Abstract
Plant growth promoting rhizobacteria (PGPR) provide an effective and environmentally sustainable method to protect crops against pathogens. The spore-forming Bacilli are attractive PGPR due to their ease of storage and application. Here, we characterized two rhizosphere-associated Bacillus velezensis isolates (Y6 and F7) that possess strong antagonistic activity against Ralstonia solanacearum and Fusarium oxysporum under both laboratory and greenhouse conditions. We identified three lipopeptide (LP) compounds (surfactin, iturin and fengycin) as responsible for the antimicrobial activity of these two strains. We further dissected the contribution of LPs to various biological processes important for rhizosphere colonization. Although either iturin or fengycin is sufficient for antibacterial activity, cell motility and biofilm formation, only iturin plays a primary role in defense against the fungal pathogen F. oxysporum. Additionally, we found that LP production is significantly stimulated during interaction with R. solanacearum. These results demonstrate the different roles of LPs in the biology of B. velezensis and highlight the potential of these two isolates as biocontrol agents against phytopathogens.
Similar content being viewed by others
Introduction
There is a global need to meet rising food demand from a growing human population in the face of rising energy costs, pressure on natural resources, and concern over global warming1. Pressure to meet increased demand has led to the use of chemical pesticides and fertilizers2, and the development of genetically resistant plant cultivars3,4. Both strategies have limitations. Agrochemicals are not effective against all diseases and can pose a health and environmental risk as toxic residues can accumulate in the soil and enter the food supply. The manufacture of conventional nitrogen-based fertilizers is dependent on fossil fuel energy resources and can have adverse effects on environmental and human health. Disease resistance of genetically modified plants is often overcome by the pathogen within a few years and there is a general lack of public acceptance for genetically modified plants in the food supply5. The application of plant growth promoting rhizobacteria (PGPR) and fungi offers an environmentally sustainable alternative to the use of genetically modified plants or synthetic chemicals6.
Gram-positive Bacillus species are attractive PGPR7. First, they form heat and desiccation resistant endospores that can be formulated as a stable dry white powder with a long shelf life8. Second, Bacillus species are common inhabitants of the microflora of crops including tomato (Solanum lycopersicum), banana (Musa acuminata), sweet corn (Zea mays convar. saccharata var. rugosa), grape (Vitis spp.), and wheat (Triticum aestivum). Thus, PGPR can suppress phytopathogens by outcompeting them for colonization of the rhizosphere. More importantly, PGPR produce a wide range of antibiotic compounds including non-ribosomally encoded lipopeptides (LPs)9,10. These LPs are the major contributor to Bacillus biocontrol activity. For instance, B. amyloliquefaciens FZB42, recently reassigned as B velezensis11, produces fengycin and bacillomycin D, which show synergistic antagonistic activity against the fungal pathogen Fusarium oxysporum9. B. velezensis SQR9 produces bacillomycin D, which contributes to biocontrol activity against F. oxysporum12.
Ralstonia solanacearum is an economically important bacterial phytopathogen13 and infects a broad range of host plants including crops such as potato (Solanum tuberosum), eggplant (Solanum melongena), and tomato (Solanum lycopersicum)14,15. Tomato wilt caused by R. solanacearum is one of the most damaging tomato diseases, particularly during the hot-humid summer season in subtropical regions such as South China. No completely effective chemical controls are available16,17. A few resistant tomato cultivars have been developed such as FL7514 and BHN46618,19; however, host resistance is often linked with unwanted traits such as reduced fruit size20. Some LP-producing Bacillus species have been reported as promising biocontrol agents21,22,23. However, the role of LPs in their biocontrol activity against R. solanacearum is unclear.
F. oxysporum f. sp. cubense 4 (Foc), a soil-borne fungus, is the causative agent of banana Fusarium wilt (also called Panama disease). Fusarium wilt is a global threat to the banana industry. It has seriously affected banana plantations in many countries and caused severe crop losses24,25. In South China, Fusarium wilt is the major disease faced by many banana plantations26. The disease incidence is in the range of 20 to 40%, with some plantations reaching a rate of 90%27. Most commercial fungicides are ineffective at controlling the disease. Currently, the best solution is the use of genetically resistant cultivars, but these plants are not resistant to all races of F. oxysporum. Emerging evidence has shown that LP-producing PGPR offers a potential environmentally-friendly biocontrol method against F. oxysporum9,12. However, PGPR isolated from non-native locations may not perform well, since they may not survive in the local soil environment due to circumstantial variables (e.g. pH, moisture, temperature), may be outcompeted by indigenous bacteria, or may not be effective against local pathogens28,29. These considerations motivated us to isolate and characterize PGPR endemic to the area where they will be applied.
Here we isolated and characterized two rhizosphere-associated B. velezensis strains (Y6 and F7) from a local farm in South China. These strains exhibit potent biocontrol activity against tomato bacterial wilt and banana Fusarium wilt under greenhouse conditions. We identified and quantified the antimicrobial LPs secreted by the two strains and defined their roles in biological processes relevant to rhizosphere colonization and plant protection. Either iturin or fengycin is sufficient for antibacterial activity, cell motility and biofilm formation, whereas iturin but not fengycin plays an essential role in antagonism against Foc. These two potential biocontrol agents offer a promising strategy to combat plant pathogens.
Results
Isolation and identification of two Bacillus strains with significant antibacterial activity
Many rhizosphere-associated Bacillus isolates exhibit antimicrobial activity towards phytopathogens10,12,30. To isolate antagonistic Bacillus species for use in the subtropical regions of China, we collected rhizosphere soil from healthy tomato plants in a local farm in Guangzhou. Among 60 isolated strains, two exhibited the strongest inhibitory effects against R. solanacearum GMI1000 on Casamino acid-Peptone-Glucose (CPG) plates using an optimized spot-on-lawn assay, where a high-density culture of the test strain is spotted on a low-density lawn of the indicator strain (R. solanacearum) (Fig. 1A). These two isolates were named Y6 and F7. Phylogenetic trees, constructed based on 16S rRNA and gyrA gene sequences, indicate that Y6 and F7 cluster with plantarum subspecies of B. velezensis such as S499, UMAF6614, SQR9 and FZB42 (Fig. S1).
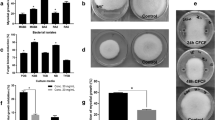
The two B. velezensis isolates (Y6 and F7) exhibit strong antibacterial activity against R. solanacearum in vitro and in vivo. (A) To evaluate the inhibitory activity of the two isolates against R. solanacearum, a spot-on-lawn assay was performed. The clearance zone indicated by the black lines was measured after 24 h incubation at 30 °C (Y6, 10 ± 0.2 mm, F7, 8 ± 0.2 mm, mean ± SD with n = 3). B megaterium X3 and B. subtilis 168 served as controls and showed no inhibition activity. The scale bar is 5 mm. (B) The biocontrol ability of the two isolates to suppress tomato bacterial wilt was evaluated in pot experiments during spring and autumn using non-sterile local soil (see Additional Methods in the SI materials). Three groups were included: control (CK), only inoculated with R. solanacearum (Rs); Y6 + Rs, inoculated with Y6 and Rs; and F7 + Rs, inoculated with F7 and Rs. The wilt incidence was calculated on the 30th day after transplanting as described in Materials and Methods. The data are expressed as the mean ± SD (n = 24). Significant differences between the control and treated group (Y6 or F7) are determined by Tukey’s Studentized Range (HSD) Test as indicated: α = 0.05, n = 24. (C) Representative photographs of the tomato plants to show typical wilt symptoms in CK group while only minor-to-negligible symptoms in the two treated groups after 25 days of transplanting in the spring pot experiments.
These two isolates showed strong antimicrobial activity against R. solanacearum in vitro. Thus, we tested their ability to suppress tomato wilt caused by R. solanacearum in pot experiments under greenhouse settings during spring and autumn. After treatment with either Y6 or F7, the wilt incidence was significantly reduced in both sets of experiments (Fig. 1B,C). For example, in pot experiments done in spring, the wilt incidence in the control group (~65%) was reduced to 29% or 35% for Y6- or F7-treated groups, respectively (Fig. 1B,C). These results reveal that the disease control ability (biocontrol efficacy) of these two isolates approaches 50%.
Identification of the lipopeptide compounds secreted by Y6 and F7
In some PGPR, the biocontrol activity is linked to their ability to produce lipopeptide (LP) compounds21,31. We isolated the secreted metabolites produced by the Y6 and F7 strains grown in LB medium and both extracts showed very strong activity against R. solanacearum with a clearance zone of 13–14 mm in diameter (Fig. 2A). We then identified the LP compounds from the extracts using ultra-performance liquid chromatography coupled with mass spectrometry (UPLC-MS). Three major LPs were detected: iturin, fengycin, and surfactin (Fig. 2B). Multiple isoforms of each LP were observed, including five for surfactin (C12 to C16), three for iturin (C14 to C16), and six for fengycin (C12 to C16) (Fig. 2B and Table S3). Consistent with the observation that Y6 showed higher antimicrobial activity against R. solanacearum than F7 (Fig. 1A), Y6 produces significantly higher levels of iturin and fengycin than F7 (Fig. 2C,D and Table S3).
Identification and quantification of LP compounds from the two isolates. (A) The methanol extracts from the two isolates showed strong antibacterial activity against R. solanacearum, as judged by an agar diffusion assay. Five µl of the methanol extracts of Y6, F7, B. megaterium X3 (X3; control), and methanol only (control) were tested. The latter two served as controls and did not show any inhibitory effects. The clearance zone indicated by the black lines is evident when treated with the methanol extract from either isolate (Y6, 14 ± 0.1 mm; F7, 13 ± 0.1 mm; mean ± SD with n = 3). The scale bar is 5 mm. (B) Identification of LP compounds from the methanol extract of Y6 or F7 using UPLC-MS analysis. Mass spectrum [M+H]+ of LPs are shown, surfactin ((i, Y6) and (iv, F7)), fengycin((ii, Y6) and (v, F7)), and iturin((iii, Y6) and (vi, F7)). (C) Representative chromatograms of the LPs (iturin, fengycin and surfactin) in methanol extract (3 μl) from strain Y6 or F7 using UPLC-MS analysis. (D) Quantification of the LP compounds (surfactin, iturn and fengycin) secreted by Y6 or F7. Cell were grown in LB medium at 30 °C for 48 h, harvested by centrifugation, and subjected to methanol extraction for LP analysis. The LPs were then quantified based on the standard curves of commercial standards surfactin, iturin and fengycin. Significant differences between Y6 and F7 were determined by two-tailed t-test as indicated: **P < 0.01, n = 3.
To further evaluate the antimicrobial potency of these isolated LPs, we tested the minimum inhibitory concentration (MIC) of the LP standards and compared them with the extracts from Y6 or F7. Both iturin and fengycin standards showed relatively strong antibacterial activity against R. solanacearum with a MIC of 100 µg ml−1 (Fig. S2B,C), while surfactin showed moderate activity with a MIC of 500 µg ml−1 (Fig. S2A). By contrast, the MIC of the extract from either Y6 or F7 is 75 µg ml−1 (Fig. S3D,E), considerably lower than that of any single LP (Fig. S2A–C), suggesting that these three LPs may have synergistic antagonistic activity against pathogenic organisms.
LP production is strongly stimulated in the presence of R. solanacearum
LP production by PGPR can be modulated by the presence of a pathogenic organism32. Thus, we evaluated the influence of R. solanacearum on the production of LPs by Y6 and F7. CPG agar samples were harvested from the clearance zone (Fig. 1A) and LP compounds were extracted using an acetonitrile/water based method and then subjected to UPLC-MS analysis. Co-culture with R. solanacearum significantly stimulated LP production, particularly fengycin and iturin. When encountering the pathogen, iturin production increased 11- and 20-fold (F7 and Y6 respectively) and fengycin production increased 15- and 38-fold (F7 and Y6, respectively) (Fig. 3A,B and Table 1). A moderate increase of ~4-fold was observed for surfactin production by Y6 and F7 (Fig. 3A,B and Table 1).
R. solanacearum strongly stimulates production of LPs. (A) LP compounds were extracted from the inhibition zone (Fig. 1A) and quantified by UPLC-MS analysis as described in Materials and Methods. Briefly, after the inhibition zone against R. solanacearum (Rs) became evident after 24 h incubation at 30 °C, a 300 mg of agar sample was harvested from the inhibition zone, mixed with 1 ml of acetonitrile/water (1:1 v/v), and sonicated for 30 s followed by centrifugation and filtration. The supernatant was collected as the acetonitrile/water extract and subjected to UPLS-MS analysis. The control was performed in a similar way but without the pathogen Rs in the top soft agar layer. (A) 300 mg of agar sample was collected around the well (2–4 mm). The two isolates produce more LPs, particularly fengycin and iturin, when they encounter the pathogen R. solanacearum. B. Representative chromatograms illustrate the differences in LP production by strain Y6 or F7 in the presence or absence of the co-culturing pathogen R. solanacearum. (C) The presence of R. solanacearum affects expression of LP biosynthesis genes. Expression of the LP biosynthesis genes (srfAB, ituC and fenD for synthesis of surfactin, iturin, and fengycin, respectively) was monitored in the two isolates in the presence or absence of the co-culturing pathogen Rs. RNA extraction and qPCR were performed as described in Materials and Methods. The relative gene expression (in fold change) was calculated using the 2−ΔΔCt method. For (A,C), significant differences between the Rs infected and control groups were determined by two-tailed t-test as indicated: **P < 0.01, *P < 0.05, n = 3.
Each LP contains multiple isoforms (Fig. 2B, Table 1 and Table S3) that may possess different antimicrobial activity and which may change upon the presence of other bacteria33,34. Indeed, upon R. solanacearum challenge, the isoform profile of all three LPs changed: the C15 isoform increased in all three LPs and became the dominant iturin and surfactin species (more than 50%) (Table 1). Whether or not the C15 isoform is most effective against R. solanacearum is unknown.
The elevated LP production by F6 and Y7 in response to R. solanacearum might be due to an increase in expression of LP biosynthesis genes. In fact, the expression of LP genes is highly upregulated in several Bacillus isolates during interaction with phytopathogens35,36, although LP production was not quantified in these studies. To test this notion, we monitored LP gene expression in the presence of R. solanacearum. Indeed, the expression of fenD (encoding fengycin synthetase D) in both Y6 and F7 was significantly upregulated (5-6-fold), whereas ituC (encoding iturin A synthetase C) and srfAB (encoding surfactin synthetase A) expression increased 2-3-fold upon encountering R. solanacearum (Fig. 3C). These results are consistent with the LP quantification results (Fig. 3A,B and Table 1) and further confirm that interaction with R. solanacearum induces expression of LP genes resulting in stimulated LP production.
Iturin and fengycin play a redundant role in antagonism against R. solanacearum
Other compounds in addition to the LPs produced by Y6 and F7 may contribute to antagonistic activity against phytopathogens. Moreover, it is difficult to accurately assess the level of LP production and activity in the soil environment. Therefore, to dissect the contribution of each individual LP compound to antimicrobial activity, we constructed mutants lacking each LP individually or in combination by disrupting LP biosynthesis genes required for iturin, fengycin and surfactin production (ituA, fenC, and srfAA, respectively) in Y6. Y6 was chosen for further study because F7 is naturally resistant to some antibiotics such as spectinomycin, which makes mutant construction more difficult. Loss of any individual LP (ituA, fenC, or srfAA) did not affect the capacity of Y6 to suppress R. solanacearum growth on CPG plates, whereas the ituA fenC double mutant completely lost the ability to inhibit R. solanacearum growth (Fig. 4). These results suggest that iturin and fengycin are the two major antimicrobial compounds and are functionally redundant in suppression of R. solanacearum.
Iturin A and fengycin play a redundant role in defense against R. solanacearum. The antagonistic activity of Y6 (WT) and its derived mutants [including srfAA (deficient in surfactin synthesis), ituA (deficient in iturin synthesis), fenC (deficient in fengycin synthesis) and ituA fenC (deficient in both iturin and fengycin synthesis)] against the pathogen R. solanacearum was tested using a spot-on-lawn assay. The clearance zone indicated by the black lines was evaluated after incubation at 30 °C for 24 h. The experiments were performed at least three times. Representative photographs are shown. The scale bar is 5 mm.
Iturin and fengycin have been proposed to have synergistic antagonistic activity against phytopathogens9. However, neither ituA nor fenC single mutants showed reduced antibacterial activity against R. solanacearum, suggesting either a lack of synergy in this case or that deficiency in biosynthesis of one LP might be compensated for by increase in production of another LP upon encountering pathogens. Indeed, quantification of LP production in wild-type (Y6), ituA, and fenC single mutants revealed that iturin production in the fenC mutant increased 2-fold (Fig. S3B,C). To test if the crosstalk between LPs occurs at the transcriptional level, we monitored the expression of ituC and fenD genes in wild-type (Y6), ituA, and fenC single mutants by qPCR during co-culture with R. solanacearum. Expression of fenD in the ituA mutant was upregulated about 2-fold compared to wild-type, and expression of ituC in the fenC mutant also increased about 2-fold (Fig. S3A). These results suggest that interactions between the LP biosynthesis pathways at the transcriptional level can allow one LP to compensate, at least in part, for the defective production of another.
Iturin is the primary factor responsible for antifungal activity against Foc in vitro and in vivo
PGPR can also provide protection from fungal pathogens8,32. To evaluate the antifungal ability of these two isolates, we tested their antagonistic activity against several common fungal plant pathogens using a plate confrontation assay. Both Y6 and F7 significantly inhibited the growth of Fusarium oxysporum f.sp. cubense (Foc), F. oxysporum f.sp. cucumerinum, and Colletotrichum gloeosporioides on Potato-Dextrose-Agar (PDA) plates (Fig. S4). We then assessed the contribution of iturin and fengycin to antifungal activity using an optimized spot-on-lawn assay (Fig. 5A), which is more sensitive compared to the traditional plate confrontation assay (see Materials and Methods). The fenC single mutant did not affect the capacity of Y6 to suppress the wildgrowth of the fungal pathogen Foc (Fig. 5A). Surprisingly, both the ituA single mutant and the ituA fenC double mutant almost completely lost the ability to inhibit Foc growth, although there is a sensitivity zone with reduced cell growth around the well where the ituA single mutant was inoculated (Fig. 5A). Similar results were observed in a plate confrontation test (Fig. S5).
Iturin A is the primary antifungal factor against F. oxysporum. (A) The antagonistic activity of Y6 (WT) and its derived mutants [including ituA (deficient in iturin synthesis), fenC (deficient in fengycin synthesis) and ituA fenC (deficient in both iturin and fengycin synthesis)] were tested against F. oxysporum (Foc) using a spot-on-lawn assay evaluated after incubation at 30 °C for 24 h. The experiments were performed at least three times. Representative photographs are shown. The scale bar is 5 mm. (B) To understand the contribution of iturin and fengycin in the biocontrol efficacy against banana Fusarium wilt in vivo, banana pot experiments were performed under greenhouse condition using non-sterile local soil (see Additional Methods in the SI materials). Same strains listed in panel (A) were tested. Two control groups were included as follows: CK1 (no Foc) and CK2 (Foc). Four treated groups were included: WT (Foc + Y6), ituA (Foc + ituA single mutant), fenC (Foc + fenC single mutant) and ituA fenC (Foc + ituA fenC double mutant). The wilt incidence of the banana plants was monitored every 3 days after transplantation. (C) Data presented are the wilt incidence of the banana plants among the same six different groups as described in Fig. 5B on the 30th day after transplanting. The data are expressed as the mean ± SD (n = 30). Significant differences among different groups indicated as different letters on top of the data bars are determined by Tukey’s Studentized Range (HSD) Test (α = 0.05). (D) Representative photographs of the banana plants to show the wilt incidence in six different groups including CK1 (No Foc), CK2 (Foc), Foc + WT, Foc + ituA, Foc + fenC and Foc + ituA fenC on the 30th day after transplanting.
In addition to their fungicidal activity, LPs are known to inhibit fungal growth by inhibiting spore germination. We conducted a spore germination assay using LP commercial standards and Y6 extract. As expected, the Y6 extract showed strong antagonistic activity against Foc and 75 μg ml−1 of the LP extract prevented ~60% of the spores from germinating. A similar level of inhibition was observed for the iturin standard (Fig. S6). By contrast, fengycin only showed minor antifungal activity even with the highest concentration tested (200 μg ml−1) and surfactin showed no evident inhibitory activity (Fig. S6). These results indicate that iturin is the major LP responsible for antifungal activity in vitro and fengycin plays a minor role.
To further investigate the biocontrol activity of Y6 and its derived LP-defective mutants against banana Fusarium wilt, we set up banana pot experiments in greenhouse settings. Treatment with WT (Y6) significantly reduced the wilt incidence, 48% by day 30 after transplanting compared to 86% in the untreated group (Fig. 5B–D). Deletion of fenC did not significantly affect the wilt incidence (51% by day 30). By contrast, the ituA single and ituA fenC double mutants only retained minor suppression ability (Fig. 5B–D). The biocontrol efficacy of Y6 was 44% against banana Fusarium wilt and iturin alone contributes about 90% of the total biocontrol efficacy. These results suggest that iturin is the major contributor against Foc in vivo under controlled-environmental conditions.
Either iturin or fengycin is sufficient for biofilm formation
Bacteria often exist in the environment as cell communities called biofilms, which are required to effectively colonize plant roots and protect cells against unfavorable conditions37,38. Thus, biofilm formation is a prerequisite for effective Bacillus PGPR activity22,39. Surfactin is well known for its role in biofilm formation in the phylloplane and rhizosphere22,40,41. However, the contribution of the other LPs to biofilm formation is unclear. Therefore, we assessed the contribution of all three LPs produced by Y6 to this multicellular process by monitoring pellicle formation and colony morphology in biofilm stimulating media (LBGM and MSgg), where no evident growth defects were observed for all the strains tested (Fig. S7). As expected, WT (Y6) forms robust, wrinkled pellicles on LBGM (Fig. 6A) and MSgg (Fig. 6C) liquid medium. WT also forms complex colony patterns on both LBGM (Fig. 6B) and MSgg plates (Fig. 6D). The srfAA single mutant, which is defective in surfactin production, showed apparent defects in colony morphology on both LBGM and MSgg plates, but no noticeable defects in pellicle formation (Fig. 6). Mutation of ituA or fenC alone has no evident effects in biofilm formation. By contrast, the ituA fenC double mutant displays a flat and non-wrinkled biofilm architecture and an unstructured pellicle in comparison to the wild-type strain (Fig. 6), indicating iturin and fengycin, like surfactin, play an important but redundant role in facilitating biofilm formation.
Either iturin A or fengycin is sufficient for biofilm formation. Pellicle formation on LBGM (A) and MSgg (C) liquid media and colony morphology on LBGM (B) and MSgg (D) plates was monitored for WT (Y6) and its derived mutants including srfAA (deficient in surfactin synthesis), ituA (deficient in iturin synthesis), fenC (deficient in fengycin synthesis) and ituA fenC (deficient in both iturin and fengycin synthesis). Representative photographs were taken after 24 or 36 h of incubation at 30 °C. The scale bar is 5 mm.
Either iturin or fengycin is sufficient for cell motility
Many bacteria move on surfaces towards nutrient-rich niches. In the rhizosphere, the ability of Bacillus to sense and move towards secreted plant compounds is critical for root colonization42. LPs and biosurfactants such as surfactin play important roles in the motility of some Pseudomonas and Bacillus isolates43. To understand the contribution of LPs to cell motility, we tested the surface motility of WT (Y6) and its derived mutants on LBGM medium plates prepared with varying levels of agar (Fig. S7). On “swim plates” (LBGM plates containing 0.3% agar)44, the wild-type (Y6) strain moved rapidly and completely spread over the whole plate within 4 h with a diameter of 85 mm. The ituA or fenC single mutants showed moderate defects in swimming with a diameter of 73 or 80 mm, respectively (Fig. 7A). In contrast, the ituA fenC double mutant almost completely lost its ability to swim with a diameter of 22 mm by 4 h, very similar to that of a surfactin-deficient strain (srfAA) (Fig. 7A). On plates to assess swarming motility (LBGM plates containing 0.7% agar)44, WT or either single mutant (ituA or fenC) was able to completely colonize the surface with a diameter of 85 mm by 15 h, whereas the ituA fenC double mutant showed a significant defect in swarming with a diameter of 60 mm by 15 h (Fig. 7B,C), close to that of srfAA mutant (50 mm in diameter by 15 h). These results suggest that in addition to surfactin, iturin and fengycin play important roles in surface movement, and either one is sufficient to facilitate cell motility.
Either iturin or fengycin is sufficient for cell motility. (A) Swimming ability of Y6 (WT) and its derived mutants including srfAA (deficient in surfactin synthesis), ituA (deficient in iturin synthesis), fenC (deficient in fengycin synthesis) and ituA fenC (deficient in both iturin and fengycin synthesis) was tested. Five µl of LB preculture (OD600~0.4) was spotted onto LBGM plates (0.3% agar) and plates were incubated at 37 °C for 3 h or 4 h as indicated. (B) Swarming ability of the same strains as in panel (A) was evaluated. 5 µl of LB culture (OD600~0.4) were spotted on LBGM plates containing 0.7% agar and plates were incubated at 37 °C for 4 or 15 h. For both (A,B), data are expressed as the mean ± SD (n = 3). Significant differences among different strains at each timepoint tested are indicated as different letters on top of the data bars. The statistical analysis was determined by a Tukey’s Studentized Range (HSD) test: α = 0.05, n = 3. (C) Representative photographs to show the differences in colony swarming morphology among Y6 (WT) and its derived mutants at different time points (4 and 15 h).
Discussion
Tomato bacterial wilt and banana Fusarium wilt are two destructive diseases to crops. Most chemical controls are ineffective against these diseases. Some lipopeptide (LP)-antibiotic producing Bacillus species offer a promising strategy in plant disease management. Here, we isolated two rhizosphere-associated B. velezensis strains (Y6 and F7) that exhibit potent antagonistic activity against bacterial and fungal pathogens in both laboratory and greenhouse settings. We demonstrated that the two LPs (iturin and fengycin) secreted by these two isolates are, chiefly if not solely, responsible for their antimicrobial activity. We then observed that expression of LP biosynthesis genes is upregulated and production of LPs is greatly stimulated in the presence of R. solanacearum. Modulation of LP production by pathogenic organisms may be a common natural phenomenon32,45,46. Indeed, the expression of LP genes including ituC, fenD, and srfAA in B. amyloliquefaciens D29 is significantly upregulated during interaction with R. solanacearum45 and in B. amyloliquefaciens Bk7 in response to Pseudomonas fuscovaginae46. Production of iturin and fengycin is strongly enhanced in B. subtilis 98S encountering fungal phytopathogens32. Interestingly, only fenC but no other LP genes is upregulated by the fungal pathogen Rhizomucor variabilis47. Thus, modulation of LP production might be a species-specific response between antagonist and pathogen. Although the triggering mechanism is largely unknown, it may involve the DegS/U two component sensing system, which is involved in the switch from a motile to sessile lifestyle and is thought to regulate lipopeptide production in B. subtilis NCD-2 and B. velezensis FZB4248,49.
The relative contribution of different LPs to antimicrobial activity may be dependent on the species of plant pathogen encountered. Our results showed that iturin and fengycin are functionally redundant in antagonism against R. solanacearum. By contrast, we observed that iturin, not fengycin, produced by Y6 plays the primary role, both in vitro and in vivo, in defense against the fungal pathogen Foc. Similar observations have been reported previously in other Bacillus isolates. In B. velezensis FZB42 and SQR9, bacillomycin D (a LP antibiotic related to iturin) is major contributor to antifungal activity against Foc while fengycin plays a minor role9,12. Fengycin has been observed to play an important role in antagonism against various fungal phytopathogens in B. subtilis UMAF6639 and EA-CB001550,51. Additionally, in B. subtilis UMAF6639, iturin but not fengycin inhibits the pathogens Pectobacterium carotovorum and Xanthomonas campestris31.
Surfactin has been shown to have antagonistic activity in vitro and play protective roles in the rhizosphere22,39,40. We investigated the contribution of surfactin to the protective effects of the isolate Y6. Our results show that surfactin plays a minor role in antagonism against R. solanacearum (Fig. 4). We observed that fengycin and iturin production by Y6 is dramatically induced during interaction with R. solanacearum while surfactin levels remain low (Figs 2 and 3). Furthermore, surfactin is the least active antibacterial compound among all three LPs tested (Fig. S2).
The contribution of LPs to biocontrol outside of their direct antimicrobial activity is not well understood. We constructed LP deficient mutants and dissected the contribution of each individual LP to different biological processes. Surfactin may contribute to traits required for effective rhizosphere colonization such as biofilm formation and cell motility as reported previously39,40,52,53. Indeed, surfactin is important for biofilm formation, particularly on a solid surface (Fig. 6B,D), and is critical for cell motility (Fig. 7). Our results also show that iturin and fengycin contribute to biofilm formation and motility, as observed in B. subtilis 91648, B. velezensis LL3 and SQR912,54. Thus, we propose that the biocontrol activity of Y6 is the result of coordinated actions of all three LPs.
Collectively, this study demonstrates that two LP-producing B. velezensis isolates (Y6 and F7) have potent antagonistic activity against R. solanacearum and Foc under both laboratory and greenhouse conditions. LP production in these two isolates is strongly stimulated in the presence of the pathogenic strain R. solanacearum and is critical for antagonism and colonization in controlled-environmental conditions. Most importantly, these two isolates, as potential biocontrol agents, may provide an effective strategy to combat plant pathogens in the target environment.
Materials and Methods
Isolation of antagonistic Bacillus species
Samples were collected from the rhizosphere soil of tomato plants in Yuejin Farm, Guangzhou, China (23° 08′N 113° 16′E). Bacillus spp. were isolated as reported previously55. Briefly, a soil sample (10 g) was shaken in 90 ml of sterilized water for 30 min, heated for 30 min at 80 °C, serially diluted and then spread over lysogeny broth (LB)56 plates. Single bacterial colonies were streaked onto fresh LB plates after 48 h of incubation at 30 °C. Frozen stocks of the purified colonies were prepared using 15% glycerol and kept at −80 °C for further study.
Antibacterial activity test
The antibacterial activity of these isolates and their derived mutants against R. solanacearum GMI100057,58 was evaluated using an optimized spot-on-lawn assay. 200 μl of R. solanacearum cell culture (OD600 ~0.4) grown in Casamino acid-Peptone-Glucose (CPG, 0.1% peptone, 0.01% casamino acids, 0.05% glucose) was mixed with 4 ml of 0.7% CPG soft agar and directly poured onto a CPG plate (1.5% agar). Plates were dried for 50 min and a well (5 mm in diameter) was made in the center of each plate, and 50 μl of each Bacillus strain (OD600~0.4) grown in LB medium was added into each well. Antagonistic activity was evaluated based on the inhibition zone after 24 h incubation at 30 °C. Antibacterial activity of the methanol extracts of Y6 and F7 was tested using an agar diffusion assay. Briefly, 200 μl of R. solanacearum CPG culture (OD600 ~0.4) was mixed with 4 ml of 0.7% CPG soft agar and directly poured onto a CPG plate (1.5% agar), and 5 µl of methanol extract was added into each well (5 mm in diameter). The methanol extracts (5 µl) of B. megaterium X3 (Genbank accession number KJ526881) and methanol alone were used as controls. The inhibition zone was measured after 24 h of incubation at 30 °C. The experiments were performed at least three times.
Antifungal activity test
We performed two assays, a plate confrontation assay and a spot-on-lawn assay, to test activity of the isolates (Y6 and F7) and derived mutants against common fungal pathogens including Fusarium oxysporum f.sp. cubense 4 strain XJZ2 (Foc4, GenBank accession number JX090598)59, F. oxysporum f.sp. cucumerinum (isolated from cucumerium rhizosphere, no accession number available yet), and Colletotrichum gloeosporioides (Genbank accession number MWUF00000000)60. In the plate confrontation assay, the fungi were cultivated on potato-dextrose-agar plates (PDA, 20% potato infusion, 2% dextrose, and 1.5% agar) at 30 °C for 5 days. A 5-mm-diameter block of mycelium agar was cut and transferred into the center of a fresh PDA plate. After 1 day of incubation, 3 µl of Bacillus spp. cells (F7, Y6, or mutants derived from Y6) (OD600 ~0.4) grown in LB medium was spotted on the PDA plate 2.5 cm away from the center, where the mycelium agar block was placed. The antifungal activity was evaluated by measuring the diameter of the inhibition zone (the distance between the Foc mycelium and the bacterial colony) after 7 days of incubation at 30 °C.
We also developed a spot-on-lawn assay, which is more sensitive compared to the plate confrontation assay and only requires one-day incubation instead of seven days. Briefly, the fungal hyphae Foc4 were streaked and inoculated with 5 ml of PDA broth. After 2-day incubation at 30 °C with 180 rpm shaking, 50 μl of fungal culture was re-inoculated into 5 ml of fresh PDA broth, incubated for additional 12 h, filtered using a cheesecloth to remove hyphae, and then the concentration of spores was determined using a hemocytometer under a light microscope. Fifty µl of a spore suspension (~106 spores ml−1) was mixed with 4 ml of 0.7% soft PDA agar and directly poured onto a PDA plate (1.5% agar). Plates were dried for 50 min and a well (5 mm in diameter) was made in the center of each plate, and 50 µl of Bacillus spp. cells (OD600 ~0.4; F7, Y6, or mutants derived from Y6) grown in LB medium was added into each well. The antifungal activity was evaluated by measuring the diameter of the inhibition zone (mm) after 24 h of incubation at 30 °C. The experiments were performed at least three times.
Lipopeptide (LP) extraction from the isolated strains
LPs were extracted according to previous protocol61. Briefly, 150 μl of Y6 or F7 cell culture (OD600 ~0.4) was added into 15 ml of fresh LB medium. After 2 days of incubation at 30 °C with 180 rpm shaking, 10 ml of cell culture was spun down by centrifugation for 5 min at 14000 × g, and the cell-free supernatant was collected and subjected to filtration using 0.22-μm filters, and then loaded into a C18 syringe cartridge column (Bond Elut C18, Agilent, USA). The column was washed with 10 ml of ddH2O and the LP compounds were eluted with 2 ml of methanol.
To understand the influence of pathogen on LP production, we extract LPs from the inhibition zone (Fig. 1A)32. 200 μl of R. solanacearum cell culture (OD600 ~0.4) grown in CPG medium was mixed with 4 ml of 0.7% CPG soft agar and directly poured onto a CPG plate (1.5% agar). Plates were dried for 50 min and a well (5 mm in diameter) was made in the center of each plate, and 50 μl of Bacillus isolates (Y6 or F7) (OD600 ~0.4) grown in LB medium was added into the well. After the inhibition zone became evident with 24 h incubation at 30 °C, a 300 mg of agar sample was harvested from the inhibition zone, mixed with 1 ml of acetonitrile/water (1:1 v/v), and sonicated for 30 s followed by centrifugation and filtration. The supernatant was collected from acetonitrile/water extract32. As a control, LPs were isolated in a similar way but without R. solanacearum in the top soft agar layer. Agar sample (300 mg) was collected around the well (2–4 mm).
Identification and quantification of LPs by UPLC–MS
Both methanol and acetonitrile/water extracts were analyzed by reverse phase Ultra-Performance Liquid Chromatography coupled with a triple quadrupole MS (UPLC-MS) (Waters, Acquity, XEVO-TQD). The lipopeptide compounds were identified based on the mass-to-charge ratio (m/z) and quantitated based on the standard curves generated using commercial LP standards (Sigma-Aldrich, USA). The column temperature was maintained at 40 °C and a gradient elution with (A) acetonitrile (containing 0.1% formic acid) and (B) water (containing 0.1% formic acid) was used. The gradient program was used as follows: 0–0.5 min, 40% A; 0.5–3.5 min, 40–80% A; 3.5–4.0 min, 80% A; 4.0–6.0 min, 80–95% A; 6.0–7.0 min, 95–98% A. The flow rate was set at 0.4 ml min−1. The Electrospray Ionization (ESI) source was set in positive ionization mode with 3.26 kV of capillary voltage, and the source temperature was maintained at 150 °C. Nitrogen flow was 600 L h−1. Argon flow was 50 L h−1.
Construction of mutants deficient in LP synthesis
In initial studies, we determined that Y6 is sensitive to spectinomycin (spc; 100 μg ml−1), kanamycin (kan; 15 μg ml−1), chloramphenicol (cat; 10 μg ml−1), tetracycline (tet; 5 μg ml−1), and macrolide lincosoamide-streptogramin B (mls; contains 1 μg ml−1 erythromycin and 25 μg ml−1 lincomycin) antibiotics, whereas F7 was sensitive to kan, cat and mls, but resistant to spc. Therefore, strain Y6 was selected for mutant construction. The srfAA:mls, ituA:mls, and fenC:spc single mutants and the double mutants ituA:mls fenC:spc and ituA:mls fenB:spc were generated by replacing the coding region with a resistance cassette using long flanking homology PCR (LFH-PCR) followed by DNA transformation as previously described62 (Table S1). Specific primers used for PCR amplification were designed based on the gene sequences of B. velezensis FZB42 (srfAA and ituA) and DSM7 (fenC) (Table S2).
Swarming and swimming motility assays
Swimming and swarming motility of Y6 and its derived mutants were tested using standard protocols with minor modification63,64. LBGM plates containing 0.7% (swarming) or 0.3% agar (swimming) were dried in a laminar flow hood for 30 min and then 5 μl of LB precultures (OD600 ~0.4) was spotted on the center of each plate. The plates were then dried for another 15 min and incubated overnight at 37 °C.
Biofilm formation assay
To evaluate the contribution of the LP compounds to biofilm formation, we monitored colony morphology on LBGM and MSgg plates and pellicle formation on LBGM and MSgg liquid media. For colony morphology analysis, 3 μl of LB preculture (OD600 ~0.4) was spotted onto LBGM or MSgg agar plates, which were dried for 30 min in a laminar airflow prior to spotting, and incubated at 30 °C for up to 36 h. To monitor pellicle formation65, 10 μl of LB precultures (OD600 ~0.4) was inoculated into 2.5 ml of LBGM or MSgg medium in a 24 well plate and incubated at 30 °C for up to 36 h65,66.
Additional methods
Additional methods are described in the Supplementary Information, including plant pot experiments, identification of the isolates Y6 and F7 by phylogenetic analysis, minimal inhibition concentrations (MIC) determination of LPs and the extracts from the isolates, RNA extraction and quantitative PCR (qPCR), growth curves, and spore germination assay.
Data availability
All data generated during this study are included in this article (and its Supplementary Information file).
References
Chakraborty, S. & Newton, A. C. Climate change, plant diseases and food security: an overview. Plant Pathology 60, 2–14, https://doi.org/10.1111/j.1365-3059.2010.02411.x (2011).
Carvalho, F. P. Pesticides, environment, and food safety. Food and Energy Security 6, 48–60, https://doi.org/10.1002/fes3.108 (2017).
Nelson, R., Wiesner-Hanks, T., Wisser, R. & Balint-Kurti, P. Navigating complexity to breed disease-resistant crops. Nature reviews. Genetics 19, 21–33, https://doi.org/10.1038/nrg.2017.82 (2018).
Boyd, L. A., Ridout, C., O’Sullivan, D. M., Leach, J. E. & Leung, H. Plant-pathogen interactions: disease resistance in modern agriculture. Trends in genetics: TIG 29, 233–240, https://doi.org/10.1016/j.tig.2012.10.011 (2013).
McHughen, A. & Wager, R. Popular misconceptions: agricultural biotechnology. New biotechnology 27, 724–728, https://doi.org/10.1016/j.nbt.2010.03.006 (2010).
Berg, G. Plant-microbe interactions promoting plant growth and health: perspectives for controlled use of microorganisms in agriculture. Applied microbiology and biotechnology 84, 11–18, https://doi.org/10.1007/s00253-009-2092-7 (2009).
Perez-Garcia, A., Romero, D. & de Vicente, A. Plant protection and growth stimulation by microorganisms: biotechnological applications of Bacilli in agriculture. Current opinion in biotechnology 22, 187–193, https://doi.org/10.1016/j.copbio.2010.12.003 (2011).
Ongena, M. & Jacques, P. Bacillus lipopeptides: versatile weapons for plant disease biocontrol. Trends Microbiol 16, 115–125, https://doi.org/10.1016/j.tim.2007.12.009 (2008).
Koumoutsi, A. et al. Structural and functional characterization of gene clusters directing nonribosomal synthesis of bioactive cyclic lipopeptides in Bacillus amyloliquefaciens strain FZB42. Journal of bacteriology 186, 1084–1096 (2004).
Molinatto, G. et al. Complete genome sequence of Bacillus amyloliquefaciens subsp. plantarum S499, a rhizobacterium that triggers plant defences and inhibits fungal phytopathogens. J Biotechnol 238, 56–59, https://doi.org/10.1016/j.jbiotec.2016.09.013 (2016).
Fan, B., Blom, J., Klenk, H. P. & Borriss, R. Bacillus amyloliquefaciens, Bacillus velezensis, and Bacillus siamensis Form an “Operational Group B. amyloliquefaciens” within the B. subtilis Species Complex. Frontiers in microbiology 8, 22, https://doi.org/10.3389/fmicb.2017.00022 (2017).
Xu, Z. et al. Contribution of bacillomycin D in Bacillus amyloliquefaciens SQR9 to antifungal activity and biofilm formation. Appl Environ Microbiol 79, 808–815, https://doi.org/10.1128/AEM.02645-12 (2013).
Mansfield, J. et al. Top 10 plant pathogenic bacteria in molecular plant pathology. Mol Plant Pathol 13, 614–629, https://doi.org/10.1111/j.1364-3703.2012.00804.x (2012).
Poueymiro, M. et al. A Ralstonia solanacearum type III effector directs the production of the plant signal metabolite trehalose-6-phosphate. mBio 5, https://doi.org/10.1128/mBio.02065-14 (2014).
Coutinho, T. A. & Wingfield, M. J. Ralstonia solanacearum and R. pseudosolanacearum on Eucalyptus: Opportunists or PrimaryPathogens? Frontiers in plant science 8, 761, https://doi.org/10.3389/fpls.2017.00761 (2017).
Yuliar, Nion, Y. A. & Toyota, K. Recent Trends in Control Methods for Bacterial Wilt Diseases Caused by Ralstonia solanacearum. Microbes and Environments 30, 1–11, https://doi.org/10.1264/jsme2.ME14144 (2015).
Kurabachew, H. & Wydra, K. Characterization of plant growth promoting rhizobacteria and their potential as bioprotectant against tomato bacterial wilt caused by Ralstonia solanacearum. Biological Control 67, 75–83, https://doi.org/10.1016/j.biocontrol.2013.07.004 (2013).
Ji, P. et al. New tactics for bacterial wilt management on tomatoes in the Southern US. Proceedings of the 1st International Symposium on Tomato Diseases, 153–159 (2005).
Pradhanang, P. M. et al. Application of acibenzolar-S-methyl enhances host resistance in tomato against Ralstonia solanacearum. Plant Disease 89, 989–993, https://doi.org/10.1094/Pd-89-0989 (2005).
Hanson, P. M., Licardo, O., Hanudin, Wang, J. F. & Chen, J. T. Diallel analysis of bacterial wilt resistance in tomato derived from different sources. Plant Disease 82, 74–78, https://doi.org/10.1094/Pdis.1998.82.1.74 (1998).
Alvarez, F. et al. The plant-associated Bacillus amyloliquefaciens strains MEP2 18 and ARP2 3 capable of producing the cyclic lipopeptides iturin or surfactin and fengycin are effective in biocontrol of sclerotinia stem rot disease. J Appl Microbiol 112, 159–174, https://doi.org/10.1111/j.1365-2672.2011.05182.x (2012).
Bais, H. P., Fall, R. & Vivanco, J. M. Biocontrol of Bacillus subtilis against infection of Arabidopsis roots by Pseudomonas syringae is facilitated by biofilm formation and surfactin production. Plant Physiol 134, 307–319, https://doi.org/10.1104/pp.103.028712 (2004).
Chowdhury, S. P., Hartmann, A., Gao, X. & Borriss, R. Biocontrol mechanism by root-associated Bacillus amyloliquefaciens FZB42 - a review. Frontiers in microbiology 6, 780, https://doi.org/10.3389/fmicb.2015.00780 (2015).
Ploetz, R. C. Fusarium Wilt of Banana. Phytopathology 105, 1512–1521, https://doi.org/10.1094/PHYTO-04-15-0101-RVW (2015).
Mostert, D. et al. The distribution and host range of the banana Fusarium wilt fungus, Fusarium oxysporum f. sp. cubense, in Asia. PLoS One 12, e0181630, https://doi.org/10.1371/journal.pone.0181630 (2017).
Lian, J. et al. Artificial inoculation of banana tissue culture plantlets with indigenous endophytes originally derived from native banana plants. Biological Control 51, 427–434, https://doi.org/10.1016/j.biocontrol.2009.08.002 (2009).
Huang, Y. H. et al. Control of Fusarium wilt in banana with Chinese leek. Eur J Plant Pathol 134, 87–95, https://doi.org/10.1007/s10658-012-0024-3 (2012).
Duffy, B. K., Ownley, B. H. & Weller, D. M. Soil Chemical and Physical Properties Associated with Suppression of Take-all of Wheat by Trichoderma koningii. Phytopathology 87, 1118–1124, https://doi.org/10.1094/phyto.1997.87.11.1118 (1997).
Aeron, A., Pandey, P. & Maheshwari, D. K. Differential response of sesame under influence of indigenous and non-indigenous rhizosphere competent fluorescent pseudomonads. Current Science 99, 166–168 (2010).
Gu, Q. et al. Bacillomycin D produced by Bacillus amyloliquefaciens is involved in the antagonistic interaction with the plant pathogenic fungus Fusarium graminearum. Appl Environ Microbiol, https://doi.org/10.1128/AEM.01075-17 (2017).
Zeriouh, H. et al. The iturin-like lipopeptides are essential components in the biological control arsenal of Bacillus subtilis against bacterial diseases of cucurbits. Mol Plant Microbe Interact 24, 1540–1552, https://doi.org/10.1094/MPMI-06-11-0162 (2011).
Cawoy, H. et al. Lipopeptides as main ingredients for inhibition of fungal phytopathogens by Bacillus subtilis/amyloliquefaciens. Microbial biotechnology 8, 281–295, https://doi.org/10.1111/1751-7915.12238 (2015).
Liu, X. et al. Optimization for the production of surfactin with a new synergistic antifungal activity. PloS one 7, e34430, https://doi.org/10.1371/journal.pone.0034430 (2012).
Nihorimbere, V. et al. Impact of rhizosphere factors on cyclic lipopeptide signature from the plant beneficial strain Bacillus amyloliquefaciens S499. FEMS Microbiol Ecol 79, 176–191, https://doi.org/10.1111/j.1574-6941.2011.01208.x (2012).
Das, P., Mukherjee, S. & Sen, R. Genetic regulations of the biosynthesis of microbial surfactants: an overview. Biotechnol Genet Eng Rev 25, 165–185 (2008).
de Bruijn, I. & Raaijmakers, J. M. Regulation of cyclic lipopeptide biosynthesis in Pseudomonas fluorescens by the ClpP protease. Journal of bacteriology 191, 1910–1923, https://doi.org/10.1128/JB.01558-08 (2009).
Cairns, L. S., Hobley, L. & Stanley-Wall, N. R. Biofilm formation by Bacillus subtilis: new insights into regulatory strategies and assembly mechanisms. Molecular microbiology 93, 587–598, https://doi.org/10.1111/mmi.12697 (2014).
Mielich-Suss, B. & Lopez, D. Molecular mechanisms involved in Bacillus subtilis biofilm formation. Environmental microbiology 17, 555–565, https://doi.org/10.1111/1462-2920.12527 (2015).
Chen, Y. et al. Biocontrol of tomato wilt disease by Bacillus subtilis isolates from natural environments depends on conserved genes mediating biofilm formation. Environmental microbiology 15, 848–864, https://doi.org/10.1111/j.1462-2920.2012.02860.x (2013).
Zeriouh, H., de Vicente, A., Perez-Garcia, A. & Romero, D. Surfactin triggers biofilm formation of Bacillus subtilis in melon phylloplane and contributes to the biocontrol activity. Environmental microbiology 16, 2196–2211, https://doi.org/10.1111/1462-2920.12271 (2014).
Aleti, G. et al. Surfactin variants mediate species-specific biofilm formation and root colonization in Bacillus. Environmental microbiology 18, 2634–2645, https://doi.org/10.1111/1462-2920.13405 (2016).
Allard-Massicotte, R. et al. Bacillus subtilis Early Colonization of Arabidopsis thaliana Roots Involves Multiple Chemotaxis Receptors. mBio 7, https://doi.org/10.1128/mBio.01664-16 (2016).
Raaijmakers, J. M., de Bruijn, I., Nybroe, O. & Ongena, M. Natural functions of lipopeptides from Bacillus and Pseudomonas: more than surfactants and antibiotics. FEMS microbiology reviews 34, 1037–1062, https://doi.org/10.1111/j.1574-6976.2010.00221.x (2010).
Kearns, D. B. & Losick, R. Swarming motility in undomesticated Bacillus subtilis. Molecular microbiology 49, 581–590 (2003).
Almoneafy, A. A. et al. Tomato plant growth promotion and antibacterial related-mechanisms of four rhizobacterial Bacillus strains against Ralstonia solanacearum. Symbiosis 63, 59–70, https://doi.org/10.1007/s13199-014-0288-9 (2014).
Kakar, K. U. et al. A novel rhizobacterium Bk7 for biological control of brown sheath rot of rice caused by Pseudomonas fuscovaginae and its mode of action. European Journal of Plant Pathology 138, 819–834, https://doi.org/10.1007/s10658-013-0356-7 (2014).
Li, B. et al. Responses of beneficial Bacillus amyloliquefaciens SQR9 to different soilborne fungal pathogens through the alteration of antifungal compounds production. Frontiers in microbiology 5, 636, https://doi.org/10.3389/fmicb.2014.00636 (2014).
Luo, C., Liu, X., Zhou, H., Wang, X. & Chen, Z. Nonribosomal peptide synthase gene clusters for lipopeptide biosynthesis in Bacillus subtilis 916 and their phenotypic functions. Applied and environmental microbiology 81, 422–431, https://doi.org/10.1128/AEM.02921-14 (2015).
Koumoutsi, A., Chen, X. H., Vater, J. & Borriss, R. DegU and YczE positively regulate the synthesis of bacillomycin D by Bacillus amyloliquefaciens strain FZB42. Applied and environmental microbiology 73, 6953–6964, https://doi.org/10.1128/AEM.00565-07 (2007).
Romero, D. et al. The iturin and fengycin families of lipopeptides are key factors in antagonism of Bacillus subtilis toward Podosphaera fusca. Mol Plant Microbe Interact 20, 430–440, https://doi.org/10.1094/MPMI-20-4-0430 (2007).
Gonzalez-Jaramillo, L. M., Aranda, F. J., Teruel, J. A., Villegas-Escobar, V. & Ortiz, A. Antimycotic activity of fengycin C biosurfactant and its interaction with phosphatidylcholine model membranes. Colloids Surf B Biointerfaces 156, 114–122, https://doi.org/10.1016/j.colsurfb.2017.05.021 (2017).
Ongena, M. et al. Surfactin and fengycin lipopeptides of Bacillus subtilis as elicitors of induced systemic resistance in plants. Environmental microbiology 9, 1084–1090, https://doi.org/10.1111/j.1462-2920.2006.01202.x (2007).
Falardeau, J., Wise, C., Novitsky, L. & Avis, T. J. Ecological and mechanistic insights into the direct and indirect antimicrobial properties of Bacillus subtilis lipopeptides on plant pathogens. Journal of chemical ecology 39, 869–878, https://doi.org/10.1007/s10886-013-0319-7 (2013).
Gao, W. et al. Mutations in genes encoding antibiotic substances increase the synthesis of poly-gamma-glutamic acid in Bacillus amyloliquefaciens LL3. Microbiologyopen 6, https://doi.org/10.1002/mbo3.398 (2017).
Xiong, H. Q., Li, Y. T., Cai, Y. F., Cao, Y. & Wang, Y. Isolation of Bacillus amyloliquefaciens JK6 and identification of its lipopeptides surfactin for suppressing tomato bacterial wilt. Rsc Advances 5, 82042–82049, https://doi.org/10.1039/c5ra13142a (2015).
Bertani, G. Lysogeny at mid-twentieth century: P1, P2, and other experimental systems. Journal of bacteriology 186, 595–600 (2004).
Decroix, G., Piquet, J. J., Pilliaert, J. M. & Desaulty, A. [Bronchial foreign bodies. Statistics of the clinique O. R. L. de Lille]. Lille medical: journal de la Faculte de medecine et de pharmacie de l’Universite de Lille 15, 1033–1038 (1970).
Salanoubat, M. et al. Genome sequence of the plant pathogen Ralstonia solanacearum. Nature 415, 497–502, https://doi.org/10.1038/415497a (2002).
Ding, Z. et al. Mitogen-activated protein kinases are associated with the regulation of physiological traits and virulence in Fusarium oxysporum f. sp. cubense. PLoS One 10, e0122634, https://doi.org/10.1371/journal.pone.0122634 (2015).
Meng, X., Li, J., Bi, F., Zhu, L. & Ma, Z. Antifungal Activities of Crude Extractum from Camellia semiserrata Chi (Nanshancha) Seed Cake Against Colletotrichum musae, Colletotrichum gloeosporioides and Penicillium italicum in vitro and in vivo FruitTest. The plant pathology journal 31, 414–420, https://doi.org/10.5423/ppj.oa.06.2015.0098 (2015).
Razafindralambo, H. et al. Purification of antifungal lipopeptides by reversed-phase high-performance liquid chromatography. Journal of chromatography 639, 81–85 (1993).
Mascher, T., Margulis, N. G., Wang, T., Ye, R. W. & Helmann, J. D. Cell wall stress responses in Bacillus subtilis: the regulatory network of the bacitracin stimulon. Molecular microbiology 50, 1591–1604 (2003).
Kearns, D. B. A field guide to bacterial swarming motility. Nature Reviews Microbiology 8, 634–644, https://doi.org/10.1038/nrmicro2405 (2010).
Morales-Soto, N. et al. Preparation, imaging, and quantification of bacterial surface motility assays. J Vis Exp, https://doi.org/10.3791/52338 (2015).
Branda, S. S., Gonzalez-Pastor, J. E., Ben-Yehuda, S., Losick, R. & Kolter, R. Fruiting body formation by Bacillus subtilis. Proc Natl Acad Sci USA 98, 11621–11626, https://doi.org/10.1073/pnas.191384198 (2001).
Shemesh, M. & Chai, Y. A combination of glycerol and manganese promotes biofilm formation in Bacillus subtilis via histidine kinase KinD signaling. Journal of bacteriology 195, 2747–2754, https://doi.org/10.1128/JB.00028-13 (2013).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China to YC (41471214, U1401234, 2015B090903077, and 2016B020242005) and a grant from the National Institutes of Health to JDH (R35GM122461).
Author information
Authors and Affiliations
Contributions
H.P., J.D.H. and Y.C. designed the study. C.Y., Y.L., Y.W., H.Z., H.X., and Y.C. conducted experiments. H.P. and Y.C. prepared the figures and tables. H.P., P.C., J.D.H. and Y.C. analyzed the data and wrote the paper. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cao, Y., Pi, H., Chandrangsu, P. et al. Antagonism of Two Plant-Growth Promoting Bacillus velezensis Isolates Against Ralstonia solanacearum and Fusarium oxysporum. Sci Rep 8, 4360 (2018). https://doi.org/10.1038/s41598-018-22782-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-22782-z
- Springer Nature Limited
This article is cited by
-
The beneficial endophytic microbes enhanced tobacco defense system to resist bacterial wilt disease
Chemical and Biological Technologies in Agriculture (2024)
-
Biological control of the shot-hole disease in flowering cherry tree using antimicrobial compounds produced by Bacillus velezensis 8–2
Chemical and Biological Technologies in Agriculture (2024)
-
Inhibitory activity of bacterial lipopeptides against Fusarium oxysporum f.sp. Strigae
BMC Microbiology (2024)
-
A Bacillus velezensis strain isolated from oats with disease-preventing and growth-promoting properties
Scientific Reports (2024)
-
Diversity, Characterization, and Potential Applications of Bacterial Endophytes Isolated from the Halophyte Limonium axillare
Journal of Plant Growth Regulation (2024)