M, Tween-20 0.5% in 0.1 M phosphate buffered solution) was added to Eppendorf tubes containing Schirmer strips.After one hour, a puncture was made at the bottom of an 0.5 mL tube and the Schirmer was secured inside. The 0.5 mL tube was then transferred into a 1.5 mL tube for centrifugation at 12,000 rpm for 15 min. The gathered tear fluid from the strip was mixed in a sample-loading buffer, boiled for 10 min, and centrifuged. BCA Protein Assay kit (Thermo Fisher Scientific, Rockford, IL, USA) was used for the quantification of the proteins in the sample lysates. Samples buffer with equal amount of protein were separated by 15% SDS-polyacrylamide gel electrophoresis (SDS-PAGE) under reducing conditions and electro-transferred to a PVDF membrane (Millipore, Billerica, MA, USA). PBS with 5% nonfat milk and 0.1% Tween-20 was used for blocking the membrane and incubated at 4 °C for 18 h with primary rabbit polyclonal antibodies targeting LC3B-I and II (1:400, Cell Signaling Technology, Boston, MA, USA) or ATG5 (1:400, Novus Biologicals, Littleton, CO). With the completion of three washes, anti-rabbit horseradish peroxidase-conjugated secondary antibodies (1:10,000, Thermo Fisher Scientific) were used for membrane incubation at RT for 1 h. With another wash 3 times, enhanced chemiluminiscence reagent (ECL; Amersham Biosciences, Sweden) was used to detect protein bands. After, all the membranes underwent a stripping and reprobing process with mouse monoclonal anti-β-actin antibody (Santa Cruz Biotechnology, Dallas, TX, USA) to provide a normalizing reference. Each experiment normalized the relative expression levels by image analysis by including a stain for β-actin. We performed 4–6 independent experiments.
Impression Cytology
We performed impression cytology (IC) at least 15 min after all ocular examination. Polyethersulfone filters (Suopor 200 membrane, Pall Corporation, Port Washington, NY, USA) were used by cutting into two halves (13 × 6.5 mm) and applying them to both the superior-nasal and superior-temporal non-exposed bulbar conjunctiva. The filter samples of the superior-temporal conjunctivas were placed in tubes that contained 2 ml of PBS with 0.05% paraformaldehyde (PFA) to be used for immunostaining. Filters from the superior-nasal conjunctivas were used for RNA extraction within 3 hours of their sampling.
Immunofluorescent Staining
IC filter papers with the detached epithelial cells were firmly pressed to silane-coated slides (Muto Pure Chemicals co., ltd. Tokyo, Japan), and epithelial cells transferred to the slides were used for immunofluorescent staining according to Baudouin’s protocol50. Briefly, cold methanol was used to fix the cells and 0.1% Triton X-100 was applied for cell permeabilization. Then, nonspecific reactions were blocked by incubation with 10% goat serum for 1 h. After, they were then incubated with anti- LC3B-I, II or ATG5 antibody (1:400) and washed twice with PBS. Finally they were incubated with goat Alexa Flour 488–conjugated anti-rabbit IgG Ab (1:400, ThermoFisher scientific, Waltham, MA, USA) in PBS and the stain was captured by confocal microscopy (LSM 510 Meta (Carl Zeiss Meditec Inc. Dublin, CA) and transferred to Photoshop (Adobe systems, Santa Clara, CA).
RNA Isolation and Real-Time PCR
The RNA from IC was isolated by TRIzol reagent (Gibco-Invitrogen, Grand Island, NY, USA) according to a modified protocol51. SuperScript IIITM reverse transcriptase (Invitrogen) was used with random hexamers to synthesize the first strand of complementary DNA (cDNA) and a SYBR Green I real-time PCR method was followed. The 2−ΔΔCt was calculated for relative quantification and for internal calibration GAPDH was used. The primers used in this study were LC3B-II (Forward: 5′-CTT TGG GTG CGA CTT GAC G-3′, Reverse: 5′-GTC GAC CCC GCT CCT TTT-3′) and ATG5 (Forward: 5′-AGG AGA GCC TGT ACC TAT GGA-3′, Reverse: 5′-TTC TCT GTT GCG CTT TTC TGA-3′).
Animals
Because a human lacrimal gland biopsy is a relatively invasive procedure, we evaluated the expression levels of ATG5 and LC3B-II from the lacrimal gland of NOD/LtJ mice, a SS animal model. Lacrimal glands of NOD/LtJ was provided by Prof. Mi-La Cho (The Catholic University of Korea, Seoul, Korea) and breeding pairs of NOD/LtJ mice were originally acquired from Charles River Laboratory (Wilmington, MA, USA). The NOD mouse is a widely used model for diabetes mellitus type I, but also for SS. It has been reported that lymphocytic infiltrates in NOD mice were detected in the exocrine glands in addition to the pancreas, resulting in sialadenitis and dacryoadenitis34, and also several major autoantibodies (ANA, anti-SSA/Ro, anti-SSB/La) in common with SS patients were exhibited52. BALB/c mice (Oriental Bio, Inc., Korea) were used for control animal. Both lacrimal glands were removed from six mice of each 8- and 16-week-old NOD/LtJ, and 16-week-old BALB/c mouse. One of lacrimal glands was immediately frozen for western plot. Western blot analysis and immunostaining of ATG5 and LC3B-II were performed as described above. The other of lacrimal glands was fixed in 10% formalin solution for overnight at room temperature. Samples were then immersed in OCT embedding compound (Tissue-Tek, Sakura, Osaka, Japan) and frozen at −80 °C. Frozen samples were cut in 6-μm-thick sections using a cryostat. The subsequent immunostaining process was as described above. Animal protocols in this current experiment were approved by the Institutional Animal Care and Use Committee (IACUC) at the College of Medicine, The Catholic University of Korea. Additionally, all procedures performed in animal studies were in alignment with the standards for the Use of Animals in Ophthalmic and Vision Research in the Association for Research in Vision and Ophthalmology Statement.
Statistical Analysis
All descriptive data in this study were analyzed with the SPSS software (version 15.0, SPSS Inc, Chicago, IL) and GraphPad Prism (version 5.0, GraphPad Inc. La Jolla, CA), and are presented as the mean and SD. Shapiro-Wilk test was utilized to assess the normality of the data, while group comparisons were analyzed by One-way analysis of variance (ANOVA) and the t-test. Pearson correlation coefficients between autophagy markers and clinical parameters were calculated to explore the relationships between the expression level of autophagy markers and clinical variables. Two-sided P values < 0.05 were considered to be significant.
References
The definition and classification of dry eye disease: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop. The ocular surface 5, 75–92 (2007).
Fox, R. I. Sjogren’s syndrome. Lancet 366, 321–331, https://doi.org/10.1016/S0140-6736(05)66990-5 (2005).
Moutsopoulos, H. M. Sjogren’s syndrome: autoimmune epithelitis. Clinical immunology and immunopathology 72, 162–165 (1994).
Skopouli, F. N. et al. T cell subpopulations in the labial minor salivary gland histopathologic lesion of Sjogren’s syndrome. The Journal of rheumatology 18, 210–214 (1991).
Stern, M. E. et al. Conjunctival T-cell subpopulations in Sjogren’s and non-Sjogren’s patients with dry eye. Investigative ophthalmology & visual science 43, 2609–2614 (2002).
Pflugfelder, S. C. et al. Conjunctival cytologic features of primary Sjogren’s syndrome. Ophthalmology 97, 985–991 (1990).
Xu, K. P., Katagiri, S., Takeuchi, T. & Tsubota, K. Biopsy of labial salivary glands and lacrimal glands in the diagnosis of Sjogren’s syndrome. The Journal of rheumatology 23, 76–82 (1996).
Morgan-Bathke, M. et al. Deletion of ATG5 shows a role of autophagy in salivary homeostatic control. Journal of dental research 92, 911–917, https://doi.org/10.1177/0022034513499350 (2013).
Katsiougiannis, S., Tenta, R. & Skopouli, F. N. Endoplasmic reticulum stress causes autophagy and apoptosis leading to cellular redistribution of the autoantigens Ro/Sjogren’s syndrome-related antigen A (SSA) and La/SSB in salivary gland epithelial cells. Clinical and experimental immunology 181, 244–252, https://doi.org/10.1111/cei.12638 (2015).
Seo, Y. et al. Activation of HIF-1alpha (hypoxia inducible factor-1alpha) prevents dry eye-induced acinar cell death in the lacrimal gland. Cell death & disease 5, e1309, https://doi.org/10.1038/cddis.2014.260 (2014).
Casciola-Rosen, L. A., Anhalt, G. & Rosen, A. Autoantigens targeted in systemic lupus erythematosus are clustered in two populations of surface structures on apoptotic keratinocytes. The Journal of experimental medicine 179, 1317–1330 (1994).
Okuma, A. et al. Enhanced apoptosis by disruption of the STAT3-IkappaB-zeta signaling pathway in epithelial cells induces Sjogren’s syndrome-like autoimmune disease. Immunity 38, 450–460, https://doi.org/10.1016/j.immuni.2012.11.016 (2013).
Halse, A., Harley, J. B., Kroneld, U. & Jonsson, R. Ro/SS-A-reactive B lymphocytes in salivary glands and peripheral blood of patients with Sjogren’s syndrome. Clinical and experimental immunology 115, 203–207 (1999).
Hu, S. et al. Identification of autoantibody biomarkers for primary Sjogren’s syndrome using protein microarrays. Proteomics 11, 1499–1507, https://doi.org/10.1002/pmic.201000206 (2011).
Yang, Z., Goronzy, J. J. & Weyand, C. M. Autophagy in autoimmune disease. Journal of molecular medicine 93, 707–717, https://doi.org/10.1007/s00109-015-1297-8 (2015).
Pierdominici, M. et al. Role of autophagy in immunity and autoimmunity, with a special focus on systemic lupus erythematosus. FASEB journal: official publication of the Federation of American Societies for Experimental Biology 26, 1400–1412, https://doi.org/10.1096/fj.11-194175 (2012).
Zhou, X. J. & Zhang, H. Autophagy in immunity: implications in etiology of autoimmune/autoinflammatory diseases. Autophagy 8, 1286–1299, https://doi.org/10.4161/auto.21212 (2012).
Han, J. W. et al. Genome-wide association study in a Chinese Han population identifies nine new susceptibility loci for systemic lupus erythematosus. Nature genetics 41, 1234–1237, https://doi.org/10.1038/ng.472 (2009).
Scharl, M. et al. Protein tyrosine phosphatase nonreceptor type 2 regulates autophagosome formation in human intestinal cells. Inflammatory bowel diseases 18, 1287–1302, https://doi.org/10.1002/ibd.21891 (2012).
Mizushima, N., Yoshimori, T. & Levine, B. Methods in mammalian autophagy research. Cell 140, 313–326, https://doi.org/10.1016/j.cell.2010.01.028 (2010).
Dengjel, J. et al. Autophagy promotes MHC class II presentation of peptides from intracellular source proteins. Proceedings of the National Academy of Sciences of the United States of America 102, 7922–7927, https://doi.org/10.1073/pnas.0501190102 (2005).
Li, C. et al. Autophagy is induced in CD4+T cells and important for the growth factor-withdrawal cell death. Journal of immunology 177, 5163–5168 (2006).
Lu, B., Capan, E. & Li, C. Autophagy induction and autophagic cell death in effector T cells. Autophagy 3, 158–159 (2007).
Towns, R. et al. Sera from patients with type 2 diabetes and neuropathy induce autophagy and colocalization with mitochondria in SY5Y cells. Autophagy 1, 163–170 (2005).
Gros, F. et al. Macroautophagy is deregulated in murine and human lupus T lymphocytes. Autophagy 8, 1113–1123, https://doi.org/10.4161/auto.20275 (2012).
Jia, W. & He, Y. W. Temporal regulation of intracellular organelle homeostasis in T lymphocytes by autophagy. Journal of immunology 186, 5313–5322, https://doi.org/10.4049/jimmunol.1002404 (2011).
Miller, B. C. et al. The autophagy gene ATG5 plays an essential role in B lymphocyte development. Autophagy 4, 309–314 (2008).
Zhou, X. J. et al. Gene-gene interaction of BLK, TNFSF4, TRAF1, TNFAIP3, and REL in systemic lupus erythematosus. Arthritis and rheumatism 64, 222–231, https://doi.org/10.1002/art.33318 (2012).
Wang, C., Khalil, M., Ravetch, J. & Diamond, B. The naive B cell repertoire predisposes to antigen-induced systemic lupus erythematosus. Journal of immunology 170, 4826–4832 (2003).
Nedjic, J., Aichinger, M., Mizushima, N. & Klein, L. Macroautophagy, endogenous MHC II loading and T cell selection: the benefits of breaking the rules. Current opinion in immunology 21, 92–97, https://doi.org/10.1016/j.coi.2009.01.013 (2009).
Pua, H. H., Dzhagalov, I., Chuck, M., Mizushima, N. & He, Y. W. A critical role for the autophagy gene Atg5 in T cell survival and proliferation. The Journal of experimental medicine 204, 25–31, https://doi.org/10.1084/jem.20061303 (2007).
Ireland, J. M. & Unanue, E. R. Autophagy in antigen-presenting cells results in presentation of citrullinated peptides to CD4 T cells. The Journal of experimental medicine 208, 2625–2632, https://doi.org/10.1084/jem.20110640 (2011).
Raychaudhuri, S. et al. Genetic variants at CD28, PRDM1 and CD2/CD58 are associated with rheumatoid arthritis risk. Nature genetics 41, 1313–1318, https://doi.org/10.1038/ng.479 (2009).
Hu, Y., Nakagawa, Y., Purushotham, K. R. & Humphreys-Beher, M. G. Functional changes in salivary glands of autoimmune disease-prone NOD mice. Am J Physiol 263, E607–614 (1992).
Shiboski, C. H. et al. 2016 American College of Rheumatology/European League Against Rheumatism Classification Criteria for Primary Sjogren’s Syndrome: A Consensus and Data-Driven Methodology Involving Three International Patient Cohorts. Arthritis Rheumatol 69, 35–45, https://doi.org/10.1002/art.39859 (2017).
Enriquez-de-Salamanca, A. et al. Tear cytokine and chemokine analysis and clinical correlations in evaporative-type dry eye disease. Molecular vision 16, 862–873 (2010).
Lam, H. et al. Tear cytokine profiles in dysfunctional tear syndrome. American journal of ophthalmology 147, 198–205 e191, https://doi.org/10.1016/j.ajo.2008.08.032 (2009).
Akpek, E. K. et al. Treatment of Sjogren’s syndrome-associated dry eye an evidence-based review. Ophthalmology 118, 1242–1252, https://doi.org/10.1016/j.ophtha.2010.12.016 (2011).
Marsh, P. & Pflugfelder, S. C. Topical nonpreserved methylprednisolone therapy for keratoconjunctivitis sicca in Sjogren syndrome. Ophthalmology 106, 811–816, https://doi.org/10.1016/S0161-6420(99)90171-9 (1999).
Jones, D. T., Monroy, D., Ji, Z. & Pflugfelder, S. C. Alterations of ocular surface gene expression in Sjogren’s syndrome. Advances in experimental medicine and biology 438, 533–536 (1998).
Jones, D. T., Monroy, D., Ji, Z., Atherton, S. S. & Pflugfelder, S. C. Sjogren’s syndrome: cytokine and Epstein-Barr viral gene expression within the conjunctival epithelium. Investigative ophthalmology & visual science 35, 3493–3504 (1994).
Ji, Y. W. et al. Lacrimal gland-derived IL-22 regulates IL-17-mediated ocular mucosal inflammation. Mucosal Immunol 10, 1202–1210, https://doi.org/10.1038/mi.2016.119 (2017).
Management and therapy of dry eye disease: report of the Management and Therapy Subcommittee of the International Dry Eye WorkShop (2007). The ocular surface 5, 163-178 (2007).
Shiboski, S. C. et al. College of Rheumatology classification criteria for Sjogren’s syndrome: a data-driven, expert consensus approach in the Sjogren’s International Collaborative Clinical Alliance cohort. Arthritis care & research 64, 475–487 (2012).
Delaleu, N., Jonsson, M. V., Appel, S. & Jonsson, R. New concepts in the pathogenesis of Sjogren’s syndrome. Rheumatic diseases clinics of North America 34, 833–845, https://doi.org/10.1016/j.rdc.2008.08.004 (2008). vii.
Tomlinson, A. et al. The international workshop on meibomian gland dysfunction: report of the diagnosis subcommittee. Investigative ophthalmology & visual science 52, 2006–2049, https://doi.org/10.1167/iovs.10-6997f (2011).
Hwang, J. et al. Comparison of clinical efficacies of autologous serum eye drops in patients with primary and secondary Sjogren syndrome. Cornea 33, 663–667, https://doi.org/10.1097/ICO.0000000000000147 (2014).
Whitcher, J. P. et al. A simplified quantitative method for assessing keratoconjunctivitis sicca from the Sjogren’s Syndrome International Registry. American journal of ophthalmology 149, 405–415, https://doi.org/10.1016/j.ajo.2009.09.013 (2010).
Posa, A. et al. Schirmer strip vs. capillary tube method: non-invasive methods of obtaining proteins from tear fluid. Ann Anat 195, 137–142, https://doi.org/10.1016/j.aanat.2012.10.001 (2013).
Baudouin, C., Haouat, N., Brignole, F., Bayle, J. & Gastaud, P. Immunopathological findings in conjunctival cells using immunofluorescence staining of impression cytology specimens. Br J Ophthalmol 76, 545–549 (1992).
Barabino, S. et al. Immune response in the conjunctival epithelium of patients with dry eye. Exp Eye Res 91, 524–529, https://doi.org/10.1016/j.exer.2010.07.008 (2010).
Winer, S. et al. Primary Sjogren’s syndrome and deficiency of ICA69. Lancet 360, 1063–1069, https://doi.org/10.1016/S0140-6736(02)11144-5 (2002).
Acknowledgements
Funding/Support: This work was supported by the Korea Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (HI13C0016) and by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2016R1A6A1A03010528). We thank to Prof. M-L. Cho (The Catholic University of Korea, Seoul, Korea) for kindly providing the animal samples.
Author information
Authors and Affiliations
Contributions
Yong-Soo Byun wrote the main manuscript text. Hyun Jung Lee performed the experiments. Yong-Soo Byun and Hyun Jung Lee performed the statistical analysis. Soojung Shin conducted the animal experiments. The critical revision of the manuscript was done by So-Hyang Chung. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Byun, YS., Lee, H., Shin, S. et al. Elevation of autophagy markers in Sjögren syndrome dry eye. Sci Rep 7, 17280 (2017). https://doi.org/10.1038/s41598-017-17128-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-17128-0
- Springer Nature Limited