Abstract
Macular degeneration (MD) affects central vision and represents the leading cause of visual diseases in elderly population worldwide. As a consequence of central vision loss, MD patients develop a preferred retinal locus (PRL), an eccentric fixation point that replaces the fovea. Here, our aim was to determine whether and to what extent spontaneous plasticity takes place in the cortical regions formerly responding to central vision and whether a visual training based on perceptual learning (PL) can boost this plasticity within the PRL area. Spontaneous and PL-induced cortical plasticity were characterized by using lateral masking, a contrast sensitivity modulation induced by collinear flankers. This configuration is known to be sensitive to neural plasticity and underlies several rehabilitation trainings. Results in a group of 4 MD patients showed that collinear facilitation was similar to what observed in age- and eccentricity-matched controls. However, MD patients exhibited significantly reduced collinear inhibition, a sign of neural plasticity, consistent with the hypothesis of partial cortical reorganization. Three AMD patients from the same group showed a further reduction of inhibition after training, but not controls. This result suggests that PL might further boost neural plasticity, opening promising perspectives for the development of rehabilitation protocols for MD patients.
Similar content being viewed by others
Introduction
Macular degeneration (MD) is a visual pathology that affects central vision and represents the leading cause of blindness in the elderly population1. Loss of central vision strongly impairs everyday life activities, such as locomotion, reading or face recognition. MD patients usually develop an eccentric retinal spot outside the scotoma, the preferred retinal locus (PRL), as a new fixation point2. The consequences of the PRL development on visual perception remain unclear. A recent study3 measured crowding, the difficulty to identify peripheral targets when surrounded by flanking elements4 in MD patients. This study showed that the shape of the crowding zone for targets presented at the PRL resembled that of the fovea rather than the periphery. This result could reflect spontaneous reorganization in cortical regions connected to the PRL, most likely in early visual areas4. However, other studies found that performances in the PRL did not differ from those usually observed in normal peripheral vision5,6. This discrepancy challenges the extent of spontaneous reorganization in MD patients. Perceptual learning (PL) has recently gained considerable attention for its ability to improve sensory performances in normal and clinical populations7. In the last years, several studies have shown that this technique can be used to improve, to some extent, residual peripheral vision in MD patients8,9,10,11,12,13. However, in most of these studies, improvements were small and remained mostly specific to the trained task, with little transfer to other visual functions, in contrast to what was observed in other clinical populations (e.g., Amblyopia14, Presbyopia15 or Myopia16). More recently, MD patients were trained using a lateral masking paradigm9. In this configuration, a low contrast central target (a Gabor patch) is flanked collinearly by two elements with the same spatial frequency and orientation. Depending on the target-to-flankers separation, lateral masking can lead either to collinear inhibition, a decrease of contrast sensitivity, or to facilitation, an increase of contrast sensitivity for the central target17. The separation within which the flankers induce suppression provides an estimation of the perceptual field (PF), the psychophysical equivalent of the classical physiological receptive field (CRF)18. In this context, the suppression effect is considered as a within-PF effect, while collinear facilitation is a between-PFs effect19. Recent studies have shown that this effect exists outside foveal vision, with the facilitatory target-to-flankers separation (and consequently, the PF size) increasing with eccentricity, most likely following the cortical magnification factor (see Fig. 1).
Schematic of the interaction between perceptual field (PF) size and collinear configuration as a function of eccentricity. X-axis represents eccentricity from central fixation. In foveal vision, the standard target-to-flankers distance that induces facilitation is 3λ (i.e., 3 times the wavelength of the Gabor patches of the configuration). In the present study, the spatial frequency was kept fixed at 1 cpd, therefore λ = 1 cpd. In this case, this distance also corresponds to the PF size and facilitation is considered as a between-PFs effect. When the same stimulus is presented in peripheral vision, the PF size increases, so that the 3λ configuration elicits within-PF, collinear inhibition. In order to restore facilitation, flankers must be placed outside the PF. Previous studies19,20,21 showed that this critical distance is 8λ at an eccentricity of 4°.
This distinction between foveal and peripheral collinear facilitation is of critical importance in MD patients. Indeed, evidence for facilitation in the PRL at shorter separations than in healthy peripheral vision would be consistent with the idea of a spontaneous cortical reorganization after retinal lesion, similar to what is reported in crowding4. While they share similar features, such as the increase in magnitude with eccentricity, and resemblance between target and flanking elements17,22, there is no consensus in the literature regarding the relationship between crowding and lateral masking23. Consequently, the evidence for spontaneous cortical reorganization in reshaping the crowding zone does not necessarily mean that the neural substrates underlying cortical inhibition are equally affected.
Horizontal connections between units sharing similar orientation and spatial frequency tunings in early visual cortex24,25,26 are known to be sensitive to PL-induced plasticity as suggested by lateral masking paradigm27. Enhancing these first stages improves in turn higher level visual processing that can rely on more efficient inputs after training15,21.
The question is still open whether collinear facilitation exists in MD patients’ PRL and whether it can be modulated through PL, as reported for other clinical populations14. Indeed, a previous study did not find collinear facilitation in MD patients28, possibly because given the tested target-to-flankers distance (4λ), the flankers remained within the inhibitory region of the PF. In the present paper, we aimed at addressing these questions in a small sample of AMD patients (and age-matched controls) using a lateral masking configuration as a baseline measure for spontaneous cortical reorganization (Experiment 1) and as a training configuration for guided cortical reorganization (Experiment 2). Previous PL studies based on collinear facilitation showed a reduction of inhibition after training21,27, an effect attributed to neural plasticity in early visual areas24,25,26. Consequently, reduced inhibition at baseline would be consistent with the hypothesis of spontaneous cortical reorganization similar, to what was observed in previous studies after training.
Method
In Experiment 1, we measured collinear facilitation (and inhibition) over a range of target-to-flankers distances in the PRL of 5 MD patients and 5 age- and eccentricity-matched controls. In Experiment 2 we trained a subgroup of participants from Experiment 1 (4 MD patients and 3 controls) to test whether facilitation and/or inhibition can be modulated through practice as observed in previous studies21,27. Because training based on PL are very long, a small sample size is not uncommon in PL studies. Nonetheless, a number of studies showed that reliable and robust results could still be obtained by comparing perfomances before and after training29,30,31,32,33,34.
Stimuli were three aligned Gabor patches which were displayed either in a collinear (flankers having the same local orientation as the target) or in an orthogonal (flankers having an orthogonal orientation with respect to the target i.e., a 90 deg offset) configuration (see20). The stimuli were aligned vertically for all participants except for MD3 and her matched control, for whom they were oriented horizontally, because of the shape of MD3’s scotoma. Each Gabor patch had a spatial frequency of 1 cpd. In a previous study, we showed that this frequency maximizes peripheral facilitation magnitude35. The target-to-flanker separations were 3λ, 4λ, 6λ and 8λ. Stimuli were displayed on a 17″ Dell M770 CRT monitor with a resolution of 1024 × 768 pixels, a refresh rate of 60 Hz and mean luminance of 47.6 cd/m2. Stimuli were generated with Matlab Psychtoolbox36,37. Each pixel subtended 2.14 arcmin. A digital-to-analogue converter (Bits#, Cambridge Research Systems, Cambridge UK) was used to increase the dynamic contrast range (13-bit luminance resolution). A 12-bit gamma-corrected lookup table (LUT) was used to linearize the monitor. Experiments were carried on at the Centre de la Retine, Hôpital Pierre-Paul Riquet, Purpan Hospital, Toulouse (France).
Participants
Patients and controls’ information is detailed in Table 1. Importantly, in order to keep our sample of patients homogeneous, we only included in the study patients with a single and well-defined PRL. MD patients were instructed to fixate with their PRL the center of the screen where a fixation point was always present. They were asked to adapt their head position in order to fixate as naturally as possible. All participants were volunteers and gave written informed consent prior to their inclusion in the study. They received an allowance for their participation. This study was conducted in accordance with the Declaration of Helsinki (1964). The experimental protocol was approved by a national ethical committee before the beginning of the study (CPP, Comité de Protection des Personnes, protocole 13018–14/04/2014). Four patients (MD1, MD2, MD3 and MD4) and 3 controls (C1, C2 and C4) took part in Experiment 2. MD5, C3 and C5 were not able to participate in the training study for logistical reasons.
Inclusion criteria
Absolute central binocular scotoma (as measured by 30° and 12° central visual fields analysed with Octopus® 300 perimeter; Haag-Streit, Köniz, Switzerland), age >60, residual vision in both eye < 2/10, presence of a single PRL (as detected with OCT measurement, see the ‘PRL location’ section). Exclusion criteria: Concomitant presence of other visual diseases (cataract, retinal detachment, glaucoma), cognitive impairment (Mini-Mental State Examination (MMSE) score <25), monocular MD.
PRL localization
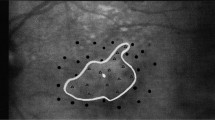
For each MD patient, we determined the PRL position using Spectral-Domain Optical Coherence Tomography (Spectralis OCT, Heildelberg Enginering, Heidelberg, Germany). First, high resolution scans of the retinal fundus, centered on the atrophic macular area, were acquired via OCT in order to localize the fovea (Fig. 2a). Three additional landmarks (i.e., crossing blood vessels) were identified on the infrared image of the macula and their coordinates were measured with respect to the fovea. In a successive acquisition, patients were asked to fixate with their PRL the central fixation point presented by the OCT system while high speed X-line OCT B-scans were acquired (Fig. 2b). To avoid the inclusion of patients with more than one PRL, this operation was repeated at least three times, and between each acquisition, the OCT system was defocalized while the patient was asked to fixate elsewhere. The same acquisition protocol was systematically conducted at each examination. As noted previously, patients with more than one PRL were excluded from the study. Afterwards, the distance between the PRL and the fovea was derived by comparing its position with respect to the three landmarks (Table 1 and Fig. 3). The small SD range of the three separate measures of PRL coordinates (Table 1) is a further indication that the patients were using a single PRL for fixation tasks.
OCT procedure for PRL localization. (a) Retinal analysis via OCT: Infrared 2D macular image of the ‘cross fixation’ acquisition showing the macular atrophy (ligther rounded area). In this image, the center of the red dashed cross corresponds to the preferred retinal locus (PRL), the eccentric fixation point replacing the fovea. (b) Autofluorescence imaging with the location of the anatomical fovea (green dot) and the PRL position (red dot). (c) Schematic retinal projection of the stimulus on the PRL location close to the scotoma (dark area); green dot is the location of the fovea.
Procedure
In Experiment 1, we tested eight conditions per participant, one for each target-to-flankers distance (3λ, 4λ, 6λ, 8λ) and orientation (collinear and orthogonal). MD patients performed the task monocularly with their dominant eye. They fixated with their PRL while the configuration was presented at the centre of the screen. A fixation point was always present to indicate the center of the configuration. Patients were instructed to keep their fixation on the central dot with their PRL and to use the three discs to adjust their gaze on the central dot (Fig. 4). A similar approach was already taken in previous studies that used visual aids to stabilize fixation in this clinical population38. Each control participant was assigned to a patient of similar age and trained at the eccentricity corresponding to the PRL position of his/her paired MD patient. Control participants had to fixate a central dot while the position of the testing configuration was randomized along the x axis. Position along the y-axis was fixed. Participants had to perform a contrast detection task with a temporal-2AFC (two alternative forced choice) procedure, with one interval containing both target and flankers and the other interval containing only the flankers (see Fig. 4). Each interval lasted 133 ms and the ISI was 500 ms. Participants had to report in which interval they detected the target. Target contrast varied according to a 3down/1up staircase, in which three consecutive correct responses reduced the target contrast of 0.1 log units and each wrong response increased the contrast of the same amount. The staircase terminated after 120 trials or 14 reversals. Contrast thresholds (Michelson contrast), corresponding to 79% of correct detection, were estimated from the algebric mean of the last 6 reversals. Acoustic signals (50 ms tone of 1000 Hz) were provided to signal the beginning of each interval and as a feedback for uncorrect responses (50 ms tone of 500 Hz). Participants sat in a dark room at a distance of 57 cm from the screen. Testings were performed monocularly. The testing configuration was always vertical (see Fig. 4), except for MD3 and her corresponding control, for whom the global and local orientation of the triplet was horizontal. This was due to the position of the PRL and the shape of the scotoma of MD3 that would have made it difficult to allow for the three Gabor patches to be visible all the time in a horizontal configuration. All 8 conditions (4 target-to-flankers separations x2 orientations) were performed within a single day. In Experiment 2, participants were trained for 12 sessions (4 blocks per day, 3 sessions per week) on the same configuration as in Experiment 1. Target to flankers distances were varied between blocks in each daily session, starting from the largest separation (8λ, 6λ, 4λ, 3λ). Spatial frequency of the stimuli was 1cpd34. Participants were trained only with the collinear configuration and tested on the orthogonal configuration before and after the training. Before each experiment, we ran a practice block of 15 trials to ensure that the patients were able to see the two flankers while fixating in the center of the screen with their PRL. All participants (patients and controls) reported that both the target and the flankers were clearly visible under these experimental conditions.
Experimental paradigm. Patients were instructed to fixate monocularly the point in the center of the screen with their PRL. In order to maximize fixation stablity, we displayed three additional red disks along the internal border of their scotoma (dark grey on the figure), selected individually for each patient within 2 deg from the border of the absolute scotoma. Patients had to adjust their eye position so that these points remained invisible. After 1000 ms, three vertically presented Gabors, either in a collinear (or orthogonal) configuration, appeared at this central position for 133 ms with an Inter Stimulus Interval (ISI) of 500 ms. In the collinear configuration, the flankers were presented above and below the target with the same (vertical) local orientation. In the orthogonal configuration, the flankers appeared above and below the target but their local orientation was horizontal (90° offset with respect to the target). Patients had to report which interval contained the target. Each control participant had to fixate foveally while the configuration was displayed at the eccentricity used in his/her paired MD patient.
Statistical analyses
Given the small sample of participants in the Experiments, we conducted non-parametric statistical analyses. In particular, to explore the main effects, we used an Aligned Rank Transformation (ART) as described by Wobbrock, Findlater, Gergle and Higgins39 with the r package ARTool40. To test for interactions, we used a non-parametric ART test for interactions in designs with repeated measures as described by Beasley & Zumbo41 with the r package “npIntFactRep”42. However, while this last package permits to test effects for mixed models with one ‘within’ and one ‘between’ factors, it does not allow for testing interactions with more than one ‘within’ factors, hence we only ran interaction tests for the Threshold elevation data. Post-hoc tests on the main effects were performed using linear models of the aligned data, as reported in Wobbrock et al.39. Finally, post-hoc tests on interactions were performed using differences of differences as suggested by Kay (supplementary documents in40). Indeed, directly comparing levels of factors in a non-parametric model is not a proper way of dealing with post hocs tests on interaction, and it is rather preferred to compare differences of differences, e.g., A-B|C vs A-B|D, where A and B are two levels of one factor and C and D are two levels of the other factor. Of note, ART tests do not allow for post-hoc tests on interactions when more than one within factor is present. These post-hoc tests were conducted using packages ARTool and phia in r.
Results
Pre training results (Experiment 1)
Contrast thresholds
Results for Experiment 1 are presented in Fig. 5 and Table 2. A Mixed Model ART on the contrast thresholds (Michelson contrast) of between-subjects factor Group (MD participants vs Controls) and within subjects factors Orientation (Collinear vs Orthogonal) and Target-to-flankers distances (3λ, 4λ, 6λ, 8λ) showed a significant main effect of Group (F 1,8 = 10.604, p = 0.011) and of Target-to-flankers distances (F 3,56 = 9.77, p < 0.001). As reported earlier, we could not test for the interaction effects (see Statistical analyses section). Post-hoc analysis, in the form of differences of differences, showed that the difference between collinear and orthogonal thresholds was significantly different for 3λ with respect to 8λ (p < 0.001, iso-ortho|3λ vs iso-ortho| 8λ) and for 3λ vs 6λ (p < 0.001, iso-ortho|3λ vs iso-ortho| 6λ).
Box-and-whisker diagrams for collinear and orthogonal contrast thresholds (Michelson contrast) for MD (red boxes) and Control participants (green boxes) as a function of the target-to-flankers distances (λ). In each boxplot, the central mark is the median. The edges of the box are the 25th and 75th percentiles and the whiskers are the interquartile range (i.e., Q3–Q1). The black circles represent the mean and the vertical bars are +/− SEM.
Since the effect of the group was significant, we conducted separated analysis on MD participants and controls. In patients, a linear model ART with Orientation (Collinear vs Orthogonal) and Target-to-flankers distance (3λ, 4λ, 6λ, 8λ) as factors, showed a significant effect of Target-to-flankers distance (F 3,28 = 3.11, p = 0.042). Post-hoc analysis showed a significant difference between 3λ and 6λ (p = 0.025).
In controls, a linear model ART with Orientation (Collinear vs Orthogonal) and Target-to-flankers distances (3λ, 4λ, 6λ, 8λ) as factors showed a significant main effect of Orientation (F 1,28 = 6.06, p = 0.021) and Target-to-flankers distances (F 1,28 = 14.642, p < 0.001).
Post-hoc analysis showed a significant difference between thresholds at 3λ and 6λ (p < 0.001) and between 3λ and 8λ (p < 0.001).
Threshold elevation
Following previous studies17,21, collinear inhibition and facilitation were computed as ratios of contrast thresholds (CT), using the following formula, separately for each target-to-flankers separation:
We used the orthogonal condition rather than the Gabor alone as a baseline measure of facilitation to distinguish actual orientation-specific increase in contrast sensitivity from reduction of spatial uncertainty. This approach is standard for peripheral collinear facilitation measurements19,21,35.
For Experiment 1, we conducted a mixed model Aligned Rank Test on threshold elevations with between factor Group (MDs vs Controls) and within factor. Target-to-flankers distances (3λ, 4λ, 6λ, 8λ). The interaction was calculated using the r package npInt for Nonparametric Interaction Tests for Factorial Designs with Repeated Measures (Feys, 2015). Results showed a main effect of Target-to-flankers distance (F 3,24 = 17.808, p < 0.001) and the interaction between factors (F 3,24 = 5.39, p = 0.00554).
Post-hoc analysis (ART pairwise comparisons, holm adjusted) showed that overall threshold elevation at 3λ was significantly higher than all the other separations (p = 0.0391, p < 0.001 and p < 0.001 for 4λ, 6λ and 8λ, respectively). Additionally, we explored post-hoc tests for the interaction. Since tests of differences of combinations of levels between factors have issues in ART (Kay, 2014), it is recommendable to test for differences of differences; (i.e., for the interaction Group x Target-to-flankers distance, we might test whether the difference AMD - control is different for 3λ vs 4λ, Wobbrock et al., 2011). This analysis showed that the difference AMD-controls was significant between 3λ and 8λ (p = 0.014973) and 3λ and 6λ (p = 0.00193), suggesting that patients exhibited both less inhibition (0.213+/−0.156 vs 0.464+/−0.171) and less facilitation (−0.0836+/− 0.0391 vs −0.165+/−0.109) than controls at baseline.
MD patients
We estimated collinear facilitation by conducting a series of one sample t-tests (threshold elevation vs zero value) similarly to previous studies20,21. Results showed a significant threshold reduction for target-to-flankers separations of 6λ (t 4 = 3.05, p = 0.038) and 8λ (t 4 = 4.78, p = 0.009) and a significant threshold elevation for 3λ (t 4 = 3.04, p = 0.039).
Controls
Independent one sample t-tests revealed a significant threshold reduction for target-to-flankers separations of 6λ (t 4 = 4.78, p = 0.008) and 8λ (t 4 = 3.38, p = 0.027) and a significant threshold elevation for 3λ (t 4 = 6.04, p = 0.004).
Finally, comparing threshold elevation in the two groups, at 3λ MD patients showed less inhibition than controls (two-sample t-test, t 8 = 0 2.42, p = 0.042), as well as lessr facilitation at 6λ (t 8 = 1.17, p = 0.018).
Training Results (Experiment 2)
Contrast thresholds
For Experiment 2 (Fig. 6 and 7, Table 3), a Mixed Model ART with within factors Training (Pre vs Post), Orientation (Iso vs Ortho) and Target-to-flankers distances (3λ vs 4λ vs 6λ vs 8λ) and between factor group (MDs vs controls) showed a main effect of Group (F 1,5 = 16.91, p = 0.009), Training (F 3,75=121.44, p < 0.001) and Target-to-flankers distances (F 3,75=5.57, p = 0.002): Again, it was not possible to run interaction tests. Post-hoc analysis showed that training significantly reduced contrast thresholds (p < 0.001). Since the between-subjects factor was significant, we conducted two separate ART for the two groups.
Box-and-whisker diagrams for collinear (above) and orthogonal (below) Michelson contrast thresholds for MD (red boxes) and Control participants (green boxes), before and after training as a function function of the target-to-flankers distances. See Fig. 5 for the details of the legend.
MD Patients
A Mixed Model ART on contrast thresholds with factors Training (Pre vs Post), Orientation (Iso vs Ortho) and Target-to-flankers distances (3λ vs 4λ vs 6λ vs 8λ) showed a main effect of Training (F 1,45 = 88.69, p < 0.001).
Controls
A repeated on contrast thresholds with factors Training (pre vs post), Orientation (iso vs ortho) and Target-to-flankers distance (3λ vs 4λ vs 6λ vs 8λ) showed a main effect of Orientation (F 1,30 = 9.07, p = 0.005) and Target-to-flankers distance (F 1,30 = 9.43, p < 0.0001).
Threshold elevation
A mixed model ART with within factors Training (Pre vs Post) and target-to-flankers distances (3λ vs 4λ vs 6λ vs 8λ) and between factor Group (MD vs Control) showed a main effect of Target-to-flankers distances (F 3,35 = 21.65, p < 0.001), a main effect of Group (F 1,5 = 11.82, p = 0.018). The MD group showed lower threshold elevation (0.1+/−0.025 vs −0.041+/−0.021), indicating overall greater facilitation for all the separations tested. Moreover, paired t-tests comparing pre- and post- training performance separately for each target-to-flankers separation showed that controls worsened their performance at 6λ after training (p = 0.004), while pre- post- paired t-test in MD patients approached significance for 3λ (p = 0.062). However, control group showed low contrast thresholds for the large target-flanker distances, suggesting a small room for improvement during the training. This might have reduced the differences between collinear and orthogonal thresholds and could thereby be the cause of this apparent performance deterioration.
Discussion
Cortical reorganization in patients with central vision loss, either spontaneous or training induced, is a topic of great interest in visual and clinical neuroscience. Recent results from psychophysics, neurophysiology and neuroimaging studies remain unclear. Indeed, previous studies using psychophysics approaches reported evidence for and against spontaneous cortical reorganization4,6, respectively. Similarly, some neuroimaging studies supported the hypothesis of a spontaneous neural reorganization in the lesion projection zone of MD patients43. This effect existed for stimuli presented outside the scotoma, around the PRL or not44. However, other fMRI studies did not find evidence for spontaneous cortical reorganization in MD patients45 or monkeys with retinal lesion46. In Experiment 1, we showed that in MD, both collinear inhibition and facilitation emerged at target-to-flankers separations that are consistent with those measured in previous studies in normal peripheral19,20,21,35 but not foveal17 vision. With respect to control participants, MD patients had overall higher contrast thresholds, and presented both reduced collinear inhibition at short separations (3λ) and reduced facilitation at larger separations (6λ and 8λ). On the one hand, reduced inhibition is consistent with the hypothesis of a spontaneous or use-dependent cortical reorganization. Previous training studies on collinear facilitation reported reduced inhibition after training21,27 suggesting neural plasticity in the early visual areas as possible neural substrate of this phenomenon24,25,26. On the other hand, the reduced facilitation and the emergence of the effect at target-to-flankers separations consistent with normal periphery rather than fovea might indicate that spontaneous cortical plasticity did not produce major reorganization in the PRL projecting zone, in agreement with recent psychophysical studies5,6. Overall, despite being tested at similar eccentricities, MD patients showed lower contrast sensitivity with respect to their age-matched controls. Previous studies on this population have shown that even in the absence of a clear lesion in the peripheral retina, a central scotoma might impair peripheral vision through a reduction of fixation stability and/or damages in the lateral connections of the retina3. Moreover, it has been shown that the PRL chosen by patients for fine vision and fixation might not always the most sensitive or adequate region of the spared retina47,48. Consistently, Van der Stigchel and colleagues5 showed that in a visual search task, AMD patients had inferior performances than controls tested with an artificial scotoma, suggesting that the switch from a fovea-based to a PRL-based reference frame might impair efficiency in a number of visual tasks.
Spontaneous cortical reorganization and the size of the perceptual field in MD patients
As reported in the introduction, the border between collinear inhibition and facilitation can be used as an estimate of the PF size. Previous studies showed that the inhibitory region, and consequently the size of PF, increases with eccentricity19,21, suggesting a relationship with the cortical magnification factor. Measuring the size of PF might thus offer hints about the cortical reorganization processes in the MD brain, i.e., whether units formerly responding to foveal stimulation have been recruited to respond to stimuli around the PRL. Consequently, a reduction in size of the PF might be consistent with a large scale, spontaneous cortical reoganization. However, results from the present study does not seem to support this hypothesis, given that facilitation emerged at the same target-to-flankers distance for both patients and controls. Nonetheless, MD patients showed a significantly reduced inhibition at short separation, an effect associated with neural plasticity in peripheral collinear facilitation21. Future studies might train patients in their PRLs on shorter target-to-flankers separations with respect to those used here (<3λ), to test whether collinear inhibition (and consequently PF size) can be reduced to the spatial extent observed in the fovea. Succeeding in reducing facilitation at shorter separations than those used in the present study would actually reshape lateral interactions in the PRL in the same way as those of the healthy fovea.
Training-induced cortical reorganization
Regarding training-induced cortical reorganization, the few existing studies report small or inconsistent improvements and limited transfer8,10,11,12. However, a recent training paradigm based on lateral interaction showed that patients improved in the trained task and learning transferred to visual acuity and, partially, to contrast sensitivity function (but not to crowding)9. In general, trainings based on lateral masking might be more effective because of the plasticity hypothesis and hierarchical mechanisms they rely upon27. In Experiment 2, we trained MD patients and controls for 12 sessions on the lateral interaction configuration. The rationale was that several rehabilitative approaches are based on similar training-induced modulation of collinear facilitation14,16. Results showed that MD patients (but not age matched controls) reduced their collinear inhibition at short flankers separations, to the point that on average, inhibition was no longer observed for all the separations tested. If the reduced inhibition observed in Experiment 1 is consistent with the hypothesis of spontaneous cortical reorganization, PL further amplified this reduction. This improvement is even larger than the one observed in younger, healthy participants21. Because the neural substrates of collinear facilitation lay in early visual cortex, a portion of which does not receive foveal input anymore, it is possible that a training specifically designed to stimulate these regions might further promote plasticity, e.g., by recruiting new neural units or by reshaping the PF size in a more fovea-like fashion. This result is particularly promising because the majority of rehabilitation approaches in MD patients currently focuses on improving reading speed and training-induced reduction of cortical inhibition is correlated with reduction of visual crowding21, one of factors contributing to slower letter recognition (and consequently reading speed) in peripheral vision48. However, one cannot rule out the possibility that low level neural plasticity is not the sole or main responsible of the observed performance changes. Other mechanisms, such as attentional modulation or read-out models, could also account for the training results49,50.
Limitations of the study
Given the clinical population involved, the study presents a number of limitations. The main limitation of this study is the small number of patients. This small number is largely explained by the fact that PL trainings are often long. In our case, sessions extended over several weeks. It made it more difficult to recruit a large number of participants, and specifically of MD patients. However, several studies showed that reliable and robust results could still be obtained in this case by comparing perfomances before and after training27,29,30,31,32,33,34. In our study, patients consistently showed reduced inhibition with respect to controls at baseline as well as larger learning effect after training with respect to controls.
Additionally, we did not directly measure eye position (e.g., by imaging the retina or tracking the eye) during the training. Nonetheless, we used visual aids to facilitate fixation. This approach was shown to improve eye stability in previous studies on MD patients38. In our case, all the patients had a unique and well-defined PRL. Thus, they could not rely on a secondary PRL to perform the task. In addition, they all reported that both the flankers and the targets were visible througout the whole experiment. We are therefore confident that our results are very unlikely affected by fixation unstability. A direct measure of eye position would however have permitted to better characterize this aspect.
References
Velez-Montoya, R. et al. Current knowledge and trends in age-related macular degeneration: genetics, epidemiology, and prevention. Retina 34, 423–441 (2014).
Timberlake, G. T., Peli, E., Essock, E. A. & Augliere, R. A. Reading with a macular scotoma. II. Retinal locus for scanning text. Investigative ophthalmology & visual science 28, 1268–1274 (1987).
Chung, S. T. Cortical reorganization after long-term adaptation to retinal lesions in humans. J Neurosci 33, 18080–18086 (2013).
Bouma, H. Interaction effects in parafoveal letter recognition. Nature 226, 177–178 (1970).
Van der Stigchel, S. et al. Macular degeneration affects eye movement behavior during visual search. Front psychol 4, 579 (2013).
Haun, A. M. & Peli, E. Similar Sensitivity to Ladder Contours in Macular Degeneration Patients and Controls. PloS one 10, e0128119 (2015).
Campana, G. & Maniglia, M. Editorial: Improving visual deficits with perceptual learning. Front Psychol 6, 491 (2015).
Chung, S. T. Improving reading speed for people with central vision loss through perceptual learning. Investigative ophthalmology & visual science 52, 1164–1170 (2011).
Maniglia, M. et al. Perceptual learning leads to long lasting visual improvement in patients with central vision loss. Restorative neurology and neuroscience 34, 697–720 (2016).
Plank, T. et al. Perceptual learning in patients with macular degeneration. Front psychol 5, 1189 (2014).
Rosengarth, K. et al. Functional and structural brain modifications induced by oculomotor training in patients with age-related macular degeneration. Front psychol 4, 428 (2013).
Tarita-Nistor, L., Brent, M. H., Steinbach, M. J., Markowitz, S. N. & Gonzalez, E. G. Reading training with threshold stimuli in people with central vision loss: a feasibility study. Optometry and vision science 91, 86–96 (2014).
Maniglia, M., Cottereau, B., Soler, V. & Trotter, Y. Rehabilitation approaches in macular degeneration patients. Front Syst Neurosci (2016)
Polat, U., Ma-Naim, T., Belkin, M. & Sagi, D. Improving vision in adult amblyopia by perceptual learning. Proc Natl Acad Sci USA 101, 6692–6697 (2004).
Polat, U. Making perceptual learning practical to improve visual functions. Vision research 49, 2566–2573 (2009).
Tan, D. T. & Fong, A. Efficacy of neural vision therapy to enhance contrast sensitivity function and visual acuity in low myopia. Journal of cataract and refractive surgery 34, 570–577 (2008).
Polat, U. & Sagi, D. Lateral interactions between spatial channels: suppression and facilitation revealed by lateral masking experiments. Vision research 33, 993–999 (1993).
Neri, P. & Levi, D. M. Receptive versus perceptive fields from the reverse-correlation viewpoint. Vision Res 46, 2465–2474 (2006).
Lev, M. & Polat, U. Collinear facilitation and suppression at the periphery. Vision research 51 (2011).
Maniglia, M., Pavan, A., Aedo-Jury, F. & Trotter, Y. The spatial range of peripheral collinear facilitation. Scientific reports 5, 15530 (2015).
Maniglia, M. et al. Reducing crowding by weakening inhibitory lateral interactions in the periphery with perceptual learning. PloS one 6, e25568 (2011).
Levi, D. M. Crowding-An essential bottleneck for object recognition: A mini-review. Vision Res 48, 635–654 (2008).
Lev, M. & Polat, U. Space and time in masking and crowding. Journal of vision 15, 10 (2015).
Gilbert, C. D. & Wiesel, T. N. Intrinsic connectivity and receptive field properties in visual cortex. Vision Res 25, 365–374 (1985).
Grinvald, A., Lieke, E. E., Frostig, R. D. & Hildesheim, R. Cortical point-spread function and long-range lateral interactions revealed by real-time optical imaging of macaque monkey primary visual cortex. J Neurosci 14, 2545–2568 (1994).
Ts’o, D. Y., Gilbert, C. D. & Wiesel, T. N. Relationships between horizontal interactions and functional architecture in cat striate cortex as revealed by cross-correlation analysis. J Neurosci 6, 1160–1170 (1986).
Polat, U. & Sagi, D. Spatial interactions in human vision: from near to far via experience-dependent cascades of connections. Proc Natl Acad Sci USA 91, 1206–1209 (1994).
Alcalá-Quintana, R., Woods, R. L., Giorgi, R. G., Peli,E. Lateral Interactions in People with Central Visual Field Loss. Perception 40 ECVP 2011 abstract (2011).
Ahissar, M. & Hochstein, S. Attentional Control of Early Perceptual Learning. Proc Nat Acad Sci USA 90, 5718–5722 (1993).
Chirimuuta, M., Burr, D. & Morrone, M. C. The role of perceptual learning on modality-specific visual attentional effects. Vision Res 47, 60–70 (2007).
Dosher, B. A., Han, S. & Lu, Z. L. Perceptual learning and attention: Reduction of object attention limitations with practice. Vision Res 50, 402–415 (2010).
Huang, T. R. & Watanabe, T. Task attention facilitates learning of task-irrelevant stimuli. PLoS ONE 7, e35946 (2012).
Paffen, C. L., Naber, M. & Verstraten, F. A. The spatial origin of a perceptual transition in binocular rivalry. PLoS ONE 3, e2311 (2008).
Szpiro, S. F., Wright, B. A. & Carrasco, M. Learning one task by interleaving practice with another task. Vision Res 101, 118–124 (2014).
Maniglia, M., Pavan, A. & Trotter, Y. The effect of spatial frequency on peripheral collinear facilitation. Vision research 107, 146–154 (2015).
Brainard, D. H. The Psychophysics Toolbox. Spatial vision 10, 433–436 (1997).
Pelli, D. G. The VideoToolbox software for visual psychophysics: transforming numbers into movies. Spatial vision 10, 437–442 (1997).
Astle, A. T., Blighe, A. J., Webb, B. S. & McGraw, P. V. The effect of normal aging and age-related macular degeneration on perceptual learning. J Vis 15, 16 (2015).
Wobbrock, J.O., Findlater, L., Gergle, D. & Higgins, J.J. The aligned rank transform for nonparametric factorial analyses using only ANOVA procedures. Proc SIGCHI Conf Hum Factors Comput Syst (2011).
Kay, M. & Wobbrock J. ARTool: Aligned Rank Transform for Nonparametric Factorial ANOVAs (2016).
Beasley, T. M. & Zumbo, B. Aligned rank tests for interactions in split-plot designs: Distributional assumptions and stochastic homogeneity. J Mod Appl Stat Methods 8, 16–50 (2009).
Feys, J. Nonparametric Tests for the Interaction in Two-way Factorial Designs Using R. R J. 8, 367–378 (2016).
Baker, C. I., Peli, E., Knouf, N. & Kanwisher, N. G. Reorganization of Visual Processing in Macular Degeneration. J Neurosci 25, 614–618 (2005).
Dilks, D. D., Baker, C. I., Peli, E. & Kanwisher, N. Reorganization of visual processing in macular degeneration is not specific to the “preferred retinal locus”. J Neurosci 29, 2768–2773 (2009).
Baseler, H. A. et al. Large-scale remapping of visual cortex is absent in adult humans with macular degeneration. Nature neuroscience 14, 649–655 (2011).
Smirnakis, S. M. et al. Lack of long-term cortical reorganization after macaque retinal lesions. Nature 435, 300–307 (2005).
Fine, E. M. & Rubin, G. S. Reading with simulated scotomas: Attending to the right is better than attending to the left. Vision Research 39(5), 1039–1048 (1999).
Petre, K. L., Hazel, C., Fine, E. M. & Rubin, G. S. Reading with eccentric fixation is faster in inferior visual field than in left visual field. Optometry & Vision Science 77(1), 34–39 (2000).
Bouma, H. Visual interference in the parafoveal recognition of initial and final letters of words. Vision Res 13, 767–782 (1973).
Maniglia, M. & Seitz, A. Towards a whole brain model of Perceptual Learning. Current Opinion in Behavioral Science 20, 47–55 (2018).
Acknowledgements
Author MM was supported by the ≪ Fondation Fouassier (2013 00039351 and 2014 00048124) and Fondation de l’Avenir (AP-RMA-2015-003) ≫. Authors would like to thank Jean Pierre Jaffrézou for corrections on the manuscript.
Author information
Authors and Affiliations
Contributions
M.M. designed, implemented the experiments, collected and analysed the data. All authors interpreted the results and wrote the main manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Maniglia, M., Soler, V., Cottereau, B. et al. Spontaneous and training-induced cortical plasticity in MD patients: Hints from lateral masking. Sci Rep 8, 90 (2018). https://doi.org/10.1038/s41598-017-18261-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-18261-6
- Springer Nature Limited
This article is cited by
-
The effect of duration between sessions on microperimetric biofeedback training in patients with maculopathies
Scientific Reports (2024)
-
Contextual influences in the peripheral retina of patients with macular degeneration
Scientific Reports (2019)