Abstract
The clinical presentations of skin diseases produced by different pathogens, as American tegumentary leishmaniasis (ATL) and sporotrichosis can be similar and possibly influenced by the skin immune system (SIS). The aim of the study was to understand the underlying mechanisms of skin inflammation produced by different pathogens. We used immunohistochemistry to analyze 96 patients: a- localized cutaneous leishmaniasis (LCL-ATL); b- sporotrichoid cutaneous leishmaniasis (SCL-ATL); c-lymphocutaneous (LC-SP); d- fixed (F-SP) sporotrichosis. LCL-ATL and SCL-ATL had a significantly higher percentage of CD8, FasL and NOS2 than sporotrichosis. In contrast, LC-SP had a substantially higher percentage of CD4, BCl2 and neutrophils than ATL lesions. These results indicated some differences in the profile of the in situ immune response suggesting that SIS is a complex, adaptable system capable of different responses to intracellular or extracellular pathogens. However, regardless of the etiological agents, the inflammatory reaction and clinical manifestations can be similar. SCL-ATL and LC-SP presented similarities in both clinical presentation and in situ inflammatory profile (CD3, CD22, neutrophils, macrophages). The clinical presentation of ATL and sporotrichosis could be explained by a combination of factors both of the host SIS and the etiological agent. The unbalanced host parasite relationship could result in atypical manifestations of skin disease.
Similar content being viewed by others
Introduction
Immunologists have recently paid more attention to the importance of the skin for immune surveillance. In humans, the skin, which covers approximately 2 m2 and accounts for 16% of the body weight, is the largest organ of the body1. This organ has several immune systems, such as the skin immune system (SIS), skin-associated lymphoid tissue (SALT), and hair follicle immune system (HFIS)1,2,3,4,5,6. Consequently, the skin is now deemed essential for the development and selection of the immune response to several agents7,8,9,10,11,12,13,14.
As an immune surveillance organ, the skin continuously interacts with various infectious agents. Not surprisingly, some infectious and parasitic diseases primarily or secondarily target the skin15,16,17,18,19,20,21,22. For example, Leishmania spp. and Sporothrix spp cause two granulomatous skin diseases: American tegumentary leishmaniasis (ATL) and sporotrichosis (SP), respectively. Although these diseases share clinical similarities as ulcerated lesions that arise frequently in the limbs23, they differ in their duration and the number of lesions as well as the degree to which the skin is involved20,23,24,25. Since SP is caused by an extracellular fungus that occasionally enters phagocytic cells whereas ATL is caused by an obligatory intracellular parasite of mononuclear phagocytes, we hypothesized that these differences could elicit different SIS responses and, therefore, cause different clinical symptoms and signs. To test this hypothesis, we used immunohistochemistry to compare the in situ inflammatory reaction of active lesions in ATL and SP patients presenting a dissimilar clinical presentation in order to elucidate some aspects underlying the mechanisms of localized inflammation of the skin by different infectious agents.
Results
Sporotrichosis and American tegumentary leishmaniasis patients only partially differ in the aspect of lesions and duration of infection before diagnosis
LCL-ATL and F-SP commonly presented single and localized lesions without lymphatic involvement (Fig. 1A,B). SCL-ATL and LC-SP presented multiple lesions frequently associated with lymphangitis and more extensive lesions (Fig. 1C,D). All 4 groups of patients had similar age distributions (p > 0.05; Table 1). The duration of infection, time elapsed between the beginning of the cutaneous lesions and the attendance of the patient at the Instituto Nacional de Infectologia Evandro Chagas – (INI) and the diagnostic procedures, was different among the groups. Sporotrichosis patients showed more acute development since their evolution was shorter than that of the LCL-ATL patients (Table 1). However, SCL-ATL presented similar duration as LC-SP (p > 0.05). F-SP showed the shorter time of evolution (Table 1).
Comparison of the clinical presentations of patients with (A) a fixed form of sporotrichosis (F-SP), (B) localized cutaneous leishmaniasis (LCL-ATL), (C) lymphocutaneous (LC-SP) form of sporotrichosis and (D) sporotrichoid cutaneous leishmaniasis (SCL-ATL). Note the similarities between the different clinical forms of sporotrichosis and ATL.
Sporotrichosis and American tegumentary leishmaniasis lesions can differ in the distribution of their inflammatory reactions
The inflammatory infiltrate in sporotrichosis lesions strongly predominated in the reticular dermis, near sweat glands and ducts, and around blood vessels, while that in ATL lesions it prevailed in the papillary dermis and was diffusely distributed throughout the lesion. However, since the inflammatory cells were heterogeneously distributed, there were no apparent differences in the cell density per mm2 of tissue in any of the four groups of patients with ATL or SP. Moreover, they were different from healthy skin that showed reduced cellularity organized as cell nests around cutaneous adnexa more concentrated in the papillary dermis (data not shown).
The percentage composition of cell types and markers of in situ inflammatory reactions only partially differ when ATL and SP lesions are compared
We observed significant differences as well as similarities in the percentage composition of cell types and markers between LCL-ATL, SCL-ATL, F-SP and LC-SP lesions (Tables 2–3 and Suplemmentary Table S1) (Figs 2–4). All four patient groups presented a higher percentage of CD3+ cells than healthy skin (p < 0.05). LCL-ATL patients had the highest percentage of CD3+ cells and it was significantly different from F-SP patients (p = 0.012; Mann-Whitney test) (Fig. 3A and Table 2). Finally, CD3+ cells were similar in SCL-ATL and LC-SP.
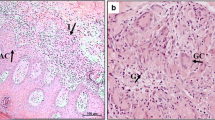
NOS2 expression (A,C,E,G) and neutrophils (B,D,F,H) in active lesions of LCL-ATL (A,B), SCL-ATL (C,D), F-SP (E,F) and LC-SP (G,H) patients. Magnification (A,C,E,G) 200× scale bar = 50 µm and (B,D,F,H) 400× scale bar = 25 µm. LCL-ATL: localized cutaneous leishmaniasis; SCL-ATL: sporotrichoid cutaneous leishmaniasis; F-SP: fixed sporotrichosis; LC-SP: Lymphocutaneous sporotrichosis. Arrows demonstrate examples of positive cells.
Percentage of (A) CD3+, (B) CD4+, (C) CD8+, (D) CD22+ cells, (E) macrophages (CD68+ cells) and (F) neutrophilic elastase-positive cells (neutrophils) Data represented as mean and SEM. P-value < 0.05 was considered statistically significant. LCL-ATL: localized cutaneous leishmaniasis; SCL-ATL: sporotrichoid cutaneous leishmaniasis; F-SP: fixed sporotrichosis; LC-SP: Lymphocutaneous sporotrichosis. Anova test was used for variables with normal distribution, otherwise Kruskal-Wallis test was applied. Bonferroni test was used as post hoc test. Pis modi aliquatem ad magnate erupis nulpari busam, nimus aut aut odi comniatur sita.
Percentage of (A) Ki67+, (B) BCl2+, (C) CD95+ (Fas+ cells), (D) CD95L+ (FasL+ cells). Data represented as mean and SEM. P-value < 0.05 was considered statistically significant. LCL-ATL: localized cutaneous leishmaniasis; SCL-ATL: sporotrichoid cutaneous leishmaniasis; F-SP: fixed sporotrichosis; LC-SP: Lymphocutaneous sporotrichosis. Anova test was used for variables with normal distribution, otherw ise Kruskal-Wallis test was applied. Bonferroni test was used as post hoc test.
The highest percentage of CD4+ cells was found in LC-SP lesions (Fig. 3B and Table 2). CD4+ cells were similar in LCL-ATL and F-SP. SCL-ATL lesions showed the lowest percentage of CD4+ cells in lesions of ATL or SP patients, similar to that of healthy skin. On the other hand, SCL-ATL had more CD8+ than CD4+ cells, and more CD8+ cells than the other groups (Fig. 3C and Table 2). In general, the percentage of CD8+ cells was higher in the leishmaniasis than in the sporotrichosis lesions. Moreover, the number of CD8+ cells in the sporotrichosis lesions was similar to that of the healthy skin.
B lymphocytes concentration was more intense in LCL-ATL lesions (Fig. 3D and Table 2). It was similar in SCL-ATL and LC-SP, and higher than that in F-SP and in healthy skin. These last two were comparable regarding B lymphocytes concentration.
Excepting F-SP, there were no differences in the percentage of macrophages among the groups including healthy skin (Fig. 3E and Table 2). However, it is noteworthy emphasizing that cellularity per tissue area in healthy skin was markedly reduced when compared to the lesion groups, producing a difference when the absolute number of cells were counted. On the other hand, the percentage of neutrophils was higher in SCL-ATL and LC-SP than in the other groups (Fig. 2B,D,F,H and F and Table 2). It was lower and similar in LCL-ATL and F-SP groups. Finally, all lesions groups showed a higher percentage of neutrophils than healthy skin.
NOS2 expression was higher in leishmaniasis than in sporotrichosis lesions (Table 3 and Fig. 2A,C,E,G). The highest expression was found in SCL-ATL lesions and 80% were classified as intense or very intense expression, followed by LCL-ATL (50%) and LC-SP (33.3%). F-SP lesions showed a similar expression of NOS2 when compared to healthy skin.0
All lesion groups showed more BCl-2+ and Ki67+ cells than healthy skin (Fig. 4A,B and Table 2). BCl-2+ cells were more common in LC-SP and less frequent in SCL-ATL. Although Fas expression was similar among the groups (Fig. 4C), FasL was more expressed in leishmaniasis lesions than sporotrichosis or healthy skin (Fig. 4D). There were no differences between sporotrichosis lesions and healthy skin, regarding FasL.
Discussion
In this study, we compared the in situ inflammatory response of ATL and SP, cutaneous lesions in order to evaluate how the skin immune system reacts to pathogens with different natures and in patients with diverse or similar clinical aspects. Our results have pointed to a general and similar inflammatory skin reaction with some differences according to the infection/clinical presentation. Although both infections primarily target the skin, they can present different degrees of in situ granulomatous reactions and different courses of disease. SP is often a subacute infection, which is characterized by an exudative reaction, whereas ATL is a chronic disease, which is characterized by a long duration of infection and the presence of non-exudative ulcers26,27,28. Furthermore, their infectious agents mainly affect different cellular compartments (extracellular and intracellular). As a result, we hypothesized that differences in the cellular composition and markers of the in situ inflammatory activity in ATL and SP lesions could reveal two different patterns: intracellular and extracellular responses. However, our results pointed that sometimes their arrangements can be similar as observed for SCL-ATL and LC-SP. In the present study, even pathogens with different natures could elicit similar immune responses and clinical aspects of lesions, which could reflect a host background. Furthermore, the same pathogen can also elicit different degree of immune response and severity of clinical lesions. Although the potential of clones/isolates from the same pathogens in eliciting different degrees of tissue damage should be considered29,30,31, it is now well established that the clinical presentation is a consequence of host-parasite balance32,33,34. In this context, it has been recently demonstrated35 the role of Th17 cells in pathogenesis and fungal burden control in sporotrichosis.
We also observed differences in the clinical presentation of ATL and SP. For example, ATL lesions developed more slowly than the SP lesions. In fact, after only 2 weeks of infection, 6 SP patients had already developed lesions. On the other hand, in ATL lesions, the infection usually occurred more than 2 months before the patient’s first medical visit. These observations suggest a relationship between the nature of the etiological agent and the immune response leading to the development of acute, subacute, or chronic lesions.
Our results showed that ATL lesions elicited a pattern of inflammatory response that is characterized by CD3+, CD8+, and FasL+ cells as well as higher NOS2 expression than SP lesions. As Leishmania spp. infects and thrives inside phagocytes, these cells should be activated or eliminated to reduce the parasite burden9,24,36. Since the cellular immune response and cytotoxicity have major roles in the effector immune response to control intracellular pathogens37, this profile would characterize the intracellular pattern of the skin immune response against intracellular pathogens. However, excess of NOS2 expression as observed in SCL-ATL and LC-SP when compared to counterpart’ lesions (LCL-ATL and F-SP) could be associated to more extensive and severe lesions suggesting an unbalanced/uncontrolled immune response. As macrophages were similar in all studied groups, we can suggest that these cells might be distinctly activated. In this context, M1 and M2 macrophages have already been demonstrated in literature38,39,40,41, but other analyses should be done to better clarify this hypothesis.
On the other hand, our results demonstrated that LC-SP lesions predominantly elicited an pattern of response that is characterized by more neutrophils and CD4+ cells, and fewer markers of cytotoxic response and NOS2 expression than ATL lesions. In response to extracellular pathogens like helminthes, Gram-negative bacteria, some fungi or some protozoa, it is thought that secretory factors, antibodies, enzymes, antibiotic peptides, and various cells, such as mast cells, neutrophils, and eosinophils, are responsible for reducing the microorganism burden42. However, there are few studies on the immune response in sporotrichosis. In a murine model of sporotrichosis infection, thymus-derived cells are involved in resistance to this infection, which suggests that cellular immunity plays an important role in host resistance to this pathogen43,44,45. Interestingly, SCL-ATL showed similar quantities of neutrophil elastase expression to LC-SP, and in both they were more intense than in the other groups. Neutrophil elastase can cause tissue liquefaction (pus)25. Although tissue damage is usually harmful, small amounts of neutrophil-mediated tissue damage are beneficial because they destabilize collagen fibers leading to the collapse of capillaries and lymphatic vessels and confine the infectious agent to a local, toxic environment where can be destroyed46. In addition, neutrophils also secrete chemotactic factors for T cells and can induce T cell activation via interferon γ47. The cooperation of macrophages and neutrophils in order to eliminate parasites or fungal cells respectively in leishmaniasis or sporotrichosis, has also been suggested25,48,49,50,51,52. However, our results showed that the highest concentration of neutrophils occurred in LC-SP and SCL-ATL lesions, which are both characterized by a regional lymphatic spread. It is worth note that neutrophil enzymatic contents are able to degrade the extracellular matrix, leading to an easier migration of immune cells53. However, this function also could facilitate fungus spread, through lymphatic vessels54,55. As a result, even in the presence of the correct stimulus, an unbalanced inflammatory reaction could increase tissue destruction and facilitate fungal or parasite spread.
Interestingly, despite the low number of B-lymphocytes their concentration was higher in LCL-ATL lesions, followed by SCL-ATL and LC-SP both in a similar degree. B lymphocytes and their secreted antibodies could play a role in parasite burden control in leishmaniasis opsonizing occasional extracellular amastigotes, inducing phagocytosis, complement activation or NK cells stimulation and interaction with T cells56,57,58,59. In sporotrichosis, the role of B cells and imunoglobulins were verified. The expression of specific immunoglobulins increases as the fungal load decreases in mice, which suggests that the humoral immune response could be related to pathogen elimination or to secondary protective mechanisms60.
Collectively, our results suggested that the SIS is a complex, adaptable structure that is capable of optimizing the response to intracellular or extracellular pathogens. However, an unbalanced inflammatory reaction could increase tissue destruction and worsen the disease.
Although these response models can account for the classical clinical presentation of LCL-ATL and LC-SP, some patients have atypical forms25,43,44,45 with different degrees of inflammation when compared with the typical clinical presentation. In this regard, F-SP lesions have an intermediate severity and development time when compared to LC-SP and ATL lesions. Consistent with these characteristics, the in situ inflammatory reaction in F-SP lesions exhibited fewer macrophages, CD3+, CD8+, and FasL+ cells as well as lower NOS2 expression than ATL lesions and fewer neutrophils and B cells than LC-SP lesions. In addition, our previous study24 showed that F-SP lesions have lower fungal burden than LC-SP lesions as well as clinical similarities (localized lesions without lymphatic alterations) with LCL-ATL lesions. On the other hand, when compared to F-SP, LC-SP lesions presented higher fungal burden and are clinically characterized by numerous skin nodules or plaques, following regional lymphatic dissemination, with an elevated degree of necrosis. On the basis of these findings, we hypothesize that the SIS response in patients with the F-SP rapidly controls the fungal burden, which reduces the severity of lesions and the exudative reaction, thereby promoting a more balanced immune response. Although this model of the immune response might not be completely applicable to the sporotrichoid form of ATL, it should be noted that even in SCL-ATL, the lesions are less exudative and develop more chronically than LC-SP lesions.
We cannot eliminate the possibility that different pathogen isolates could affect the SIS response to infection. However, at Rio de Janeiro State, Leishmania braziliensis is the causative agent of almost all cases and all isolates from ATL lesions included in our study were characterized as Leishmania braziliensis. Although the suggestion that different human Sporothrix spp isolates can affect both, the immune response and the development of infection in experimental sporotrichosis29, there is no clear relationship between human isolates and clinical presentation when genotype, protein expression, or antifungal susceptibility patterns are considered33,61. Moreover, in epidemic of sporotrichosis in Rio de Janeiro previous studies identified S. brasiliensis as most frequent species (up to 93.5% of the cases) by molecular analyses62,63. These results corroborate with data from Marimon and coworkers (2007) in a study that described the new species of complex Sporothrix being of 127 Sporothrix strains collected from several parts of the world reported only S. brasiliensis among the tested isolates from Rio de Janeiro64.
In addition, we cannot exclude the possibility that, during infection, Leishmania amastigotes and Sporothrix spp yeasts can be transiently localized in the extracellular and intracellular milieu, respectively. This possibility could be important to control the parasite and fungal burden. However, the duration of the transience and the effects on the skin immune response are not known. Though, our results suggested that the clinical presentation of infectious skin diseases could be due to a combination of factors from both, the host SIS and the etiological agent.
Therefore, we could draw two conclusions. First, the SIS is a complex, adaptable structure that is capable of responding with plasticity to intracellular or extracellular pathogens in order to control microorganism burden. Second, the clinical presentation of infectious skin diseases could result from a combination of factors from both, the host SIS and the etiological agent. Since more severe lesions from both SP and ATL presented an important concentration of neutrophils, or CD8+ T cells and NOS2 expression, respectively, our results also suggest that, unbalanced host SIS - parasite relationship can lead to more severe manifestations of skin infectious diseases.
Material and Methods
Patients
Patients were diagnosed and followed up in the outpatient clinic of the Laboratório de Pesquisa Clinica e Vigilância em Leishmanioses (LaP Clin VigiLeish), Instituto Nacional de Infectologia Evandro Chagas (INI), Fundação Oswaldo Cruz (FIOCRUZ), Rio de Janeiro, Brazil. A total of 105 individuals were divided into 5 groups: (1) Localized cutaneous leishmaniasis (LCL-ATL n = 30); (2) Sporotrichoid cutaneous leishmaniasis (SCL-ATL n = 18); (3) Fixed cutaneous sporotrichosis (F-SP n = 24); (4) Lymphocutaneous sporotrichosis (LC-SP n = 24); (5) Healthy skin (n = 9) from esthetical surgery. The study was approved by the Ethics Committee of INI-FIOCRUZ (04/2001) and all patients provided informed written consent. All methods were performed in accordance with the relevant guidelines and regulations related to researches using humans.
Tissue samples
Samples were obtained from the primary lesion from patients at the time of their investigative procedures for diagnosis. Normal skin was obtained from healthy individuals during esthetical surgery procedures. These samples were prepared for the following analyses: (1) histopathology (stored in 10% formalin buffer), (2) immunohistochemistry (stored at −196 °C in OCT medium (Tissue-Tek, Sakura Finetek, Torrance, CA, USA), and (3) collected in sterile saline and cultured to isolate fungal cells and Leishmania spp. In order to isolate fungal cells, the samples were cultured at 28 °C in Sabouraud Dextrose Agar supplemented with 20 U·mL−1 penicillin and 40 µg·mL−1 streptomycin (Sigma, St. Louis, MI, USA) during the hyphal phase. Then followed by growth at 37 °C in Brain Heart Infusion medium (Sigma) until the yeast like phase. The fungal strains obtained of six patients used in this study were characterized at species level according to Oliveira and collaborators62 using polifasic taxonomy. Briefly, fungi were sub-cultured on Potato dextrose agar and Mycobiotic agar (both from DifcoTM BD/Sparks MD, USA), and then identified by phenotypic and genotypic characteristics (macro and micromorphology, thermotolerance, carbohydrate assimilation and molecular assay). All of them were characterized as S. brasiliensis”To isolate Leishmania spp., the samples were cultured in biphasic medium Novy-MacNeal-Nicolle (NNN)/Schneider’s insect medium (Sigma) at 28 °C. Twenty-five isolates were typing through multilocus enzyme electrophoresis as L. braziliensis.
Histopathology
Formalin-fixed samples were stained with hematoxylin-eosin, and examined with a light microscope (Carl Zeiss Axioskop, Jena, Germany).
Immunohistochemistry
Immunohistochemistry was performed as described previously24. Three-micrometer-thick sections were mounted on silanized slides (Dakocytomation, Carpinteria, CA, USA), fixed in acetone, and hydrated in phosphate buffered saline (PBS, pH 7.4). After blocking endogenous peroxidase with peroxidase blocking reagent (Dakocytomation) and nonspecific staining with normal goat serum (Zymed, San Francisco, CA, USA), the slides were incubated with the following antibodies: CD3+ (clone UCHT1), CD4+ (clone MT310) and CD8+ (clone DK25) T lymphocytes, CD22+ (clone 4KB128) B lymphocytes, CD68+ (clone KP1) macrophages, Bcl-2+ (clone 124), Ki67+ (clone Ki-S5) and neutrophil elastase (clone NP57 - neutrophils) (Dakocytomation); nitric oxide synthase 2 (NOS2) (clone 6) (BD Transduction Laboratories, KY, USA); Fas (clone DX2), and FasL (clone G247–4) (BD Biosciences Pharmingen, San Jose, CA, USA). After that, the specimens were incubated with a biotinylated secondary antibody (goat anti-mouse IgG or goat anti-rabbit IgG – both from Zymed) followed by a streptavidin-biotin-peroxidase complex (ABC kit, Dakocytomation) and aminoethylcarbazole (AEC kit, Zymed). Subsequently, the slides were counterstained with Mayer’s hematoxylin (Dako) and examined under a light microscope (Zeiss). The percentage of stained cells was determined among 500 mononuclear cells. The intensity of NOS2 staining was measured in 10 microscope fields (200× magnification) and scored as discrete (1 positive), moderate (2 or 3 positive areas), intense (4 or 5 positive areas), or very intense (>5 positive areas), as described previously24. All experiments were replicated at least twice and the suppression of the primary antibody served as a negative control.
Statistical analysis
Statistical analyses were calculated with SPSS24 for Windows (SPSS, Inc., Chicago, IL, USA). Kolmogorov Smirnov test was used to evaluate the distribution of variables. The Mann-Whitney or t-Student tests and Kruskal Wallis or Anova tests and Bonferroni post hoc test were used to compare the groups. Data are reported as median, SEM and range. The p-value cutoff for statistical significance was 0.05.
References
Spellberg, B. The cutaneous citadel: a holistic view of skin and immunity. Life Sci. 67, 477–502 (2000).
Streilein, J. W. Lymphocyte traffic, T-cell malignancies and the skin. J Invest Dermatol. 71, 167–71 (1978).
Bos, J. D. & Kapsenberg, M. L. The skin immune system: progress in cutaneous biology. Immunol Today 14, 5–8 (1993).
Christoph, T. et al. The human hair follicle immune system: cellular composition and immune privilege. Br J Dermatol. 142, 862–73 (2000).
Paus, R., Nickoloff, B. J. & Ito, T. A “hairy” privilege. Trends Immunol. 26, 32–40 (2005).
Ito, T. et al. Immunology of the human nail apparatus: the nail matrix is a site of relative immune privilege. J Invest Dermatol. 125, 1139–48 (2005).
Conceição-Silva, F., Dórea, R. C., Pirmez, C., Schubach, A. & Coutinho, S. G. Quantitative study of Leishmania braziliensis braziliensis reactive T cells in peripheral blood and in the lesions of patients with American mucocutaneous leishmaniasis. Clin Exp Immunol. 79, 221–6 (1990).
da Conceição-Silva, F., Perlaza, B. L., Louis, J. A. & Romero, P. Leishmania major infection in mice primes for specific major histocompatibility complex class I-restricted CD8+ cytotoxic T cell responses. Eur J Immunol. 24, 2813–7 (1994).
Conceição-Silva, F., Hahne, M., Schröter, M., Louis, J. & Tschopp, J. The resolution of lesions induced by Leishmania major in mice requires a functional Fas (APO-1, CD95) pathway of cytotoxicity. Eur J Immunol. 28, 237–45 (1998).
Pirmez, C. et al. Immunologic responsiveness in American cutaneous leishmaniasis lesions. J Immunol Baltim Md 1950. 145, 3100–4 (1990).
Ahmed, A. A., Nordlind, K., Schultzberg, M. & Lidén, S. Interleukin-1 alpha- and beta-, interleukin-6- and tumour necrosis factor-alpha-like immunoreactivities in chronic granulomatous skin conditions. Acta Derm Venereol. 74, 435–40 (1994).
Khanolkar-Young, S., Snowdon, D. & Lockwood, D. N. Immunocytochemical localization of inducible nitric oxide synthase and transforming growth factor-beta (TGF-beta) in leprosy lesions. Clin Exp Immunol. 113, 438–42 (1998).
Orteu, C. H. et al. The role of apoptosis in the resolution of T cell-mediated cutaneous inflammation. J Immunol Baltim Md 1950. 161, 1619–29 (1998).
Bertho, A. L., Santiago, M. A., Da-Cruz, A. M. & Coutinho, S. G. Detection of early apoptosis and cell death in T CD4+ and CD8+ cells from lesions of patients with localized cutaneous leishmaniasis. Braz J Med Biol Res Rev Bras Pesqui Medicas E Biol. 33, 317–25 (2000).
Bravo, F. G. & Gotuzzo, E. Cutaneous tuberculosis. Clin Dermatol. 25, 173–80 (2007).
Narang, T., Dogra, S. & Kaur, I. Localized lepromatous leprosy in household contact of multibacillary disease. J Cutan Med Surg. 12, 139–41 (2008).
Araújo, M. G. Leprosy in Brazil. Rev Soc Bras Med Trop. 36, 373–82 (2003).
Marques, A. F. et al. Additive effect of P10 immunization and chemotherapy in anergic mice challenged intratracheally with virulent yeasts of Paracoccidioides brasiliensis. Microbes Infect. 10, 1251–8 (2008).
Restrepo, A., Benard, G., de Castro, C. C., Agudelo, C. A. & Tobón, A. M. Pulmonary paracoccidioidomycosis. Semin Respir Crit Care Med. 29, 182–97 (2008).
Schubach, A., Barros, M. B., de, L. & Wanke, B. Epidemic sporotrichosis. Curr Opin Infect Dis. 21, 129–33 (2008).
Lindoso, J. A. L. & Lindoso, A. A. B. P. Neglected tropical diseases in Brazil. Rev Inst Med Trop Sao Paulo. 51, 247–53 (2009).
Gomes, A. H. S., Armelin, I. M., Menon, S. Z. & Pereira-Chioccola, V. L. Leishmania (V.) braziliensis: detection by PCR in biopsies from patients with cutaneous leishmaniasis. Exp Parasitol. 119, 319–24 (2008).
Barros, M. B. et al. Cat-transmitted sporotrichosis epidemic in Rio de Janeiro, Brazil: description of a series of cases. Clin Infect Dis Off Publ Infect Dis Soc Am. 38, 529–35 (2004).
Morgado, F. N. et al. Is the in situ inflammatory reaction an important tool to understand the cellular immune response in American tegumentary leishmaniasis? Br J Dermatol. 158, 50–8 (2008).
Morgado, F. N., Schubach, A. O., Barros, M. B. L. & Conceição-Silva, F. The in situ inflammatory profile of lymphocutaneous and fixed forms of human sporotrichosis. Med Mycol. 49, 612–20 (2011).
Schubach, A. et al. Retrospective study of 151 patients with cutaneous leishmaniasis treated with meglumine antimoniate. Rev Soc Bras Med Trop. 38, 213–7 (2005).
Schubach, A. O., Schubach, T. M. P. & Barros, M. B. L. Epidemic cat-transmitted sporotrichosis. N Engl J Med. 353, 1185–6 (2005).
Barros, M. B. L., Schubach, A. O., Schubach, T. M. P., Wanke, B. & Lambert-Passos, S. R. An epidemic of sporotrichosis in Rio de Janeiro, Brazil: epidemiological aspects of a series of cases. Epidemiol Infect. 136, 1192–6 (2008).
Brito, M. M. S. et al. Comparison of virulence of different Sporothrix schenckii clinical isolates using experimental murine model. Med Mycol. 45, 721–9 (2007).
de Oliveira, F. S. et al. Genetic polymorphism in Leishmania (Viannia) braziliensis detected in mucosal leishmaniasis of HIV-infected and non-HIV-infected patients. Trans R Soc Trop Med Hyg. 106, 683–7 (2012).
Cantanhêde, L. M. et al. Further Evidence of an Association between the Presence of Leishmania RNA Virus 1 and the Mucosal Manifestations in Tegumentary Leishmaniasis Patients. PLoS Negl Trop Dis., e0004079 (2015).
Reithinger, R. et al. Cutaneous leishmaniasis. Lancet Infect Dis. 7, 581–96 (2007).
Galhardo, M. C. G. et al. Molecular epidemiology and antifungal susceptibility patterns of Sporothrix schenckii isolates from a cat-transmitted epidemic of sporotrichosis in Rio de Janeiro, Brazil. Med Mycol. 46, 141–51 (2008).
Remadi, L. et al. Clinical Presentation of Cutaneous Leishmaniasis caused by Leishmania major. Dermatol Basel Switz. 232, 752–9 (2016).
Gonçalves, A. C. et al. The NLRP3 inflammasome contributes to host protection during Sporothrix schenckii infection. Immunology. 151, 154–66 (2017).
Da-Cruz, A. M. et al. T-cell-mediated immune responses in patients with cutaneous or mucosal leishmaniasis: long-term evaluation after therapy. Clin Diagn Lab Immunol. 9, 251–6 (2002).
Louis, J. A., Conceiçao-Silva, F., Himmelrich, H., Tacchini-Cottier, F. & Launois, P. Anti-leishmania effector functions of CD4+ Th1 cells and early events instructing Th2 cell development and susceptibility to Leishmania major in BALB/c mice. Adv Exp Med Biol. 452, 53–60 (1998).
Martinez, P. et al. Macrophage polarization alters the expression and sulfation pattern of glycosaminoglycans. Glycobiology. 25, 502–13 (2015).
Ferrante, C. J. & Leibovich, S. J. Regulation of Macrophage Polarization and Wound Healing. Adv Wound Care. 1, 10–6 (2012).
Mukhopadhyay, D. et al. M2 Polarization of Monocytes-Macrophages Is a Hallmark of Indian Post Kala-Azar Dermal Leishmaniasis. PLoS Negl Trop Dis. 9, e0004145 (2015).
Siewe, N., Yakubu, A. A., Satoskar, A. R. & Friedman, A. Immune response to infection by Leishmania: A mathematical model. Math Biosci. 276, 28–43 (2016).
Fietta, P. & Delsante, G. The effector T helper cell triade. Riv Biol. 102, 61–74 (2009).
Iftikhar, N., Bari, I. & Ejaz, A. Rare variants of Cutaneous Leishmaniasis: whitlow, paronychia, and sporotrichoid. Int J Dermatol. 42, 807–9 (2003).
López-Escobar, M., Drake-Monfort, M., Salesa-Gutiérrez, de, R. R. & Hermana-Ramírez, S. Sporotrichoid cutaneous leishmaniasis. Actas Dermosifiliogr. 98, 444–5 (2007).
Bari, A. U. & Rahman, S. B. Many faces of cutaneous leishmaniasis. Indian J Dermatol Venereol Leprol. 74, 23–7 (2008).
Nathan, C. Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol. 6, 173–82 (2006).
Venuprasad, K., Chattopadhyay, S. & Saha, B. CD28 signaling in neutrophil induces T-cell chemotactic factor(s) modulating T-cell response. Hum Immunol. 64, 38–43 (2003).
Guimarães-Costa, A. B. et al. Leishmania amazonensis promastigotes induce and are killed by neutrophil extracellular traps. Proc Natl Acad Sci USA 106, 6748–53 (2009).
Morgado, F. N. et al. Are Neutrophil Extracellular Traps Playing a Role in the Parasite Control in Active American Tegumentary Leishmaniasis Lesions? PloS One. 10, e0133063 (2015).
Braian, C., Hogea, V. & Stendahl, O. Mycobacterium tuberculosis- induced neutrophil extracellular traps activate human macrophages. J Innate Immun. 5, 591–602 (2013).
Novais, F. O. et al. Neutrophils and macrophages cooperate in host resistance against Leishmania braziliensis infection. J Immunol Baltim Md 1950. 183, 8088–98 (2009).
Ribeiro-Gomes, F. L. et al. Neutrophils activate macrophages for intracellular killing of Leishmania major through recruitment of TLR4 by neutrophil elastase. J Immunol Baltim Md 1950. 179, 3988–94 (2007).
Boaventura, V. S. et al. Human mucosal leishmaniasis: neutrophils infiltrate areas of tissue damage that express high levels of Th17-related cytokines. Eur J Immunol. 40, 2830–6 (2010).
Nishikaku, A. S. et al. Burger, E. Matrix metalloproteinases with gelatinolytic activity induced by Paracoccidioides brasiliensis infection. Int J Exp Pathol. 90, 527–37 (2009).
de Araújo, M. S. et al. Evaluation of in situ expression of effector and regulatory cytokines, TLR, galectins and matrix metalloproteinases in oral manifestations of paracoccidioidomycosis. Immunobiology. 220, 154–63 (2015).
Cunha, C. F. et al. Cytotoxic cell involvement in human cutaneous leishmaniasis: assessments in active disease, under therapy and after clinical cure. Parasite Immunol. 38, 244–54 (2016).
Gibson-Corley, K. N. et al. Promotion of a functional B cell germinal center response after Leishmania species co-infection is associated with lesion resolution. Am J Pathol. 180, 2009–17 (2012).
Gibson-Corley, K. N., Boggiatto, P. M., Mukbel, R. M., Petersen, C. A. & Jones, D. E. A deficiency in the B cell response of C57BL/6 mice correlates with loss of macrophage-mediated killing of Leishmania amazonensis. Int J Parasitol. 40, 157–61 (2010).
Moore, J. W. J. et al. B cell: T cell interactions occur within hepatic granulomas during experimental visceral leishmaniasis. PloS One. 7, e34143 (2012).
Nascimento, R. C. & Almeida, S. R. Humoral immune response against soluble and fractionate antigens in experimental sporotrichosis. FEMS Immunol Med Microbiol. 43, 241–7 (2005).
Fernandes, G. F., Do Amaral, C. C., Sasaki, A., Godoy, P. M. & De Camargo, Z. P. Heterogeneity of proteins expressed by Brazilian Sporothrix schenckii isolates. Med Mycol. 47, 855–61 (2009).
Oliveira, M. M. E., Almeida-Paes, R., Muniz, M. M., Gutierrez-Galhardo, M. C. & Zancope-Oliveira, R. M. Phenotypic and molecular identification of Sporothrix isolates from an epidemic area of sporotrichosis in Brazil. Mycopathologia. 172, 257–67 (2011).
Almeida-Paes, R. et al. Sporotrichosis in Rio de Janeiro, Brazil: Sporothrix brasiliensis is associated with atypical clinical presentations. PLoS Negl Trop Dis. 8, e3094 (2014).
Marimon, R. et al. Sporothrix brasiliensis, S. globosa, and S. mexicana, three new Sporothrix species of clinical interest. J Clin Microbiol. 45, 3198–206 (2007).
Acknowledgements
This study was funded by IOC and INI-FIOCRUZ, PAEF-IOC-Fiocruz (IOC-008-FIO-15), FAPERJ (E26/111.230/2014), and CNPq, Brasil. A.O.S. is a fellowship beneficiary from CNPq and FAPERJ.
Author information
Authors and Affiliations
Contributions
F.N.M., L.M.V.C. and F.C.S. conceived and designed the research; F.N.M., L.M.V.C., J.L.S., A.J.S., M.I.F.P., M.L.R., M.M.O., A.F., M.F.M.,. and A.O.S. performed experiments; F.N.M., L.M.V.C., J.L.S., and F.C.S. prepared figures and wrote the manuscript.; F.N.M., L.M.V.C. and F.C.S. analyzed data.; F.N.M., L.M.V.C. and F.C.S. edited the manuscript and F.N.M., L.M.V.C., M.I.F.P., M.L.R., A.F., M.F.M., J.L.S., A.O.S. and F.C.S. revised and approved the final manuscript version.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Morgado, F.N., de Carvalho, L.M.V., Leite-Silva, J. et al. Unbalanced inflammatory reaction could increase tissue destruction and worsen skin infectious diseases – a comparative study of leishmaniasis and sporotrichosis. Sci Rep 8, 2898 (2018). https://doi.org/10.1038/s41598-018-21277-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-21277-1
- Springer Nature Limited
This article is cited by
-
cFLIPS regulates alternative NLRP3 inflammasome activation in human monocytes
Cellular & Molecular Immunology (2023)
-
Gallic and Vanillic Acids as Promising Succinate Dehydrogenase Inhibitors and Antigenotoxic Agents
Revista Brasileira de Farmacognosia (2021)