Abstract
The C-repeat binding factor (CBF)/dehydration-responsive element binding (DREB1) proteins play a prominent role in freezing tolerance and are highly conserved in higher plants. Here we performed a genome-wide search of the CBF/DREB1 gene family in lettuce (Lactuca sativa L.) and identified 14 members of the family with one member gene containing a non-sense mutation within the AP2 DNA-binding domain. A comprehensive phylogenetic analysis of the CBF/DREB1 family members in 20 plant species from the Asterid or Rosid clade provided evidence that tandem duplication played an important role in the expansion of the CBF/DREB1 family. Expression analysis showed that twelve of the lettuce CBF genes were responsive to low temperature (4 °C), and that three and six of them could also be responsive to salt and heat stresses, respectively. Unlike Arabidopsis thaliana whose members of the CBF/DREB1 family respond only to a particular stress, lettuce CBFs provide wider protection from combinations of abiotic stresses. A global transcriptome analysis revealed distinctive temporal expression patterns among the cold-regulated genes in lettuce plants exposed to low temperature. Genes induced throughout the cold treatment are enriched in functions associated with protection from UV and high-light intensity and the genes suppressed after 7 days of cold exposure are enriched in photosynthesis-associated functions. These results provide insight into the molecular evolutionary properties of the CBF/DREB1 gene family in lettuce and a reference for genetic improvement of the lettuce response to cold acclimation.
Similar content being viewed by others
Introduction
Freezing stress is a primary environmental factor that limits productivity and growth of plants. Many plants in temperate regions show an increase in freezing tolerance after exposure to low, non-freezing temperatures, a phenomenon called cold acclimation. The C-repeat binding factors (CBFs)/dehydration-responsive element binding (DREB1) proteins have been identified as key transcription factors in cold acclimation. In Arabidopsis thaliana (hereafter referred to as Arabidopsis), CBF1, CBF2, and CBF31,2—also known as DREB1b, DREB1c and DREB1a, respectively3—are rapidly induced when plants are exposed to low temperatures, and they subsequently induce about 130 genes, referred to as the CBF regulon4,5,6. Expression of the CBF regulon brings about an increase in freezing tolerance, which is supported by the findings that overexpression of CBF1, CBF2, or CBF3 leads to an increase in freezing tolerance without pre-exposing the transgenic plants to low temperature5,7,8 and attenuation of the CBF pathway in plants exposed to low temperature results in a decrease in freezing tolerance6,9. The mechanisms by which the CBF regulon increases freezing tolerance are not fully understood, but involve the function of genes encoding cryoprotective polypeptides10,11 and enzymes associated with the synthesis of low molecular weight cryoprotectants such as proline and raffinose12,13.
Arabidopsis CBF1, CBF2, and CBF3 are linked tandemly in chromosome 4 and are very similar to each other in protein sequences with > 86% identity in full-length and >95% over the AP2-domain, suggesting that the CBF genes evolved through tandem duplication. In addition, the nonsynonymous (Ka) and synonymous (Ks) substitution ratio between them is significantly less than 114. In general, a Ka/Ks ratio greater than one indicates positive selection, and the ratio close to 1 indicates neutral selection, while the ratio less than 1 indicates purifying selection that leads to limited functional divergence of the duplicated genes. Thus, the result implies that the CBFs are under the influence of purifying selection and have not diverged much in function after duplication. Consistently, CBF1, CBF2, and CBF3 regulate similar sets of genes as overexpression of CBFs induces similar sets of genes6 and the mutation studies using CRISPR genome-editing technology suggests their functional redundancy as the three Arabidopsis CBFs are required for a full capacity of freezing tolerance15,16,17.
The CBF/DREB1 genes are well conserved in flowering plants18. Homologs of Arabidopsis CBF genes have been identified and characterized in several species including maize19, rice20, barley21,22, wheat23, tomato and rapeseed18, soybean24, and oat25. The CBF/DREB1 homologs contain an APETALA2/ethylene-responsive element binding factor (AP2/ERF) DNA-binding motif26. The CBF pathway has been identified as a locus contributing to freezing tolerance in Arabidopsis27,28, Medicago29, barley30, and wheat31. Thus identification of the CBF pathway genes would be a critical step towards improvement of freezing tolerance in agricultural crops.
The Arabidopsis genome encodes three other genes closely related to the CBF genes, CBF4, DDF1, and DDF2. Previously, Nakano et al.32 showed through a phylogenetic analysis that these six genes constituted a subfamily, designated IIIc, which is part of a larger monophyletic group III in the AP2/ERF superfamily in Arabidopsis. The members of IIIc subfamily (i.e. CBF/DREB1 subfamily) are separated from other AP2/ERF family members by the CBF signature sequence motifs (PKK/RPAGRxKFxETRHP and DSAWR) flanking the AP2 domain18. Despite high sequence similarity and similar sets of target genes between the family members33, they appeared to be involved in different abiotic stresses. For example, CBF1, CBF2, and CBF3 respond only to low temperature and are involved in cold acclimation; CBF4 is involved in drought tolerance33; and DDF1 and DDF2 are involved in high-salinity tolerance34. These results suggest that much of their functional divergence lies in different responsiveness to stress rather than protein sequences. Thus, the identification of the CBF pathway associated with freezing tolerance would require both phylogenetic analysis and expression analysis.
Lettuce is an important vegetable crop, with health benefits attributed to vitamin C, phenolic compounds, and fiber content35,36 and recently the genome sequence of a lettuce cultivar ‘Salinas’ has been released37. Lettuce is a cool season vegetable that can grow in regions with mild winters with an optimal temperature range of 13 °C to 18 °C38. However, brief exposure to frost during the winter season may reduce quality of lettuce. Frost damage often occurs in the winter lettuce production areas of Imperial Valley, California and Yuma, Arizona39,40. Freezing temperatures cause blisters and peeling of lettuce leaves, leading to decay and rot. Damaged leaves also provide entrance for plant pathogens. Improved freezing tolerance is an important long-term lettuce breeding goal for winter production. As a first step toward the goal, we identified the CBF/DREB1 gene family in lettuce through a comprehensive phylogenetic analysis and characterized expression of the genes under various stress conditions to determine their associations with abiotic stress response. In addition, we investigated the cold-regulated (COR) genes that might play important roles in the process of cold acclimation in lettuce. These results provide insight into the mechanism of freezing tolerance as well as evolution of the CBF/DREB1 genes in lettuce.
Results
Identification of CBF/DREB1 gene family in lettuce
The CBF/DREB1 genes in Arabidopsis belong to a subfamily, namely, the group IIIc with six gene members32. We used all six Arabidopsis genes as queries to search the protein database of lettuce (genome version 8) using the BLASTP method. To determine the CBF/DREB1 gene family through a comparative phylogenetic analysis, we expanded the search to 19 other plant species. The 19 species were selected from the Asterid and Rosid clades that represent the two largest clades in flowering plants41: nine species including Arabidopsis were selected from the Rosid clade, and ten species were selected from the Asterids, which also includes lettuce. (Table S1).
Based on sequence similarity (E-value <1e-20) with the Arabidopsis proteins and the presence of an AP2 DNA-binding domain, we detected 652 CBF-like genes from the 19 species and lettuce (Table S2; see Materials and methods). The number of genes varied among species, ranging from 19 genes from chili pepper (Capsicum annuum) or sesame (Sesamum indicum) to 65 genes from cotton (Gossypium hirsutum). Additionally, 38 genes were identified from lettuce; 22 genes were identified from Arabidopsis that correspond to the previously identified genes of the Arabidopsis group III except a pseudo gene32. In general, the number of CBF-like genes in each species correlated with the total number of genes in its genome with an R2 of 0.75 (Table S3).
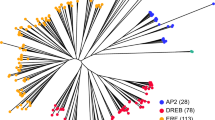
To determine the CBF/DREB1 gene family in the CBF-like genes, we conducted a phylogenetic tree analysis with the amino acid sequences of the genes. The phylogenetic tree distinguished five clusters (IIIa, IIIb, IIIc, IIId, and llle) (Fig. 1), consistent with the previous classification in Arabidopsis32. All five clusters contained genes from both Asterid and Rosid species, indicating that the clusters diverged prior to evolutionary separation of the Asterid and Rosid clades. The cluster IIIc corresponding to the CBF/DREB1 family had 198 genes in total including the six Arabidopsis CBF/DREB1 genes (Table S2). There were 13 lettuce genes in the IIIc cluster, twelve of which had predicted protein sizes ranging from 197 to 231 amino acids, whereas one gene (LsCBF7; Ls9g54981) had a predicted protein size of 65 amino acids. The LsCBF7 had a nonsense mutation within the AP2-DNA binding domain that resulted in a truncated protein (Fig. S1). To ensure that all the CBF/DREB1 family members in lettuce genome were identified, we searched the lettuce genome sequence using the tBLASTN method with protein sequences of the identified 13 genes as queries. A new gene (Ls9g54101.1) was found with a significant E-value of 3E-98, which was annotated as a splicing variant of the representative Ls9g54101.3 locus in the lettuce genome v8. The two splicing variants are, however, located in separate genomic regions (Fig. S2a) and encode similar but distinct proteins (Fig. S3), suggesting that they are different genes. Consistently, our RNA-seq analysis indicated independent transcriptions from the splicing variants as RNA-seq reads were mapped exclusively to each of the variants (Fig. S2b). Thus, we included the new gene as a member of the lettuce CBF/DREB1 family. In addition, the LsCBF8 gene sequence was incomplete in the genomic database, and we corrected it by Sanger sequencing (Fig. S4). Finally, a total 14 genes were identified and named consecutively from LsCBF1 to LsCBF14, according to their chromosomal locations (Table 1).
Phylogenetic analysis of the 652 CBF-like genes from 20 species including lettuce and Arabidopsis. The eleven species in the Asterid clade including lettuce are marked in blue circles; the nine species in the Rosid clade including Arabidopsis, in red circles; Arabidopsis CBF1–4, in gold circles; and Arabidopsis DDF1–2, in yellow circles. Five clusters, IIIa-e are named following the classification of the Arabidopsis AP2/ERF family by Nakano et al.32.
The 14 LsCBF genes showed 41–98% identity to each other at the amino acid level in full-length, and 71–100% identity for the AP2 DNA-binding domain (Fig. S5). The CBF/DREB1 signature sequence motifs18 were also highly conserved with a few variations. An arginine (R) was present in place of the proline (P) at the fourth residue of the first motif (PKK/RPAGRxKFxETRHP), and in the second motif (DSAWR), a valine (V) was more common at the third position.
Phylogenetic analysis of the CBF/DREB1 gene family
To determine orthologous or paralogous relationships of the CBF/DREB1 genes from the 20 species, a phylogenetic tree was constructed using the neighbor joining (NJ) method based on the encoded protein sequences of the genes. The resulting tree categorized the genes into three clades, designated A, B, and C (Fig. 2). The three clades included genes from both Asterid and Rosid species, indicating that ancestral genes of the three clades diverged before the Asterids and Rosids separated. Clade A included the four Arabidopsis CBFs and clade B included the two Arabidopsis DDFs, while clade C did not have any Arabidopsis gene. Thus, the ancestral gene of clade C appeared to be lost in the Arabidopsis lineage. Clade A could be further divided into two subclades, A1 and A2. A1 included genes from both Asterid and Rosid species, whereas A2 included genes only from the Asterid species. Thus, subclade A1 appeared to predate a separation of the Asterids and Rosids and be ancestral to the paralogous A2 subclade (Fig. 2).
Phylogenetic analysis of 198 CBF/DREB1 genes from 20 plant species. The phylogenetic tree was constructed using the NJ method62. The blue and red circles on the tree indicate Asterid and Rosid species, respectively. The CBF genes of Arabidopsis (At) and lettuce (Ls) are presented in black and genes from other species are color-coded according to species. The other species are: Cucumber (Cs: Cucumis sativus); Rubber Tree (Hb: Hevea brasiliensis); Medicago (Mt: Medicago truncatula); Cotton (Gh: Gossypium hirsutum); Eucalyptus (Eug: Eucalyptus grandis); Apple (Md: Malus domestica); Cottonwood (Pt: Populus trichocarpa); Wine grape (Vv: Vitis vinifera); Chili Peppers (Ca: Capsicum annuum); Artichoke (Cc: Cynara cardunculus); Carrot (Dc: Daucus carota); Monkey flower (Erg: Erythranthe guttata); Sunflower (Ha: Helianthus annuus); Morning glory (Ln: Ipomoea nil); Tobacco (Nt: Nicotiana tabacum); Olive (Oe: Olea europaea); Sesame (Si: Sesamum indicum); Potato (St: Solanum tuberosum) (See Table S1).
The lettuce CBF/DREB1 genes were distributed unevenly over the clades. Clade A had 13 lettuce CBF/DREB1 genes and clade B had only one gene, LsCBF2, while Clade C had no lettuce genes (Fig. 2). This result suggested that the 13 LsCBFs in clade A were orthologous to AtCBFs, and LsCBF2 was orthologous to AtDDFs, whereas, like Arabidopsis, the ancestral gene of clade C seemed to be lost in the lettuce lineage. Within clade A, most of the lettuce genes (10) formed a distinct monophylogenetic group while the two genes, LsCBF1 and LsCBF4, were clustered with those of the Asteraceae family–artichoke (Cc) and sunflower (Ha)–to which lettuce also belongs. This topology was also observed among other plant species. For instance, the majority of the genes of artichoke (Cc), sunflower (Ha), and carrot (Dc) within subclade A2 were formed into separate phylogenetic groups by species, and likewise were genes of Eucalyptus (Eug), monkey flower (Erg), Arabidopsis (At), Medicago (Mt), and tobacco (Nt) in subclade A1 and genes of cotton (Gh), morning glory (Ln), and olive (Oe) in the B clade (Fig. 2). The results implied that the paralogous duplication in the CBF/DREB1 gene family occurred in each species lineage.
Chromosomal locations and tandem duplication of the lettuce CBF/DREB1 genes
We examined the chromosomal locations of the 14 lettuce CBF/DREB1 genes. The LsCBFs were unevenly distributed: chromosome 2 and 6 contained only one LsCBF gene, while chromosome 9 contained 12 of LsCBFs. Of the 12 LsCBFs, 10 genes were located in a cluster (Fig. 3), suggesting tandem duplication. In support of this idea is that the tandemly arrayed LsCBF genes (i.e., LsCBF5–12) fall into a single clade in the phylogenetic tree (Fig. 2). Thus, these results suggest that those genes on chromosome 9 evolved through tandem duplication in the lettuce lineage.
To see if selective constraints acted on the duplicated genes, we examined Ka/Ks ratio using full-length protein sequences of the genes. The pairwise comparison between the 14 LsCBF genes displayed a range of 0.07–0.49 of Ka/Ks ratio, and comparison of those to Arabidopsis CBF/DREB1 genes displayed a range of 0.08–0.18, which are significantly lower than 1. The results indicated that the LsCBF genes were under purifying selection pressure with limited functional divergence (Table S4).
For those CBF/DREB1 genes from the same species that fall into a phylogenetic clade as shown in Fig. 2, we investigated whether the genes were tandemly distributed in the genome. We examined the chromosomal location of the CBF/DREB1 genes of carrot, Medicago, Eucalyptus, olive, and sunflower, in which chromosomal locations of these genes were available. We found that a majority of the genes in the five species were indeed tandemly arrayed (Fig. S6). These results support the idea that tandem duplication was a major process in the evolution of the CBF/DREB1 gene family.
Transcriptome analysis of the response to low temperature in lettuce cv. Salinas
To survey the global transcriptome change during cold acclimation in lettuce, we conducted RNA-seq on ‘Salinas’ lettuce plants exposed to low temperature for 0 h, 4 h, 24 h, and 7 days. We determined the COR genes with the criteria of fold change >2 and FDR = 0.01. We identified 5,449 COR genes, of which 2,957 were cold‐induced and 2,492 were cold‐repressed in lettuce plants (Fig. 4a; Table S5). The greatest number of genes (1,628) were significantly upregulated at 4 h, followed by 1,408 genes at 7 days and 1,387 genes at 24 h, whereas 1,800 of genes were significantly downregulated at 7 days, followed by 1,471 genes at 24 h, and 325 at 4 h (Fig. 4a). Venn diagram analysis indicated that 341 genes were commonly upregulated at all three time-points, which accounted for 21–25% of the upregulated genes at the three time-points, whereas only 133 genes were commonly downregulated at all three time-points, which accounted for 41% at 4 h, 9% at 24 h, and 7% at 7 days, respectively. These results also indicated that there were substantial differences in the COR gene sets between the different time-points. However, the differences could be due to the arbitrary cut-offs used to define the COR genes. To determine whether there were distinctive temporal expression patterns for the COR genes, we conducted a hierarchical clustering on the transcript levels of the COR genes. The heat map showed that there were indeed groups of genes with different temporal expression patterns. According to the time-points of peak expression, the COR genes could be divided into six groups: the cold-induction of G1 genes peaked at 4 h, G2 at 24 h, G3 at 7 days, and G4 throughout the cold treatment; cold-suppression of G5 and G6 genes peaked at 24 h and 7 days, respectively (Fig. 4).
The COR genes of lettuce display distinctive temporal expression patterns. (a) Venn diagrams comparing the number of up- or down-regulated genes in plants exposed to low temperature for 4 h, 24 h, 7 d. (b) Heat map with differentially expressed genes in lettuce plants exposed to low temperature (4 °C) for 4 h, 24 h, or 7 days (7 d). The groups of genes labeled G1–6 show different temporal patterns of expression in response to low temperature. The color scale represents log2‐fold change.
Enrichment analysis of GO biological functions indicated that the G1 and G2 genes were enriched in functions relating to stresses including “defense response”, “response to stress”, “response to wounding”, and “proline biosynthesis process”. The G3 genes peaking at a later stage of cold treatment showed a significant enrichment in functions associated with protein synthesis including “rRNA processing,” “RNA processing,” and “tRNA processing” (Table 2). The G4 genes that were upregulated throughout cold treatment were highly enriched in functions associated with protection from high-light intensity including “response to UV-A”, “cellular response to high light intensity,” “response to light stimulus,” and “photoprotection.” In the case of downregulated COR genes, the G5 genes that were suppressed most at relatively earlier stage of cold acclimation, i.e., 24 h, were enriched in functions associated with active growth including “hormone-mediated signaling pathway,” “cell wall organization or biogenesis,” and “signal transduction,” while the G6 genes that were suppressed most at later stage of cold acclimation, i.e., 7 days, were enriched in functions associated with photosynthesis or metabolism including “photosynthesis light harvesting,” “oxidation-reduction process,” “metabolic process,” and “carbohydrate metabolic process.” These results suggest that lettuce plants require the coordinated regulation of different sets of genes in the process of cold acclimation.
Expression analysis of the lettuce CBF/DREB1 genes under low temperature
To determine their association with cold acclimation, we examined expression levels of the 14 LsCBF genes in plants exposed to low temperature for 0 h, 4 h, 24 h, 7 days in the RNA-sequencing. Three LsCBF genes–LsCBF2, LsCBF6, and LsCBF13–did not show any significant change in transcript levels. In contrast, the remaining 11 LsCBF genes exhibited a significant increase in transcript levels in response to low temperature, with their expression peaking at the time point of 4 h as do Arabidopsis CBF1–3 genes in response to low temperature6 (Table S6). For instance, LsCBF10 exhibited the highest increase with a 651-fold change (log2 = 9.3) relative to 0 h, followed by LsCBF8 with 159-fold change (log2 = 7.3) and LsCBF12 with 151-fold change (log2 = 7.2), while LsCBF14 exhibited the least increase with a 2.8- fold change (log2 = 1.5) at 4 h (Fig. 5; Table S6). We confirmed the cold responsiveness of those 11 genes using quantitative real‐time PCR (qRT‐PCR) (Fig. 6). As in the RNA-seq experiments, the transcript levels of the genes peaked at 4 h of cold treatment. In addition, we tested the three LsCBF genes that could not be detected by RNA-seq. Consistently, the LsCBF2 and LsCBF13 transcript levels did not change in respond to cold. However, LsCBF6 was significantly upregulated with its transcript levels peaking at 4 h. Thus, of the 14 LsCBF genes, 12 LsCBF genes appeared to be induced in response to low temperature, thereby suggesting a role in cold acclimation.
Expression of lettuce CBF/DREB1 genes in response to low temperature. Expression levels were determined by quantitative RT-PCR in lettuce plants grown at 20 °C (0 h) or exposed to 4 °C for 4 h, 24 h, or 7 days (7 d). Average expression level for plants grown at 20 °C was set to one. LsCBF8, LsCBF9, and LsCBF10 were amplified using a common set of primers. Error bars indicate S.E. of three biological replicates. Different letters indicate significant differences between time-points of cold treatment. Multiple comparisons of means were performed using Tukey HSD test at the 0.05 significance level68.
Expression responses of the lettuce CBF/DREB1 family to abiotic stress
We explored the possibility that the LsCBFs are associated with other abiotic stresses. We examined expression of the 14 LsCBFs under drought, salt, and heat stress conditions using qRT‐PCR. Of the 12 LsCBFs that could respond to low temperature (Fig. 6), four of them–LsCBF3, LsCBF8, LsCBF9, and LsCBF10–did not show any significant change in transcript levels under the abiotic stresses tested, whereas two–LsCBF4, and LsCBF6–were induced in response to salt stress, five–LsCBF5, LsCBF7, LsCBF11, LsCBF12, and LsCBF14–were induced by heat, and LsCBF1 was induced by both salt and heat stress (Fig. 7). Thus, unlike Arabidopsis, the lettuce CBF genes were able to respond to multiple stresses, suggesting that they could be involved in these stresses. In addition, of the two genes that did not respond to low temperature, LsCBF2 exhibited a significant increase of transcript levels in response to salt stress while LsCBF13 did not show any change under the abiotic stresses tested.
Expression of lettuce CBF/DREB1 genes in response to abiotic stresses. Expression levels were determined by quantitative RT-PCR in lettuce plants grown at 20 °C (0 h) or exposed to drought (Dr), salt, and heat for 5 h, 24 h, or 48 h. The average expression level for plants grown at 20 °C was set to one. Error bars indicate S.E. of three biological replicates. Different letters indicate significant differences between time-points for each stress treatment. Multiple comparisons of means were performed using Tukey HSD test at the 0.05 significance level68.
Discussion
The CBF/DREB1 genes were identified first in Arabidopsis as key transcription factors in cold acclimation1,42. Since then, the CBF homologs and their contribution to enhancement of freezing tolerance have been reported in many plant species22,28,29,30. In this study, we conducted a genome-wide search for the CBF/DREB1 orthologs in lettuce and identified a total of 14 CBF orthologous genes.
Our phylogenetic analysis comparing the CBF/DREB1 genes from 19 of other plant species revealed three monophyletic groups, designated A, B, and C, and the group A can be further divided into two subgroups, A1 and A2 (Fig. 2). The A1, B, and C groups contain genes from Asterid and Rosid species, and the A2 group contains genes only from Asterid species (Fig. 2). Thus, this topology suggests that there were at least four multiplication events in evolution of the CBF/DREB1 gene family in the Asterid lineage: three ancient events in a common ancestor of the Asterid and Rosid species and more recent one in the Asterid lineage. Many studies provide strong evidence that whole genome triplication (WGT), so called gamma event, has occurred in core eudicot lineage that is placed before the split of Asterids and Rosids43,44,45. Thus, the ancient three multiplications (i.e., A, B, and C groups) in the CBF/DREB1 gene family might result from the WGT in core eudicot lineage. This notion is further supported by the observation that the genes from different clade groups are mostly located in different chromosomes in the diverse plant species. For example, Arabidopsis AtCBF1–3 in clade A are in chromosome 4, whereas AtDDF1–2 in clade B are in chromosome 1; lettuce genes in clade A and clade B are located in chromosome 9 and 6, respectively; and so do genes from carrot, Medicago, sunflower, olive, and Eucalyptus (Figs. 2, S6). As gene duplication could provide the raw genetic material for functional novelty46, one of the ancient multiplications resulted in the functional divergence of ancestral genes of CBFs and DDFs, two subgroups of the CBF/DREB1 family that are involved in different abiotic stresses1,33,34: the four AtCBFs fall into group A and the two AtDDFs fall into group B. With regard to the lettuce CBF/DREB1 genes, LsCBF2 is the only one that belongs to the DDF orthologous phylogenetic group (Group B in Fig. 2) while all other LsCBFs belong to the CBF orthologous phylogenetic group (Group A in Fig. 2). Consistent with the orthologous relationships, LsCBF2 exhibited a responsiveness to salt stress but not to low temperature as AtDDFs do, and all the other LsCBFs except LsCBF13 in the group A exhibited a responsiveness to low temperature. Furthermore, the cold-responsive LsCBFs exhibited a similar expression kinetics to that of Arabidopsis CBF genes, with expression peaking at around 4 h and reduced gradually at later time points in response to low temperature42, supporting the orthologous role of the lettuce CBF genes in cold acclimation (Fig. 5). However, there is a striking difference. Unlike Arabidopsis in which the members of the CBF/DREB1 family can respond only to a particular abiotic stress1,34, eight of these 12 cold-responsive LsCBFs show significant induction by salt or heat stress (Fig. 7): two LsCBFs can respond to salt; five LsCBFs can respond to heat stress; and one LsCBF can respond to both heat and salt stress. The LsCBFs responsive to both cold- and heat-stress might be a result of adaption to a broad range of environmental temperature changes. For instance, the all members of CBF family in Eucalyptus are induced significantly by both cold and heat conditions, which would contribute to adaptation of the woody species to cold and dry climate in Australia where the species have originated47. Thus, the responsiveness of LsCBFs to multiple stresses (cold and heat; cold and salt; or cold, heat and salt) suggests that those genes may function as master regulator over abiotic stresses, such that lettuce could adapt to a wider range of climate conditions.
The 13 LsCBFs in the group A are asymmetrically distributed over the subgroups A1 and A2: subgroup A1 contains one gene, LsCBF3; and subgroup A2 contains the remaining 12 LsCBF genes (Fig. 2). Within the subgroup A2, the LsCBF1 and LsCBF4 are more closely related to genes from the other Asteraceae species–sunflower and artichoke–than to any other genes of lettuce, whereas the other 10 lettuce genes are more closely related to each other (Fig. 2). Moreover, eight of these LsCBFs are located in a tandem array in chromosome 9 (Fig. 3), which suggests that these genes evolved through tandem duplication in the lettuce lineage. This notion is further supported by the fact that those genes in tandem array are more similar to each other in protein sequence: the tandem genes display a range of 68–98% identity while they display a range of 41–59% identity in comparison with the other LsCBFs (Fig. S5). This tandem duplication of CBF/DREB1 genes is also observed in other species including Arabidopsis2, tomato48, barley22, carrot, Eucalyptus, Medicago, olive, and sunflower (this study). Taken together, these results suggest that tandem duplication has played a significant role in the expansion of the CBF/DREB1 gene family across diverse plant species. In addition, our phylogenetic analysis shows that the duplicates of different species are distributed over different phylogenetic clades, i.e., groups A1, A2, B, and C in Fig. 2. Duplicates of Eucalyptus (Eug) or Medicago (Mt) are in group A1 and those of sunflower (Ha), carrot (Dc) or lettuce (Ls) are in A2 while those of olive (Oe) are in group B. Thus, it seems that the tandem duplications in different species occurred independently from rather phylogenetically distinct ancestral genes. Sandve and Fjellheim49 provided evidence through phylogenetic and molecular clock analysis that the expansion of CBF/DREB1 family in the Pooideae family species coincides with a global super-cooling period at the Eocene–Oligocene boundary (33.5–26 Ma), suggesting that the expansion resulted from the adaptation to the cooling climate. It is intriguing to investigate whether the diverse plant species has undergone the lineage-specific tandem duplications as a result of adaption to the supercooling climate.
Duplication is widely believed to be a major source of genetic variation through sub- or neofunctionalization that can lead to species divergence50. Generally, functional redundancy created by gene duplication allows the genes to accumulate mutations, leading to increase of divergence and subsequently expansion of the gene family. However duplicate genes can also be preserved by natural selection. Our Ka/Ks ratio analysis supports the latter scenario for the CBF/DREB1 gene family in lettuce. The ratio is in the range of 0.07–0.49, which is significantly lower than 1, suggesting that purifying selection predominated among the lettuce CBF/DREB1 genes. Purifying selection would eliminate deleterious mutations and preserve the ancestral function of duplicated genes, which could provide benefit with increased production of ancestral gene product through a gene dosage effect51,52,53. That most members of the lettuce CBF/DREB gene family are responsive to low temperatures and are induced with similar kinetics, peaking at 4 h of cold treatment, supports that duplicated LsCBF genes may be under this dosage effect. Recent studies of CRISPR-mediated CBF mutation in Arabidopsis15,16,17 also support this idea. The reports showed that the CBF mutations resulted in a gradual decrease in freezing tolerance with the triple cbf mutation exhibiting the least freezing tolerance and single mutations exhibiting a moderate reduction in freezing tolerance compared with wildtype control. The results indicate that the three Arabidopsis CBF genes act additively to increase of freezing tolerance and all three genes are required for full-capacity of freezing tolerance in Arabidopsis. Thus, it is worthy of noting that one lettuce CBF gene, LsCBF7, encodes a non-functional allele due to a premature stop codon, which could lead to reduced freezing tolerance in ‘Salinas’ plants. Given that the cold acclimation process is costly in cold but non-freezing environments, it is not surprising that the accessions adapted to warmer climate have sometimes a nonfunctional CBF28,54. If that is the case in lettuce, the CBF locus would be a good target for breeding and genetic engineering for improved freezing tolerance in lettuce. In addition to dosage effect, gene expression divergence may also play a role in the preservation of CBF duplicates. The LsCBF2 is responsive predominantly to salt stress (Fig. 7), and this subfunctionalization would contribute to its retention by providing selective advantage in new environments55.
Materials and methods
Plant material and growth conditions
The lettuce cultivar ‘Salinas’ was grown on potting mix soil in pots in a growth chamber at 20 °C under a 16-h photoperiod with a light intensity of 350–400 μmol m−2 sec−1. The 18-day-old plants were used for stress treatments. For cold acclimation treatment42, plants were exposed to 4 °C for 0 h, 4 h, 24 h, or 7 days with a light intensity of 100 μmol m−2 sec−1. The other stress treatments were carried out for 0 h, 5 h, 24 h, and 48 h with a light intensity of 300 μmol m−2 sec−1. For salt and heat stress treatments, plants were treated with 250 mM NaCl56,57 or exposed to 34 °C58, respectively. For drought stress treatment, the excess water was removed from the pots and water was withheld up to 48 h. The 0 h samples were used as a control in all treatments.
Identification of CBF-like genes from 20 plant species including lettuce
In this study, 20 plant species including lettuce were selected from the Asterid or Rosid clade and their protein databases were used to identify CBF-like genes. The protein databases of 19 species (except lettuce) were downloaded from the NCBI database and the lettuce protein database (genome v8: id37106) was obtained at https://genomevolution.org/coge (Table S1). Where a gene had multiple isoforms in the protein databases, the longest protein was selected as a representative for the gene. The protein sequences of the Arabidopsis CBF/DREB1 family genes (i.e., CBF1, CBF2, CBF3, CBF4, DDF1, and DDF2) were used as queries to search the protein databases using the BLASTP method with an E-value threshold of <1E-20. In addition, the presence of the AP2-domain was examined using the hmmscan function of HMMER3 v3.2.1 (http://hmmer.org)59 with the AP2 domain profile (Pfam accession, PF00847) as a query for the protein sequences selected from the BLASTP search. The protein sequences were excluded from further consideration if the AP2 domain was truncated, or the AP2 domain match E-value was greater than 1e-5. As a result, 652 CBF-like genes were identified from the 20 plant species (Table S2).
Phylogenetic analysis
Multiple protein sequences were aligned using ClustalW260 with default parameters, and the alignment was further inspected and manually adjusted with BioEdit61. Phylogenetic trees were generated based on the alignment using the NJ method in MEGA X version 1062 with the parameters of Jones-Taylor-Thornton model, uniform rates among sites, and complete deletion of gaps. The trees were visualized using FigTree version 1.4.3 (http://tree.bio.ed.ac.uk/software/figtree).
Chromosomal mapping and Ka/Ks ratio calculation
Lettuce CBF/DREB1 genes were mapped onto the nine lettuce chromosomal linkage groups according to their physical positions (bp). The R/LinkageMapView package63 was used to draw their locations onto the physical map of each chromosome. To estimate the evolution rate between the CBF genes, we calculated Ka/Ks ratios using a bioperl utility, ‘aa_to_dna_aln’ (https://bioperl.org) and ‘KaKs_calculator’ version 1.264 with a method of model-average. The significance of Ka/Ks that deviated from neutrality (=1) was tested using the Fisher’s exact test, and the ratios with a P-value of at least 0.01 were considered as significant.
RNA-seq analysis
Above-ground tissues were collected from ‘Salinas’ plants exposed to 4 °C for 0 h, 4 h, 24 h and 7 days. Total RNA was isolated for each biological replicate using Plant RNeasy kit (Qiagen) and submitted to Novogene Corporation (https://en.novogene.com/) for RNA-seq library preparation and sequencing. Sequencing was performed on the Illumina HiSeq platform with 150 bp paired-end reads (http://www.illumina.com). The RNA-seq reads were mapped to the Lactuca sativa reference genome (version 8) using STAR v2.5.265. The resulting BAM files were used to count reads at the gene-level using ‘featureCounts’66. Differential expression analysis was implemented using the ‘edgeR’ package67 in R software, version 3.5.0 (https://www.r-project.org/). Because estimates of differential gene expression can be inflated by lowly expressed genes, we included only genes with at least 0.5 read per million (<0.5 CPM) in at least two samples, which resulted in 23933 genes. Genes with a two-fold change (log2 = 1) or more and an FDR = 0.01 were designated as differentially expressed. Hierarchical clustering analyses were performed using the hcluster method of ‘amap’ in R software and the resulting clusters were visualized with ‘treeview’ (http://rana.lbl.gov/EisenSoftware.htm). The RNA-seq data have been deposited in the Gene Expression Omnibus under accession number GSE134012.
Quantitative Real-Time PCR
Total RNA was extracted from above-ground tissues of lettuce seedlings grown on soil in pots using the RNeasy Plant Mini kits (Qiagen, http://www.qiagen.com/). cDNA was synthesized with 200 ng total RNA and random primers using the Reverse Transcription System (Promega, https://www.promega.com). qRT‐PCR was performed using fast SYBR Green master mix (Life Technologies, http://www.lifetechnologies.com). Two housekeeping genes, eukaryotic translation initiation factor 2 A (EIF2a;Ls6g95581) and isopentenyl diphosphate isomerase 2 (IPP2;Ls2g17540), were used as reference genes. Lettuce homologs of those genes were identified through the BLASTN method using Arabidopsis EIF2a (At5g05470) and IPP2 (At3g02780) as queries. Primers used for qRT‐PCR are shown in Table S7. Relative expression values were calculated by ddCt method using the average of two reference genes and normalized to control treatment for fold-changes. Tukey Honestly Significant Difference (HSD) test for multiple comparisons was performed using TukeyHSD function in R environment68.
Gene ontology enrichment assay
Gene ontology (GO) functional annotation of lettuce genes was conducted using the Trinotate pipeline (https://trinotate.github.io/) and custom PERL scripts. The lettuce protein sequences were BLASTP-searched against UniProtKB/Swiss-Prot, which is a manually annotated, non-redundant protein sequence database. The GO terms and biological functions for lettuce genes were derived from those of the UniprotKB database if the genes had a match with an E -value threshold of <1e-20. GO term enrichment was performed on the differentially expressed genes with the 23,933 expressed gene set as a background. The significance of enrichment was tested by a hypergeometric test, which was conducted using ‘phyper’ function in R environment68.
Data availability
The RNA-seq data are available in the Gene Expression Omnibus (www.ncbi.nlm.nih.gov/geo/) under accession number GSE134012. All relevant data are included in the manuscript and the Supporting Information files.
References
Stockinger, E. J., Gilmour, S. J. & Thomashow, M. F. Arabidopsis thaliana CBF1 encodes an AP2 domain-containing transcriptional activator that binds to the C-repeat/DRE, a cis-acting DNA regulatory element that stimulates transcription in response to low temperature and water deficit. Proc. Natl. Acad. Sci. 94, 1035–1040 (1997).
Medina, J., Bargues, M., Terol, J., Pérez-Alonso, M. & Salinas, J. The Arabidopsis CBF gene family is composed of three genes encoding AP2 domain-containing proteins whose expression is regulated by low temperature but not by abscisic acid or dehydration. Plant Physiol. 119, 463–470 (1999).
Liu, Q. et al. Two transcription factors, DREB1 and DREB2, with an EREBP/AP2 DNA binding domain separate two cellular signal transduction pathways in drought- and low-temperature-responsive gene expression, respectively, in Arabidopsis. Plant Cell 10, 1391–1406 (1998).
Gilmour, S. J. et al. Low temperature regulation of the Arabidopsis CBF family of AP2 transcriptional activators as an early step in cold-induced COR gene expression. Plant J. 16, 433–442 (1998).
Kasuga, M., Liu, Q., Miura, S., Yamaguchi-Shinozaki, K. & Shinozaki, K. Improving plant drought, salt, and freezing tolerance by gene transfer of a single stress-inducible transcription factor. Nat. Biotechnol. 17, 287 (1999).
Park, S. et al. Regulation of the Arabidopsis CBF regulon by a complex low-temperature regulatory network. Plant J. 82, 193–207 (2015).
Jaglo-Ottosen, K. R., Gilmour, S. J., Zarka, D. G., Schabenberger, O. & Thomashow, M. F. Arabidopsis CBF1 overexpression induces COR genes and enhances freezing tolerance. Science 280, 104–106 (1998).
Gilmour, S. J., Sebolt, A. M., Salazar, M. P., Everard, J. D. & Thomashow, M. F. Overexpression of the Arabidopsis CBF3 transcriptional activator mimics multiple biochemical changes associated with cold acclimation. Plant Physiol. 124, 1854–1865 (2000).
Novillo, F., Medina, J. & Salinas, J. Arabidopsis CBF1 and CBF3 have a different function than CBF2 in cold acclimation and define different gene classes in the CBF regulon. Proc. Natl. Acad. Sci. 104, 21002–21007 (2007).
Steponkus, P. L., Uemura, M., Joseph, R. A., Gilmour, S. J. & Thomashow, M. F. Mode of action of the COR15a gene on the freezing tolerance of Arabidopsis thaliana. Proc. Natl. Acad. Sci. 95, 14570–14575 (1998).
Battaglia, M., Olvera-Carrillo, Y., Garciarrubio, A., Campos, F. & Covarrubias, A. A. The enigmatic LEA proteins and other hydrophilins. Plant Physiol. 148, 6–24 (2008).
Gilmour, S. J., Fowler, S. G. & Thomashow, M. F. Arabidopsis transcriptional activators CBF1, CBF2, and CBF3 have matching functional activities. Plant Mol. Biol. 54, 767–781 (2004).
Kaplan, F. et al. Exploring the temperature-stress metabolome of Arabidopsis. Plant Physiol. 136, 4159–4168 (2004).
Lin, Y.-H. et al. Molecular population genetics and gene expression analysis of duplicated CBF genes of Arabidopsis thaliana. BMC Plant Biol. 8, 111 (2008).
Jia, Y. et al. The cbfs triple mutants reveal the essential functions of CBFs in cold acclimation and allow the definition of CBF regulons in Arabidopsis. New Phytol. 212, 345–353 (2016).
Zhao, C. et al. Mutational evidence for the critical role of CBF transcription factors in cold acclimation in Arabidopsis1. Plant Physiol. 171, 2744–2759 (2016).
Park, S., Gilmour, S. J., Grumet, R. & Thomashow, M. F. CBF-dependent and CBF-independent regulatory pathways contribute to the differences in freezing tolerance and cold-regulated gene expression of two Arabidopsis ecotypes locally adapted to sites in Sweden and Italy. PLoS One 13 (2018).
Jaglo, K. R. et al. Components of the Arabidopsis C-repeat/dehydration-responsive element binding factor cold-response pathway are conserved in Brassica napus and other plant species. Plant Physiol. 127, 910–917 (2001).
Shen, Y.-G. et al. Characterization of a DRE-binding transcription factor from a halophyte Atriplex hortensis. Theor. Appl. Genet. 107, 155–161 (2003).
Dubouzet, J. G. et al. OsDREB genes in rice, Oryza sativa L., encode transcription activators that function in drought-, high-salt- and cold-responsive gene expression. Plant J. 33, 751–763 (2003).
Choi, D.-W., Rodriguez, E. M. & Close, T. J. Barley cbf3 gene identification, expression pattern, and map location. Plant Physiol. 129, 1781–1787 (2002).
Skinner, J. S. et al. Mapping of barley homologs to genes that regulate low temperature tolerance in Arabidopsis. Theor. Appl. Genet. 112, 832–842 (2006).
Badawi, M., Danyluk, J., Boucho, B., Houde, M. & Sarhan, F. The CBF gene family in hexaploid wheat and its relationship to the phylogenetic complexity of cereal CBFs. Mol. Genet. Genomics 277, 533–554 (2007).
Kidokoro, S. et al. Soybean DREB1/CBF-type transcription factors function in heat and drought as well as cold stress-responsive gene expression. Plant J. 81, 505–518 (2015).
Bräutigam, M. et al. Generation and analysis of 9792 EST sequences from cold acclimated oat, Avena sativa. BMC Plant Biol. 5, 18 (2005).
Sakuma, Y. et al. DNA-binding specificity of the ERF/AP2 domain of Arabidopsis DREBs, transcription factors involved in dehydration- and cold-inducible gene expression. Biochem. Biophys. Res. Commun. 290, 998–1009 (2002).
Ågrena, J., Oakley, C. G., McKay, J. K., Lovell, J. T. & Schemske, D. W. Genetic mapping of adaptation reveals fitness tradeoffs in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 110, 21077–21082 (2013).
Gehan, M. A. et al. Natural variation in the C-repeat binding factor cold response pathway correlates with local adaptation of Arabidopsis ecotypes. Plant J. 84, 682–693 (2015).
Tayeh, N. et al. A tandem array of CBF/DREB1 genes is located in a major freezing tolerance QTL region on Medicago truncatula chromosome 6. BMC Genomics 14, 814 (2013).
Francia, E. et al. Copy number variation at the HvCBF4–HvCBF2 genomic segment is a major component of frost resistance in barley. Plant. Mol. Biol. 92, 161–175 (2016).
Würschum, T., Longin, C. F. H., Hahn, V., Tucker, M. R. & Leiser, W. L. Copy number variations of CBF genes at the Fr-A2 locus are essential components of winter hardiness in wheat. Plant J. 89, 764–773 (2017).
Nakano, T., Suzuki, K., Fujimura, T. & Shinshi, H. Genome-wide analysis of the ERF gene family in Arabidopsis and rice. Plant Physiol. 140, 411–432 (2006).
Haake, V. et al. Transcription Factor CBF4 Is a Regulator of Drought Adaptation in Arabidopsis. Plant Physiol. 130, 639–648 (2002).
Magome, H., Yamaguchi, S., Hanada, A., Kamiya, Y. & Oda, K. Dwarf and delayed-flowering 1, a novel Arabidopsis mutant deficient in gibberellin biosynthesis because of overexpression of a putative AP2 transcription factor. Plant J. 37, 720–729 (2004).
Mou, B. Nutritional quality of lettuce. Curr. Nutr. Food Sci. 8, 177–187 (2012).
Mulabagal, V. et al. In vitro evaluation of red and green lettuce (Lactuca sativa) for functional food properties. Food Chem. 118, 300–306 (2010).
Reyes-Chin-Wo, S. et al. Genome assembly with in vitro proximity ligation data and whole-genome triplication in lettuce. Nat. Commun. 8, 14953 (2017).
Simko, I., Hayes, R. J., Mou, B. & McCreight, J. D. Lettuce and spinach. Yield Gains in Major U.S. Field Crops cssaspecialpubl, 53–86 (2014).
Luna, N. Lettuce prices jump due to cold snap. Orange County Register, https://www.ocregister.com/2013/01/23/lettuce-prices-jump-due-to-cold-snap/ (2013).
Desert frosts nip lettuce. Packer, https://www.thepacker.com/article/desert-frosts-nip-lettuce (2014).
Moore, M. J., Soltis, P. S., Bell, C. D., Burleigh, J. G. & Soltis, D. E. Phylogenetic analysis of 83 plastid genes further resolves the early diversification of eudicots. Proc. Natl. Acad. Sci. 107, 4623–4628 (2010).
Thomashow, M. F. Molecular basis of plant cold acclimation: insights gained from studying the CBF cold response pathway. Plant Physiol. 154, 571–577 (2010).
Jiao, Y. et al. Ancestral polyploidy in seed plants and angiosperms. Nature 473, 97–100 (2011).
Jiao, Y. et al. A genome triplication associated with early diversification of the core eudicots. Genome Biol. 13, R3 (2012).
Ren, R. et al. Widespread Whole Genome Duplications Contribute to Genome Complexity and Species Diversity in Angiosperms. Mol. Plant 11, 414–428 (2018).
Adams, K. L. & Wendel, J. F. Polyploidy and genome evolution in plants. Curr. Opin. Plant Biol. 8, 135–141 (2005).
Martin, H. A. Cenozoic climatic change and the development of the arid vegetation in Australia. J. Arid Environ. 66, 533–563 (2006).
Zhang, X. et al. Freezing-sensitive tomato has a functional CBF cold response pathway, but a CBF regulon that differs from that of freezing-tolerant Arabidopsis. Plant J. 39, 905–919 (2004).
Sandve, S. R. & Fjellheim, S. Did gene family expansions during the Eocene–Oligocene boundary climate cooling play a role in Pooideae adaptation to cool climates? Mol. Ecol. 19, 2075–2088 (2010).
Davies, T. J. et al. Darwin’s abominable mystery: Insights from a supertree of the angiosperms. Proc. Natl. Acad. Sci. 101, 1904–1909 (2004).
Kaessmann, H. Origins, evolution, and phenotypic impact of new genes. Genome Res. 20, 1313–1326 (2010).
Conant, G. C., Birchler, J. A. & Pires, J. C. Dosage, duplication, and diploidization: clarifying the interplay of multiple models for duplicate gene evolution over time. Curr. Opin. Plant Biol. 19, 91–98 (2014).
Panchy, N., Lehti-Shiu, M. & Shiu, S.-H. Evolution of gene duplication in plants. Plant Physiol. 171, 2294–2316 (2016).
Kang, J. et al. Natural variation of C-repeat-binding factor (CBFs) genes is a major cause of divergence in freezing tolerance among a group of Arabidopsis thaliana populations along the Yangtze River in China. New Phytol. 199, 1069–1080 (2013).
Ganko, E. W., Meyers, B. C. & Vision, T. J. Divergence in Expression between Duplicated Genes in Arabidopsis. Mol. Biol. Evol. 24, 2298–2309 (2007).
Garrido, Y. et al. Physiological, phytochemical and structural changes of multi-leaf lettuce caused by salt stress. J. Sci. Food Agric. 94, 1592–1599 (2014).
Bartha, C., et al Sodium accumulation contributes to salt stress tolerance in lettuce cultivars. J. Appl. Bot. Food Qual. 88 (2015).
Jenni, S. Rib Discoloration: A Physiological Disorder Induced by Heat Stress in Crisphead Lettuce. HortScience 40, 2031–2035 (2005).
Potter, S. C. et al. HMMER web server: 2018 update. Nucleic Acids Res. 46, W200–W204 (2018).
Larkin, M. A. et al. Clustal W and Clustal X version 2.0. Bioinformatics 23, 2947–2948 (2007).
Hall, T. A. et al. BioEdit: a user-friendly biological sequences alignment editor and analysis program for Window 95/98/NT, https://doi.org/10.1021/bk-1999-0734.ch008 (1999).
Kumar, S., Stecher, G., Li, M., Knyaz, C. & Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 35, 1547–1549 (2018).
Ouellette, L. A., Reid, R. W., Blanchard, S. G. & Brouwer, C. R. LinkageMapView—rendering high-resolution linkage and QTL maps. Bioinformatics 34, 306–307 (2018).
Zhang, Z. et al. KaKs_Calculator: Calculating Ka and Ks Through Model Selection and Model Averaging. Genomics Proteomics Bioinformatics 4, 259–263 (2006).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Liao, Y., Smyth, G. K. & Shi, W. FeatureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014).
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. EdgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2010).
R Core Team. R: a language and environment for statistical computing. R Foundation for statistical computing, https://www.R-project.org/ (2018).
Acknowledgements
We are grateful to Drs. James D. McCreight (USDA-ARS) and Sarah J. Gilmour (Michigan State University) for critically reading the manuscript. This research was funded by a grant from USDA-AMS Specialty Crop Multistate Program (Award No. 16SCCMAR0001) and USDA-NIFA Specialty Crop Research Initiative (Award No. 2015-51181-24283). Mention of a trade name, proprietary product, or vendor does not constitute an endorsement, guarantee, or warranty by the USDA and does not imply its approval to the exclusion of other products or vendors that may be suitable.
Author information
Authors and Affiliations
Contributions
S.P. and B.M. conceptualized the study and designed experiments. B.M. and A.S. obtained funding for this investigation. S.P conducted the experiments and analyzed the results with help of B.M and A.S. S.P., A.S., and B.M. wrote the manuscript. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Park, S., Shi, A. & Mou, B. Genome-wide identification and expression analysis of the CBF/DREB1 gene family in lettuce. Sci Rep 10, 5733 (2020). https://doi.org/10.1038/s41598-020-62458-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-62458-1
- Springer Nature Limited
This article is cited by
-
Integrated transcriptomic and metabolomic analysis provides insights into cold tolerance in lettuce (Lactuca sativa L.)
BMC Plant Biology (2024)
-
Low frequency of the wild-type freezing-tolerance LsCBF7 allele among lettuce population suggests a negative selection during domestication and breeding
Theoretical and Applied Genetics (2024)
-
Genome-wide characterization and evolutionary analysis of the AP2/ERF gene family in lettuce (Lactuca sativa)
Scientific Reports (2023)
-
Overexpression of MbICE3 increased the tolerance to cold and drought in lettuce (Lactuca sativa L.)
In Vitro Cellular & Developmental Biology - Plant (2023)
-
Parthenolide reduces the freezing tolerance of hydrated lettuce seeds by inhibiting the proteolysis of seed storage globulins
Plant Cell, Tissue and Organ Culture (PCTOC) (2021)