Abstract
Steroids are frequently used for postoperative pain relief without definite evidence. This study was conducted to assess the pain management effect of the addition of steroids to a multimodal cocktail periarticular injection (MCPI) in patients undergoing knee arthroplasty and evaluate their safety. Pubmed, Embase, and Cochrane Library were searched through April, 2018. A total of 918 patients from ten randomized controlled trials (RCTs) were ultimately included. Compared with placebo groups, steroids application could effectively relieve pain on postoperative day (POD)1; decrease C-Reactive protein (CRP) level on POD3; improve range of motion (ROM) in postoperative 5 days; reduce morphine consumption, achieve earlier straight leg raising (SLR), and shorten the length of stay (LOS) in hospital. With regards to adverse effects, it did not increase the risk of postoperative infection, postoperative nausea and vomiting (PONV), or other complications. However, no significant difference in pain relief, ROM, or increased Knee Society Knee Function Scores were found during long-term follow up. Overall, this meta-analysis ensured the efficiency and safety of steroids with MCPI in knee arthroplasty patients during the early postoperative period.
Similar content being viewed by others
Introduction
Knee osteoarthritis (KOA) is a degenerative disease involving the intra-articular tibiofemoral and patellofemoral cartilage1. It causes chronic pain, functional limitation, and emotional disturbance which may lead to disability and poor quality of life2. A knee arthroplasty (KA) is a reliable and suitable surgical procedure for end-stage OA patients to relieve pain, to recover function and to improve health related quality of life3. With the creative designs and improved skills, we are now entering a new phase in which partial osteoarthritic changes can be treated with partial resurfacing prosthetic solutions such as unicompartmental, bi-unicompartmental or patellofemoral arthroplasty4. However, no matter what type of KA is performed, this surgery has commonly been associated with severe postoperative pain as well as postoperative nausea and vomiting (PONV). This can result in immense discomfort, emotional distress, low satisfaction, and further leads to poor surgical outcomes and delayed postoperative recovery5,6. Therefore, effective management of postoperative pain is essential for early rehabilitation and better functional outcomes. Various procedures including continuous epidural anesthesia, local infiltration anesthesia, peripheral nerve block, and patient controlled analgesia (PCA) are taken to control pain.
As an orthopedic surgeon, we place great concerns on the multimodal cocktail periarticular or intra-articular injection (MCPI) strategy after KA for analgesia. Not only this treatment is proven to be a safe and cost-effectiveness measure7,8, but also this intervention is easy to perform. Although the gold standard for the cocktail formula has not yet been set up, the components always contains local anesthetics (ropivacaine and bupivacaine), epinephrine, steroids, opioids and nonsteroidal anti-inflammatory drugs (NSAIDs)9,10,11. However, whether those compositions are all necessary is still to be investigated considering the efficacy and safety.
Steroids have been extensively used in various perioperative settings due to their potent anti-inflammatory and antiemetic effects12,13. Clinical trials have indicated that the addition of steroids to MCPI might decrease edema and blood loss and achieve permanently better range of motion (ROM) due to reduced local inflammatory response following surgical trauma14,15. Some studies have advocated the use of steroids for the benefits of postoperative pain relief and reduced risk of POVN16,17,18, while others have found no significant decrease in pain and PONV with administration of steroids19,20. Moreover, periarticular steroids use may arouse concerns for possible postoperative infection and patellar tendon rupture21. To sum up, it remains inconclusive whether the addition of steroids in the MCPI is necessary.
Several meta-analyses have been launched to address the efficacy and safety of steroids application in knee joint arthroplasty. The major drawback of these studies is not distinguishing local usage of steroid from systemic application12,22,23,24,25,26,27,28, which may hamper the reliability of the conclusion. Only three studies focused on the periarticular application of steroids in KA26,27,29. In Tran’s study, only five studies were included and there was no overwhelming data to suggest the addition of steroids to MPCPI improves postoperative total knee arthroplasty (TKA) pain26. In Zhao’s study, only six RCTs met the inclusion criteria and it was found that the addition of steroids to MCPI could improve the analgesic effect and was proved to be highly safe in patients undergoing TKA, but it couldn’t increase the postoperative ROM of knee joint27. Though eight studies were evaluated in Xing’s meta-analysis, it didn’t assess the analgesia effect of the steroid in patients underwent joint arthroplasty, the results showed that intraarticular steroid injections may lead to increased deep infection rates of subsequent joint arthroplasty29. Moreover, all of these studies were confined to TKA with a limited number of studies included. Actually, for the patients suffering KA, no matter whether partial knee arthroplasty (PKA) or TKA are performed, the mechanisms of pain, stress and other reactions that caused by KA might be similar30,31,32. Consequently, the aim of this study is to systematically summarize the available scientific literatures on all kinds of KA to evaluate the efficacy and safety the addition of steroids addition to MCPI by conducting a quantitative meta-analysis including randomized controlled trials (RCTs).
Results
Search results
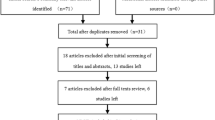
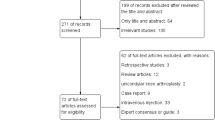
A total of 918 potentially relevant articles wereidentified from the databases, including 315 duplicated articles; 698 studies were excluded after screening titles and abstracts. 34 full-text articles were assessed for eligibility, of which 24 were excluded after assessment of the full-text articles. Eventually, ten14,21,33,34,35,36,37,38,39,40 articles that fulfilled the inclusion criteria were identified for synthetic evaluation. The flow diagram is presented in Fig. 1.
Study characteristics
Demographic characteristics, the details about the included studies are summarized in Table 1. Ten RCTs were performed in 7 countries, involving 820 participants. Eight21,33,34,35,36,37,38,39 of the ten trials performed TKA surgery, while the other two21,40 performed UKA. Both experimental and control groups were tightly matched in sample size, age, sex, and body mass index (BMI). Experimental groups received single-dose postoperative intraarticular injection or periarticular infiltration of steroids, while control groups received placebo or none. Four studies33,34,35,38 performed spinal anesthesia (SA), two36,37 opted for general anesthesia (GA), one21 received endotracheal anaesthesia (EA), and the other three14,39,40
received SA or GA. PCA with opioid, was given to all participates for concomitant pain management. The follow-up period ranged from 1 week to 2 years.
Risk of bias assessment
Cochrane Collaboration tool was used to assess the RCTs (Table 2). Double blinding was provided in all RCTs. The randomization algorithm was mentioned in 6 trials14,21,33,37,38,39, including random number generator, block randomization, random numbers, and randomization table. Four trials34,35,36,40 recorded the allocation concealment information by sealed envelopes. Other assessments, such as blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, and selective reports, were all in good performance.
Visual analogue pain score (VAS)
All studies recorded pain score, but only 4 trials21,33,37,38 provided detailed data as means and standard deviations (SDs). Two studies33,38 reported both VAS at rest (RVAS) and during activity (AVAS). AVAS could not be collected for analysis due to unsuitable format of the data. Therefore, if not specifically mentioned, VAS at different time points were all considered as RVAS. Finally, postoperative day (POD) 1, 3 and postoperative week (POW) 2 were eligible for meta-analysis. The result showed that VAS was significantly lower in the steroids group than the control group on POD1 (MD = −1.52, 95% CI −2.94 to −0.10, P = 0.04). However, no significant difference was found between the two groups on POD3 and POW 2. Random-effect models were used to perform meta-analysis for POD1 (I2 = 82%, P = 0.004) and POD3 (I2 = 76%, P = 0.04) as the substantial heterogeneity was high (Fig. 2).
Range of motion (ROM)
Seven studies14,21,33,35,37,38,40 provided detailed data. Eleven time points were analyzed which included POD1, 2, 3, 4, 5, POW1, 2, 4, 6, and postoperative month (POM) 3, 6, respectively. The results showed that the steroids group had significantly better ROM on the first postoperative 5 days: POD1 (MD = 11.57, 95% CI 9.85 to 13.30, P < 0.00001), POD2 (MD = 9.03, 95% CI 6.67 to 11.38, P < 0.00001), POD3 (MD = 5.73, 95% CI 0.85 to 10.60, P = 0.02), POD4 (MD = 5.53, 95% CI 0.68 to 10.38, P = 0.03), and POD5 (MD = 5.90, 95% CI 0.87 to 10.93, P = 0.02). However, the difference was not significant after POW1. The substantial heterogeneity was high at POW4 (I2 = 94%, P < 0.00001), POM3 (I2 = 93%, P < 0.00001), and POM6 (I2 = 81%, P = 0.006). Therefore, the random-effect models were used for these outcomes. Fixed-effect models were applied at other time points (Fig. 3).
Morphine consumption
Three studies14,21,37 provided detailed data for postoperative morphine consumption and the total amount used during hospitalization was accumulated for analysis. A random-effect model was used due to significant heterogeneity (I2 = 68%, P = 0.02). The overall effect showed that morphine consumption for the steroid groups were significantly less than that for the control groups (MD = −7.94, 95% CI −14.35 to −1.53, P = 0.02; Fig. 4a).
C-reactive protein (CRP)
Three studies37,38,40 provided detailed data for CRP level, but only one time point included two studies that were eligible for meta-analysis. Pooled results showed that the CRP level on POD3 in the steroid groups was significantly lower than that in control groups (WMD = −4.82, 95% CI −7.41 to −2.23, P = 0.0003). Random-effect models were used for this study (I2 = 84%, P = 0.01; Fig. 4b).
Straight leg raising (SLR)
The interval required to perform a SLR was collected from three studies14,35,37 involving 282 knees. The result indicated that the patients in the steroid groups could perform a SLR significantly earlier than those in the control groups (MD = −0.65, 95% CI −0.86 to −0.44, P < 0.00001). Statistical heterogeneity was not found in SLR (I2 = 43%, P = 0.17). A fixed-effects model was performed (Fig. 4c).
Length of stay (LOS)
Two studies21,40 provided detailed data with regard to length of stay in hospital. The results indicated that the patients in the steroid groups had significantly shorter LOS than that of the control groups (MD = −0.98, 95% CI −1.25 to −0.71, P < 0.00001). The fixed-effects model was selected as no significant heterogeneity was found (I2 = 0%, P = 0.78; Fig. 4d).
Knee society knee and function scores (KSS)
Two studies21,35 provided detailed data of KSS knee scores and function scores, respectively. Only 1 time point was eligible for meta-analysis. During the follow-up evaluation POM6, the patients in the steroid groups did not achieve higher KSS knee scores than the control groups (MD = 0.54, 95% CI −0.64 to 1.72, P = 0.37, Fig. 4e). A fixed-effects model was performed because of low heterogeneity (I2 = 0%, P = 0.55). Likewise, the KSS function scores were also similar between the two groups (MD = −2.47, 95% CI −11.62 to 6.68, P = 0.60). A random-effect model was used due to significant heterogeneity (I2 = 56%, P = 0.13; Fig. 4f).
Complications
All ten included studies reported the incidence of various complications such as PONV, infection, and wound oozing. There was no significant difference in total incidence of complications between the steroid and control groups (OR = −1.07, 95% CI 0.63 to 1.82, P = 0.81; Fig. 5a).
There were only two types of complications mentioned in at least two papers. Three studies14,21,39 involving 225 patients reported the occurrence of postoperative infection, and the steroid groups did not have an increased number of patients with infection (OR = 1.29, 95% CI 0.31 to 5.38, P = 0.72; Fig. 5b). Four trials33,34,35,37 involving 352 patients were included that assessed the occurrence of PONV. According to the outcomes, the incidence of PONV in the two groups was also similar (OR = 0.78, 95% CI 0.46 to 1.34, P = 0.37; Fig. 5c). The fixed-effects models were applied in these 4 cases due to the low heterogeneity (I2 = 0%). All the results of this meta-analysis are presented in Table 3.
Discussion
This meta-analysis assessed the efficacy and safety of the addition of steroids to MCPI for pain management and physical dysfunction of TKA and UKA patients. The current evidence shows that periarticular steroids injection is certainly effective in pain relief and improved ROM during the early stage after surgery, and is able to reduce morphine consumption, decrease CRP level, achieve earlier SLR, and shorten LOS, but the analgesic effect and functional outcomes were not significant compared to placebo groups during the late postoperative period. The most encouraging result is that the periarticular steroids injection did not increase the risk of postoperative infection, PONV, or other complications. Therefore, we conclude that steroids are more likely to play a positive role during the early postoperative period.
The forest plots indicated that the addition of steroids could further improve the analgesic efficacy of MCPI during the hyper-acute phase as we observed VAS score significantly decreased on the POD1 in the steroids group. The suppression of the inflammatory response by steroids application can be explained by the signaling pathways that suppress the cytoactivity of immune cells by inhibiting the production of phospholipase A2 (PLA2) and hence decreasing the production of inflammatory cytokines and chemokines41. Previous studies had detected lowered interleukin (IL)-6 and serum CRP level in cases treated with systemic and local steroids37,40. Our results also found decreased serum CRP level on the third day after surgery, proving the anti-inflammatory effects of steroids. Consistently, with the relief of postoperative pain, morphine consumption during the hospitalization was significantly decreased, and the time required to perform a SLR and LOS were also shortened in the steroids group. However, analgesic effect in the steroids group was not significant at POD3 and POW2, which indicated that MCPI with steroids could not provide a sustained analgesic effect. Steroids might play a pivotal role, but the effect of local anesthetics would disappear gradually over time.
In this meta-analysis, significant improvement in ROM was observed during the early postoperative period in those patients who received steroid injections during surgery, but the effects subsequently vanished. We inferred that the analgesic effect is sufficient to suppress the postoperative pain caused by surgical trauma, which is consistent with achievement of early SLR and shorter LOS as verified by our results. The difference in ROM between steroids and the control groups was insignificant at the POW1, suggesting that the addition of steroidsto MCPI was unable to increase the ROM, which might be explained by its inability to eliminate postoperative fibrosis and scarring within their respective time frame. Likewise, we observed no improvement of KSS knee or functional scores, which is a comprehensive system rating both the knee prosthesis function and patients’ functional abilities after TKA, at POW6. Three included RCTs reported the KSS scores. No group differences were noted in studies conducted by Christian et al. and Kown et al.21,35. Yue et al. observed significant improvement in postoperative KSSscores in the steroids group as compared with that in control groupat POM1 and POM3. However, at POM6, the KSS scores showed no significant difference between these two groups36. All these results indicate that long term effects of steroids application on functional outcomes are still uncertain.
For safety concerns, many surgeons remain leery of using steroids that may increase the risk of catastrophic complications such as infection and patellar tendon rupture42,43. A series of complications, including postoperative infection, wound oozing, PONV, pruritis, and transient peroneal nerve palsy were mentioned in the included studies. Our results indicate there were no difference in the total incidence of complications between the two groups. Steroids addition to MCPI would not increase the incidence of postoperative infection and PONV. Furthermore, there were no differences in the incidences of complicationsbetweenthe steroids and placebo groups. No case of tendon rupture was reported in the included RCTs.
Limitations of the present meta-analysis should also be acknowledged. Firstly, the sample size is relatively small and some of the indexes were objectively judged which may reduce the reliability of the study. The second limitation is the lack of consensus on the type and doses of the drugs applied in MCPI which also undermine the consolidation of the analysis. Third, some data were missing or could not be extracted. Some of the results appeared heterogeneous, and couldn’t be eliminated by sensitivity or subgroup analyses. We included these high-quality studies and applied random-effect models for meta-analysis, which may mildly influence the reliability of the results. Finally, the reported side effect is scarce and duration of follow-up is relatively short, which might lead to underestimation of complications and uncertainty of long-term efficacy44. Despite the above limitations, this is the first meta-analysis that included RCTs to evaluate the efficiency and safety of periarticular steroids addition to MCPI in TKA and UKA. Large well-designed RCTs are still needed to validate this research moving forward.
In conclusion, this meta-analysis of RCTs suggested that the administration of periarticular steroids injection with MCPI appears to effectively relieve postoperative pain, improve functional outcomes, reduce morphine consumption, and decrease inflammatory reaction without causing a higher incidence of complications in the early postoperative period.
Methods
Search strategy
This quantitative meta-analysis was in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines45. Two researchers searched the relevant studies independently including MEDLINE/PubMed database, the Cochrane Central Register of Controlled Trials (CENTRAL), and EMBASE databases from inception to April, 2018 for relevant RCTs that compared postoperative periarticular injection of steroids with placebo in the knee arthroplasty. Search terms included “knee”, “arthroplasty”, “replacement”, “steroids”, “corticosteroids”, “cortisol”, “cortisone”, “dexamethasone”, “glucocorticoid”, “betamethasone”, “hydrocortisone”, “prednisone”, “prednisolone”, “ethylprednisolone”, “riamcinolone”, and “adrenal cortex hormone” (supplymentary files). Search terms were combined using the Boolean operators ‘AND’ or ‘OR’. No restrictions were imposed, and reference lists of retrieved articles and reviews were also searched. A third reviewer acted as a judge if there was any disagreement.
Inclusion and exclusion criteria
Only RCTs comparing the clinical efficacy between postoperative periarticular or intraarticular injection of steroids with placebo among adults of any sex undergoing primary KA (including TKA, PKA, UKA, et al.) for any indication (osteoarthritis, rheumatoid arthritis, osteonecrosis or acute trauma) were included in our study. Studies that reported at least one outcome were included. The Primary outcomes being VAS and ROM. With secondary outcomes being morphine consumption, CRP, SLR, LOS, and KSS knee and function scores. Studies were excluded from present meta-analysis in view of incomplete data, letters, comments, editorials, case reports, conference abstracts, or review articles.
Data extraction
The relevant data was extracted from included studies by using a standard data extraction. Two researchers used this form to collect the information from studies independently. The main characteristics included the following items: first author’s name, publication year, country, sample size, mean age, sex ratio, BMI, steroids intervention (dose and type), anesthetic techniques, perioperative analgesia intervention, and duration of follow-up. Other relevant data were extracted from included studies. Differences were resolved by consensus. If a study reported the outcomes of multiple doses of steroids for treatment, only data of the low dose group was extracted for analysis. The first main index was VAS score that has 11 pain levels (0 = no pain, 10 = extreme pain). All other types of VAS were converted to this 11 levels score according to ratio. The second index was ROM, composed of both flexion and extension angles of the knee. The flexion angle was used to represent ROM if flexion and extension angles cannot be combined. The third index contained the postoperative morphine consumption. The total amount of morphine consumption during hospitalization was accumulated for analysis. The other indexes included CRP, SLR, LOS, KSS knee and function scores, and complications. CRP is an acute-phase protein synthesized by the liver which reflects the postoperative inflammation46. All the data on CRP were converted to the unit of mg/dl. SLR is a test-conducted post knee surgery to gauge how high a patient is able to elevate his/her leg off of an exam table, reflecting pain control as well as muscle strength recovery26. KSS, composed of knee score and function score, is considered as an objective scoring system to rate the knee and patient’s functional abilities such as walking and stair climbing before and after TKA47. The total incidence of complications after surgery was calculated. If any special type of complication was mentioned more than twice, its incidence was pooled for subgroup analysis48. For missing or incomplete data, we extracted information from diagrams or contacted the corresponding authors to ensure that the information was integrated. Otherwise, we estimated the data wherever possible using the recommendations in the Cochrane Handbook49. Formats that were not suitable included studies reporting means with interquartile ranges, suggesting the data were non-normally distributed making conversion to means and SDs controversial49. Furthermore, data only reporting means without SDs, standard errors (SEs) or confidence intervals (CIs) could not be extracted considering that one of these is required with the mean for meta-analysis.
Quality assessment
The risk of bias of the included studies was assessed using the Cochrane Handbook for Systematic Reviews of Interventions version 5.1.049, based on the following items: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, and selective reporting. The assessment items were either categorized as low risk of bias, high risk of bias, or unclear risk of bias. Discrepancies between assessments were resolved by discussion with a third reviewer when necessary.
Statistical analysis
For continuous data, like VAS scores, ROM and so on, we calculated mean differences (MDs) and 95% CIs if outcomes were measured in the same way between studies, or we used the standardized mean differences (SMDs). Continuous data reported as means and ranges were transformed into means and SDs using Hozo’s formula50. Dichotomous data such as incidence of complications were expressed as odds ratios (ORs) with 95% CIs indicating the effect on intervention. Subgroup analysis was performed for different time points. The Q and chi-squared test were used to assess statistical heterogeneity with the value of P and I2. If heterogeneity was low (P > 0.1, I2 < 50%), a fixed-effects model was used. If heterogeneity was significant (P < 0.1, I2 > 50%), a random-effects model was used. When possible, sensitivity and subgroup analyses were conducted to find the source of the heterogeneity. Publication bias was visually examined by funnel plots. We used Review Manager 5.2 software (Rev Man 5.2, The Cochrane Collaboration, Oxford, UK) and STATA, version 12.0 (Stata Corp LP, College Station, TX) to perform statistical analyses. P < 0.05 was considered statistically significant.
References
Lane, N. E. et al. OARSI-FDA Initiative: Defining the Disease State of Osteoarthritis. Osteoarthritis Cartilage. 19, 478–482 (2011).
Heidari, B. Knee Osteoarthritis Prevalence, Risk Factors, Pathogenesis and Features: Part I. Caspian J Intern Med. 2, 205–212 (2011).
Ethgen, O., Bruyere, O., Richy, F., Dardennes, C. & Reginster, J. Y. Health-Related Quality of Life in Total Hip and Total Knee Arthroplasty. A Qualitative and Systematic Review of the Literature. J. Bone Joint Surg. Am. 86-A, 963–974 (2004).
Benazzo, F., Rossi, S. M. & Ghiara, M. Partial Knee Arthroplasty: Patellofemoral Arthroplasty and Combined Unicompartmental and Patellofemoral Arthroplasty Implants–General Considerations and Indications, Technique and Clinical Experience. Knee. 21(Suppl 1), S43–S46 (2014).
Parvizi, J., Miller, A. G. & Gandhi, K. Multimodal Pain Management After Total Joint Arthroplasty. J. Bone Joint Surg. Am. 93, 1075–1084 (2011).
Dorr, L. D. & Chao, L. The Emotional State of the Patient After Total Hip and Knee Arthroplasty. Clin. Orthop. Relat. Res. 463, 7–12 (2007).
Barrington, J. W., Lovald, S. T., Ong, K. L., Watson, H. N. & Emerson, R. J. Postoperative Pain After Primary Total Knee Arthroplasty: Comparison of Local Injection Analgesic Cocktails and the Role of Demographic and Surgical Factors. J. Arthroplasty. 31, 288–292 (2016).
Joo, J. H., Park, J. W., Kim, J. S. & Kim, Y. H. Is Intra-Articular Multimodal Drug Injection Effective in Pain Management After Total Knee Arthroplasty? A Randomized, Double-Blinded, Prospective Study. J. Arthroplasty. 26, 1095–1099 (2011).
Zhou, Y. et al. Single-Dose Intra-Articular Ropivacaine After Arthroscopic Knee Surgery Decreases Post-Operative Pain without Increasing Side Effects: A Systematic Review and Meta-Analysis. Knee Surg. Sports Traumatol. Arthrosc. 24, 1651–1659 (2016).
Zeng, C. et al. Single-Dose Intra-Articular Morphine After Arthroscopic Knee Surgery: A Meta-Analysis of Randomized Placebo-Controlled Studies. Arthroscopy. 29, 1450–1458 (2013).
Wei, J., Yang, H. B., Qin, J. B., Kong, F. J. & Yang, T. B. Single-Dose Intra-Articular Bupivacaine After Knee Arthroscopic Surgery: A Meta-Analysis of Randomized Placebo-Controlled Studies. Knee Surg. Sports Traumatol. Arthrosc. 22, 1517–1528 (2014).
Fan, Z. et al. The Efficacy of Dexamethasone Reducing Postoperative Pain and Emesis After Total Knee Arthroplasty: A Systematic Review and Meta-Analysis. Int. J. Surg. 52, 149–155 (2018).
Gilron, I. Corticosteroids in Postoperative Pain Management: Future Research Directions for a Multifaceted Therapy. Acta Anaesthesiol. Scand. 48, 1221–1222 (2004).
Pang, H. N., Lo, N. N., Yang, K. Y., Chong, H. C. & Yeo, S. J. Peri-Articular Steroid Injection Improves the Outcome After Unicondylar Knee Replacement: A Prospective, Randomised Controlled Trial with a Two-Year Follow-Up. J. Bone Joint Surg. Br. 90, 738–744 (2008).
Holte, K. & Kehlet, H. Perioperative Single-Dose Glucocorticoid Administration: Pathophysiologic Effects and Clinical Implications. J. Am. Coll. Surg. 195, 694–712 (2002).
Sculco, P. K. & Pagnano, M. W. Perioperative Solutions for Rapid Recovery Joint Arthroplasty: Get Ahead and Stay Ahead. J. Arthroplasty. 30, 518–520 (2015).
Jules-Elysee, K. M. et al. Use of Low-Dose Steroids in Decreasing Cytokine Release During Bilateral Total Knee Replacement. Reg. Anesth. Pain Med. 36, 36–40 (2011).
McLawhorn, A. S. et al. Effects of Steroids On Thrombogenic Markers in Patients Undergoing Unilateral Total Knee Arthroplasty: A Prospective, Double-Blind, Randomized Controlled Trial. J. Orthop. Res. 33, 412–416 (2015).
Bergeron, S. G., Kardash, K. J., Huk, O. L., Zukor, D. J. & Antoniou, J. Perioperative Dexamethasone Does Not Affect Functional Outcome in Total Hip Arthroplasty. Clin. Orthop. Relat. Res. 467, 1463–1467 (2009).
Jules-, E. K. et al. Steroid Modulation of Cytokine Release and Desmosine Levels in Bilateral Total Knee Replacement: A Prospective, Double-Blind, Randomized Controlled Trial. J. Bone Joint Surg. Am. 94, 2120–2127 (2012).
Christensen, C. P., Jacobs, C. A. & Jennings, H. R. Effect of Periarticular Corticosteroid Injections During Total Knee Arthroplasty. A Double-Blind Randomized Trial. J. Bone Joint Surg. Am. 91, 2550–2555 (2009).
Liu, G., Gong, M., Wang, Y. & Xiang, Z. Effect of Methylprednisolone on Pain Management in Total Knee or Hip Arthroplasty: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Clin. J. Pain. 34, 967–974 (2018).
Li, X., Xu, G., Xie, W. & Ma, S. The Efficacy and Safety of Dexamethasone for Pain Management After Total Knee Arthroplasty: A Systematic Review and Meta-Analysis. Int. J. Surg. 53, 65–71 (2018).
Mohammad, H. R. et al. Perioperative Adjuvant Corticosteroids for Postoperative Analgesia in Knee Arthroplasty. Acta Orthop. 89, 71–76 (2018).
Meng, J. & Li, L. The Efficiency and Safety of Dexamethasone for Pain Control in Total Joint Arthroplasty: A Meta-Analysis of Randomized Controlled Trials. Medicine (Baltimore). 96, e7126 (2017).
Tran, J. & Schwarzkopf, R. Local Infiltration Anesthesia with Steroids in Total Knee Arthroplasty: A Systematic Review of Randomized Control Trials. J Orthop. 12, S44–S50 (2015).
Zhao, X. et al. Efficacy of Steroid Addition to Multimodal Cocktail Periarticular Injection in Total Knee Arthroplasty: A Meta-Analysis. J. Orthop. Surg. Res. 10, 75 (2015).
Cui, Z. et al. The Efficacy of Steroid Injection in Total Knee Or Hip Arthroplasty. Knee Surg. Sports Traumatol. Arthrosc. 23, 2306–2314 (2015).
Xing, D. et al. Dose Intraarticular Steroid Injection Increase the Rate of Infection in Subsequent Arthroplasty: Grading the Evidence through a Meta-Analysis. J. Orthop. Surg. Res. 9, 107 (2014).
Witjes, S. et al. Return to Sports and Physical Activity After Total and Unicondylar Knee Arthroplasty: A Systematic Review and Meta-Analysis. Sports Med. 46, 269–292 (2016).
Pollock, M., Somerville, L., Firth, A. & Lanting, B. Outpatient Total Hip Arthroplasty, Total Knee Arthroplasty, and Unicompartmental Knee Arthroplasty: A Systematic Review of the Literature. JBJS Rev. 4 (2016).
Chughtai, M. et al. Cryotherapy Treatment After Unicompartmental and Total Knee Arthroplasty: A Review. J. Arthroplasty. 32, 3822–3832 (2017).
Tsukada, S., Wakui, M. & Hoshino, A. The impact of including corticosteroid in a periarticular injection for pain control after total knee arthroplasty: a double-blind randomised controlled trial. Bone Joint J. 98-B, 194–200 (2016).
Kim, T. W. et al. Which analgesic mixture is appropriate for periarticular injection after total knee arthroplasty? Prospective, randomized, double-blind study. Knee Surg. Sports Traumatol. Arthrosc. 23, 838–845 (2015).
Kwon, S. K., Yang, I. H., Bai, S. J. & Han, C. D. Periarticular injection with corticosteroid has an additional pain management effect in total knee arthroplasty. Yonsei Med. J. 55, 493–498 (2014).
Yue, D. B., Wang, B. L., Liu, K. P. & Guo, W. S. Efficacy of multimodal cocktail periarticular injection with or without steroid in total knee arthroplasty. Chin. Med. J. (Engl.). 126, 3851–3855 (2013).
Ikeuchi, M. et al. Effects of dexamethasone on local infiltration analgesia in total knee arthroplasty: a randomized controlled trial. Knee Surg. Sports Traumatol. Arthrosc. 22, 1638–1643 (2014).
Chia, S. K. et al. Peri-articular steroid injection in total knee arthroplasty: a prospective, double blinded, randomized controlled trial. J. Arthroplasty. 28, 620–623 (2013).
Sean, V. W. et al. Single-dose periarticular steroid infiltration for pain management in total knee arthroplasty: a prospective, double-blind, randomised controlled trial. Singapore Med. J. 52, 19–23 (2011).
Ng, Y. C. et al. Effects of periarticular steroid injection on knee function and the inflammatory response following Unicondylar Knee Arthroplasty. Knee Surg. Sports Traumatol. Arthrosc. 19, 60–65 (2011).
Creamer, P. Intra-articular corticosteroid injections in osteoarthritis: do they work and if so, how? Ann. Rheum. Dis. 56, 634–636 (1997).
Marsland, D., Mumith, A. & Barlow, I. W. Systematic review: the safety of intra-articular corticosteroid injection prior to total knee arthroplasty. Knee. 21, 6–11 (2014).
Sauerland, S., Nagelschmidt, M., Mallmann, P. & Neugebauer, E. A. Risks and benefits of preoperative high dose methylprednisolone in surgical patients: a systematic review. Drug Saf. 23, 449–461 (2000).
Cutolo, M. et al. Use of glucocorticoids and risk of infections. Autoimmun Rev. 8, 153–155 (2008).
Liberati, A. et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J. Clin. Epidemiol. 62, e1–34 (2009).
Pepys, M. B. & Hirschfield, G. M. C-reactive protein: a critical update. J. Clin. Invest. 111, 1805–1812 (2003).
Scuderi, G. R. et al. The new Knee Society Knee Scoring System. Clin. Orthop. Relat. Res. 470, 3–19 (2012).
Deng, Z. H. et al. Topical diclofenac therapy for osteoarthritis: a meta-analysis of randomized controlled trials. Clin. Rheumatol. 35, 1253–1261 (2016).
Julian, H. & Sally, G. Cochrane handbook for systematic reviews of interventions (2011).
Hozo, S. P., Djulbegovic, B. & Hozo, I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med. Res. Methodol. 5, 13 (2005).
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 81874030), the Guangdong Province Science and Technology R&D project planning (No. 2014A020212656), the Provincial Science Foundation of Hunan (No. 2015JJ3139) and China Scholarship Council (student ID: 201606370164 and 201606375101).
Author information
Authors and Affiliations
Contributions
All authors had full access to the data in the study and take responsibility for the integrity of the data and theaccuracy of the data analysis. Y.S.L., W.L., S.S.G. and Z.H.D. were responsiblefor conception and design of the study and drafted the manuscript. Z.H.D., Y.S.L., X.Y.S. and G.H.L. contributed to data collection. R.N.K. and G.R.S. contributed to language editing. Z.H.D. contributed to manuscriptpreparation and data analysis. Z.H.D. and S.S.G. contributed tostudy retrieval. W.L., Y.S.L. and Z.H.D. contributed to revision of the manuscript. All the authors contributed to the interpretationof the data and critically reviewed the manuscript for publication.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Deng, Z., Li, Y., Storm, G.R. et al. The efficiency and safety of steroid addition to multimodal cocktail periarticular injection in knee joint arthroplasty: a meta-analysis of randomized controlled trials. Sci Rep 9, 7031 (2019). https://doi.org/10.1038/s41598-019-43540-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-43540-9
- Springer Nature Limited
This article is cited by
-
Perioperative multimodal analgesic injection for patients with adolescent idiopathic scoliosis undergoing posterior spinal fusion surgery
Spine Deformity (2023)
-
Periarticular injection and hamstring block versus placebo for pain control in anterior cruciate ligament reconstruction: A randomized controlled trial
European Journal of Orthopaedic Surgery & Traumatology (2022)
-
Adding corticosteroids to periarticular infiltration analgesia improves the short-term analgesic effects after total knee arthroplasty: a prospective, double-blind, randomized controlled trial
Knee Surgery, Sports Traumatology, Arthroscopy (2021)
-
Relationship between dexamethasone added to periarticular anesthetic infiltration and postoperative nausea and vomiting following total knee arthroplasty under general anesthesia: a retrospective observational study
JA Clinical Reports (2020)