Abstract
Host biological factors and habitat influence the faunal assemblages and biodiversity worldwide, including parasite communities of vertebrate and invertebrate hosts. The ecological relationship between hosts and parasites can be mediated by interaction of host’s biological factors, as their physiological condition, diet and size, with the environmental components, somehow influencing the features of parasite infection in host populations. Here, we used boosted regression tree models to study the parasite communities of two sympatric sparid fishes, the salema Sarpa salpa and the white seabream Diplodus sargus, to investigate the role of specific host’s traits in two contiguous coastal areas along the southern-western Tyrrhenian coast of Italy characterized by different degree of deterioration. Results showed that overall and across all parasite groups (ecto-, endo- and ecto- plus endo-parasites), sampling localities were the most important predictors of abundance, species richness, and diversity for salema. Moreover, seasonality was the main predictor of endo-parasite abundance, while size-related factors explained most of the variation in species richness and diversity. In the white seabream, size-related factors and reproductive cycle-related factors were the most important predictors for the overall parasite abundance and parasite richness, respectively. Our findings suggest that the parasite community of salema and white seabream responded differently to specific biological factors, highlighting how the environmental conditions under which they live may exert a strong influence on the parasite communities of each host fish.
Similar content being viewed by others
Introduction
The parasite community of marine hosts is influenced by the interactions that occur between hosts, parasites, and their environment. Biological (or biotic) factors affecting the parasite community structure in a given host and locality may be altered by several stressors of both natural and anthropogenic nature1,2,3,4,5. High impact due to human activities can cause changes to the assemblages and biodiversity of coastal fauna, including parasite community of vertebrate and invertebrate hosts1,2,3,4,6,7. Metazoan parasites are considered to be sensitive to environmental stress and potential bio-indicators of water quality and environment stability. Their community’s descriptors have been considered as effective indicators that reflect habitat alterations and have been used to evaluate the environment health status and anthropogenic impact in coastal habitats2,3,4,8,9,10,11,12,13.
The Gulf of Naples and Gulf of Salerno are both located along the Tyrrhenian coast of southern Italy. The first area is a semi-closed basin affected by significant degradations, especially along the coast. The strong pressure due to high population density and agricultural and industrial activities has resulted in the general deterioration of the marine environment by urban and industrial sewages and river discharges14,15,16. In particular, on its south-eastern part, the basin is strongly affected by the pulsing runoff of the Sarno River, which is considered the most polluted European river featuring a mix of sewage and untreated agricultural and industrial wastes and chemicals14,15. Its sediments cause eutrophication that in turn can enhance changes in the composition of coastal fauna and its food web 14,15. The communication with the southern contiguous basin of the Gulf of Salerno, which in comparison with the Gulf of Naples shows a reduced anthropogenic pressure14,15,16, is through the passage between the Island of Capri and the Sorrento Peninsula.
The salema Sarpa salpa and the white seabream Diplodus sargus (Sparidae) are demersal and sympatric species that inhabit coastal rocky reef areas and Posidonia oceanica meadows. Both species are among the most common and abundant sparid fishes in shallow waters of the Mediterranean Sea, so that they could easily be used as sentinels of environment stability. Moreover, because they show different feeding ecology, the simultaneous study of their parasite communities provides the opportunity to obtain information from different trophic levels. Salema is largely herbivorous grazing on aquatic plants17, in contrast, invertebrates and different algal species compose the diet of the white seabream18. To our knowledge, no study has been conducted on the whole metazoan parasite community of salema and white seabream from the Tyrrhenian Sea or in other sparid fishes from off southern Italian coast.
Herein, we use boosted regression tree models (BRTm) to investigate the influence of biological factors and geographic localities on the descriptors of parasite communities in salema and white seabream in two contiguous basins characterized by different ecosystems and degrees of alteration along the southern-western Tyrrhenian coast of Italy. Our results show that biological factors and geographic locality affect the parasite community of the two fishes, supporting the idea that the deterioration of ecosystems may play an important role on fish hosts that, in turn, could be used as biological indicators.
Methods
Study area
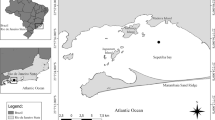
For comparative purposes, two areas known to have different degree of deteriorations due to human impact have been selected for this study: the first area is located along the coastline between Vico Equense and Massa Lubrense in the Gulf of Naples, and the second between Recommone Bay and the Rock of Isca in the Gulf of Salerno (Fig. 1). The two areas will further be referred to as GN (Gulf of Naples) and GS (Gulf of Salerno), respectively. GN and GS are located along the Tyrrhenian Sea, Campania region, (southern Italy), separated by the Sorrento Peninsula. GN is strongly influenced by heavy pollution due to anthropogenic impact14,15,16, while GS is located just outside the marine protected area of Punta Campanella, where the anthropogenic pressure is known to be strongly reduced14.
Collection and fish examination
All the fish specimens were collected during normal fishery procedures with no additional experimental catches being performed. According to Italian law DL16/92 and European directive 2010/63/EU, this study did not require a specific permit. Procedures for this study were performed in accordance with the guide for the care and use of animals by the Italian Ministry of Health.
Between February 2017 and October 2018, a total of 242 individual fish of two species of Sparidae were collected by net at benthic depths ranging from 15 to 30 m. Sampling included 64 and 56 specimens of white seabream from GN and GS, respectively and 60 and 62 specimens of salema from GN and GS, respectively. However, due to extreme weather events in the GS, it was not possible to obtain white seabream individuals in winter 2017. For this latter reason, the timing factors including season (winter, spring, summer and autumn) and year have been not considered in the statistical analysis for the white seabream.
Fish were weighed to the nearest 0.1 g and measured (fork length-FL) to nearest 0.1 cm; sex was determined before parasitological study by gonadal examination19. A macroscopic gonadal maturity score (GMS) was recorded to investigate the phase of the reproductive cycle (1 = inactive; 2 = developing; 3 = ripe; 4 = post spawning)19. Body condition index (BCI, whole weight/fork length3) was calculated as described by Le Cren20. The gonadosomatic (GSI, gonad weight/host eviscerated weight × 100) and hepatosomatic indices (HSI, liver weight/host eviscerated weight × 100) were calculated as suggested by Mouine et al.19.
The skin, musculature, gills, mouth cavity, digestive tract, liver, heart, gonads, visceral cavity and mesenteries of each fresh individual fish were examined under a dissecting microscope for parasites. For each organ, ecto- and endo-parasites were collected, counted, washed in physiological saline solution, and preserved in 70% ethanol21. For identification, crustaceans and nematodes were clarified in 20% potassium hydroxide and Amman’s lactophenol respectively, and then returned to 70% ethanol, trematodes were stained with Mayer’s acid carmine and mounted in Canada balsam. Parasites were studied by a compound microscope.
Descriptors of parasite community
A component community comprises all the species of parasites recovered from a sample of a particular host species, while infracommunity refers to the assemblage of parasites in one host individual. Prevalence was defined as the number of hosts infected with 1 or more individuals of a particular parasite species; parasite species with prevalence higher than 10% in any of the host samples will further be referred to as common. Abundance was measured as the number of individuals of a particular parasite in/on a single host regardless of whether or not the host is infected; intensity was the number of individuals of a particular parasite species in a single infected host22.
The mean total abundance, species richness and the Simpson and Shannon–Wiener indices of diversity were used as overall descriptors of infracommunities for each host species examined. Mean total abundance was measured as the mean number of individuals of all parasite species, and species richness the number of parasite species harboured by each individual fish. We used both diversity indices because Simpson's diversity index is able to detect differences in abundant species, whilst the Shannon–Wiener diversity index detects differences in rare species23. Descriptors of community were compared between hosts using the Mann–Whitney U- test.
Parasite species by host specificity were classified as ‘‘specialists’’, defined narrowly as having the bulk of reproducing adults found only in a single host species or having been reported from a single host species and ‘‘generalists’’ when reported from a variety of related host species.
Statistical analysis
We used BRTm to assess the influence of host biological factors (sex, weight, FL, GMS, BCI, GSI, and HSI), timing (sampling season and year, available only for salema) and sampling areas (which we considered as proxies of different abiotic conditions) on parasites abundance, species richness (including zeros22), and diversity, measured by Shannon and Simpson indices23. BRT models are characterized by a great flexibility in model evaluation, allowing for non-linear responses, collinear predictors, and variable interactions24. Since sampling in salema took a longer period of time encompassing different seasons and years, we included those variables into the BRTm. To avoid biases in model fitting due to possible interactions between different predictors (e.g. sampling year and season in salema), we re-ran BRTm for salema by excluding those terms. Comparative analyses were conducted on gastro-intestinal plus liver parasites (endo-parasites), and external parasites (ecto-parasites; from gills and skin) separately, and then on all parasites (endo- plus ecto-parasites). Parasite descriptors (total abundance, species richness, Shannon and Simpson indices) were subdivided in total, endo- and ecto-parasites, and models were trained on all groups by means of the ‘gbm.step’ function in the ‘dismo’ package of R25.
Models were trained by using common values of learning rate, step size and bag fraction (i.e. the proportion of observations used in selecting variables24), allowing for interactions among covariates. To avoid overfitting due to the limited amount of data (nSarpa = 90, nDiplo = 102), the optimal number of trees was chosen by means of a cross-validation procedure (max trees = 10,000). The relative importance of each predictor was determined by measuring the increase in model fitting after accounting for a given predictor, scaled in a 0–100% scale where larger values correspond to larger (relative) influence in model fitting26. The relative influence of each predictor on the abundance/richness and diversity indices was measured by means of partial dependence plots visualizing the relationship between predictor and the fitted function. Model accuracy was examined using the Spearman’s rank correlation values between fitted and observed abundance, richness and diversity values for each parasite group. The strength of interaction effects in BRT models was measured by means of the H-statistic, ranging between 0 and 1, with higher values corresponding to larger interaction effects24.
Results
Host and parasite data
Biological data (including sex, weight, BCI, GMS, GSI, and HIS) of salema and white seabream individuals according to sampling localities are reported in Table 1. A total of 3,861 individual parasites belonging to 20 taxa (11 in white seabream and 10 in salema) were identified in two host species. Only larval forms of the isopod Gnathia sp. were found in both host species. Basic parameters of infection for each parasite taxon from both localities are presented in Table 2. All ecto-parasites were obtained from the gills, and Gnathia sp. from the skin also, while endo-parasites were obtained from the intestine, except for larvae of Hysterothylacium sp. collected from the liver. The clearly predominant group of parasites with respect to species diversity was the Digenea (8 species) followed by Monogenea (4 species). The other groups were represented by fewer species: Copepoda (3 species), Hirudinidae and Nematoda (2 species each), and Isopoda (1 species). The local parasite fauna showed a low representation of larval parasite stages (2 species: one nematode and one isopod). Only five parasite species in white seabream and four species in salema were present from both localities (Table 2).
Out of the 1,481 parasite specimens found in white seabream, 567 (38.2%) were ecto-parasites and 914 (61.8%) endo-parasites. Adult parasites of white seabream were all generalist in Sparidae. Out of the 2,380 parasite specimens found in salema, 468 (19.6%) were ecto-parasites and 1,912 (80.4%) endo-parasites. Adult parasites of salema were all specialist species. Overall prevalence of infection was 71.6% and 88.5% in white seabream and salema, respectively.
In white seabream, the most prevalent and abundant species was Lamellodiscus ignoratus (Monogenea), while in general endo-parasites showed low prevalence and abundance (Table 2). In salema, the most prevalent and abundant species was Robphildollfusium fractum (Digenea) with endo-parasite prevalence ranging from 14.7 to 66.3, depending on parasite species (Table 2). Both endo-parasite communities were dominated by digeneans. Endo-parasites were all trophically transmitted helminths.
Parasite communities
In white seabream, the number of parasite species ranged from 1 to 5, with the maximum number of species observed in a single individual host. The most frequent numbers of parasite species observed per host were one and two in 32 and 31 individual hosts, respectively.
In salema, the number of parasite species ranged from 1 to 6, with the maximum number of species observed in nine individuals from the GS. The most frequent numbers of parasite species observed per host were two and four in 32 and 22 individual hosts, while the abundance ranged from 1 to 153 and from 1 to 140 in white seabream and salema, respectively.
Descriptors of parasite infracommunities for both host species are listed in Table 3. The white seabream and salema showed significant differences in parasite richness, abundance and diversity when considering the total and endo-parasite community composition, showing higher values in salema. The ecto-parasite communities did not show differences between host species (Table 3).
BRTm were able to accurately predict the abundance, richness and diversity of all parasite groups (i.e. total, ecto- and endo-parasites, p < 0.001) having, on average, a higher prediction accuracy in salema (\(\overline{\rho } > 0.6\)) than in white seabream (\(\overline{\rho } < 0.5\)) and, for both species, a lower accuracy in identifying the most important predictors for endo-parasites communities (salema, \(\overline{\rho } = 0.56\); white seabream, \(\overline{\rho } = 0.466\)).
Overall and across all parasite groups, sampling localities were the most important predictors in salema (Table 4, Fig. 2). This was especially true for the ecto-parasite community, which showed higher values in terms of abundance, species richness and Shannon diversity in the GS (Fig. 2 and Supplementary figures S2, S5 and S8). Seasonality was the main predictor of endo-parasite abundance (Fig. 2 and Supplementary figure S3), while size-related factors (e.g. FL, weight and BCI) explained most of the variation in species richness and diversity (Fig. 2 and Supplementary figures S6 and S9). FL showed a step-like relationship with both species’ richness and diversity, characterized by a sudden decrease for intermediate values of FL followed by a plateau (Fig. 2 and Supplementary figures S6 and S7), suggesting a given stability in terms of number and diversity of endo-parasites harboured by small-sized individuals. Overall, factors related to sexual maturity (e.g. gonadal stage, HSI and GSI) were less reliable predictors of the parasitic load in salema (Fig. 2).
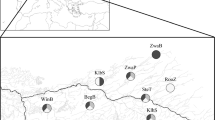
The relative contribution of locality, season, year, and biological (BCI, weight, HSI, FL, GSI, GMS and sex) predictors on parasite abundance, richness, Shannon and Simpson indices of salema Sarpa salpa resulting from the boosted regression tree model (BRTm). BCI: body condition index; HSI: hepatosomatic index; FL: fork length; GSI: gonadosomatic index; GMS: gonadal maturity score. The salema drawing in this figure has been obtained from Schneider51 and downloaded by https://fishbase.org.
In white seabream, size-related factors (e.g. FL and weight) were the most important predictors for the overall parasite abundance (Fig. 3), with larger individuals harbouring a higher number of parasite individuals. Covariates of size-related factors were largely positively associated with parasite abundance, although some non-linear relationships can be observed in the partial dependence plots (Supplementary figure S13). Besides FL, factors related to the host reproductive cycle were important predictors for parasite richness (Fig. 3). A positive linear relationship was found between FL and GSI of white seabream and endo-parasite species richness, suggesting how large, sexually mature individuals harboured a high number of endo-parasites species (Supplementary figure S18). HSI was the main predictor for ecto-parasite richness (Fig. 3), following a non-linear relationship with a U-shaped partial dependence plot characterized by lower values of richness for intermediated HSI (Supplementary figure S17). Ecto-parasite diversity was mainly predicted by factors related to the reproductive cycle, observing a negative linear relationship with GSI and a non-linear U-shaped relationship with HSI (Supplementary figure S20). GSI and, to a lesser extent, FL were the main predictors of endo-parasite diversity in white seabream (Fig. 3), both characterized by a sudden increase followed by a plateau (i.e. a step-like distribution). This suggests a threshold limit (independently of sex) in the dimension and sexual maturity of individuals beyond which parasite diversity stabilizes (Supplementary figures S21 and S24).
The relative contribution of locality and biological (BCI, weight, HSI, FL, GSI, GMS and sex) predictors on parasite abundance, richness, Shannon and Simpson indices of white seabream Diplodus sargus resulting from the boosted regression tree model (BRTm). BCI: body condition index; HSI: hepatosomatic index; FL: fork length; GSI: gonadosomatic index; GMS: gonadal maturity score. The white seabream drawing in this figure has been obtained from Bauchot52 and downloaded by https://fishbase.org.
The most important predictors of parasite community descriptors in salema and white seabream (Figs. 2 and 3) produced relatively strong interaction effects on the abundance of the overall, ecto- and endo-parasite communities. In particular, factor locality in salema had a relatively strong interaction effect with season (H-statistic 0.25), a lower effect with both FL and HSI (H-statistic 0.015 and 0.021, respectively) and only a marginal effect with BCI (H-statistic 0.001). The interaction effect between biological factors was mostly negligible (H-statistic < 0.006), with the only exception for the interaction between HSI and weight (H-statistic 0.1).
Conversely, in white seabream biological factors were the only having strong interaction effects. Weight was found to have a relatively high interaction with both FL and GSI (H-statistic 0.24 and 0.14, respectively) and only marginal with HSI (H-statistic 0.03). Sex had an interaction effect of 0.19 with GSI and only marginal with FL and BCI (H-statistic 0.08 and 0.02, respectively). Other interaction effects between biological factors in white seabream were only negligible (H-statistic < 0.01). The strength of interactions between covariates in BRT models for the Shannon and Simpson indices of both species was near zero when compared with other descriptors.
Discussion
Parasite communities of salema and white seabream seem to respond differently to host biological factors and sampling locality. The endo-helminths of salema are classified into two families of digeneans restricted to this fish species27. In the present study, we found four species of Mesometridae (Mesometra brachycoelia, M. orbicularis, Elstia stossichianum, and Wardula capitellata) and one of Gyliauchenidae (Robphildollfusium fractum). First intermediate hosts of Mesometridae are gastropod molluscs (Prosobranchia); infection of the definitive host occurs by ingestion of cercariae encysted on algae or marine flowering plants28. No data exists on the life cycle of members within Gyliauchenidae, however the concentration of its members in herbivorous fishes strongly suggests that the life cycle might incorporate a cercaria that encysts on algae, as it has been shown for Mesometridae28,29.
The endo-helminths of white seabream are largely generalist parasites. In the present study we found only two common parasite species in both localities (prevalence ≤ 16%): the nematode Cucullanus campanae (Cucullanidae) and the digenean Lepocreadium pegorchys (Lepocreadiidae). Infective larvae of Cucullanus spp. develop in polychaetes and copepods30, while Lepocreadium spp. use sorbeoconchan gastropods as first intermediate host, and a wide range of invertebrates including medusa, ctenophores, polychaetes, turbellarians as second intermediate hosts31. Ecto-parasites of both fish hosts included members within three main groups (leeches, crustaceans and monogeneans) with different ecological and biological features, however all characterized by direct life cycle.
When comparing descriptors of communities between the two fish hosts, we found significant differences only in endo-parasites, with the herbivorous-feeding salema showing higher values (Table 3). Results from studies of helminth communities of fishes show that the richest enteric helminth assemblages are found in carnivorous fishes, whereas algal feeders, herbivores and detritivores showed species poor helminth communities32,33. The results presented in this study are in apparent deviation from our expectations, considering that helminth communities of white seabream in other Mediterranean areas are richest than that found here33,34,35. To explain this finding, we hypothesize that selective feeding on specific poorly infected food items in sampling areas may have led to the poor endo-parasites communities in the white seabream. Similarly, a study from the Tyrrhenian coast of southern Italy showed that in the last decade, the white seabream changed its feeding habit having become the invasive green alga Caulerpa racemosa the most important component of its diet36. In addition, Bartoli et al.37 suggested for the labrid fish Syrnphodus ocellatus living in sites colonized by the invasive alga Caulerpa taxifolia that secondary metabolites as caulerpenyne and other terpenes synthesized by this alga and released into the environment or transmitted along the food web, might be responsible for the near-complete disappearance of digeneans in S. ocellatus. In the present study, we did not focus the attention on the quantification and identification to the lowest taxonomic level of the gastro-intestinal contents. However, gross analysis of gastro-intestinal material during the parasitological analysis revealed that remains of Caulerpa spp. were common food items found in the white seabream from both localities, strongly supporting the hypothesis regarding the possible depletion of digenean community due to the new acquired feeding habit.
Geographical features and, possibly, the degree of alteration of the two studied basins could account for differences in the parasite communities found in salema and, partially, white seabream (only for the ecto-parasites), with the GN showing poorest communities. Parasite communities may be good indicators of environmental disturbance because they reflect the interactions between a possible stressor and either free-living larval stages or populations of their intermediate and final hosts2,3,4,9,10,13. The Gulf of Castellammare in the south-eastern part of GN (where fish samples were obtained) is strongly affected by the sewages of the Sarno River14,15,16. The poor values of communities in GN suggest an unstable ecosystem with a decrease in the biomass and densities of hosts. In contrast, the higher values of parasite communities found in GS, where samples were obtained close to a marine reserve, could be related to the relative stability of its ecosystem2,3,11,12,13, which shows abundant Posidonia meadows and a more preserved benthonic ecosystem with richer communities of intermediate hosts for digeneans. Regarding the ecto-parasite community, it has been demonstrated that crustaceans and monogeneans are more sensitive than endo-parasites to the environmental deterioration, anthropogenic or other pressures (e.g. high levels of conductivity, nutrients and hypoxia)2,4,6,7. This is because they are directly exposed to water and to environmental changes and may be strongly affected by reducing their survival and reproduction rates 2,4,6,7.
For instance, Kostarev38 observed that industrial waste discharge into two reservoirs affected negatively the species richness of monogeneans in fishes. Similarly, it has been documented that the prevalence and abundance of crustacean parasites decreased considerably in polluted sites39,40, and no ecto-parasites have been found on fishes at the closest location to the effluent discharge40 or at polluted sites when compared with un-polluted sites41,42.
In general, size-related factors including BCI, FL and weight explained most of predictor variables in parasite communities for both host species, and interaction effects suggested the influence of localities and season on specific biological traits in salema but not in white seabream, ultimately influencing the abundance of both ecto- and endo-parasites. FL was the most important predictor for species richness and diversity of endo-parasite community in salema and for the overall parasite abundance and species richness in the white seabream, respectively. Our results are in accordance with previous investigations8,43,44,45,46, which suggested that larger body size increases the vagility and habitat exploitation by hosts8,47,48, therefore making them a better target for the infective stage of a parasite and enhancing the exposure to more parasite species on a regional scale. In white seabream, the ecto-parasite diversity was also predicted by factors related to the reproductive cycle, supporting the view that the vagility and aggregation of individuals during the reproductive season may facilitate the infection of parasites with direct life-cycles, leading to more diverse ecto-parasite assemblages5,47,48,49,50. Moreover, in salema, seasonality was the main predictor of endo-parasite abundance with highest value in winter (December/February), suggesting the seasonal variation in availability of encysted larval digeneans.
Conclusion
The present study shows that the structure of parasite community of salema and white seabream from two contiguous areas is the result of complex interactions between environment, fish host populations and free-living larval stages or populations of their intermediate hosts. Host size-related factors are important natural predictors of parasite community in both host species. However, geographic locality may affect differently the parasite community depending on host species and sensitivity of their parasite stages to environmental features and their changes and pressures. Compared to GS, the poorer parasite communities found in GN suggests that changes and deterioration of environmental conditions can play an important role, affecting the parasite communities of these fishes, that in turn, could be used as biological indicators. However, further investigations are needed to provide conclusive data about the nature and importance of the potential effects of environmental degradation, by studying the temporal fluctuation of parasite community descriptors in these common sparid fishes.
References
Lafferty, K. D. Environmental Parasitology: what can parasites tell us about human impacts on the environment?. Parasitol. Today13, 251–255 (1997).
Blanar, C. A., Munkittrick, K. R., Houlahan, J., MacLatchy, D. L. & Marcogliese, D. J. Pollution and parasitism in aquatic animals: a meta-analysis of effect size. Aquat. Toxicol.93, 18–28. https://doi.org/10.1016/j.aquatox.2009.03.002 (2009).
Vidal-Martínez, V. M., Pech, D., Sures, B., Purucker, S. T. & Poulin, R. Can parasites really reveal environmental impact?. Trends Parasitol.26, 44–51. https://doi.org/10.1016/j.pt.2009.11.001 (2010).
Sures, B., Nachev, M., Selbach, C. & Marcogliese, D. J. Parasite responses to pollution: what we know and where we go in “Environmental Parasitology”. Parasit. Vectors.10(1), 65. https://doi.org/10.1186/s13071-017-2001-3 (2017).
Carrassón, M. et al. Drivers of parasite community structure in fishes of the continental shelf of the Western Mediterranean: the importance of host phylogeny and autecological traits. Int. J. Parasitol.49(9), 669–683. https://doi.org/10.1016/j.ijpara.2019.04.004 (2019).
Khan, R. A. & Thulin, J. Influence of pollution on parasites of aquatic animals. Adv. Parasitol.30, 201–238 (1991).
MacKenzie, K., Williams, H. H., Williams, B., McVicar, A. H. & Siddall, R. Parasites as indicators of water quality and the potential use of helminth transmission in marine pollution studies. Adv. Parasitol.35, 128–144 (1995).
Sasal, P., Morand, S. & Guegan, J. F. Determinants of parasite species richness in Mediterranean marine fishes. . Mar. Eco1 Prog. Ser.149, 61–71 (1997).
Marcogliese, D. J. Food webs and the transmission of parasites to marine fish. Parasitology124, 83–99. https://doi.org/10.1017/s003118200200149x (2002).
Sures, B. Environmental parasitology: relevancy of parasites in monitoring environmental pollution. Trends Parasitol.20, 170–177. https://doi.org/10.1016/j.pt.2004.01.014 (2004).
Bartoli, P., Gibson, D. I. & Bray, R. A. Digenean species diversity in teleost fish from a nature reserve of Corsica, France (Western Mediterranean), and a comparison with other Mediterranean regions. J. Nat. Hist.39, 47–70. https://doi.org/10.1080/00222930310001613557 (2005).
Derbel, H., Châari, M. & Neifar, L. Digenean species diversity in teleost fishes from the Gulf of Gabes, Tunisia (Western Mediterranean). Parasite19, 129–135. https://doi.org/10.1051/parasite/2012192129 (2012).
Mattiucci, S. et al. Temporal stability of parasite distribution and genetic variability values of Contracaecum osculatum sp. D and C. osculatum sp E (Nematoda: Anisakidae) from fish of the Ross Sea (Antarctica). Int. J. Parasitol. Parasites Wildl.4, 356–367. https://doi.org/10.1016/j.ijppaw.2015.10.004 (2015).
Tornero, V. & Ribera d’Alcalà, M. Contamination by hazardous substances in the Gulf of Naples and nearby coastal areas: a review of sources, environmental levels and potential impacts in the MSFD perspective. Sci. Total. Environ.466–467, 820–840. https://doi.org/10.1016/j.scitotenv.2013.06.106 (2014).
Lofrano, G., Libralato, G., Acanfora, F. G., Pucci, L. & Carotenuto, M. Which lesson can be learnt from a historical contamination analysis of the most polluted river in Europe?. Sci. Total Environ.524–525, 246–259. https://doi.org/10.1016/j.scitotenv.2015.04.030 (2015).
Arienzo, M., Adamo, P., Bianco, M. R. & Violante, P. Impact of land use and urban runoff on the contamination of the Sarno River basin in South-Western Italy. Water Air Soil Pollut.131, 349–366. https://doi.org/10.1023/A:1011908019933 (2001).
Havelange, S., Lepoint, G., Dauby, P. & Bouquegneau, J. M. Feeding of the sparid fish Sarpa salpa in a seagrass ecosystem: diet and carbon flux. Mar. Ecol.18, 289–297. https://doi.org/10.1111/j.1439-0485.1997.tb00443.x (1997).
Sala, E. & Ballesteros, E. Partitioning of space and food resources by three fish of the genus Diplodus (Sparidae) in a Mediterranean rocky infralittoral ecosystem. Mar. Ecol. Prog. Ser.152, 273–283 (1997).
Mouine, N., Francour, P., Ktari, M. H. & Chakroun-Marzouk, N. The reproductive biology of Diplodus sargus sargus in the Gulf of Tunis (Central Mediterranean). Sci. Mar.71, 461–469. https://doi.org/10.1017/S0025315411000798 (2007).
Le Cren, E. D. The length-weight relationship and seasonal cycle in gonad weight and condition in the perch (Perca fluviatilis). J. Anim. Ecol.20, 201–219 (1951).
Santoro, M. et al. Parasite communities of icefish (Chionodraco hamatus) in the Ross Sea (Antarctica): influence of the host sex on the helminth infracommunity structure. PLoS ONE9(2), e88876. https://doi.org/10.1371/journal.pone.0088876 (2014).
Bush, A. O., Lafferty, K. D., Lotz, J. M. & Shostak, A. W. Parasitology meets ecology on its own terms: Margolis et al. revisited. J. Parasitol.83(4), 575–583 (1997).
Krebs, C.J. Ecological Methodology (ed. Krebs, C.J.) 620 (Addison-Wesley Educational Publishers, 1999).
Elith, J., Leathwick, J. R. & Hastie, T. A working guide to boosted regression trees. J. Anim. Ecol.77, 802–813. https://doi.org/10.1111/j.1365-2656.2008.01390.x (2008).
R Core Team. R: a language and environment for statistical computing. https://www.R-project.org/ (2017).
Breiman, L. Random forests. Mach. Learn.45, 5–32 (2001).
Bartoli, P. Caractères adaptatifs originaux des Digènes intestinaux de Sarpa salpa (Teleosti, Sparidae) et leur interprétation en termes d’évolution. Ann. Parasitol. Hum. Comp.62, 542–576 (1987).
Jousson, O. & Bartoli, P. The life-cycle of three species of the Mesometridae (Digenea) with comments on the taxonomic status of this family. Syst. Parasitol.44, 217–228 (1999).
Hall, K. A. & Cribb, T. H. Family Gyliauchenidae Fukui, 1929. In Keys to the Trematoda Vol. 2 (eds Jones, A. et al.) 665–678 (London, UK, The Natural History Museum, 2005).
Anderson, R.C., Chabaud, A.G. & Willmott, S. CIH keys to the nematode parasites of vertebrates: Archival volume (eds. Anderson, R.C., Chabaud, A.G. & Willmott, S.) 480 (CAB International, 2009).
Bray, R. A. Family Lepocreadiidae Odhner, 1905. In Keys to the Trematoda Vol. 2 (eds Jones, A. et al.) 545–602 (London, UK, The Natural History Museum, 2005).
Choudhury, A. & Dick, T. Richness and diversity of helminth communities in tropical freshwater fishes: empirical evidence. J. Biogeogr.27(4), 935–956 (2000).
Pérez-Del-Olmo, A., Kostadinova, A. & Gibson, D. I. The Mediterranean: high discovery rates for a well-studied trematode fauna. Syst. Parasitol.93, 249–256. https://doi.org/10.1007/s11230-016-9626-z (2016).
Bellal, A., Brahim Tazi, N. A., Hadjou, Z. & Boutiba, Z. First records of digenean trematodes of two fishes (Teleostei Sparidae) from the West Algerian coast and comparative study with Tunisian coast (Mediterranean Sea). Biodivers. J.7, 233–240 (2016).
Antar, R. & Gargouri, L. The diversity of teleost fish trematodes in the Bay of Bizerte, Tunisia (Western Mediterranean). Helminthologia55(2), 146–156. https://doi.org/10.2478/helm-2018-0004 (2018).
Terlizzi, A. et al. Detrimental physiological effects of the invasive alga Caulerpa racemosa on the Mediterranean white seabream Diplodus sargus. Aquat. Biol.12, 109–117. https://doi.org/10.3354/ab00330 (2011).
Bartoli, P. & Boudouresque, C. F. Transmission failure of digenean parasites (Digenea) in sites colonized by the recently introduced invasive alga Caulerpa taxifolia. Mar. Ecol. Progr. Ser.154, 253–260. https://doi.org/10.3354/meps154253 (1997).
Kostarev, G.F. The effects of pollution on the fish parasite fauna in the Kama reservoirs (ed. Kostarev, G.F.) 147–153 (Perskii Gosudarstvennyi Universitet, 1980).
Kuperman, B. Fish parasites as bioindicators of the pollution of the bodies of water. Parassitologia26, 479–482 (1991).
Overstreet, R. M. & Thulin, J. Influence of pollution on parasites of aquatic animals. Ann. Parasitol. Hum. Comp.66, 49–51 (1991).
Zharikova, T. I. Effect of water pollution on ectoparasites of bream (Abramis brama). J. Ichthyol.33, 50–62 (1993).
Madanire-Moyo, G. & Barson, M. Diversity of metazoan parasites of the African catfish Clarias gariepinus as indicators of pollution in a subtropical African river system. J. Helminthol.84, 216–227. https://doi.org/10.1017/S0022149X09990563 (2010).
Poulin, R. Phylogeny, ecology, and the richness of para- site communities in vertebrates. Ecol. Monogr.65, 283–302 (1995).
Grutter, A. S. & Poulin, R. Intraspecific and interspecifi c relationships between host size and the abundance of parasitic larval gnathiid isopods on coral reef fishes. Mar. Ecol. Prog. Ser.164, 263–271 (1998).
Poulin, R. The functional importance of parasites in animal communities: many roles at many levels?. Int. J. Parasitol.29(6), 903–914. https://doi.org/10.1016/S0020-7519(99)00045-4 (1999).
George-Nascimento, M., Muñoz, G., Marquet, P. & Poulin, R. Testing the energetic equivalence rule with helminth endoparasites of vertebrates. Ecol. Lett.7, 527–531. https://doi.org/10.1111/j.1461-0248.2004.00609.x (2004).
Price, P.W. Evolutionary biology of parasites (ed. Price, P.W.) 256 (Princeton University Press, 1980).
Rohde, K. Marine parasitology (ed. Rohde, K.) 565 (Csiro Publishing, 2005).
Caro, A., Combes, C. & Euzet, L. What makes a fish a suitable host for Monogenea in the Mediterranean?. J. Helminthol.71, 203–210 (1997).
Raibaut, A., Combes, C. & Benoit, F. Analysis of the parasitic copepod species richness among Mediterranean fish. J. Mar. Syst.15(1–4), 185–206. https://doi.org/10.1016/S0924-7963(97)00079-1 (1998).
Schneider, W. FAO species identification sheets for fishery purposes. Field guide to the commercial marine resources of the Gulf of Guinea (FAO, 1990).
Bauchot, M.L. (1987). Poissons osseux (eds. Fischer, W., Bauchot, M.L., Schneider, M.) 891–1421 (Commission des Communautés Européennes and FAO, 1987).
Acknowledgements
We thank Fabio Di Nocera and Giuseppe Di Vincenzo (Istituto Zooprofilattico Sperimentale del Mezzogiorno) for collaborating in the conceptualization of the first step of this study, and providing the support during the dissection of fish, respectively. We are grateful to the marine protected area of Punta Campanella and two local fishermen for providing logistic support and the fish, respectively. The study was financed by the Italian Ministry of Health (IZS-ME 05/15RC).
Author information
Authors and Affiliations
Contributions
MS conceived the study and performed parasitological analysis. MS and DI performed fish dissection and collected biometrical and biological data of fish. BB performed statistical analysis. MS and BB wrote the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Santoro, M., Iaccarino, D. & Bellisario, B. Host biological factors and geographic locality influence predictors of parasite communities in sympatric sparid fishes off the southern Italian coast. Sci Rep 10, 13283 (2020). https://doi.org/10.1038/s41598-020-69628-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-69628-1
- Springer Nature Limited
This article is cited by
-
Drivers of richness and abundance of parasites of fishes from an intermittent river before and after an interbasin water transfer in the Brazilian semi-arid region
Parasitology Research (2024)
-
Endohelminth diversity in the invasive Clarias gariepinus (Burchell, 1822) from two freshwater lakes: Naivasha and Ol’Bolossat, Kenya
Parasitology Research (2024)
-
Fish parasites as proxy bioindicators of degraded water quality of River Saraswati, India
Environmental Monitoring and Assessment (2023)
-
Drivers of parasite communities in three sympatric benthic sharks in the Gulf of Naples (central Mediterranean Sea)
Scientific Reports (2022)
-
Parasites as Emerging Biomonitoring Tools-Promises and Pitfalls
Proceedings of the National Academy of Sciences, India Section B: Biological Sciences (2022)