Abstract
Breast background parenchymal enhancement (BPE) is an increasingly studied MRI parameter that reflects the microvasculature of normal breast tissue, which has been shown to change during neoadjuvant chemotherapy (NAC) for breast cancer. We aimed at evaluating the BPE in patients undergoing NAC and its prognostic value to predict recurrence. MRI BPE was visually and quantitatively evaluated before and after NAC in a retrospective cohort of 102 women with unilateral biopsy-proven invasive breast cancer. Pre-therapeutic BPE was not predictive of pathological response or recurrence. Quantitative post-therapeutic BPE was significantly decreased compared to pre-therapeutic value. Post-therapeutic quantitative BPE significantly predicted recurrence (HR = 6.38 (0.71, 12.06), p < 0.05).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Neoadjuvant chemotherapy (NAC) is increasingly used in locally advanced breast cancers and in women at high risk of occult distant metastases1.
Tumor response after NAC is assessed pathologically2, but imaging biomarkers based on breast dynamic-contrast enhanced MRI (DCE-MRI), such as the size of residual enhancing tumor after NAC, have shown very promising leads for non-invasive monitoring of NAC efficacy and prediction of long-term prognosis3,4,5.
Background parenchymal enhancement – the enhancement of non-tumor breast tissue – is a promising MRI biomarker that is linked to overall breast cancer risk6,7. Its variation during NAC has been related to pathological response or patient outcome8,9.
However, the biological determinants of BPE are poorly known, except for its hormonal dependence, that accounts for the high variability of BPE in premenopausal women10.
We hypothesized that post-chemotherapy BPE would evaluate residual biological activity of the breast and help define a subgroup of patients at high risk for recurrence.
In this study, we studied the ability of post-chemotherapy BPE to predict the risk of recurrence. We used quantitative evaluation of BPE to determine if subtle changes in BPE is associated with different prognosis.
Material and Method
Population
All consecutive women with a biopsy-proven breast cancer, diagnosed between 01.01.2012 and 01.01.2017, and treated with NC plus surgery in our institution, with available pre- and post-chemotherapy MRI, were retrospectively included.
3 patients were excluded from the analysis because of missing radiology or pathology data.
On the 102 remaining patients, 84 received a combination of Epirubicine, 5-Fluoro-uracile, Cyclophosphamide, 51 received additional weekly treatment with taxane and 33 patients received Trastuzumab therapy.
Pre-operative recorded data included: patient’s age, clinical TNM stage at diagnosis, tumor pathology and molecular subtype11.
All surgical breast specimens were analyzed in the pathology department of our institution. A pathological complete response (pCR) was defined as the absence of residual invasive tumor in both the breast and the axillary nodes.
Each patient underwent post-operative annual follow-up including clinical examination, mammography, thoraco-abdominal imaging and determination of serum CA 15-3 value.
Recurrence was characterized as local (recurrence in the original tumor bed with the same histopathologic features of the primary tumor), regional (recurrence in the ipsilateral axillary, internal mammary or supraclavicular or infraclavicular nodes) or distant (metastasis in all other locations), and the length of recurrence-free survival (RFS) was defined as the time from surgery to recurrence or to the last follow-up in patients without evidence of recurrence.
MRI acquisition
All included patients underwent dynamic contrast-enhanced (DCE) MRI before treatment and after the last cycle of chemotherapy, with a mean time to surgery of 15 days (2–35).
MRI were performed on a 1.5 T unit with a dedicated breast coil (Aera, Siemens Healthcare). The standard protocol included an unenhanced axial 3D fat-suppressed T1-weighted sequence and five consecutive post-contrast series (0.1 mmol/kg of gadoteric acid, Dotarem, Guerbet, Roissy, France). The first post-contrast acquisition was centered at 45 seconds after contrast material injection, with a temporal resolution of 90 seconds, the late phase was centered at 6 minutes. Scanning parameters were: acquisition time 90 s, repetition time (TR)/echo time (TE): 4.41/1.78, flip angle 10°, field of view (FOV) 359, slice thickness 1.5 mm.
MRI visual analysis
Breast MRI were independently analyzed by two readers (SM: reader 1, IO: reader 2 with respectively 6 and 3 years of breast MRI experience). Readers were blinded to pathology data.
A radiological complete response was defined as the absence of residual tumor enhancement at the early phase. Disagreements between readers were resolved by consensus.
Both readers independently assessed MRI breast density (fibroglandular tissue, FGT) by using the four BI-RADS categories (A, B, C and D) and BPE by using the four BI-RADS categories (1 or minimal, 2 or mild, 3 or moderate, 4 or marked) on subtracted enhanced series12. A second visual evaluation of FGT and BPE was also done by reader 1 after a 6-month wash-out period. Disagreements between readers were resolved by consensus.
Inter-reader and intra-reader variability of FGT and BPE visual assessment were evaluated.
MRI quantitative analysis
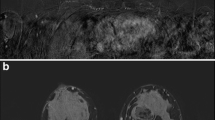
For quantitative assessment of FGT we used a semi-automated computer segmentation of the non-tumor breast on T1-weighted pre-contrast images (Fig. 1), including: i correction of field inhomogeneities using N4 method13, ii deformable registration of the postcontrast time series to the precontrast time series, using ANTS software14, iii supervised delimitation of chest wall, in consensus by two operators, and iv. segmentation of FGT by thresholding based on fuzzy C-means.
We then defined quantitative FGT as the proportion of FGT volume (VFGT) to the whole breast volume (VBreast):
For BPE quantitative assessment, we used a previously published method [15].
For each voxel, we evaluated Rvoxel, the ratio between the voxel’s intensity in the substracted image of the early phase (Ipost − Ipre) and its intensity in the pre-contrast image (Ipre):
We then calculated VBPE, the total volume of the enhancing voxels over the FGT region that had a relative difference equal to or greater than a predefined enhancement ratio threshold, that we set at 20% in accordance with previous studies15,16:
BPE20% was then defined as the proportion of this volume to the whole FGT volume.
Intrasubject change of BPE20% was defined as:
where BPE20%pre is the value of BPE on the pretherapeutic MRI, and BPE20%post the value of BPE on the post-therapeutic MRI.
Statistics
Comparisons were done using Student t test. Inter- and intra-rater reliability were assessed with Fleiss’ kappa method. Correlation between visual and quantitative FGT and BPE was analyzed with Pearson’s method.
Multivariate Cox regression was used for disease-free survival (DFS) analysis. Patients lost to follow-up were excluded from the analysis. All Cox proportional hazards results were reported as estimated hazard ratios, 95% confidence intervals and likelihood ratio test p-values. Statistical analyses were performed using Python’s module Statsmodels (https://www.statsmodels.org) and Lifelines (http://lifelines.readthedocs.io/) for DFS analysis. A statistical significance level of p = 0.05 was used throughout.
Ethics
This study received Institutional Review Board approval (Comité de Protection des Personnes Est). Informed consent was obtained for each participant and the study followed the relevant guidelines: Strengthening the Reporting of Observational Studies in Epidemiology (STROBE).
Results
Characteristics of the population
Most of the patients had intermediate- or high-grade tumors (96%), histological subtypes were either luminal (41%), HER2-enriched (31%) or basal (28%).
Most of the patients were either post-menopausal (47%) or had chemotherapy-induced amenorrhea (30%).
Pathological complete response was obtained in 27 patients.
Table 1 describes the characteristics of the population.
Inter and intra reader correlation of visual parameters
There was excellent agreement between readers for visual evaluation of FGT (kappa = 0.85, p < 0.001), but only fair agreement for visual evaluation of BPE (kappa = 0.44, p < 0.001 for pre-therapeutic imaging and kappa = 0.41, p < 0.001 for post-therapeutic imaging). Visual BPE had fair to good intra-rater reliability (kappa = 0.64, p < 0.001 for pre-therapeutic imaging, kappa = 0.50, p < 0.001 for post-therapeutic imaging).
Correlation between visual and quantitative parameters
Pre and post-therapeutic quantitative FGT were almost perfectly correlated (r = 0.98 p < 0.001).
Visual and quantitative evaluation of pretherapeutic FGT were strongly correlated (r = 0.78 p < 0.001).
Quantitative BPE was reduced on average by 37% after NAC. The decrease was higher in premenopausal women (48%).
No difference was found in pretherapeutic BPE, post-therapeutic BPE or intrasubject change in BPE across different tumor subtypes.
BPE decrease after chemotherapy was correlated with post-therapeutic quantitative BPE value (r = −0.57, p < 0.001).
Visual and quantitative evaluation of BPE were fairly correlated in the pre-therapeutic imaging (r = 0.51 p < 0.001) and weakly correlated in the post-therapeutic imaging (r = 0.27 p = 0.008).
Pathological response after NAC
Comparison between pCR and non-pCR groups is shown in Table 2.
Radiological complete response had a 93% (CI95% 83–100) sensitivity and 84% (CI95% 76–92) specificity for predicting pCR.
There was no significant difference between complete responders and non-complete responders in term of pre and post-therapeutic BPE.
Disease-free survival
Median follow-up was 37 months (15–59). One patient was lost to follow-up. During follow-up there were 2 local recurrences, 3 regional recurrences and 10 distant metastases. Recurrences were found in 1 luminal A tumor, 7 luminal B tumors, 2 HER2 tumors and 5 basal type tumors (Table 3).
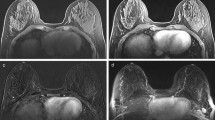
Multivariate Cox analysis (Table 4) showed a strong positive association between the quantitative post-therapeutic BPE and the risk of recurrence (HR = 6.38 (0.71, 12.06) p < 0.05). An illustration of these findings by a case is shown in Fig. 2.
Breast MRI of a 45-year-old premenopausal woman with grade II luminal B HER 2 positive cT2N2 ductal invasive carcinoma relapsing with bone metastases 2 years after surgery, despite having achieved pathological complete response with neoadjuvant chemotherapy. Pretherapeutic MRI (A) showed a 5 cm tumor in the left breast (arrowheads) and moderate BPE, as evaluated in the right breast (arrows). Post-therapeutic MRI (B) showed no residual enhancing tumor in the left breast (star) with persisting BPE (arrows).
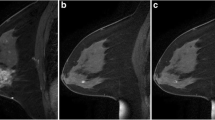
Other parameters significantly linked to local or distant recurrence risk were: mastitis at presentation, high tumor grade, HER2 negativity, larger residual enhancing tumor diameter on post-therapeutic MRI (Fig. 3).
Discussion
Post-therapeutic quantitative BPE is an independent predictor of recurrence after NAC.
This results broaden other published results, that have shown BPE to be linked with breast cancer risk16,15, breast metabolic activity17, as well as therapeutic response8,18,19,20.
The precise physiology of BPE is poorly known. While independent of breast density, it has been shown to be highly dependent on the breast fibroglandular tissue’s exposure to hormones21. Chemotherapy frequently causes transient or permanent ovarian failure, which may have per se a therapeutic effect on hormone-positive breast cancer [22]22.
In our study, amenorrhea was noted in 77% of the premenopausal women undergoing post-NAC MRI, but the degree of ovarian suppression probably differs between patients. BPE could thus give quantitative information about chemotherapy-induced ovarian suppression in premenopausal women16, as well as residual baseline estrogen levels in post-menopausal women23.
Ultimately, the intensity of BPE ultimately reflects the density and permeability of blood and lymphatic vessels in the breast, which are known to be important cofactors of breast tumor growth, angiogenesis, metastasis and immune response24,25,26. Along with their cytotoxic effect on tumor cells, chemotherapy drugs have a direct effect on endothelial cells18, thus decreasing BPE. This decrease may be an indirect marker of chemotherapy action.
Quantitative assessment of BPE allows an objective appreciation of the microvasculature of the breast gland and is able to highlight subtle changes of vessels density and permeability, not easily detected by visual evaluation27, especially in the post-chemotherapy setting.
In fact, there was only moderate correlation between quantitative and visual evaluation. This is certainly explained by the significant inter-observer variability of the 4-grade visual assessment, the presence of motion-induced artifacts (as the visual evaluation was done on subtracted uncorrected series)28 as well as the difficulty to visually assess BPE in mainly fatty breasts.
Tumor immunohistochemical subtype was not significantly linked to pretherapeutic or post-therapeutic quantitative BPE, nor BPE change under chemotherapy.
On the contrary to a previous study18, no influence of tumor subtype on the prognostic value of BPE could be showed in this study, because of the small number of events in each subtype (1 for luminal A tumor, 7 for luminal B, 2 for HER2 and 5 for basal).
While our results tend to demonstrate the potentially general prognostic value of quantitative BPE evaluation, larger studies are needed to determine in which oncological situations quantitative BPE could be an optimal biomarker.
Interestingly, we did not find any significant change in the breast density after NAC. While a decrease of breast density after chemoprevention has been reported in many publications, change in breast density after chemotherapy has rarely been studied29,30, only one study described a decrease in breast density (in average a 10–12% decrease) following neoadjuvant chemotherapy29. Some explanations can be raised. First, we did not exclude post-menopausal women and women with extremely fatty breast from the analysis, while hese women – roughly half of the study population - are highly unlikely to have a significant hormonal-related decrease in breast density. Furthermore, the change in breast density implies extensive histological changes, with fatty involution of the glandular tissue, which may take more time than the usual 4 months between pretherapeutic and post-therapeutic MRI, on the contrary to BPE which has proved to be changing on a much faster pace, during the menstruation cycle21.
We found that quantitative BPE was correlated to disease-free survival, independently of pCR. Pathological complete response did not reach statistical significance, probably due to a small number of events during the follow-up period.
Chen et al.18 showed a correlation between pathological response and early decreased of BPE after one cycle of NAC in an ER-negative subgroup. In fact, we chose to evaluate BPE at the end of the treatment, as routinely done in our institution, which may not bring the same information.
In the setting of neoadjuvant chemotherapy, the prognostic value of an imaging biomarker, independently of pathological complete response, has already been published, for MRI-assessed post-therapeutic functional tumor volume5.
Our study highlights the role of quantitative MRI assessment of normal breast tissue’s microvascularization after chemotherapy for predicting prognosis, independently of pathological analysis of the tumor. If validated by larger studies, this biomarker could in particular prove useful to tailor the type and the rate of post-operative follow-up.
Our study has limitations, among them the retrospective nature of this study, the relatively small population size and the limited number of events during the follow-up period. The inhomogeneity of treatment, with some patients having adjuvant hormone therapy during the follow-up period, may also limit the extrapolation of the results and should prompt further larger prospective studies.
Conclusion
Quantitative BPE is strongly linked to recurrence after NAC. More biological and clinical studies are needed to compare the different measurement methods with their underlying pathophysiological mechanisms.
References
Kaufmann, M. et al. Recommendations from an international consensus conference on the current status and future of neoadjuvant systemic therapy in primary breast cancer. Ann. Surg. Oncol. 19, 1508–1516 (2012).
Cortazar, P. et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet Lond. Engl. 384, 164–172 (2014).
Abramson, R. G. et al. Methods and Challenges in Quantitative Imaging Biomarker Development. Acad. Radiol. 22, 25–32 (2015).
Lobbes, M. B. I. et al. The role of magnetic resonance imaging in assessing residual disease and pathologic complete response in breast cancer patients receiving neoadjuvant chemotherapy: a systematic review. Insights Imaging 4, 163–175 (2013).
Hylton, N. M. et al. Neoadjuvant Chemotherapy for Breast Cancer: Functional Tumor Volume by MR Imaging Predicts Recurrence-free Survival-Results from the ACRIN 6657/CALGB 150007 I-SPY 1 TRIAL. Radiology 279, 44–55 (2016).
King, V. et al. Background parenchymal enhancement at breast MR imaging and breast cancer risk. Radiology 260, 50–60 (2011).
Dontchos, B. N. et al. Are Qualitative Assessments of Background Parenchymal Enhancement, Amount of Fibroglandular Tissue on MR Images, and Mammographic Density Associated with Breast Cancer Risk? Radiology 276, 371–380 (2015).
Hattangadi, J. et al. Breast Stromal Enhancement on MRI Is Associated with Response to Neoadjuvant Chemotherapy. Am. J. Roentgenol. 190, 1630–1636 (2008).
Preibsch, H. et al. Background parenchymal enhancement in breast MRI before and after neoadjuvant chemotherapy: correlation with tumour response. Eur. Radiol. 26, 1590–1596 (2016).
Pujara, A. C. et al. Comparison between qualitative and quantitative assessment of background parenchymal enhancement on breast MRI. J. Magn. Reson. Imaging JMRI, https://doi.org/10.1002/jmri.25895 (2017).
Perou, C. M. et al. Molecular portraits of human breast tumours. Nature 406, 747–752 (2000).
Morris, E., Comstock, C. & Lee, C. ACR BI-RADS Magnetic Resonance Imaging. in ACR BI-RADS® Atlas, Breast Imaging Reporting and Data System (2013).
Tustison, N. J. et al. N4ITK: improved N3 bias correction. IEEE Trans. Med. Imaging 29, 1310–1320 (2010).
Avants, B. B., Epstein, C. L., Grossman, M. & Gee, J. C. Symmetric diffeomorphic image registration with cross-correlation: evaluating automated labeling of elderly and neurodegenerative brain. Med. Image Anal. 12, 26–41 (2008).
Wu, S. et al. DCE-MRI Background Parenchymal Enhancement Quantified from an Early versus Delayed Post-contrast Sequence: Association with Breast Cancer Presence. Sci. Rep. 7, 2115 (2017).
Wu, S. et al. Quantitative assessment of background parenchymal enhancement in breast MRI predicts response to risk-reducing salpingo-oophorectomy: preliminary evaluation in a cohort of BRCA1/2 mutation carriers. Breast Cancer Res. 17, 67 (2015).
Mema, E. et al. Does breast MRI background parenchymal enhancement indicate metabolic activity? Qualitative and 3D quantitative computer imaging analysis. J. Magn. Reson. Imaging JMRI 47, 753–759 (2018).
Chen, J. H. et al. Background Parenchymal Enhancement of the Contralateral Normal Breast: Association with Tumor Response in Breast Cancer Patients Receiving Neoadjuvant Chemotherapy. Transl. Oncol. 8, 204–209 (2015).
van der Velden, B. H. M., Dmitriev, I., Loo, C. E., Pijnappel, R. M. & Gilhuijs, K. G. A. Association between Parenchymal Enhancement of the Contralateral Breast in Dynamic Contrast-enhanced MR Imaging and Outcome of Patients with Unilateral Invasive Breast Cancer. Radiology 276, 675–685 (2015).
Olshen, A. et al. Features of MRI stromal enhancement with neoadjuvant chemotherapy: a subgroup analysis of the ACRIN 6657/I-SPY TRIAL. J. Med. Imaging Bellingham Wash 5, 011014 (2018).
Kuhl, C. K. et al. Healthy premenopausal breast parenchyma in dynamic contrast-enhanced MR imaging of the breast: normal contrast medium enhancement and cyclical-phase dependency. Radiology 203, 137–144 (1997).
Zhou, Q., Yin, W., Du, Y., Shen, Z. & Lu, J. Prognostic impact of chemotherapy-induced amenorrhea on premenopausal breast cancer: a meta-analysis of the literature. Menopause N. Y. N 22, 1091–1097 (2015).
Brooks, J. D. et al. MRI background parenchymal enhancement, breast density and serum hormones in postmenopausal women. Int. J. Cancer, https://doi.org/10.1002/ijc.31370 (2018).
Artacho-Cordón, A., Artacho-Cordón, F., Ríos-Arrabal, S., Calvente, I. & Núñez, M. I. Tumor microenvironment and breast cancer progression. Cancer Biol. Ther. 13, 14–24 (2012).
Kang, D. K., Kim, E. J., Kim, H. S., Sun, J. S. & Jung, Y. S. Correlation of whole-breast vascularity with ipsilateral breast cancers using contrast-enhanced MDCT. AJR Am. J. Roentgenol. 190, 496–504 (2008).
Karaman, S. & Detmar, M. Mechanisms of lymphatic metastasis. J. Clin. Invest. 124, 922–928 (2014).
Kim, J. Y. et al. Enhancement parameters on dynamic contrast enhanced breast MRI: do they correlate with prognostic factors and subtypes of breast cancers? Magn. Reson. Imaging 33, 72–80 (2015).
You, C. et al. The Assessment of Background Parenchymal Enhancement (BPE) in a High-Risk Population: What Causes BPE? Transl. Oncol. 11, 243–249 (2018).
Chen, J.-H. et al. Decrease in breast density in the contralateral normal breast of patients receiving neoadjuvant chemotherapy: MR imaging evaluation. Radiology 255, 44–52 (2010).
Toledano, A. et al. Concurrent administration of adjuvant chemotherapy and radiotherapy after breast-conserving surgery enhances late toxicities: long-term results of the ARCOSEIN multicenter randomized study. Int. J. Radiat. Oncol. Biol. Phys. 65, 324–332 (2006).
Author information
Authors and Affiliations
Contributions
S.M. and I.O. wrote the manuscript, gathered and analysed the clinical and imaging data. V.N. did the MRI quantitative study and statistical analysis. F.V. and C.M. reviewed the manuscript and suggested significant changes.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Moliere, S., Oddou, I., Noblet, V. et al. Quantitative background parenchymal enhancement to predict recurrence after neoadjuvant chemotherapy for breast cancer. Sci Rep 9, 19185 (2019). https://doi.org/10.1038/s41598-019-55820-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-55820-5
- Springer Nature Limited
This article is cited by
-
Predictive value of background parenchymal enhancement on breast magnetic resonance imaging for pathological tumor response to neoadjuvant chemotherapy in breast cancers: a systematic review
Cancer Imaging (2024)
-
Background Parenchymal Enhancement on Breast MRI: Assessment and Clinical Implications
Current Radiology Reports (2021)