Abstract
It has been proposed that intrauterine administration of peripheral blood mononuclear cells (PBMCs) modulates maternal immune response through a cascade of cytokines, chemokines and growth factors to favor implantation. We conducted a meta-analysis to verify the effect of intrauterine PBMC administration on the outcome of embryo transfer in women with recurrent implantation failure (RIF). All relevant trials published in PubMed, Web of Science and Cochrane library databases were searched. Two randomized controlled trials and three cohort studies (1173 patients in total) matched the inclusion criteria. No differences in live birth rates were seen between the PBMC-treated patients and controls (OR: 1.65, 95% CI: 0.84–3.25; p = 0.14; I2: 66.3%). The clinical pregnancy rate was significantly higher in women who received intrauterine PBMCs before embryo transfer compared with those who did not (OR: 1.65, 95% CI: 1.30–2.10; p = 0.001, heterogeneity; I2: 60.6%). Subgroup analyses revealed a significant increase in clinical pregnancy rates with the administration of PBMCs in women with ≥3 previous failures compared with controls (OR: 2.69, 95% CI: 1.53–4.72; p = 0.001, I2: 38.3%). In summary, the data did not demonstrate an association between the administration of PBMCs into the uterine cavity before fresh or frozen-thawed embryo transfer and live birth rates in women with RIF. Whether intrauterine PBMC administration significantly changes live birth and miscarriage rates requires further investigation.
Similar content being viewed by others
Introduction
Procedures or medications with limited or unproven effectiveness are typically offered to couples with repeated implantation failure who desperately seek novel therapies. Implantation failure has been related to a myriad of problems ranging from anatomical uterine defects to more complex etiologies, such as suboptimal embryo quality or decreased endometrial receptivity; however, the etiology remains unexplained in the majority of cases1.
Despite the lack of a uniform definition, recurrent implantation failure (RIF) is generally defined as the absence of a clinical pregnancy after two consecutive fresh or frozen embryo transfer cycles involving the transfer of at least four cleavage stage embryos or two good quality blastocysts. The current treatment of RIF is largely empirical and based on poor scientific evidence. There is a sizable amount of literature relating to the management options in RIF cases targeting either the embryo (assisted hatching, aneuploidy screening), the endometrium (endometrial co-culture, scratching, receptivity assays, short-term copper intrauterine device insertion, freezing-all strategy), the maternal immune system (steroids, aspirin, heparin, immunoglobulins, anti-TNF antibodies, granulocyte colony-stimulating factor, intralipid infusions), or sperm (higher magnification sperm selection, microchips), but none of these techniques been proven beyond reasonable doubt to have a therapeutic effect2,3.
During the last decade, particular interest has focused on the role of endocrine-immune interactions in the implantation process. The interplay between the invading embryo and mononuclear cells that come into contact with trophoblasts of embryonic origin through a cascade of cytokines, chemokines and growth factors actively occurs at the implantation site. Based on this rationale, it has been proposed that modulating the maternal immune response to the invading embryo might improve the clinical outcome in women who fail to achieve pregnancy despite the transfer of good quality embryos. In 2006, researchers reported that women with RIF who received intrauterine peripheral blood mononuclear cells (PBMCs) cultured with human chorionic gonadotropin (hCG) experienced increases in implantation rates4. PBMCs are defined as any blood cell with a round nucleus, such as B and T lymphocytes, monocytes or macrophages5. Since then, a few controlled studies, including two randomized controlled trials with substantial differences in methodologies and small sample sizes, reported promising findings with the use of PBMCs in women with RIF6,7,8,9.
The primary objective of this systematic review and meta-analysis was to assess the available evidence on the intrauterine administration of PBMCs in women with implantation failures, provide an estimate of the average treatment effect, and recommend how to improve the design of future trials.
Results
Search results, study characteristics, and quality assessment
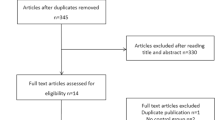
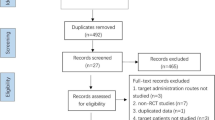
The Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) flow diagram of the review process is presented in Fig. 1. Supplementary Table S1 presents the inclusion criteria based on the PICOS aspects (participants, interventions, comparators, outcomes, and study design) as recommended by the Cochrane Handbook for Systematic Reviews. The search strategy yielded 54 citations, of which 30 were excluded as it was clear from scrutinizing the title and abstract that they did not fulfill the selection criteria. Full manuscripts of 24 articles were obtained. A total of 19 of these publications were excluded because eight were reviews5,10,11,12,13,14,15,16, four articles reported findings on animal studies17,18,19,20, three articles were conference abstracts with limited data21,22,23, three articles did not specify outcomes from assisted reproductive techniques24,25,26, and one was a case-series report27. After exclusion of the inappropriate studies, five were included in the meta-analysis4,6,7,8,9.
The eligible studies were published between 2006 and 2017, and the number of patients included in the study ranged from 35 to 663 patients. Two studies were randomized controlled trials (RCTs)7,8, and three were nonrandomized cohort studies4,6,9. The randomization method was reported in one7 of two RCTs. Inclusion criteria based on the number of previous failed IVF cycles and the primary outcome varied among studies. None of the studies performed a power analysis. Financial support was declared in one4 out of five studies. The risk of bias assessments for RCTs and observational studies are summarized in Supplementary Tables S2 and S3.
The data extracted from each included study are listed in Tables 1 and 2.
Live birth rate
The meta-analysis regarding the LBR is derived from four studies: one RCT8 and three nonrandomized cohort studies6,9 (Fig. 2). No difference in LBRs was noted between PBMC-treated and control groups (OR: 1.65, 95% CI: 0.84–3.25; p = 0.14; random effects model; heterogeneity; I2: 73.1%).
Live birth rates in frozen-thawed embryo transfer cycles
When the analysis was restricted to frozen-thawed embryo transfer cycles, LBRs were similar between PBMC-treated and control groups (OR: 1.43, 95% CI: 0.76–2.71; P = 0.26; random effects model; heterogeneity; I2: 75.4%) (Fig. 3).
Clinical pregnancy rate
All five studies were included in the meta-analysis for clinical pregnancy rate (CPR) outcome (Fig. 4). Pooling the data together, a significant improvement in the clinical pregnancy rate in women who had been given intrauterine PBMCs before embryo transfer was noted compared with those who had not (OR: 1.65, 95% CI: 1.30–2.10; p = 0.001, heterogeneity; I2: 60.6%).
Clinical pregnancy rate in women with ≥3 failures
Regarding the clinical pregnancy rate in the subgroup of patients with ≥3 failures, meta-analysis of the four studies, including two RCTs7,8 and two observational studies6,9, demonstrated a significant benefit of intrauterine PBMC administration (OR: 2.69, 95% CI: 1.53–4.72; P = 0.001; random effects model; heterogeneity; I2: 38.3%) (Fig. 5).
Miscarriage rate
Pooled estimates for the miscarriage risk revealed no significant difference between PBMC-treated and control groups (OR: 0.21, 95% CI: 0.02–2.43; P = 0.21; random effects model) (Fig. 6). However, the sample size was very low, and significant heterogeneity (I2 = 86.8%) was observed among studies.
None of the studies reported any adverse events.
Discussion
The present systematic review and meta-analysis summarizes the best available evidence regarding the intrauterine administration of PBMCs in cases with RIF. The results from the included studies demonstrated that intrauterine administration of PBMCs with or without hCG culture fails to improve live birth rates when compared to controls. However, PBMCs were associated with an improvement in clinical pregnancy rates, which was more prominent in women with ≥3 failures.
Our analysis was strengthened by a number of factors. A comprehensive search strategy was used, employing relevant research databases. Additionally, a valid data synthesis method was implemented, and no language restrictions were applied. Cochrane’s and ROBINS-I tools were used to assess the quality of the included studies, suggesting a low to moderate risk of bias28,29.
There are also weaknesses in our analysis, which mainly arise from the high level of clinical heterogeneity of the publications that were included. There were many sources of heterogeneity, including the patient population (number of previous failed IVF cycles), study design (randomized or nonrandomized), intervention (in vitro culture with or without hCG, dose, volume and timing of PBMC administration, day and number of embryo transfers, fresh or frozen-thawed embryo transfer) and primary and secondary outcomes. The small sample size and substantial differences in the study design or patient populations may also undermine the credibility of the results. Another possible limitation of the review is that although multiple scientific databases were used in the electronic search, some relevant reports might still have been missed.
Several lines of evidence from both animal models and in vitro studies suggest improvements in implantation rates following intrauterine PBMC administration. Pregnancy rates and embryonic implantation sites were significantly increased after intrauterine PBMC treatment in bovine17,18 and mouse models19. PBMCs promote murine blastocyst spreading and invasion as well as human choriocarcinoma-derived BeWo cell invasion in vitro, and these promoting effects were augmented by hCG supplementation20. Various mechanisms have been proposed to explain the implantation-promoting effects of PBMCs. PBMC treatment induces the production of several cytokines, such as IL-1α, IL-1ß, and TNF- α, which can have positive impacts on endometrial receptivity and actively contribute to blastocyst attachment and invasion5,18,19. Further, hCG activates PBMC sin vitro and promotes trophoblast invasion through enhancement of LIF and IL-1 ß secretion20,24. PBMCs cultured with hCG might modulate the implantation site and infiltrate into endometrial stroma, serving as a guide for the invading blastocyst. In addition, regulatory Th-2 cells, which are PBMC-derived immune cells, may facilitate a more permissive immune-inflammatory profile for implantation25,26. Although the biological plausibility of modulating the immune response to implantation is intriguing, the magnitude of the clinical effect is yet to be determined.
The positive impact of intrauterine PBMC administration on pregnancy rates might be attributed to two other factors. The first factor is the mechanical trauma induced on the endometrium by intrauterine catheterization before embryo transfer. The proposed mechanism here is the modulation of the local immune system by a proinflammatory response to catheter-induced injury through secretion of cytokines, growth factors, interleukins and immune cells, such as macrophages and dendritic cells, which would favor implantation30. Madkour et al. suggested that this argument could be refuted based on the lack of a positive effect on implantation rates in women with one or two IVF failures, whereas a significant enhancement was observed with the same technique in women with ≥3 IVF failures7. A Cochrane analysis, which included nine RCTs on 1496 women, showed that endometrial injury performed between day 7 of the previous cycle and day 7 of the ET cycle was associated with an increase in live birth or ongoing pregnancy rates (RR 1.42, 95% CI 1.08 to 1.85) compared with no intervention or a sham procedure31. Another Cochrane review that meta-analyzed nine trials testing the efficacy of the method in subfertile women and couples attempting to conceive through sexual intercourse or intrauterine insemination reported that it is uncertain whether it improves the chance of a live birth or ongoing pregnancy32. The most recent data from a large, multicenter, well-designed, randomized controlled trial failed to show an improvement in the clinical outcome by endometrial scratching, reporting similar LBRs for both the intervention and control groups (26.1% (180/690) vs. 26.1% (176/674), respectively; odds ratio = 1.00 [0.78 to 1.27])33.
The second factor is the effect of hCG administered into the uterine cavity prior to embryo transfer. hCG, the key molecule to initiate the communication between the embryo and the endometrium, contributes to maternal tolerance of the embryo through interactions with immune cells within the receptive endometrium and plausibly in systemic circulation. A Cochrane review including 12 RCTs of 4038 women analyzed the efficacy of the intrauterine administration of hCG in ART. The study, which also suffered from a considerable degree of heterogeneity (I2 > 75%), reported an increase in clinical pregnancy (RR 1.41, 95% CI 1.25 to 1.58) and live birth rates (RR: 1.57, 95% CI 1.32 to 1.87) in the subgroup of women having cleavage-stage embryo transfers with intrauterine hCG administration compared with women with no hCG treatment14. However, they concluded that the current evidence for intrauterine hCG treatment does not support its use in ART cycles due to the small size and the variable quality of the trials and the fact that positive findings were extracted from a subgroup analysis. The other systematic review and meta-analysis on the subject analyzed eight RCTs that included 3087 women and found no difference in the live birth (RR 1.13; 95% CI 0.84 to 1.53) and spontaneous abortion rates (RR 1.00, 95% CI 0.74 to 1.34) in women who received intrauterine hCG and those who did not15.
Could these data be considered as new evidence of a game changer in patients with RIF? Based on the results of the present meta-analysis, there is limited evidence suggesting that intrauterine administration of PBMCs with or without hCG culture improves LBRs in women with RIF. However, the reliability of the data is limited by the observational nature of the available studies.
As infertile couples and their physicians have long been awaiting opportunities to celebrate medical breakthroughs, it is crucial for infertility specialists to refrain from offering treatment options that are not evidence based. The scientific background and the available evidence should be shared with the couple prior to implementing such a procedure. The use of PBMCs should be considered within the context of clinical trials with proper randomization and patient consent. Furthermore, there is a lack of long-term safety data of PBMC administration that is needed.
Methods
Databases and search strategies
The searches and selection of the studies were performed independently by two of the reviewers (K.Y. and O.O.), and any disagreement was resolved by discussion. PubMed, Web of Science and Cochrane library databases (from inception to April 2017) were searched for all relevant articles under the following key words and/or medical subject heading (MeSH) terms: ‘assisted reproduction’ or ‘IVF’ or ‘intracytoplasmic sperm injection’ or ‘embryo transfer’, or ‘implantation failure’ or recurrent implantation failure’ or ‘intrauterine administration’ and ‘peripheral blood mononuclear cells’ or ‘peripheral mononuclear cells’. References of all included primary and review articles were examined to identify relevant articles not captured by the electronic search. In addition, reference lists of all relevant publications and review articles as well as meeting proceedings of the European Society of Human Reproduction and Embryology and the American Society for Reproductive Medicine were hand-searched for the identification of relevant studies. No restriction was applied in terms of language, geographic distribution or publication type.
This study was performed using a predetermined protocol in accordance with the PRISMA reporting guidelines (Supplementary Table S4).
Inclusion and exclusion
The study was designed a priori with inclusion of primary articles that studied women undergoing any form of ART (IVF, ICSI) who had intrauterine administration of PBMCs with or without hCG culture before fresh or frozen embryo transfer. Adjuvant medical therapy should not have been administered and asymmetric intervention should not have been performed in the control group. Eligible study designs were randomized controlled trials (RCTs), nonrandomized controlled trials, and prospective controlled cohort studies. The inclusion criteria based on the PICOS aspects are summarized in Supplementary Table S1.
Data extraction and quality assessment
Two review authors (K.Y. and O.O.) independently extracted the following data from the included studies: demographic data (citation data, country, study period, number of patients included), study design, methodology (method of randomization, allocation concealment), intervention (administration route, timing, amount, extraction, in vitro culture, culture conditions), ovarian stimulation protocol, type and starting dose of gonadotropin administered for ovarian stimulation, type and dose of medication used for triggering final oocyte maturation, criteria used for triggering, type of fertilization, endometrial preparation protocol, day of embryo transfer, embryo quality, embryo freezing and thawing techniques and protocols, type of luteal support, adverse events associated with the intervention and pregnancy outcome.
The main outcome measure chosen for the current meta-analysis was live births per patient. Secondary outcomes were the clinical pregnancy rate (evidence of an intrauterine pregnancy sac with fetal heart activity at 6–8 weeks of gestation) or miscarriage rate. In cases of missing information, the study authors were contacted to retrieve relevant data where available. Any disagreement between the two reviewers regarding data extraction was resolved by discussion.
To determine the validity of the included trials, we assessed the risk of bias by Cochrane’s tool for the ‘risk of bias’ assessment in the RCTs10 and the ROBINS-I tool in the observational studies11.
Statistical analysis
All outcomes were dichotomous, and the results were expressed for each trial as an odds ratio (OR) with a 95% confidence interval (CI). To examine the associations between intrauterine PBMC administration and outcomes of interest, the OR with 95% CI was estimated by summarizing the risk estimates of each study using the random effect model. Heterogeneity of exposure effects was evaluated graphically using forest plots34 and heterogeneity across studies using the I2 statistic. Five studies were included in the meta-analysis; therefore, a funnel plot asymmetry test was not performed35. To investigate the possible sources of heterogeneity, we performed stratified analyses for the following: fresh versus frozen embryo transfer and <3 or ≥3 previous treatment failures. Statistical analyses were performed using RevMan 5.1 software (Cochrane Collaboration, Oxford, UK).
References
Polanski, L. T. et al. What exactly do we mean by ‘recurrent implantation failure’? A systematic review and opinion. Reprod Biomed Online 28, 409–423 (2014).
Urman, B., Yakin, K. & Balaban, B. Recurrent implantation failure in assisted reproduction: how to counsel and manage. B. Treatment options that have not been proven to benefit the couple. Reprod Biomed Online 11, 382–391 (2005).
Coughlan, C. et al. Recurrent implantation failure: definition and management. Reprod Biomed Online 28, 14–38 (2014).
Yoshioka, S. et al. Intrauterine administration of autologous peripheral blood mononuclear cells promotes implantation rates in patients with repeated failure of IVF-embryo transfer. Hum Reprod 21, 3290–3294 (2006).
Fujiwara, H. Do circulating blood cells contribute to maternal tissue remodelling and embryo-maternal cross-talk around the implantation period? Mol Hum Reprod 15, 335–343 (2009).
Okitsu, O., Kiyokawa, M., Oda, T. & Miyake, K. Intrauterine administration of autologous blood mononuclear cells increases clinical pregnancy rates in frozen/thawed embryo transfer cycles of patients with repeated implantation failure. J Reprod Immunol 92, 82–87 (2011).
Madkour, A. et al. Intrauterine insemination of cultured peripheral blood mononuclear cells prior to embryo transfer improves clinical outcome for patients with repeated implantation failures. Zygote 24, 58–69 (2015).
Yu, N. et al. Intrauterine administration of autologous peripheral blood mononuclear cells (PBMCs) activated by HCG improves the implantation and pregnancy rates in patients with repeated implantation failure: a prospective randomized study. Am J Reprod Immunol 76, 212–216 (2016).
Li, S. et al. Intrauterine administration of hCG-activated autologous human peripheral blood mononuclear cells (PBMC) promotes live birth rates in frozen/thawed embryo transfer cycles of patients with repeated implantation failure. J Reprod Immunol 119, 15–22 (2017).
Hviid, M. M. & Macklon, N. Immune modulation treatments – where is the evidence? Fertil Steril 107, 1284–1293 (2017).
Makrigiannakis, A. et al. Repeated implantation failure: a new potential treatment option. Eur J Clin Invest 45, 380–384 (2015).
Makrigiannakis, A., Vrekoussis, T., Zoumakis, E., Kalantaridou, S. N. & Jeschke, U. The role of HCG in implantation: A minireview of molecular and clinical evidence. Int J Mol Sci 18, 1305–1309 (2017).
Mekinian, A. et al. Unexplained recurrent miscarriage and recurrent implantation failure: is there a place for immunomodulation? Am J Reprod Immunol 76, 8–28 (2016).
Craciunas, L., Tsampras, N., Coomarasamy, A. & Raine-Fenning, N. Intrauterine administration of human chorionic gonadotropin (hCG) for subfertile women undergoing assisted reproduction. Cochrane Database Syst Rev 20(5), CD011537 (2016).
Osman, A. et al. The effect of intrauterine HCG injection on IVF outcome: a systematic review and meta-analysis. Reprod Biomed Online 33, 350–359 (2016).
Zeyneloglu, H. B. & Onalan, G. Remedies for recurrent implantation failure. Semin Reprod Med 32, 297–305 (2014).
Ideta, A. et al. Intrauterine administration of peripheral blood mononuclear cells enhances early development of the pre-implantation bovine embryo transfer. Mol Reprod Dev 77, 954–962 (2010).
Ideta, A. et al. Administration of peripheral blood mononuclear cells into the uterine horn to improve pregnancy rate following bovine embryo transfer. Anim Reprod Sci 117, 18–23 (2010).
Yu, N. et al. Intrauterine administration of peripheral blood mononuclear cells (PBMCs) improves endometrial receptivity in mice with embryonic implantation dysfunction. Am J Reprod Immunol 71, 24–33 (2014).
Nakayama, T. et al. Human peripheral blood mononuclear cells (PBMC) in early pregnancy promote embryo invasion in vitro: HCG enhances the effects of PBMC. Hum Reprod 17, 207–212 (2002).
Zhylkova, I., Feskov, A., Feskova, I., Somova., O. & Chumakova, N. Influence of peripheral blood mononuclear cells intrauterine transfer on implantation rates in patients with unsuccessful IVF cycles. Hum Reprod 25(Suppl1), i210–i235 (2010).
Gultomruk, M. et al. Intrauterine administration of autologous mononuclear cells (AMC) before embryo transfer (ET) in blastocyst stage: Is it useful approach in cases with recurrent implantation failure (RIF)? Hum Reprod 29(Suppl 1), i230 (2014).
Sefrioui, O., Madkour, A. & Louanjli, N. Immunotherapy of patients with repeated implantation failures in randomized controlled design and its impact on luteal progesterone synthesis. Hum Reprod 29(Suppl1), i217 (2014).
Yu, N. et al. HCG-activated human peripheral blood mononuclear cells (PBMC) promote trophoblast cell invasion. PloS One 10, e0125589 (2015).
Ginsburg, E. S. et al. T-helper 2 and 3 type immunity to trophoblast in successful in vitro fertilization-embryo transfer. Fertil Steril 83, 1659–1664 (2005).
Hashii, K. et al. Peripheral blood mononuclear cells stimulate progesterone production by luteal cells derived from pregnant and non-pregnant women: possible involvement of interleukin-4 and interleukin-10 in corpus luteum function and differentiation. Hum Reprod 13, 2738–4274 (1998).
Chen, L. et al. Frozen embryo transfer combined with intrauterine administration of autologous peripheral blood mononuclear cells for repeated implantation failure: report of 3 cases. J South Med Univ 31, 724–729 (2011).
Higgins, J. P, Green, S. Cochrane Handbook for Systematic Reviews of Interventions (Version 5.1.0), http://www.cochranehandbook.org (2012).
Sterne, J. A. C. et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 12(355), i4919 (2016).
Gnainsky, Y. et al. Biopsy-induced inflammatory conditions improve endometrial receptivity: the mechanism of action. Reproduction 149, 75–85 (2015).
Nastri, C. O. et al. Endometrial injury in women undergoing assisted reproductive techniques. Cochrane Database Syst Rev 7, CD009517 (2012).
Lensen, S. et al. Endometrial injury for pregnancy following sexual intercourse or intrauterine insemination. Cochrane Database Syst Rev 6, CD011424 (2016).
Lensen, S. et al. Endometrial scratching by pipelle biopsy in IVF (the PIP study). A pragmatic randomised controlled trial. Hum Reprod 33(Suppl_1), i62 (2018).
Lewis, S. & Clarke, M. Forest plots: trying to see the wood and the trees. BMJ 322, 1479–1480 (2001).
Sterne, J. A. C. et al. Recommendations for examining and interpreting funnel plot asymmetry in meta-analyses of randomized controlled trials. BMJ 22(343), d4002 (2011).
Acknowledgements
The authors would like to acknowledge Hale Yapici Eser (Koc University School of Medicine, Department of Psychiatry) who helped with analysis and interpretation of the data.
Author information
Authors and Affiliations
Contributions
K.Y. and O.O were responsible for defining the research question, designing the strategy for the literature search and systematic review, interpreting the results, assessing the eligibility of studies for inclusion to the systematic review and manuscript preparation. B.U. was responsible for critical review, revisions, and final approval of the article. All authors approved the final version that is to be published.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yakin, K., Oktem, O. & Urman, B. Intrauterine administration of peripheral mononuclear cells in recurrent implantation failure: a systematic review and meta-analysis. Sci Rep 9, 3897 (2019). https://doi.org/10.1038/s41598-019-40521-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-40521-w
- Springer Nature Limited
This article is cited by
-
Evaluation of NKT Cell Percentage and Function and Its Relationship with Serum IFN-γ and Vitamin D Levels in Women with Recurrent Spontaneous Abortion and Recurrent Implantation Failure
The Journal of Obstetrics and Gynecology of India (2023)
-
The effectiveness of immunomodulatory therapies for patients with repeated implantation failure: a systematic review and network meta-analysis
Scientific Reports (2022)