Abstract
Clinically differentiating multiple system atrophy cerebellar (MSA-C) phenotype and spinocerebellar ataxias (SCAs) is challenging especially in the early stage. We assessed diagnostic value of brain magnetic resonance imaging (MRI) in differentiating MSA-C and SCAs based at different disease stages (<3, 3–7, and >7 years of disease duration). Overall, 186 patients with probable MSA-C and 117 with genetically confirmed SCAs were included. Hot cross bun (HCB) signs and middle cerebellar peduncle (MCP) hyperintensities were exclusively prevalent in MSA-C compared to SCAs at <3 years (HCB, 44.6% versus 0.9%; MCP hyperintensities, 38.3% versus 0.9%, respectively). Sensitivity, specificity, and positive predictive value (PPV) for HCB signs to differentiate MSA-C from SCAs were 45%, 99%, and 99% and those for MCP hyperintensities were 68%, 99%, and 99%, respectively; considering both HCB signs and MCP hyperintensities, specificity and PPV were 100%. However, the differential value of MRI signs decreased over time. MCP widths were smaller and showed more significant decrease in MSA-C than in SCAs. In conclusion, pontine and MCP changes were exclusively prominent in early stage MSA-C rather than in SCAs. Therefore, we should consider disease duration when interpreting pontine and MCP changes in brain MRIs, which will help better differentiate MSA-C and SCAs.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Ataxia is a clinically heterogeneous group of disorders, in which multiple system atrophy cerebellar (MSA-C) phenotype denotes idiopathic degenerative cerebellar ataxia, while spinocerebellar ataxias (SCAs) represent autosomal dominant cerebellar ataxia1,2. Although aetiologies, diagnostic processes, and prognoses are totally different3,4, differentiation should be done very carefully, especially in the early stage of disease. Ataxia could be the only sign of both diseases in the early stage, as autonomic dysfunction or parkinsonism appears 2–5 years later in MSA patients with cerebellar ataxia5,6,7. Some patients with SCAs could also manifest autonomic dysfunction or parkinsonism8,9. Furthermore, family history could be negative in patients with SCAs10.
Brain magnetic resonance imaging (MRI) could be helpful when making a differential diagnosis, as hot cross bun (HCB) signs and hyperintensities in middle cerebellar peduncles (MCPs) are considered characteristic for MSA-C11,12,13. However, recent reports showed that those signs were found in ataxic patients with other etiologies including SCAs14,15,16. HCB signs and MCP changes reflect sequential gliosis and degenerative process of the pons and related afferent fibers17,18. As disease progression rates are different in MSA and SCAs5,7,19, the effect of disease duration should be considered. To our knowledge, radiologic changes depending on disease duration of MSA and SCAs have not been studied so far.
We hypothesised that pontine and MCP changes would differ in early-stage brain MRIs of patients with MSA-C and SCAs, which would have a diagnostic value in differentiating MSA-C from SCAs. Thus, we explored the prevalence of HCB signs and MCP changes in brain MRIs based on the duration of MSA-C and SCAs.
Methods
Patient selection and baseline data collection
This study was approved by the Institutional Review Board (IRB) of Samsung Medical Centre, Seoul, Korea. All methods were performed in accordance with the relevant guidelines and regulations, and a waiver of informed consent was granted by the IRB. Medical records of patients with probable MSA-C and genetically confirmed SCAs from Samsung Medical Centre in Seoul were retrospectively reviewed. From January 2012 to March 2018, 196 MSA-C patients were initially identified; eventually, 186 patients were included based on the second consensus diagnostic criteria of MSA4. Patients who showed a static course, lacked autonomic failure as described in the criteria, reported >75 years as age of onset, or presented with other explicable causes of cerebellar ataxia were excluded. Furthermore, for those who manifested initial symptom before 50 years of age, SCA panel tests were performed to exclude the possibility of autosomal dominant cerebellar ataxia. Regarding SCAs, 157 patients with positive genetic tests visited our centre from January 2011 to March 2018. Patients whose MRI findings were not available or inappropriate for assessment were excluded; finally, 117 patients from 84 families with SCAs were included. Baseline characteristics such as sex, age of onset, and disease duration at the time of MRI acquisition were obtained. Disease duration was calculated from the symptom onset based on medical records, when any of the following features manifested: gait disturbance due to imbalance or slowness, dysarthria, hand incoordination, postural dizziness or syncope, urinary incontinence or retention, erectile dysfunction, dizziness unexplained by otologic problems or postural change; decreased visual acuity, parkinsonism and dystonia were considered initial symptoms as well20,21.
MRI acquisition and analysis
Conventional brain MRIs of the patients were retrospectively reviewed. MRIs were grouped according to the duration of SCAs and MSA-C (<3, 3–7, and >7 years of disease duration) with reference to the clinical course of the disease. The median time period from onset to the point of assisted walking was approximately 3 years and that to bedridden state or death was around 7 years in MSA-C patients5,7. Two independent movement disorder specialists (M Kim and JW Cho) who were blinded to the clinical information evaluated all MRIs. In cases of discrepancy between grades assessed by the two neurologists, the final grade for analysis was decided by consensus between the two.
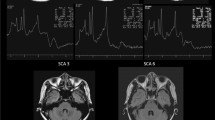
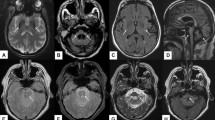
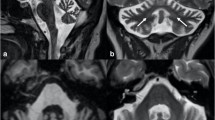
All patients were imaged on 3.0 T system. Presence of HCB signs, hyperintensities in MCPs, dorsolateral hyperintensities in the putamen (putaminal rim signs), or putaminal hypointensities were assessed on T2-weighted axial images (3000–6960 ms repetition time, 80–130 ms echo time, and 3.0–5.0 mm thickness). For MCP atrophy, MCP widths were measured by averaging the distances between the superior and inferior borders of MCPs in both sides on T1-weighted sagittal images [250–717 ms repetition time, 2.9–12.5 ms echo time, 4.0–5.0 mm thickness] (Supplementary Fig. S1)22. Pontine signals were identified on three categories, namely, no HCB sign or midline hyperintensities (MH), MH, and HCB signs23. HCB signs were considered present when both horizontal and vertical lines were observed24. MCP signs were marked as absent or present11,12,13. Regarding putaminal abnormalities, high signals alongside the dorsolateral margin of the putamen (putaminal rim signs) and signal intensities lower than those of adjacent globus pallidus (putaminal hypointensities) were considered significant25.
Statistical analysis
Statistical analyses were performed with a commercially available software package (IBM SPSS Statistic version 25, SPSS Inc., Chicago, IL, USA). Age of onset, sex ratio, and disease duration at MRI acquisition were compared between patients with MSA-C and SCAs. For categorical variables, Pearson’s χ2 test was used; for continuous variables, data distribution was first evaluated using Shapiro-Wilk analysis, and independent t-test or Mann-Whitney test was performed as appropriate. P-values of the difference in frequencies of HCB signs and MCP hyperintensities were evaluated using Pearson’s χ2 test. Independent t-test or Mann Whitney test was used to compare MCP widths between two groups, and Wilcoxon rank test was used for comparison between <3 years and 3–7 years of disease duration within each group. Inter-rater reliability for all images between the two readers was assessed with intra-class correlation coefficient (ICC). p-values < 0.05 were considered statistically significant.
In addition, multinomial regression analysis was performed in the initial and second MRIs of MSA-C and SCAs to investigate if there was any correlation between presence of HCB or MH and clinical variables: age of onset, sex, Scale for the Assessment and Rating of Ataxia, or accompanying symptoms such as gait disturbance, dysarthria, autonomic dysfunction, rapid eye movement sleep behaviour disorder, hand incoordination, dizziness, dysphagia, sensory symptoms, and parkinsonism. Bonferroni’s method was applied to p-value and confidence level to correct the increased size of the type I error due to multiple testing.
Baseline characteristics were compared within SCA subgroups (SCA1, SCA2, SCA3, SCA6, SCA7, SCA8 and SCA17) using Fisher’s exact test for categorical variables and one-way analysis of variance for continuous variables.
Results
Patient characteristics and MRI distribution
Baseline characteristics of the subjects are described in Table 1. Sex ratios were not different between the two groups. Age of onset was significantly higher in patients with MSA-C than in those with SCAs (t = 12.225, p = 0.000). In both MSA-C and SCA groups, gait disturbance due to imbalance or slowness was the most common initial symptom (Supplementary Table S2 online). Demographic and genetic characteristics depending on SCA subgroups are demonstrated in Supplementary Table S3.
All patients had initial MRIs within 3 years of disease duration. Follow-up MRIs were performed in 40 MSA-C patients and in 33 SCA patients at 3–7 years of disease duration. No MSA-C patient had follow-up study after 7 years of disease duration, while 44 patients with SCAs took the second MRI during the same period. Initial MRI was performed at similar disease duration (1.3 ± 0.8 versus 1.5 ± 0.9 years, respectively), while the second MRI was performed earlier in MSA-C patients at 3–7 years of disease duration (4.1 ± 0.9 versus 4.9 ± 1.0 years, respectively). At > 7 years of disease duration, MRIs were available only in the SCA group, as MRI was performed at an average of 14.0 ± 7.1 years.
Radiologic findings in MSA-C and SCA patients
The distribution of HCB signs and MCP hyperintensities are illustrated in Fig. 1. At <3 years of disease duration, 83 (44.6%) of 186 MSA-C patients demonstrated HCB signs and 66 (35.5%) showed MH; however, among 117 SCA patients, only one (0.9%) patient with SCA2 manifested definite HCB sign and 30 (25.6%) exhibited MH (Fig. 1a). At 3–7 years of disease duration, 29 (72.5%) MSA-C patients showed HCB signs; 23 (57.5%) patients newly manifested HCB signs and 6 (15.0%) patients had already shown HCB signs in the initial study. MH was observed in 11 (27.5%) patients including 3 (7.5%) patients who had initially shown MH within 3 years of disease duration. Of the 33 SCA patients, HCB signs and MH appeared in four (12.1%) and 18 (54.5%) patients, of which four (12.1%) had already shown MH at <3 years of disease duration. After 7 years of disease duration, 10 (22.7%) SCA patients newly presented HCB signs. MCP hyperintensities were initially detected in 127 (68.3%) of 186 MSA-C patients at <3 years of disease duration, while they appeared only in one (0.9%) of 117 SCA patients (Fig. 1b). Overall, 30 (75.0%) MSA-C patients exhibited MCP hyperintensities at 3–7 years of disease duration; among them, 15 (37.5%) patients newly manifested high signals in MCPs for the first time. In SCAs, MCP hyperintensities newly appeared in five (15.2%) of 33 patients at 3–7 years and in 9 (20.5%) of 44 patients at >7 years of disease duration. Furthermore, when HCB and MCP signs were both considered, the difference between MSA-C and SCAs became more evident (Fig. 1c). The concurrence of HCB signs and MCP hyperintensities was detected in 71 (38.2%) versus 0 (0%) patients at <3 years, and 24 (60.0%) versus 2 (6.1%) patients at 3–7 years of disease duration in MSA-C and SCAs, and the concurrence of both signs in SCA patients at >7 years of disease duration was six (13.6%). T1-weighted sagittal images were available in 124 patients at <3 years and in 38 patients at 3–7 years of disease duration in the MSA-C group and in 51 and 28 patients, respectively, in the SCA group during the same periods. The average MCP width was smaller in MSA-C patients than in SCA patients (7.4 ± 1.5 versus 8.3 ± 1.7 mm, p = 0.000) at <3 years; the difference became more evident (5.4 ± 1.4 versus 7.6 ± 1.8 mm, p = 0.000) at 3–7 years of disease duration (Fig. 2). When the difference between <3 years and 3–7 years of disease duration was compared within each group, MCP widths for MSA-C significantly decreased overtime from 7.2 ± 1.5 to 5.0 ± 1.2 mm (p = 0.000); MCP widths in SCAs also showed marked decrease from 9.2 ± 1.2 to 7.3 ± 1.3 mm (p = 0.001).
MRI findings in MSA-C and SCAs depending on disease duration. (a) HCB signs and (b) MCP hyperintensities were more prevalent in MSA-C than in SCAs. (c) When HCB signs and MCP hyperintensities were considered together, the differentiation between MSA-C and SCAs became more evident. HCB, hot cross bun; MCP, middle cerebellar peduncle; MSA-C, multiple system atrophy cerebellar phenotype; SCAs, spinocerebellar ataxias.
MCP widths in MSA-C and SCAs. MCP widths were smaller in MSA-C than in SCAs at <3 years and 3–7 years of disease duration. The MCP widths for MSA-C and SCA groups significantly decreased overtime when <3 and 3–7 year groups were compared within each group. MCP, middle cerebellar peduncle; MSA-C, multiple system atrophy cerebellar phenotype; SCAs, spinocerebellar ataxias.
Putaminal rim signs and putaminal hypointensities were observed in overall 7 (3.8%) and 12 (6.5%) patients with MSA-C, respectively. In our cohort, no SCA patients exhibited putaminal changes.
Statistical measures
Sensitivity, specificity, and positive predictive value (PPV) of HCB signs and MCP hyperintensities are shown in Table 2. At <3 years of disease duration, sensitivity of HCB signs to differentiate MSA-C from SCAs was below 50%, while the specificity and PPV were 99.1% and 98.8%, respectively. During the next 3–7 years, sensitivity increased to 72.5%, while specificity and PPV decreased to 87.9%. MCP hyperintensities showed similar pattern. Sensitivity was 68.3%; specificity and PPV were 99.1% and 99.2%, respectively, at <3 years of disease duration, which were then 75.0%, 84.8%, and 85.7% at 3-7 years of disease duration, respectively. When HCB and MCP signs were both considered, the PPV for differentiating MSA-C from SCAs was 100% at <3 years and 92.3% at 3–7 years of disease duration, respectively. Although the p value of difference in MCP widths between MSA-C and SCAs were 0.000 both at <3 years and 3–7 years of disease duration, a large overlap made it unable to set a cut-off value that effectively differentiated the two groups. At 3–7 years of disease duration, sensitivity, specificity and positive predictive value to distinguish MSA-C from SCAs were 76.3%, 78.6% and 82.9% when a cutoff of 6 mm or less was used.
The ICC between the two investigators was 0.985 for HCB, 0.977 for MCP, and 0.979 for MCP width. ICCs for putaminal rim signs and putaminal hypointensities were 0.946 and 0.983, respectively.
Multinomial regression analysis revealed that none of the clinical parameters significantly predicted HCB or MH.
HCB signs and MCP hyperintensities among the SCA subtypes
The overall frequencies of HCB and MCP hyperintensities in SCA patients are described in Table 3. HCB signs and high signals in MCPs were the most prevalent in SCA2, and none of the patients with SCA1 (n = 7), SCA6 (n = 20), and SCA17 (n = 1) manifested those signs.
Discussion
To our knowledge, this is the first study to compare radiologic findings of MSA-C and SCAs according to their disease duration. HCB signs and MCP hyperintensities were exclusively prevalent in MSA-C at <3 years with PPVs of nearly 100%. This value even increased to 100% at the same disease duration when HCB signs and MCP hyperintensities were considered together. As a result, the presence of HCB signs or MCP hyperintensities within 3 years of disease duration would differentiate MSA-C from SCAs. MCP widths were smaller in MSA-C with statistical significance at <3 years as well as at 3–7 years of disease duration, although the differential value of MCP widths was relatively low. Dorsolateral putaminal rim signs and putaminal hypointensities were only detected in the MSA-C group. When only SCAs were considered, HCB signs and MCP hyperintensities were the most frequent in SCA2.
Our results are in line with pathologic findings. A sequential analysis on α-synuclein pathology in MSA-C patients showed that the pontocerebellar and middle cerebellar fibres were initially affected in the pons26. While post-mortem analyses in SCAs also revealed various degrees of cerebellar and brainstem involvement, reports regarding early stage pathology are limited27. Autopsy specimens from SCA2 and SCA8 patients with 3 years of disease duration demonstrated no involvement of pontocerebellar fibres28,29. Furthermore, these radiologic and pathologic findings appear to be related with the clinical course. Functional state of the patients was also assessed in terms of walking ability during MRI acquisition, in which more MSA-C patients required assistance or were wheelchair bound compared to SCA patients (Supplementary Table S4). This result was in accordance with the results of previous studies, wherein MSA-C patients showed more rapid deterioration in activities of daily living compared to those with SCAs30. Therefore, early appearance of HCB signs and MCP changes paralleled the pathologic changes involving pontocerebellar fibres, which may be related with clinical findings based on the time course.
The prevalence of putaminal rim signs or hypointensities in this study was relatively lower to that in previous studies11,12,13. The low incidence of putaminal change in this study may have been due to the correlation of parkinsonism with putaminal changes in brain MRIs and that subjects were limited to those with predominant cerebellar phenotype12.
The overall frequencies of HCB signs were 12.8% in SCA patients, which was similar to that reported in a Taiwanese study (8.7%) wherein SCA2 was also the most common subtype15. Moreover, the similar results in both populations may have been attributed to the high involvement of the pontine nucleus in SCA2 than in any other subtypes27,31. However, disease duration was not specified in the Taiwanese report15. Other case series reporting HCB signs in SCAs did not take into account disease duration; in fact, MRIs were taken at more than 5 years of disease duration32,33,34. In our SCA cohort, the overall HCB signs appeared at 11.1 ± 6.0 years of disease duration.
For those who had follow-up MRIs, we additionally investigated how the pontine and MCP signals had changed in MSA-C and SCAs (Supplementary Table S5). We observed a common process of HCB formation in both disease groups: vertical lines appeared first, followed by horizontal lines23,24. More MSA-C patients eventually came to have HCB signs (67.6% versus 18.2%) while SCA patients turned to have MH without progression to HCB signs. Similar to HCB signs, more MSA-C patients exhibited MCP hyperintensities than SCA patients in follow-up studies (60.0% versus 18.2%).
The most critical limitation of the current study was that pathological study results was not available and SCA panel tests were performed in a limited number of MSA-C patients4,35. However, all patients fulfilled the diagnostic criteria of probable MSA4. As the age of onset in genetically confirmed SCAs was around 40 years in our cohort, SCA diagnoses were ruled out in MSA-C patients with <50 years of age of onset using SCA panel test. The possibility of SCA6, which is known for late-onset cerebellar ataxia, was discarded as well, since pure cerebellar ataxia distinguished SCA6 from MSA-C. Second, age of onset, which was based on medical records, might have been inaccurate. Gait disturbance was initially the most common chief complaint and was accompanied by various motor or non-motor symptoms in MSA (Supplementary Table S2); however, it was not easy to identify accurately when all those symptoms started. In fact, a retrospective review of questionnaires revealed that postural dizziness and erectile dysfunction were the most prevalent initial symptoms in MSA, which were found in 58.1% and 38.7% of our MSA-C patients at initial visit, respectively; urinary symptoms prevailed among non-motor symptoms20. However, as gait disturbance due to postural instability or slowness was detected during the first year of disease onset20, we thought that it was possible to approximate disease duration. In the SCA group, gait disturbance was the most common initial symptom as previously described21.
We compared radiologic signs between MSA-C and SCAs for the first time, especially focusing on the disease duration. All patients in our cohort underwent MRI within 3 years after symptom onset; however, none of MSA-C patients had an MRI after 7 years, whereas SCA patients had MRI at >10 years of disease duration. This reflects that ambiguous results are often encountered in the early stage of ataxias, while clinical features become more evident after 7 years of disease duration, requiring fewer tests to differentiate MSA-C from SCAs. Obviously, a thorough history taking, neurologic examination, and other diagnostic modalities are necessary, but early HCB signs and MCP hyperintensities have diagnostic value in MSA-C and SCAs; if MCP shows marked atrophy, which also suggests MSA-C rather than SCAs.
In conclusion, we should take into account disease duration when interpreting pontine and MCP changes in brain MRIs, which helps better differentiate MSA-C and SCAs in synopsis with clinics and genetics. This would provide chances not only to anticipate prognosis but also to apply symptomatic and even disease modifying therapies.
Data availability
All data generated or analyzed during this study are included in this published article (and its Supplementary Information Files).
References
Klockgether, T. Sporadic ataxia with adult onset: classification and diagnostic criteria. Lancet Neurol. 9, 94–104 (2010).
Kim, J. S. & Cho, J. W. Hereditary cerebellar ataxias: A Korean perspective. J Mov Disord. 8, 67–75 (2015).
TD., B. Hereditary ataxia overview. GeneReviews®, https://www.ncbi.nlm.nih.gov/books/NBK1138/, (accessed 2019).
Gilman, S. et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 71, 670–676 (2008).
Tada, M. et al. Early development of autonomic dysfunction may predict poor prognosis in patients with multiple system atrophy. Arch Neurol 64, 256–260 (2007).
Gilman, S. et al. Evolution of sporadic olivopontocerebellar atrophy into multiple system atrophy. Neurology 55, 527–532 (2000).
Watanabe, H. et al. Progression and prognosis in multiple system atrophy: an analysis of 230 Japanese patients. Brain 125, 1070–1083 (2002).
De Joanna, G. et al. Autonomic nervous system abnormalities in spinocerebellar ataxia type 2: a cardiovascular neurophysiologic study. J Neurol Sci 275, 60–63 (2008).
Furtado, S. et al. Profile of families with parkinsonism-predominant spinocerebellar ataxia type 2 (SCA2). Mov Disord 19, 622–629 (2004).
Kim, J. S., Kwon, S., Ki, C. S., Youn, J. & Cho, J. W. The etiologies of chronic progressive cerebellar ataxia in a Korean population. J Clin Neurol 14, 374–380 (2018).
Bürk, K. et al. Clinical and magnetic resonance imaging characteristics of sporadic cerebellar ataxia. Arch Neuro 62, 981–985 (2005).
Lee, E. A., Cho, H. I., Kim, S. S. & Lee, W. Y. Comparison of magnetic resonance imaging in subtypes of multiple system atrophy. Parkinsonism Relat Disord 10, 363–368 (2004).
Brooks, D. J. & Seppi, K. Proposed neuroimaging criteria for the diagnosis of multiple system atrophy. Mov Disord 24, 949–964 (2009).
Christopher, W., David, P. & Amie, H. The ‘Hot Cross Bun’ sign is not always multiple system atrophy: etiologies of 11 Cases. J Mov Disord 12, 27–30 (2019).
Lee, Y. C. et al. The ‘hot cross bun’ sign in the patients with spinocerebellar ataxia. Eur J Neurol 16, 513–516 (2009).
Okamoto, K. et al. MR features of diseases involving bilateral middle cerebellar peduncles. Am J Neuroradiol 24, 1946–1954 (2003).
Takao, M., Kadowaki, T., Tomita, Y., Yoshida, Y. & Mihara, B. ‘Hot-cross bun sign’ of multiple system atrophy. Intern Med 46, 1883 (2007).
Morales, H. & Tomsick, T. Middle cerebellar peduncles: Magnetic resonance imaging and pathophysiologic correlate. World J Radiol 7, 438–447 (2015).
Diallo, A. et al. Survival in patients with spinocerebellar ataxia types 1, 2, 3, and 6 (EUROSCA): a longitudinal cohort study. Lancet Neurol 17, 327–334 (2018).
McKay, J. H. & Cheshire, W. P. First symptoms in multiple system atrophy. Clin Auton Res 28, 215–221 (2018).
Luo, L. et al. The initial symptom and motor progression in spinocerebellar ataxias. Cerebellum 16, 615–622 (2017).
Nicoletti, G. et al. MR imaging of middle cerebellar peduncle width: differentiation of multiple system atrophy from Parkinson disease. Radiology 239, 825–830 (2006).
Higashi, M. et al. A diagnostic decision tree for adult cerebellar ataxia based on pontine magnetic resonance imaging. J Neurol Sci 387, 187–195 (2018).
Horimoto, Y. et al. Longitudinal MRI study of multiple system atrophy - when do the findings appear, and what is the course? J Neurol 249, 847–854 (2002).
Watanabe, H. et al. Putaminal magnetic resonance imaging features at various magnetic field strengths in multiple system atrophy. Mov Disord 25, 1916–1923 (2010).
Brettschneider, J. et al. Progression of alpha-synuclein pathology in multiple system atrophy of the cerebellar type. Neuropathol Appl Neurobiol 43, 315–329 (2017).
Seidel, K. et al. Brain pathology of spinocerebellar ataxias. Acta Neuropathol 124, 1–21 (2012).
Hoche, F. et al. Spinocerebellar ataxia type 2 (SCA2): identification of early brain degeneration in one monozygous twin in the initial disease stage. Cerebellum 10, 245–253 (2011).
Ito, H. et al. Clinicopathologic investigation of a family with expanded SCA8 CTA/CTG repeats. Neurology 67, 1479–1481 (2006).
Klockgether, T. et al. The natural history of degenerative ataxia: a retrospective study in 466 patients. Brain 121(Pt 4), 589–600 (1998).
Tada, M., Nishizawa, M. & Onodera, O. Redefining cerebellar ataxia in degenerative ataxias: lessons from recent research on cerebellar systems. J Neurol Neurosurg Psychiatry 86, 922–928 (2015).
Wang, Y., Koh, K., Takaki, R., Shindo, K. & Takiyama, Y. Hot cross bun sign in a late-onset SCA1 patient. Neurol Sci 37, 1873–1874 (2016).
Gooneratne, I. K., Caldera, M. C., Perera, S. P. & Gamage, R. Hot cross bun sign in a patient with cerebellar ataxia. Ann Indian Acad Neurol 16, 406 (2013).
Pedroso, J. L., Rivero, R. L. & Barsottini, O. G. “Hot cross bun” sign resembling multiple system atrophy in a patient with Machado-Joseph disease. Arq Neuropsiquiatr 71, 824 (2013).
Kim, H. J. et al. Should genetic testing for SCAs be included in the diagnostic workup for MSA? Neurology 83, 1733–1738 (2014).
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
J.W.C. was involved in conception and design of the study. M.K., J.H.A., Y.C., J.S.K., and J.Y. were responsible for acquisition of data, analysis and interpretation of data. All authors were involved in drafting the article and revising it critically for important intellectual content. All authors agree that they are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, M., Ahn, J.H., Cho, Y. et al. Differential value of brain magnetic resonance imaging in multiple system atrophy cerebellar phenotype and spinocerebellar ataxias. Sci Rep 9, 17329 (2019). https://doi.org/10.1038/s41598-019-53980-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-53980-y
- Springer Nature Limited
This article is cited by
-
Radiological hints for differentiation of cerebellar multiple system atrophy from spinocerebellar ataxia
Scientific Reports (2022)
-
“Hot cross bun” is a potential imaging marker for the severity of cerebellar ataxia in MSA-C
npj Parkinson's Disease (2021)
-
Magnetic resonance T1w/T2w ratio in the middle cerebellar peduncle might be a sensitive biomarker for multiple system atrophy
European Radiology (2021)
-
Spinocerebellar ataxia type 23 (SCA23): a review
Journal of Neurology (2021)
-
Vertical pons hyperintensity and hot cross bun sign in cerebellar-type multiple system atrophy and spinocerebellar ataxia type 3
BMC Neurology (2020)