Abstract
The United States Appalachian region harbors a higher cancer burden than the rest of the nation, with disparate incidence of head and neck squamous cell carcinomas (HNSCC), including oral cavity and pharynx (OC/P) cancers. Whether elevated HNSCC incidence generates survival disparities within Appalachia is unknown. To address this, HNSCC survival data for 259,737 tumors from the North American Association for Central Cancer Registries 2007–2013 cohort were evaluated, with age-adjusted relative survival (RS) calculated based on staging, race, sex, and Appalachian residence. Tobacco use, a primary HNSCC risk factor, was evaluated through the Behavioral Risk Factor Surveillance System from Appalachian states. Decreased OC/P RS was found in stage IV Appalachian white males within a subset of states. The survival disparity was confined to human papillomavirus (HPV)-associated oropharyngeal cancers, specifically the oropharynx subsite. This correlated with significantly higher smoking and male smokeless tobacco use in most Appalachian disparity states. Lower survival of Appalachian males with advanced-stage HPV-associated oropharyngeal cancers suggests pervasive tobacco consumption likely generates more aggressive tumors at HPV-associated oropharynx subsites than national averages. Comprehensive tobacco and HPV status should therefore be evaluated prior to considering treatment de-intensification regimens for HPV-associated oropharyngeal cancers in populations with high tobacco consumption.
Similar content being viewed by others
Introduction
HNSCC involves the epithelium of the oral cavity, pharynx and larynx. OC/P cancers are a major HNSCC subset, with nearly 11,000 deaths predicted in the US in 20201. Risk factors include tobacco and alcohol use, and high-risk human papillomavirus (HPV) infection2,3. OC/P cancers are subdivided into HPV-associated oropharynx (HPV-associated) and non-HPV-associated oral cavity, hypopharynx, and nasopharynx (non-HPV-associated) cancers4. These designations are supported by studies indicating that non-HPV-associated cancers are primarily tobacco/alcohol induced, whereas HPV-associated cancers are predominantly caused by HPV infection5. Furthermore, HPV-negative or non-HPV-associated cancers consistently have poorer outcomes than HPV-positive or HPV-associated cancers, segregating these cancers as distinct diseases with differential clinical management6,7. While national incidence of non-HPV-associated cancers is decreasing due to tobacco cessation, HPV-associated cancers are increasing due to rising infection rates8. Regarding race, blacks with HNSCC present with more advanced disease, are older and have worse survival than whites, denoting a racial disparity9,10,11. Increased screening for HPV coupled with subsite analysis indicates that blacks with HNSCC have less HPV-positive cancer than whites, explaining differences in survival12,13. Consistent with this, HPV-associated cancers continue to increase in white males and in rural areas4,14,15.
The Appalachian region encompasses 205,000 square miles across 420 counties in 13 contiguous states16. Forty-two percent of Appalachia is rural, with poverty rates in many counties 1.5 times above the national mean16. The region is over 80% white, with several states having higher percentages of oral and other HPV-associated cancers, and higher smoking and smokeless tobacco use17,18,19. Aggregate risk factors experienced by Appalachian residents contribute to a high allostatic load capable of generating cancer disparities in a region with low ethnic diversity20. Incidence disparities within Appalachia are well recognized21 with rates higher for HPV- and tobacco-related malignancies22,23,24. Elevated risk factor exposure has been suggested as the prime reason for disparate incidence in Appalachian male HNSCC24,25. However, due to gaps in data availability and small patient numbers at the county level, details on how increased incidence impacts HNSCC survival in Appalachia have not been rigorously evaluated25. Here we have evaluated the RS of all HNSCC in the majority of Appalachian states from 2007–2013 and have identified an outcome disparity in white males with oropharynx cancers that corresponds with elevated smoking and smokeless tobacco use in the region.
Materials and methods
HNSCC cohort and relative survival data
All methods were carried out in accordance with relevant guidelines and regulations. Survival data were generated by NAACCR and provided in Surveillance, Epidemiology, and End Results (SEER)*Stat approved under NAACCR IRB protocol 16–14, where all patient data was obtained following informed consent. Patients were diagnosed from 2007–2013 with tumors of International Classification of Diseases for Oncology (ICD-O)-3 histology types 8,050–8,084, 8,120–8,131 (squamous and transitional epithelium). Selecting all primary tumors matching selection criteria, the cohort includes tumors of the oral cavity (n = 55,620/259,737, 21.4%) including ICD-O-3 site groups of tongue (C02.0–02.3, 02.9), floor of mouth (C04.0–04.9), and gum and other mouth (C03.0–03.9, C05.0, C05.8–05.9, C06.0–06.9); oropharynx (n = 81,170/259,737, 31.3%) including ICD-O-3 site groups of tongue (C01.9, C02.4, C02.8), gum and other mouth (C05.1–05.2), tonsil (C09.0–09.9), oropharynx (C10.0–10.9), and “other oral cavity and pharynx” (C14.0–14.8); “other pharynx” (n = 17,731/259,737, 6.8%) including ICD-O-3 site groups of nasopharynx (C11.0–11.9) and hypopharynx (C12.9–13.9); and larynx (n = 60,296/259,737, 23.2%) including ICD-O-3 site group larynx (C32.0–32.9). Remaining tumors are additional primaries of miscellaneous ICD-O-3 site groups (n = 44,916/259,737, 17.3%). The cohort includes variables denoting whether or not patients were Appalachian county residents at the time of diagnosis, and county economic status from the Appalachian Regional Commission (ARC)26. Appalachian states with survival data fit for use through the reporting period included New York, Pennsylvania, West Virginia, Kentucky, North Carolina, South Carolina, Georgia, and Alabama27. The majority of cases were white (n = 221,939/259,737, 85.4%), stage IV (n = 84,253/259,737, 32.4%), and male (n = 193,647/259,737, 74.6%). Cases were evaluated using the Ederer II method and the “U.S. by race (W, B, AIAN, API) and Canada 1995–2012, Ages 0–99, State-county (modeled by varied state-county-ses)” life table. Included cases were microscopically confirmed for malignant behavior and followed at monthly intervals. Excluded cases were those that were diagnosed solely via death certificate or autopsy, and alive cases with no survival time. Age-adjusted RS, defined as net survival measure representing cancer survival in the absence of other causes of death28, was calculated using SEER*Stat for each patient stratification, with z-tests for significance testing between each group using an alpha level of 0.05. Cumulative (C)RS was used to compare groups with 5 years of available RS data and the maximum available follow-up RS was used to compare groups that failed to reach 5 years of follow-up.
Mapping
Maps were generated using 2014 cartographic boundary shapefiles from the United States Census Bureau29 and Quantum Geographic Information System (QGIS) 3.2.2 software30. Appalachian state-Appalachian county refers to a county within Appalachia. Appalachian state-non-Appalachian county refers to a county outside of the Appalachian region, but within a state containing Appalachian counties. Non-Appalachian states lack Appalachian counties.
TCGA cohort and associated clinical data
Data from The Cancer Genome Atlas (TCGA) were obtained from the Broad Genomic Data Analysis Center (GDAC) Firehose 2016_01_28 TCGA-HNSC cohort consisting of 529 patients with tumors of the oral cavity (n = 320/529, 60.5%), pharynx (n = 92/529, 17.4%), and larynx (n = 117/529, 22.1%). Clinical data and patient characteristics were retrieved from the level 4 TCGA clinical data file All_CDEs.txt and cBioPortal31,32. OC/P patients were stratified by HPV-associated or non-HPV-associated subsite defined by the Centers for Disease Control (CDC)33 and by confirmed HPV status. All patients were evaluated for HPV in the All_CDEs.txt file, denoted by variable “hpv_status”. Kaplan–Meier P values were calculated using Mantel-Cox log-rank test and were validated by an independent biostatistician.
Appalachian tobacco use data and analysis
Data for 2016 current smoker and smokeless tobacco user frequency, weighted frequency, prevalence, and confidence intervals for Appalachian and non-Appalachian counties were calculated and provided by BRFSS coordinators from Appalachian states. Current smoker status was calculated using CDC BRFSS Tobacco Use Question 1 and 2 from the 2016 questionnaire. Current smokeless status was calculated using CDC BRFSS Tobacco Use Question 3. Non-Appalachian state tobacco use data were acquired from CDC BRFSS19. Statistical significance was determined within states and between regions using G-tests for independence without Yates’ correction using BRFSS frequency (N) values and an alpha value of 0.05. BRFSS recommends caution interpreting results with less than 50 respondents. Bonferroni correction was used where applicable to reduce type I error. Due to changes in sample composition and weighting methodology in 2011, data from years after 2011 cannot be directly compared to previous years34.
Results
Identification of a survival disparity in white Appalachian males with stage IV oral cavity and pharyngeal cancer
The NAACCR cohort from 2007–2013 in this study contained 145,823 OC/P tumors and 57,805 laryngeal tumors (Table 1, Fig. 1a, Fig. S1). The eight NAACCR-reporting Appalachian states cover 67.6% of all Appalachian counties and 72.3% of the Appalachian population35. The OC/P and laryngeal cohorts reflect overall Appalachian demographics, with a higher percentage of white cases and lower percentage of ethnic minorities than non-Appalachia (Table 1). Except for lower CRS in Appalachian OC/P and laryngeal cancers, all other parameters were nearly identical to national averages in each disease (Table 1). The majority of OC/P and laryngeal cancer cases came from distressed or transitional Appalachian counties.
Identification of a survival disparity in white Appalachian male stage IV OC/P cancer. (a) flow diagram of procedures used to evaluate Appalachian-specific survival data. Boxes contain rationale and steps involved in the sequential stratification and cohort analysis for OC/P cancer. (b) Survival analysis of Appalachian OC/P cancer stratified by clinical stage. CRS values are plotted for each year after diagnosis, with 5-year (60 month) ratios evaluated across all AJCC-6 stages. (c) and (d). Survival analysis of white (c) and black (d) stage IV OC/P cancer stratified by Appalachian residency and sex. CRS values are plotted as in (b). Patient N, 5-year CRS with 95% CI and P values between significant groups are shown at the top of each graph; n.s., not significant. Black patients were evaluated for significance at 36 months due to lack of complete female survival by 48 months and beyond.
RS was measured among OC/P and laryngeal cancer patients by Appalachian or non-Appalachian residency at time of diagnosis (Fig. 1a, Fig S1). When stratified by American Joint Committee on Cancer (AJCC)-6 stages I to IV, stage IV patients had the highest patient numbers and lowest CRS for each disease (OC/P; n = 63,396/116,595, CRS = 45.5%; laryngeal; n = 14,963/51,362; CRS = 67.1%), and were selected for further evaluation (Fig. 1b and Fig. S1). OC/P and laryngeal patients were stratified by sex, race, and Appalachian residency (Fig. 1c, 1d; Fig S1). Survival analyses indicated that white Appalachian OC/P males (n = 5,014/53,696; CRS = 44.9%) displayed a significantly lower CRS compared to white non-Appalachian males (Fig. 1c; n = 37,843/53,696; CRS = 49.2%), P = 0.00001. In OC/P, there was no significant difference in CRS between white Appalachian females (n = 1,381/53,696; CRS = 43.1%) and white non-Appalachian females (n = 9,458/53,696; CRS = 43.7%) (Fig. 1c) or between black Appalachian males (n = 387/7,342; CRS = 35.8%) and black non-Appalachian males (n = 5,297/7,342; CRS = 34.4%) (Fig. 1d). Black Appalachian females (n = 116/7,342; 3-year RS = 46.1%) failed to reach 5-year follow-up for CRS comparison, but there was no observed significance between black non-Appalachian females at the latest available follow-up year (n = 1,540/7,342; 3-year RS = 34.8%) (Fig. 1d). No significant differences in CRS were observed for Appalachian laryngeal cancers between any sex or race (Fig. S1). White Appalachian males with stage IV OC/P were selected for further study due to their significantly different survival.
The survival disparity in white Appalachian male stage IV OC/P is specific to Appalachian counties within select Appalachian states
To elucidate whether the observed stage IV white Appalachian male OC/P survival disparity is specific to Appalachian counties within Appalachian states, patients were stratified by Appalachian or non-Appalachian county and state residency. Appalachian states-Appalachian counties (n = 5,014/42,857; CRS = 44.9%) displayed a significantly lower CRS compared to Appalachian states-non-Appalachian counties (n = 9,700/42,857; CRS = 47.9%), P = 0.00017 and non-Appalachian states (n = 28,143/42,857; CRS = 49.6%), P = 0.00032 (Fig. 2a and b). This indicates that the survival disparity is specifically due to cancer survival within Appalachia, not to contributions from adjacent non-Appalachian counties within Appalachian state borders. There was no significant difference in CRS between non-Appalachian states and Appalachian states-non-Appalachian counties (Fig. 2a).
State-level analysis of Appalachian OC/P survival. (a) Disparate stage IV white Appalachian male OC/P survival is restricted to Appalachian counties. Plotted CRS values over time are shown for non-Appalachian states (black), non-Appalachian counties within Appalachian states (green) and Appalachian counties within Appalachian states (red). Patient N, 5-year CRS with 95% CI and P values at 60 months are shown at the top of the graph; n.s., not significant. (b) Mapping of the denoted geographic regions evaluated in (a). (c) Select Appalachian states with significantly different CRS values contain a survival disparity in OC/P cancer. Appalachian states with significant CRS values are shown plotted over time. Patient N, 5-year CRS with 95% CI and P values at 60 months for states with significant survival differences between Appalachian and non-Appalachian counties are shown at the top of the graph. (d) Map of Appalachian region displaying states with significantly different stage IV white Appalachian male OC/P CRS. States with significant survival differences between Appalachian and non-Appalachian counties are in black, states with no available data are marked with a cross-hatched pattern.
To determine if the stage IV white Appalachian male OC/P survival disparity could be identified at the state level, patients within Appalachian counties were stratified by state residency. Significantly lower CRS was observed for Appalachian counties in Alabama (n = 626/31,423; CRS = 39.7%), P = 0.0006, Kentucky (n = 372/31,423; CRS = 39.6%), P = 0.0101, Pennsylvania (n = 1,695/31,423; CRS = 45.3%), P = 0.0047, and West Virginia (n = 587/31,423; CRS = 35.3%), P = 0.007 compared to non-Appalachian states (n = 28,143/31,423; CRS = 49.6%) (Fig. 2c,d). All other analyzed Appalachian states did not have a significantly different CRS (Supplementary Figure S2).
The white Appalachian male stage IV OC/P survival disparity is primarily found in HPV-associated oropharyngeal subsites
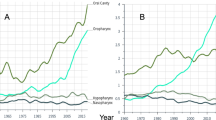
To determine if the stage IV white Appalachian male OC/P disparity is predominantly present within a specific OC/P subregion, cases were initially divided into non-HPV-associated or HPV-associated cancer subtypes for survival analysis. HPV-associated cancers frequently contain oral mucosa with HPV DNA, and are defined as HPV-associated oropharynx by the CDC33. Remaining subsites are not associated with HPV infection and represent non-HPV-associated cancer (Fig. 3a). First, we evaluated the CDC definitions as predictors of HPV infection in HNSCC by using patients in the TCGA cohort, where all patients have known HPV status. Importantly, patients were evaluated for overall survival, since HPV is not a variable collected by NAACCR and thus could not be assessed in the Appalachian cohort. TCGA patients were additionally stratified as HPV-associated and non-HPV-associated using subsite information, then compared to outcomes based on confirmed HPV status. HPV-positive white male stage IV TCGA patients have an undefined median survival with 5-year overall survival of 71%, whereas HPV-negative patients have median survival of 47.0 months and 5-year overall survival of 38% (Fig. 3b). Similarly, HPV-associated cancer patients from the same cohort have median survival of 68.4 months and 5-year overall survival of 61%, while non-HPV-associated patients have lower median survival of 30.1 months and 5-year overall survival of 37%. While the survival trends regarding HPV status are in general agreement between the NAACCR and TCGA cohorts, the difference in 5-year overall survival is greater when comparing HPV-positive (71%) and HPV-associated (61%) patients than between HPV-negative (38%) and non-HPV-associated (37%) patients. This suggests that the CDC non-HPV-associated subsite designation is a better predictor for HPV negativity than the HPV-associated subsite designation is for HPV positivity.
The stage IV white Appalachian male OC/P survival disparity is predominant in HPV-associated oropharyngeal cancers. (a) Schematic of the head and neck showing location of non-HPV-associated pharynx and oral cavity (blue), and CDC-defined HPV-associated oropharynx (green) cancers. A subset of the CDC-defined HPV-associated oropharynx is denoted in orange. ICD-O-3 site groups and corresponding site codes for each cancer type are indicated (see “Materials and methods” for detailed description of each site group). (b) Overall survival between confirmed and HPV-associated OC/P subtypes. Kaplan–Meier analysis of overall survival of stage IV white males from the TCGA HNSC cohort stratified by HPV status (HPV-positive or HPV-negative) or by ICD-O-3-coded HPV association (HPV-associated and non-HPV-associated). P values calculated using Mantel–Cox log-rank test, patient N, median and 5-year overall survival for each group are noted. (c) HPV status of OC/P subsites in stage IV white male TCGA patients. Percent of patients with confirmed HPV-negative (red) or HPV-positive (black) disease for each indicated subsite is shown. Denoted subsite groupings are indicated at the bottom as non-HPV-associated (blue), oropharynx (green), or other HPV-associated (orange). (d) The stage IV white Appalachian male OC/P survival disparity is present in HPV-associated oropharyngeal cancer. CRS values are plotted over time for stage IV white Appalachian male OC/P patients stratified by Appalachian and non-Appalachian counties and HPV- or non-HPV-associated status. (e–g) Stage IV white males stratified by HPV-associated oropharynx subsites. Oropharynx (e), tonsil (f), and tongue (g). CRS values are plotted over time with patients stratified by Appalachian counties (red), non-Appalachian counties (green) and non-Appalachian states (black). (h) Stage IV white males stratified by non-HPV-associated, oropharynx, other HPV-associated subsites and Appalachian residency. For (d–h), RS values are plotted over time with patients stratified by Patient N, 5-year CRS with 95% CI and P values at 60 months for each group are shown at the top of each respective graph; n.s., not significant.
To elucidate the discordance between HPV-positive and HPV-associated patients in explaining the observed Appalachian survival differences, TCGA patients were separated into non-HPV-associated cancers and into the specific HPV-associated oropharynx subsites, with the HPV status determined for each grouping. Using this breakdown, non-HPV-associated cancers are mostly HPV-negative, whilst HPV-associated cancers contain a mixture of HPV-positive and HPV-negative cases (Fig. 3c). These findings potentially explain the discrepancy between HPV-positive and HPV-associated outcomes, and also point to confounding factors such as tobacco/alcohol use or socioeconomic status (SES) that may exist in patient cohorts stratified solely on the basis of HPV-association.
White male stage IV cancer cases in the NAACCR cohort were next stratified by HPV-association and Appalachian residency. Appalachian patients with HPV-associated cancers (n = 3,449/33,281; CRS = 53.1%) displayed a significantly lower CRS compared to HPV-associated patients in non-Appalachian states (n = 20,399/33,281; CRS = 57.7%), P = 0.0118 (Fig. 3d). No significant difference was found in CRS between non-HPV-associated patients in Appalachian counties (n = 1,577/33,281; CRS = 29.5%) compared to non-HPV-associated patients in non-Appalachian states (n = 7,856/33,281; CRS = 30.9%). These results suggest that the stage IV white Appalachian male OC/P survival disparity is driven by lower survival of HPV-associated oropharyngeal patients. Appalachian states with the overall stage IV white Appalachian male OC/P survival disparity identified in Fig. 2c were evaluated separately and trended in a similar manner (Supplementary Fig. 3).
White male stage IV patients in the NAACCR cohort were subsequently stratified by each HPV-associated oropharyngeal region to delineate the contributions of each subsite on Appalachian survival. A significant difference in survival was observed between patients with primary oropharynx tumors in Appalachia (n = 325/2,807; CRS = 24.4%) and non-Appalachian counties (n = 649/2,807; CRS = 36.9%), P = 0.0399 as well as non-Appalachian states (n = 1,833/2,807; CRS = 37.5%), P = 0.0394 (Fig. 3e). No significant differences were observed between patients with primary tonsil tumors in Appalachia (n = 1,603/14,110; CRS = 55.8%) and non-Appalachia states (n = 9,240/14,110; CRS = 63.3%) or counties (n = 3,267/14,110; CRS = 59.6%). Similarly, no significant differences were observed between patients with primary tongue tumors in Appalachia (n = 1,579/14,345; CRS = 54.8%) and non-Appalachian states (n = 9,625/14,345; CRS = 55.9%) or counties (n = 3,141/14,345; CRS = 55.1%) (Fig. 3f,g). Other oropharyngeal subsites lacked sufficient numbers for individual analysis but were combined with tonsil and (base of) tongue to generate an “other HPV-Associated” subset (Fig. 3a).
Regarding survival driven by oropharyngeal tumors, “other HPV-associated” and non-HPV-associated stratification reveals three distinct outcomes in non-Appalachia (Fig. 3h): “Other HPV-associated” (n = 18,627/33,347; CRS = 59.7%) has the best outcome, followed by oropharynx (n = 1,833/33,347; CRS = 37.5%), then non-HPV-associated (n = 7,856/33,347; CRS = 30.9%). However, in Appalachia, only two distinct outcomes are revealed: “Other HPV-associated” (n = 3,129/33,347; CRS = 56.7%) has the best outcome, whereas oropharynx (n = 325/33,347; CRS = 24.4%), has a statistically similar outcome to non-HPV-associated (n = 1,577/33,347; CRS = 29.5%) (Fig. 3h). These results suggest that the observed Appalachian survival disparity is largely driven by lower survival in the oropharynx subregion of HPV-associated stage IV male oropharyngeal patients.
High Appalachian tobacco use correlates with stage IV white Appalachian male OC/P outcomes
The lower survival of stage IV white Appalachian male HPV-associated cancer compared to confirmed HPV-positive national cases implies that risk factors other than HPV are driving poorer survival in the Appalachian cohort (Fig. 3b). Since smoking and smokeless tobacco use are highest in Appalachia19, and current smoker prevalence rates have remained higher in West Virginia (a state with all counties within Appalachia) than the nation for several decades36 (Fig. 4a), we evaluated tobacco use prevalence rates in Appalachian states from state BRFSS registries stratified by Appalachian residency and sex using data from 2016 as a representative year.
High combined tobacco use in stage IV white Appalachian male OC/P disparity states. (a) Overall smoking prevalence rates in West Virginia (as a representation of Appalachia, gold) compared to the United States (grey). Years 2011–2018 cannot be directly compared to earlier years due to differences in weighting methodology across time. (b) Elevated tobacco use by Appalachian males (top) and females (bottom). Data for 2016 smoking weighted prevalence estimates for males in Appalachian states stratified by county status (blue, Appalachian counties; gold, non-Appalachian counties; grey, non-Appalachian states), smoking (solid) or smokeless (cross-hatched) tobacco use with P values calculated using G-test for independence without Yates’ correction are shown at top. Error bars represent 95% CI. (c) Combined Appalachia county smoking (solid) and smokeless (cross-hatched) weighted prevalence estimates with P value calculated using G-test for independence without Yates’ correction are shown above significant results. (d) Mapping of Appalachian counties smokeless tobacco use. Appalachian counties with significantly higher (red) and non-significantly different (black) smokeless tobacco use are shown compared to non-Appalachian counties within each respective Appalachian state. West Virginia (hatched; non-applicable) lacks non-Appalachian counties for comparison.
In males, smoking prevalence rates were significantly higher in Appalachian counties compared to non-Appalachian counties, P = 4.441e−16 and non-Appalachian states, P < 2.2e−16. Specifically, male smoking prevalence rates were significantly higher in Appalachian counties compared to non-Appalachian counties in Kentucky (P = 2.80e−04), New York (P = 2.55e−04), Ohio (P = 0.02965), and Virginia (P = 0.008904) (Fig. 4b, top). Male smokeless tobacco prevalence rates were significantly higher in Appalachian counties compared to non-Appalachian counties, P < 2.2e−16 and non-Appalachian states, P < 2.2e−16. Specifically, male smokeless tobacco prevalence rates were significantly higher in Appalachian counties compared to non-Appalachian counties in Kentucky (P < 2.2e−16), New York (P = 9.03e−7), North Carolina (P = 5.44e−6), Ohio (P = 1.87e−12), South Carolina (P = 0.00419), and Virginia (P = 6.63e−9). Significant differences were also observed in Maryland and Pennsylvania, but these regions had fewer than 50 respondents.
West Virginia contains no non-Appalachian counties for comparison, but the smoking tobacco rate of 25.8% and the smokeless tobacco rate of 15.9% are similar to that of other states with significant differences in the region (Fig. 4b, top). Interestingly, except for Alabama, states with significantly higher male smokeless tobacco use include every state with a significant difference in CRS for stage IV white Appalachian male OC/P and HPV-associated cancers (Fig. 4d).
In females, average smoking prevalence rates were higher in Appalachian counties compared to non-Appalachian counties, P < 2.2e−16, and non-Appalachian states, P < 2.2e−16 (Fig. 4b, bottom) and were statistically different than that of Appalachian males (20.7% males vs. 19.2% females) (Fig. 4c). Smoking prevalence rates were significantly higher in Appalachian counties compared to non-Appalachian counties in Alabama (P = 1.5e−4), Kentucky (P = 1.33e−9), Maryland (P = 0.02284), New York (P = 3.4e−9), Ohio (P = 9.38e−5), Pennsylvania (P = 1.44e−4), and Virginia (P = 0.0362). Female smokeless tobacco prevalence rates were significantly higher in Appalachian counties compared to non-Appalachian counties, P = 8.76e−4 and non-Appalachian states, P = 2.912e−05, and were significantly different than that of Appalachian males (10.9% males vs. 1.2% females), P < 2.2e−16 (Fig. 4c). Significant differences were observed in Kentucky and Maryland, although these regions had fewer than 50 respondents. These data indicate that significantly higher smoking and smokeless tobacco rates within Appalachia correlate with disparate survival of stage IV white Appalachian male OC/P patients in most Appalachian disparity states.
Discussion
With a majority of population and geographical coverage, our direct, non-exploratory analysis of HNSCC survival from available states representing all ARC-defined Appalachian subregions16 identifies stage IV white Appalachian males with OC/P cancer as having lower cancer-related survival compared to non-Appalachian males. Based on available data, this stage- and sex-specific disparity is manifest within the Appalachian areas of four states. The disparity is restricted to the ICD-O-3 defined oropharynx (C10.0–10.9) within the CDC-defined HPV-associated oropharyngeal region. This disparity predominantly occurs in states with significantly higher male smoking and smokeless tobacco use, consistent with high risk-factor exposure in Appalachia known to contribute to increased HNSCC incidence22,24,25.
Previous work evaluating Appalachian oral cancer survival using 2004 SEER data identified lower combined male and female survival in 10 states without consideration of stage or race25. When stratified by stage, race and sex, our multivariate analysis indicates that the only significant difference in CRS occurs in white Appalachian stage IV male OC/P patients diagnosed under AJCC-637. RS values for stage IV white Appalachian and non-Appalachian male OC/P are higher than that of black males or black females, in agreement with a recognized national survival disparity for black OC/P cancers attributed to lower overall SES and cultural barriers9,11. In addition, female white stage IV OC/P patients have a lower CRS than male white stage IV OC/P regardless of Appalachian status, likely reflecting the lower rate of HPV-positive oropharyngeal disease in females38,39. Emerging national trends indicate that HPV-associated oropharyngeal cancers occur more frequently in younger, white male patients with limited tobacco use, and within rural areas4,14,38. These factors, coupled with blacks and white females having higher percentages of HPV-negative disease12,13 are congruent with HPV-positive and/or HPV-associated white male oropharyngeal patients having higher survival at the national level.
OC/P patient populations consist of a mixture of HPV-negative and HPV-positive disease. White Appalachian males have a higher incidence in HPV-positive OC/P cancers, which would be expected to result in increased survival. However, stage IV-matched CRS for white Appalachian males is closer to the lower survival prevalence rates observed for white females than for non-Appalachian white males (Fig. 1c). Furthermore, male Appalachian stage IV HPV-associated oropharynx patients exhibit outcomes similar to non-HPV-associated male stage IV disease within and outside of Appalachia. Of the recognized cancer risk factors endemic to Appalachia, age at diagnosis, travel distance to critical care centers and disproportionate presentation of patients at stage IV have been reported to have no impact on survival in Appalachian subpopulations40,41 (Table 1). However, increased smoking and smokeless tobacco use within Appalachia have been linked to low SES and increased HNSCC incidence42,43,44, and thus are presumably factors contributing to the decreased male stage IV oropharyngeal cancer survival.
Our findings also indicate that Appalachian smoking tobacco prevalence rates, represented by West Virginia, have been higher than national averages for several decades (Fig. 4a), and while Appalachian smoking tobacco use is significantly higher in males and females, smokeless tobacco use is primarily higher in males compared to national averages (Fig. 4b). Smokeless tobacco use has been specifically linked to increased cancer risk in OC/P and oropharyngeal subsites45,46,47,48. Elevated smoking and smokeless tobacco use in most Appalachian states with significantly different CRS corresponds with decreased CRS in white male stage IV OC/P cancers. This has the potential to result in the male patient population garnering a higher percentage of tobacco-induced HPV-negative disease at all oral subsites, including the oropharynx. While the lack of diagnostic HPV detection by p16 staining as a variable in the NAACCR cohort precludes direct analysis of viral status, segregation of white male stage IV OC/P patients by non-HPV-associated OC/P and HPV-associated oropharynx indicates that HPV-associated oropharyngeal cancer is responsible for the Appalachian male stage IV disparity within ICD-O-3 oropharynx codes. The predominant subsites under this delineation are C10.0 (vallecula), C10.1 (anterior surface of epiglottis), C10.2 (lateral wall of oropharynx), C10.3 (posterior wall of pharynx) and C10.4 (branchial cleft). These oropharyngeal regions exclude palatine and lingual tonsils, as well as most other HPV-associated oropharyngeal sites containing the reticular epithelium lining the tonsillar crypts that constitute the primary sites of oropharyngeal HPV infection and neoplasia49,50,51. Higher aggregate tobacco use by Appalachian males may therefore result in more frequent carcinogen-induced malignancy at oropharynx subsites, resulting in a greater percentage of HPV-negative disease in the oropharynx and corresponding worse CRS, similar to that observed in other tobacco-heavy oropharyngeal cohorts3,52,53.
The Appalachian-specific OC/P and oropharynx-specific disparities found in this study were diagnosed under the AJCC-6 timeframe, having clinical and pathological staging guidelines independent of HPV status. Restaging of HPV-positive oropharynx in 2018 under AJCC-8 due to the favorable prognosis of HPV-positive oropharyngeal cancers would shift most HPV-positive oropharynx in AJCC-6 to lower clinical stages. Such restaging would have the effect of potentially eliminating or downstaging the Appalachian male disparity described in this report. However, an additional ramification from this work is that male Appalachian or other populations with multi-factorial oropharyngeal tobacco exposure may actually be under-staged using current AJCC-8 guidelines. This is an important consideration, since National Comprehensive Cancer Network (NCCN) guidelines recommend staging and treatment dependent on p16 status52,54, where treatment de-escalation of HPV-associated oropharyngeal cancers continues to be evaluated55,56,57. Future efforts towards definitively determining the extent of HPV involvement in Appalachian OC/P and oropharynx through comprehensive p16 staining and PCR58 will be required to better clarify the predominant factors underlying oropharynx-driven, stage-based disparities in past and future Appalachian cohorts.
Limitations and implications
While the current study covers the majority of the Appalachian population and is the most comprehensive study of HNSCC survival of the region to date, lack of available qualified survival data from five Appalachian states prevented complete assessment of survival in the region. The association between heavy aggregate tobacco use and poor male stage IV oropharyngeal survival in states with significantly different CRS suggests that stage IV oropharyngeal patients from states with Appalachian regions containing similar tobacco use patterns may also harbor disparate outcomes. Our findings, in conjunction with the poor survival of HPV-positive oropharynx patients with high smoking histories59,60, underscore the need for the comprehensive tobacco history of any HPV-positive oropharynx patient to be considered prior to treatment. In addition to smoking, the significantly higher smokeless tobacco use by the male Appalachian population further increases the risk of tobacco-induced cancer at HPV-associated oropharyngeal sites, leading to oropharynx tumors that are either HPV-negative, or are HPV-positive but exhibit aggressive HPV-negative tumor behavior.
Conclusions
These findings provide novel and in-depth insight into a specific demographic within a chronically underserved, rural population that is at higher clinical risk for poor OC/P outcome. Persistent high tobacco usage in Appalachia, in spite of increased tobacco cessation efforts, reinforces the need for continued targeted risk awareness. This includes emerging forms of supposed safer nitrosamine-containing products, such as e-cigarettes, that contribute to the cumulative patient tobacco load61. Inclusion of patient use of these products as standard registry variables should be considered for improved monitoring of tobacco-related disparities in future populations. The described male oropharynx disparity may also be present in male patients from other regions with heavy smoking, smokeless tobacco62 or betel-quid63 use, and should be monitored accordingly for similar poor survival and continued cessation intervention policies.
Data availability
The data that support the findings of this study are available upon application and IRB approval from NAACCR, thus these data are not in the public domain. Data are however available from the authors upon reasonable request and with prior approval by NAACCR.
References
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2020. CA. Cancer J. Clin. 70, 7–30 (2020).
Chow, L. Q. M. Head and neck cancer. N. Engl. J. Med. 382, 60–72 (2020).
Ang, K. K. et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N. Engl. J. Med. 363, 24–35 (2010).
LeHew, C. W. et al. The health system and policy implications of changing epidemiology for oral cavity and oropharyngeal cancers in the United States from 1995 to 2016. Epidemiol. Rev. 39, 132–147 (2017).
Rettig, E. M. & D’Souza, G. Epidemiology of head and neck cancer. Surg. Oncol. Clin. N. Am. 24, 379–396 (2015).
Maxwell, J. H., Grandis, J. R. & Ferris, R. L. HPV-associated head and neck cancer: unique features of epidemiology and clinical management. Annu. Rev. Med. 67, 91–101 (2016).
Marur, S. & Forastiere, A. A. Head and neck squamous cell carcinoma: update on epidemiology, diagnosis, and treatment. Mayo Clin. Proc. 91, 386–396 (2016).
Spence, T., Bruce, J., Yip, K. W. & Liu, F.-F. HPV-associated head and neck cancer. J. Natl. Cancer Inst. 107, djv344 (2015).
Goodwin, W. J. et al. Unequal burden of head and neck cancer in the United States. Head Neck 30, 358–371 (2008).
Morse, D. E. & Kerr, A. R. Disparities in oral and pharyngeal cancer incidence, mortality and survival among black and white Americans. J. Am. Dent. Assoc. 137, 203–212 (2006).
Daraei, P. & Moore, C. E. Racial disparity among the head and neck cancer population. J. Cancer Educ. 30, 546–551 (2015).
Settle, K. et al. Racial survival disparity in head and neck cancer results from low prevalence of human papillomavirus infection in black oropharyngeal cancer patients. Cancer Prev. Res. 2, 776–781 (2009).
Saba, N. F. et al. Gender and ethnic disparities in incidence and survival of squamous cell carcinoma of the oral tongue, base of tongue, and tonsils: a surveillance, epidemiology and end results program-based analysis. Oncology 81, 12–20 (2011).
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2019. CA. Cancer J. Clin. 69, 7–34 (2019).
Zahnd, W. E. et al. Rural-Urban differences in cancer incidence and trends in the United States. Cancer Epidemiol. Biomark. Prev. 27, 1265–1274 (2018).
The Appalachian Region—Appalachian Regional Commission (Accessed 5 May 2020); https://www.arc.gov/appalachian_region/TheAppalachianRegion.asp.
Pollard, K. & Jacobsen, L. A. The Appalachian Region: A Data Overview from the 2013–2017 American Community Survey. Appalachian Regional Commission (Accessed 8 August 2019); https://www.arc.gov/research/researchreportdetails.asp?REPORT_ID=159.
Viens, L. J. et al. Human papillomavirus-associated cancers—United States, 2008–2012. MMWR. Morb. Mortal. Wkly. Rep. 65, 661–666 (2016).
Behavioral Risk Factor Data: Tobacco Use (2011 to present) | Chronic Disease and Health Promotion Data & Indicators (Accessed 1 May 2020); https://chronicdata.cdc.gov/Survey-Data/Behavioral-Risk-Factor-Data-Tobacco-Use-2011-to-pr/wsas-xwh5.
Knox, S., Basu, S. & Remick, S. A systems approach to cancer health disparities in Appalachia. Austin J Public Heal. Epidemiol 1, 1004 (2014).
Paskett, E. D. et al. Disparities in underserved white populations: the case of cancer-related disparities in Appalachia. Oncologist 16, 1072–1081 (2011).
Lengerich, E. J. et al. Cancer incidence in Kentucky, Pennsylvania, and West Virginia: disparities in Appalachia. J. Rural Heal. 21, 39–47 (2004).
Reiter, P. L. et al. Assessing the burden of HPV-related cancers in Appalachia. Hum. Vaccines Immunother. 9, 90–96 (2013).
Wilson, R. J., Ryerson, A. B., Singh, S. D. & King, J. B. Cancer incidence in Appalachia, 2004–2011. Cancer Epidemiol. Biomarkers Prev. 25, 2004–2011 (2016).
Casto, B. C. et al. Oral cancer in Appalachia. J. Health Care Poor Underserved 20, 274–285 (2009).
County Economic Status in Appalachia, FY 2017. Appalachian Regional Commission (Accessed 5 May 2020); https://www.arc.gov/research/MapsofAppalachia.asp?MAP_ID=116.
Cancer in North America: 2009–2013, Volume Four: Cancer Survival in the United States and Canada 2006–2012. (2013).
Measures of Cancer Survival (Accessed 1 May 2020); https://surveillance.cancer.gov/survival/measures.html.
Cartographic Boundary Files—Shapefile | US Census Bureau (Accessed 4 April 2019); https://www.census.gov/geographies/mapping-files/time-series/geo/carto-boundary-file.2014.html.
QGIS Development Team. QGIS Geographic Information System. Open Source Geospatial Foundation Project (Accessed 4 April 2019); https://qgis.osgeo.org.
Cerami, E. et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2, 401–404 (2012).
Gao, J. et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal. 6, 1–34 (2013).
Van Dyne, E. A. et al. Trends in human papillomavirus-associated cancers—United States, 1999–2015. MMWR. Morb. Mortal. Wkly. Rep. 67, 918–924 (2018).
Centers for Disease Control and Prevention (CDC). Methodologic changes in the behavioral risk factor surveillance system in 2011 and potential effects on prevalence estimates. MMWR. Morb. Mortal. Wkly. Rep. 61, 410–413 (2012).
Annual Estimates of the Resident Population: April 1, 2010 to July 1, 2016 | 2015 Population Estimates | US Census Bureau. https://factfinder.census.gov/faces/tableservices/jsf/pages/productview.xhtml?src=bkmk.
West Virginia Behavioral Risk Factor Surveillance System. https://www.wvdhhr.org/bph/hsc/pubs/Topics/default.shtml.
Lydiatt, W. M. et al. Head and Neck cancers-major changes in the American Joint Committee on cancer eighth edition cancer staging. CA Cancer J. Clin. 67, 122–137 (2017).
Mahal, B. A. et al. Incidence and demographic burden of HPV-associated oropharyngeal head and neck cancers in the United States. Cancer Epidemiol. Biomark. Prev https://doi.org/10.1158/1055-9965.epi-19-0038 (2019).
Lawrence, M. S. et al. Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature 517, 576–582 (2015).
Ringstrom, M. J. et al. Travel distance: impact on stage of presentation and treatment choices in head and neck cancer. Am. J. Otolaryngol. Head Neck Med. Surg. 39, 575–581 (2018).
Gleich, L. L. et al. Therapeutic decision making in stages III and IV head and neck squamous cell carcinoma. Arch. Otolaryngol. Neck Surg. 129, 26 (2003).
Stanford-Moore, G. et al. Interaction between known risk factors for head and neck cancer and socioeconomic status: the Carolina Head and Neck Cancer Study. Cancer Causes Control 29, 863–873 (2018).
Welte, J. W., Barnes, G. M., Tidwell, M. C. O. & Hoffman, J. H. Tobacco use, heavy use, and dependence among adolescents and young adults in the United States. Subst. Use Misuse 46, 1090–1098 (2011).
Nemeth, J. M. et al. Factors influencing smokeless tobacco use in rural Ohio Appalachia. J. Community Health 37, 1208–1217 (2012).
Lee, P. N. & Hamling, J. Systematic review of the relation between smokeless tobacco and cancer in Europe and North America. BMC Med. 7, 36 (2009).
Zhou, J. et al. Smokeless tobacco and risk of head and neck cancer: evidence from a case-control study in New England. Int. J. Cancer 132, 1911–1917 (2013).
Wyss, A. B. et al. Smokeless tobacco use and the risk of head and neck cancer: pooled analysis of US studies in the inhance consortium. Am. J. Epidemiol. 184, 703–716 (2016).
Mello, F. W. et al. The synergistic effect of tobacco and alcohol consumption on oral squamous cell carcinoma: a systematic review and meta-analysis. Clin. Oral Investig. 23, 2849–2859 (2019).
Gillison, M. L. Evidence for a causal association between human papillomavirus and a subset of head and neck cancers. J. Natl. Cancer Inst. 92, 709–720 (2000).
Kim, S. H. et al. HPV integration begins in the tonsillar crypt and leads to the alteration of p16, EGFR and c-myc during tumor formation. Int. J. Cancer 120, 1418–1425 (2007).
Thavaraj, S. et al. Patients with HPV-related tonsil squamous cell carcinoma rarely harbour oncogenic HPV infection at other pharyngeal sites. Oral Oncol. 50, 241–246 (2014).
Kompelli, A. R. et al. Prognostic impact of high-risk pathologic features in HPV-related oropharyngeal squamous cell carcinoma and tobacco use. Otolaryngol. Head Neck Surg. (U.S.) 160, 855–861 (2019).
Grønhøj, C. et al. Impact on survival of tobacco smoking for cases with oropharyngeal squamous cell carcinoma and known human papillomavirus and p16-status: a multicenter retrospective study. Oncotarget 10, 4655–4663 (2019).
National Comprehensive Cancer Network. Head and Neck Cancers (Version 3.2019) (Accessed 10 September 2019); https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf.
Oosthuizen, J. C. & Kinsella, J. B. Is treatment de-escalation a reality in HPV related oropharyngeal cancer?. Surgeon 14, 180–183 (2016).
White, R. et al. Practice patterns and outcomes following radiation dose de-escalation for oropharyngeal cancer. Laryngoscope https://doi.org/10.1002/lary.28083 (2019).
Oosthuizen, J. C. & Doody, J. De-intensified treatment in human papillomavirus-positive oropharyngeal cancer. Lancet 393, 5–7 (2019).
Rietbergen, M. M. et al. Molecular characterization of p16-immunopositive but HPV DNA-negative oropharyngeal carcinomas. Int. J. Cancer 134, 2366–2372 (2014).
Haigentz, M. et al. Understanding Interactions of smoking on prognosis of HPV-associated oropharyngeal cancers. Adv. Ther. 35, 255–260 (2018).
Liskamp, C. P. et al. Adverse effect of smoking on prognosis in human papillomavirus-associated oropharyngeal carcinoma. Head Neck 38, 1780–1787 (2016).
Cummings, K. M. et al. E-cigarettes and cancer patients. J. Thorac. Oncol. 9, 438–441 (2014).
Critchley, J. A. & Unal, B. Health effects associated with smokeless tobacco: a systematic review. Thorax 58, 435–443 (2003).
Tangjaturonrasme, N., Vatanasapt, P. & Bychkov, A. Epidemiology of head and neck cancer in Thailand. Asia. Pac. J. Clin. Oncol. 14, 16–22 (2018).
Acknowledgments
This work was supported by pilot grants from NIH U54GM104942 and P20GM103434 (B. Papenberg, J. Allen, S. Markwell, S. Weed). Mr. Johnson was supported with Federal funds from the National Cancer Institute, NIH, HHS, under Contract No. HHSN261201800006I and the Centers for Disease Control and Prevention, HHS, under Cooperative Agreement 1NU58DP006270. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH or the Centers for Disease Control and Prevention. Additional support was provided by the West Virginia University Department of Biochemistry (S. Markwell, S. Weed), Department of Otolaryngology, Head and Neck Surgery (E. Interval, P. Montague) and by Transition Grant Support from the Office of Research and Graduate Education, West Virginia University Health Sciences Center (B. Papenberg, J. Allen, S. Weed). We thank Recinda Sherman (NAACCR) and Information Management Services for assistance with IRB and database preparation, Sijin Wen (West Virginia University School of Public Health) for biostatistical validation, Pamela Moats (West Virginia University Cancer Institute Registry) and Alana Hudson (West Virginia Cancer Registry) for early conceptual help.
Author information
Authors and Affiliations
Contributions
B.W.P: Conceptualization, writing—original draft, data curation, formal analysis, and investigation. J.L.A.: Conceptualization and writing–review and editing. S.M.M.: Conceptualization, writing—review and editing. E.T.I.: Conceptualization and writing—review and editing. P.A.M.: writing—review and editing. C.J.J.: Conceptualization, writing—review and editing, supervision, methodology, software, and resources. S.A.W.: Funding acquisition, conceptualization, project administration, writing—original draft, review, editing and project supervision.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Papenberg, B.W., Allen, J.L., Markwell, S.M. et al. Disparate survival of late-stage male oropharyngeal cancer in Appalachia. Sci Rep 10, 11612 (2020). https://doi.org/10.1038/s41598-020-68380-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-68380-w
- Springer Nature Limited
This article is cited by
-
A systematic review and meta-analysis of e-cigarette use among cancer survivors
Journal of Cancer Survivorship (2024)