Abstract
Whole brain radiotherapy (WBRT) is a mainstay of the treatment for brain metastases (BM). We evaluated prognostic factors in colorectal cancer (CRC) patients undergoing WBRT for BM. The medical records of 106 CRC patients undergoing WBRT for BM between 2000 and 2014 at three institutions were reviewed. Patient and tumor factors were analyzed to identify the prognostic factors for overall survival (OS) calculated from the date of BM diagnosis to the date of death or last follow-up. Surgical resection of BM was performed in six patients. The dose of WBRT was 30 Gy, and boost radiotherapy or stereotactic radiosurgery (8–23 Gy) was given to 15 patients. Systemic therapy for BM was administered in one patient before WBRT and 26 patients after WBRT. The median follow-up time was 3.9 months (range, 0.4–114.1 months). The median OS time was 3.9 months, and the 1-year OS rate was 18.2%. Older age (>65 years), multiple BM (≥3), elevated level of carcinoembryonic antigen (CEA, >5 ng/ml) at BM diagnosis, and extracranial metastases were adverse prognostic factors for OS. Patient with 0–1 factor showed better OS (at 1 year, 76.9%) than patients with 2 factors (16.7%) or 3–4 factors (4.2%; p < 0.001). In conclusion, we evaluated age, the number of BM, CEA level, and extracranial metastases as the prognostic factors for OS in CRC patients undergoing WBRT. Our result might be useful to develop prognostic models predicting survival for patients whom WBRT is intended for.
Similar content being viewed by others
Introduction
According to population-based studies, about 20% of patients with colorectal cancer (CRC) have distant-stage disease1. Liver and lung metastases are most frequent, found in about 20% to 30% of CRC patients, including those with synchronous and metachronous disease2,3. Brain metastasis (BM) rarely occurs in CRC patients, with an incidence of less than 3%4,5,6, but the prognosis is poor. The median survival time is less than 6 months after the diagnosis of BM4,7. Retrospective studies have reported young age, a single BM lesion, the absence of extracranial metastases, and a lower level of carcinoembryonic antigen (CEA) as good prognostic factors in CRC patients with BM4,7,8.
Validated prognostic tools that classify BM patients according to their predicted survival times can offer useful guidance for BM treatment. The Radiation Therapy Oncology Group (RTOG) performed recursive partitioning analysis (RPA) using a database from consecutive RTOG trials and suggested three classes based on performance status, age, primary tumor status, and extracranial metastases9. The Graded Prognostic Assessment (GPA) is the updated diagnosis-specific prognostic index, which includes additional prognostic factors such as number of BM and intrinsic subtypes of breast cancers10. CRC is not specified as a separate cancer subtype but is combined with other gastrointestinal (GI) cancers in the GPA indices. In this context, we previously presented a novel CRC-specific GPA index for patients with CRC who received any kind of BM treatment11. The novel index included number of BM, CEA level, and the presence or absence of neurologic symptoms11.
In terms of the treatment for BM, whole brain radiotherapy (WBRT) is a traditional and essential modality. In randomized clinical trials, more than 60% of BM patients treated with WBRT have shown complete or partial responses12. Although more aggressive local treatment modalities such as surgical resection or stereotactic radiosurgery (SRS) have been actively employed in recent decades for patients with good performance status and a limited number of BM, the addition of WBRT can reduce intracranial relapses and neurologic deaths13. However, prognostic factors in CRC patients undergoing WBRT for BM have not been evaluated in prospective trials as well as large retrospective studies. In this study, we aim to evaluate prognostic factors which could attribute to develop prognostic tools in CRC patients undergoing WBRT for BM.
Results
Participating institutions collected 113 patients who received WBRT for BM from CRC. We excluded five patients who were lost to follow-up, one patient who underwent WBRT for pachymeningeal metastasis, and one patient with insufficient information of BM. Finally, 106 patients were included in this study. When the primary tumor was diagnosed, 10 patients (9.4%) had BM and 33 patients (31.1%) had systemic disease other than BM. The median time interval between the diagnosis of primary tumor and BM was 22 months (range, 0–132 months). The median age at the time of BM diagnosis was 62 years (range, 31–85). Regarding the number of BM, 41 patients (38.7%) had a single BM, 21 (19.8%) had two lesions, 14 (13.2%) had three lesions, and 30 (28.3%) had four or more lesions. The median CEA level at the time of BM diagnosis was 29.75 ng/dL (range, 0.5–7,620 ng/dL). Neurologic symptoms were noted in 71 patients (67.0%): motor weakness (n = 45), gait disturbance (n = 17), seizure (n = 17), dysarthria (n = 14), disorientation (n = 10), and other symptoms (n = 8). Extracranial metastases were found in 86 patients (81.1%): the lungs (n = 68), lymph nodes (n = 47), liver (n = 44), bones (n = 31), and other locations (n = 26). Details of patient and tumor characteristics are summarized in Table 1.
Surgical resection of BM was performed in six patients (5.7%). The dose of WBRT was 30 Gy. Of the patients, 13 received boost radiotherapy (8 Gy to 20 Gy), and two patients underwent SRS (18 Gy and 23 Gy). One patient received systemic therapy, regorafenib, for BM before WBRT. After WBRT, systemic chemotherapy was administered in 26 patients: 11 patients received 5-fluorouracil (5-FU)- and oxaliplatin-based regimens, while the following three approaches were used in five patients each: 5-FU- and irinotecan-based, capecitabine-based, and other regimens.
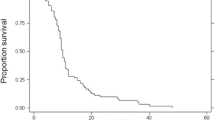
The median follow-up time was 3.9 months (range, 0.4–114.1 months). Overall, 103 patients (97.2%) died, two patients were alive, and one patient was lost to follow-up. The median overall survival (OS) time was 3.9 months, and OS rates were 28.7% at 6 months and 18.2% at 1 year (Fig. 1). Follow-up imaging studies were performed in 39 patients after WBRT. Complete response was observed in 4 patients, partial response in 9, stable disease in 11, and progressive disease in 15. Regarding salvage treatments, SRS (median 18 Gy; range, 15–22 Gy) was administered in seven patients, surgical resection and SRS (17 Gy and 18 Gy) in two patients, and WBRT (20 Gy) in another three patients.
Table 2 lists the results of the univariate analyses. Age, presence of neurologic symptoms, number of BM, presence of extracranial metastases, and level of CEA showed statistical significances. After adjusting for covariates, age, number of BM, presence of extracranial metastases, and level of CEA were confirmed as prognostic factors (Table 2). Patients with age > 65 years, BM ≥ 3, CEA level > 5 ng/mL, or extracranial metastases had lower OS (Fig. 2).
We classified patients as 3 risk groups according to the prognostic factors: high-risk (3–4 factors; n = 13), intermediate-risk (2 factors; n = 36), and low-risk (0–1 factor; n = 49) groups. The patients who had missing data of CEA level were excluded. The risk groups showed statistically significant differences in OS (Fig. 3). At 1 year, OS rates were 76.9% for low-risk group, 16.7% for intermediate-risk group, and 4.2% for high-risk group (p < 0.001).
Discussion
With the improvement in survival outcomes and imaging studies, the incidence of BM is increasing in patients with cancer. WBRT has been the most common treatment modality for BM. Although the benefit of SRS for patients with a limited number of BM has been emphasized14,15, WBRT still has an important role in the treatment of patients unsuitable for SRS or surgery, in the management of multiple BM16, and as adjuvant treatment after SRS or surgery13,17.
Traditionally, the homogeneous distribution of radiation dose to the entire brain has been the preferred WBRT plan. The hippocampi are associated with the formation and recall of new memories, and radiation to the hippocampus induces neural precursor cell dysfunction by the ablation of hippocampal neurogenesis18. Even a relatively modest radiation dose to the bilateral hippocampi can induce neurocognitive function impairment19. To decrease the dose of radiation to the hippocampal area, hippocampal-avoidance WBRT (HA-WBRT) was introduced. The RTOG performed a phase II trial (RTOG 0933) examining memory preservation using the HA-WBRT technique. Compared with historical data, patients undergoing HA-WBRT had significantly lower memory decline20.
Despite the dosimetric benefit of HA-WBRT, its high cost remains the biggest hurdle working against the wide use of this technique. In Korea, the medical cost was more than $4,000 U.S. for 10 fractions of HA-WBRT, but less than $1,000 U.S. for 10 fractions of conventional WBRT. Administration of HA-WBRT could be justified in patients with longer expected survival because the technique can mitigate neurocognitive dysfunction after WBRT. In patients with short life expectancy, however, conventional WBRT may have a better rationale considering its shorter planning time and lower cost21.
Within the context of the cost-effectiveness of WBRT techniques, prognosis prediction becomes even more important. By predicting the prognosis of these patients, we could then elaborate WBRT planning and intensify systemic treatments, or save excessive medical costs and concentrate on supportive care. However, due to the rareness of disease condition, even prognostic factors in CRC patients undergoing WBRT for BM have not been clearly evaluated.
Actually, previously reported prognostic indices, such as the RPA and GPA, are not specific for primary sites9,22. Although the GPA was refined to be diagnosis-specific, CRC was not appreciated as a separate entity but instead included with GI cancers, where the only prognostic factor is Karnofsky Performance Score (KPS)10,23. In fact, when applied to the current cohort, the RPA, GPA, and diagnosis-specific GPA were incapable of patient stratification, with p-values exceeding 0.05 (data not shown).
In this study, we revealed four prognostic factors for OS in patients with BM from CRC: age, level of CEA at BM, presence of extracranial metastases, and number of BM. Our results would be useful to develop prognostic models predicting survival for patients whom WBRT is intended for.
Patients’ performance status is the weighted prognostic factor in the RPA and GPA, but not in our study. Differences between KPS’s and Eastern Cooperative Oncology Group (ECOG)’s scoring systems could be a source of potential bias. We used the ECOG grading system, but the RPA and GPA used the KPS, which has a more subdivided scale. A KPS of 70 to 90 could be regarded as grade 1 under the ECOG Performance Status, but the GPA system scores different points for a KPS of 70, 80, or 90. The subjective aspect of performance evaluation might be another source of bias because data was retrospectively reviewed by investigators from different institutions. Although performance status has not always been reported as a prognostic factor in other studies of BM from CRC4,8,24,25,26,27,28,29,30, a prospective study needs to confirm its prognostic value.
The number of BM is another well-known prognostic factor for patients with BM. This factor, as in fewer than four BM, is incorporated in the diagnosis-specific GPA for lung cancer, melanoma, and renal cell carcinoma10,23. Practically speaking, the number of BM affects choice of treatment. For patients with a limited number of BM, SRS is preferred over WBRT alone14,15,17. The prognostic value of the number of BM has also been reported in previous studies of BM from CRC, though the studies were retrospectively designed because of the rareness of the situation30. In this context, we tried to perform log-rank tests for each BM number and found statistical significance with a cut-off of three lesions (Table 2).
The fact that patients with a limited number of BM received WBRT rather than SRS in our study could be a point of discussion. The volume effect, which we did not examine, might be an underlying cause. Like BM number, larger cumulative BM volume is regarded as a poor prognostic factor31. We could presume that our patients with a limited BM number might have had a relatively large BM volume because only eight patients underwent surgical resection or SRS rather than WBRT alone. It would be clinically meaningful if a prognostic index encompassing cumulative BM volume were investigated.
CEA level is one of the most useful tumor markers for CRC. An elevation means a larger entire tumor burden and higher probability of metastatic disease, as CEA-related cell adhesion molecules participate in tumor progression, angiogenesis, and metastasis32. CEA level can be used to estimate prognosis and monitor recurrence after complete CRC resection33. The association of CEA level with survival has also been confirmed in patients with metastatic CRC34,35. Particularly in patients with BM from CRC, a higher level of CEA was associated with lower survival rates8,29.
As expected, an elevated level of CEA at the time of BM diagnosis was associated with lower OS in this study. CEA elevation may be a surrogate for systemic tumor burden. Thus, the significance of extracranial metastases was tested, and, as expected, these metastases were found to be prognostic. Extracranial metastases were more commonly found in patients with an elevated CEA level than in patients with a CEA level within normal range (94.6% vs. 54.2%; p < 0.001). Despite the correlation, both elevated CEA level and presence of extracranial metastases independently showed prognostic significance in the multivariate analysis.
Based on four prognostic factors, we classified the patients as 3-tier risk groups and confirmed significant differences in OS. Our results are needed to be validated in prospective trials or large population studies. Another limitation of this study was the insufficient imaging information before and after WBRT for the evaluation of treatment response. This limitation represents an inherent restriction of retrospective, multicenter studies conducted for a rare disease.
In conclusion, age, level of CEA at BM, presence of extracranial metastases, and number of BM were evaluated to be prognostic factors for OS in CRC patients undergoing WBRT for BM. The prognostic factors might be helpful to develop prognostic models selecting patients who are expected to have a good prognosis. Further validation using larger independent cohorts is needed.
Methods
Three Korean institutions participated in this study after receiving approval from the institutional review board of Seoul National University Hospital, Ewha Womans University Mokdong Hospital, and Hallym University Sacred Heart Hospital. All procedures performed in studies were in accordance with the ethical standards of the institutional review board of each hospital and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Also, a waiver of informed consent was approved by each institutional review board.
Included in the study were CRC patients who underwent WBRT for BM diagnosed by imaging studies between 2000 and 2014. Magnetic resonance imaging (MRI) or computerized tomography (CT) was used for the imaging diagnosis. Patients with multiple primary cancers were excluded. The collected data included patient characteristics, clinicopathological information about primary tumors and BM lesions, and details of treatment for BM. Regarding the location of the primary tumor, ascending and transverse colon cancers were classified as right CRC, and descending colon, sigmoid colon, and rectal cancers as left CRC. Patient performance at BM diagnosis was graded using the ECOG performance scale. Motor weakness, gait disturbance, disorientation, dysarthria, visual disturbance, consciousness change, and seizure were recorded as neurologic symptoms11.
Statistical analyses were performed using Predictive Analytics Software, version 18.0 (SAP America Inc., Newtown Square, PA, USA). We calculated OS from the date of BM diagnosis to the date of death or last follow-up. The Kaplan–Meier method was used to estimate the OS rates and plot survival curves. Univariate and multivariate analyses were performed using the log-rank test and Cox proportional hazards model, respectively. The multivariate analyses included variables that showed a p value < 0.05 in the univariate analyses.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available due to personal information protection act in Republic of Korea.
References
Siegel, R. L. et al. Colorectal cancer statistics, 2017. CA Cancer J. Clin. 67, 177–193, https://doi.org/10.3322/caac.21395 (2017).
Manfredi, S. et al. Epidemiology and management of liver metastases from colorectal cancer. Ann. Surg. 244, 254–259, https://doi.org/10.1097/01.sla.0000217629.94941.cf (2006).
Mitry, E. et al. Epidemiology, management and prognosis of colorectal cancer with lung metastases: a 30-year population-based study. Gut. 59, 1383–1388, https://doi.org/10.1136/gut.2010.211557 (2010).
Jung, M. et al. Brain metastases from colorectal carcinoma: prognostic factors and outcome. J. Neurooncol 101, 49–55, https://doi.org/10.1007/s11060-010-0214-9 (2011).
Barnholtz-Sloan, J. S. et al. Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the Metropolitan Detroit Cancer Surveillance System. J. Clin. Oncol. 22, 2865–2872, https://doi.org/10.1200/JCO.2004.12.149 (2004).
Schouten, L. J., Rutten, J., Huveneers, H. A. & Twijnstra, A. Incidence of brain metastases in a cohort of patients with carcinoma of the breast, colon, kidney, and lung and melanoma. Cancer 94, 2698–2705 (2002).
Farnell, G. F. et al. Brain metastases from colorectal carcinoma. The long term survivors. Cancer 78, 711–716, doi:10.1002/(SICI)1097-0142(19960815)78:4<711::AID-CNCR3>3.0.CO;2-H (1996).
Noura, S. et al. Brain metastasis from colorectal cancer: prognostic factors and survival. J. Surg. Oncol. 106, 144–148, https://doi.org/10.1002/jso.23055 (2012).
Gaspar, L. et al. Recursive partitioning analysis (RPA) of prognostic factors in three Radiation Therapy Oncology Group (RTOG) brain metastases trials. Int. J. Radiat. Oncol. Biol. Phys. 37, 745–751 (1997).
Sperduto, P. W. et al. Summary report on the graded prognostic assessment: an accurate and facile diagnosis-specific tool to estimate survival for patients with brain metastases. J. Clin. Oncol. 30, 419–425, https://doi.org/10.1200/JCO.2011.38.0527 (2012).
Kim, B. H. et al. Novel graded prognostic assessment for colorectal cancer patients with brain metastases. Int. J. Clin. Oncol. 23, 1112–1120, https://doi.org/10.1007/s10147-018-1329-9 (2018).
Khuntia, D., Brown, P., Li, J. & Mehta, M. P. Whole-brain radiotherapy in the management of brain metastasis. J. Clin. Oncol. 24, 1295–1304, https://doi.org/10.1200/JCO.2005.04.6185 (2006).
Kocher, M. et al. Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952-26001 study. J. Clin. Oncol. 29, 134–141, https://doi.org/10.1200/JCO.2010.30.1655 (2011).
Aoyama, H. et al. Stereotactic radiosurgery plus whole-brain radiation therapy vs stereotactic radiosurgery alone for treatment of brain metastases: a randomized controlled trial. JAMA 295, 2483–2491, https://doi.org/10.1001/jama.295.21.2483 (2006).
Brown, P. D. et al. Effect of Radiosurgery Alone vs Radiosurgery With Whole Brain Radiation Therapy on Cognitive Function in Patients With 1 to 3 Brain Metastases: A Randomized Clinical Trial. JAMA 316, 401–409, https://doi.org/10.1001/jama.2016.9839 (2016).
Brown, P. D. et al. Whole-Brain Radiotherapy for Brain Metastases: Evolution or Revolution? J. Clin. Oncol. 36, 483–491, https://doi.org/10.1200/JCO.2017.75.9589 (2018).
Aoyama, H., Tago, M. & Shirato, H. & Japanese Radiation Oncology Study Group, I. Stereotactic Radiosurgery With or Without Whole-Brain Radiotherapy for Brain Metastases: Secondary Analysis of the JROSG 99-1 Randomized Clinical Trial. JAMA Oncol. 1, 457–464, https://doi.org/10.1001/jamaoncol.2015.1145 (2015).
Monje, M. L., Mizumatsu, S., Fike, J. R. & Palmer, T. D. Irradiation induces neural precursor-cell dysfunction. Nat. Med. 8, 955–962, https://doi.org/10.1038/nm749 (2002).
Gondi, V., Hermann, B. P., Mehta, M. P. & Tome, W. A. Hippocampal dosimetry predicts neurocognitive function impairment after fractionated stereotactic radiotherapy for benign or low-grade adult brain tumors. Int. J. Radiat. Oncol. Biol. Phys. 83, e487–493, https://doi.org/10.1016/j.ijrobp.2011.10.021 (2012).
Gondi, V. et al. Preservation of memory with conformal avoidance of the hippocampal neural stem-cell compartment during whole-brain radiotherapy for brain metastases (RTOG 0933): a phase II multi-institutional trial. J. Clin. Oncol. 32, 3810–3816, https://doi.org/10.1200/JCO.2014.57.2909 (2014).
Savitz, S. T., Chen, R. C. & Sher, D. J. Cost-effectiveness analysis of neurocognitive-sparing treatments for brain metastases. Cancer 121, 4231–4239, https://doi.org/10.1002/cncr.29642 (2015).
Sperduto, P. W., Berkey, B., Gaspar, L. E., Mehta, M. & Curran, W. A new prognostic index and comparison to three other indices for patients with brain metastases: an analysis of 1,960 patients in the RTOG database. Int. J. Radiat. Oncol. Biol. Phys. 70, 510–514, https://doi.org/10.1016/j.ijrobp.2007.06.074 (2008).
Sperduto, P. W. et al. Diagnosis-specific prognostic factors, indexes, and treatment outcomes for patients with newly diagnosed brain metastases: a multi-institutional analysis of 4,259 patients. Int. J. Radiat. Oncol. Biol. Phys. 77, 655–661, https://doi.org/10.1016/j.ijrobp.2009.08.025 (2010).
Matsunaga, S. et al. Gamma Knife surgery for brain metastases from colorectal cancer. Clinical article. J. Neurosurg. 114, 782–789, https://doi.org/10.3171/2010.9.JNS10354 (2011).
Nieder, C., Pawinski, A. & Balteskard, L. Colorectal cancer metastatic to the brain: time trends in presentation and outcome. Oncol. 76, 369–374, https://doi.org/10.1159/000210026 (2009).
Pietrantonio, F. et al. A new nomogram for estimating survival in patients with brain metastases secondary to colorectal cancer. Radiother. Oncol. 117, 315–321, https://doi.org/10.1016/j.radonc.2015.08.023 (2015).
Hammoud, M. A., McCutcheon, I. E., Elsouki, R., Schoppa, D. & Patt, Y. Z. Colorectal carcinoma and brain metastasis: distribution, treatment, and survival. Ann. Surg. Oncol. 3, 453–463 (1996).
Wronski, M. & Arbit, E. Resection of brain metastases from colorectal carcinoma in 73 patients. Cancer 85, 1677–1685 (1999).
Kye, B. H. et al. Brain metastases from colorectal cancer: the role of surgical resection in selected patients. Colorectal Dis. 14, e378–385, https://doi.org/10.1111/j.1463-1318.2012.02962.x (2012).
Go, P. H., Klaassen, Z., Meadows, M. C. & Chamberlain, R. S. Gastrointestinal cancer and brain metastasis: a rare and ominous sign. Cancer 117, 3630–3640, https://doi.org/10.1002/cncr.25940 (2011).
Emery, A. et al. More than Just the Number of Brain Metastases: Evaluating the Impact of Brain Metastasis Location and Relative Volume on Overall Survival After Stereotactic Radiosurgery. World Neurosurg. 99, 111–117, https://doi.org/10.1016/j.wneu.2016.11.119 (2017).
Beauchemin, N. & Arabzadeh, A. Carcinoembryonic antigen-related cell adhesion molecules (CEACAMs) in cancer progression and metastasis. Cancer Metastasis Rev. 32, 643–671, https://doi.org/10.1007/s10555-013-9444-6 (2013).
Herrera, M. A., Chu, T. M. & Holyoke, E. D. Carcinoembryonic antigen (CEA) as a prognostic and monitoring test in clinically complete resection of colorectal carcinoma. Ann. Surg. 183, 5–9 (1976).
Giacchetti, S. et al. Long-term survival of patients with unresectable colorectal cancer liver metastases following infusional chemotherapy with 5-fluorouracil, leucovorin, oxaliplatin and surgery. Ann. Oncol. 10, 663–669 (1999).
Regnard, J. F. et al. Surgical treatment of hepatic and pulmonary metastases from colorectal cancers. Ann Thorac Surg 66, 214–218; discussion 218–219 (1998).
Author information
Authors and Affiliations
Contributions
Kyubo Kim and Eui Kyu Chie designed the study. Taeryool Koo, Kyubo Kim, Hae Jin Park, Sae-Won Han, Tae-You Kim, Seung-Yong Jeong, Kyu Joo Park, Eui Kyu Chie reviewed medical records and collected data. Taeryool Koo analyzed data and wrote the first draft. Kyubo Kim and Eui Kyu Chie contributed to revise the draft. All authors read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Koo, T., Kim, K., Park, H.J. et al. Prognostic factors for survival in colorectal cancer patients with brain metastases undergoing whole brain radiotherapy: multicenter retrospective study. Sci Rep 10, 4340 (2020). https://doi.org/10.1038/s41598-020-61354-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-61354-y
- Springer Nature Limited
This article is cited by
-
Frequency of seizures in patients with metastatic brain tumors
Neurological Sciences (2023)