Abstract
In patients with atherosclerotic cardiovascular disease (ASCVD) under statin treatment, the influence of on-treatment level of high-density lipoprotein cholesterol (HDL-C) on cardiovascular (CV) events is controversial. Statin-treated patients were selected from the Taiwanese Secondary Prevention for patients with AtheRosCLErotic disease (T-SPARCLE) Registry, a multicenter, observational study of adult patients with ASCVD in Taiwan. Low HDL-C was defined as < 40 mg/dL for men and < 50 mg/dL for women. The primary outcome was a composite CV events including CV death, myocardial infarction (MI), stroke or cardiac arrest with resuscitation. A total of 3731 patients (mean age 65.6 years, 75.6% men) were included. Patients with on-treatment low HDL-C (44%, mean HDL-C 34.9 ± 6.8 mg/dL) were younger and with more diabetes and higher body weight. The mean follow-up time was 2.7 years. We used restricted cubic spline curves to examine the potential non-linear association between HDL-C and adverse outcomes. Decreased HDL-C levels were associated with a significantly increased risk of CV events in women (< 49 mg/dL in women) but not in men (< 42 mg/dL in men). However, the protective effect of elevated HDL-C levels was more prominent in men than in women. In ASCVD patients with statin therapy, low on-treatment HDL-C was common in Taiwan and associated with an increased risk of CV events in women. Higher HDL-C levels provided more protective effect in men than in women.
Similar content being viewed by others
Introduction
Multiple lines of evidence have established the role of low-density lipoprotein cholesterol (LDL-C) in the development and progression of atherosclerotic cardiovascular disease (ASCVD)1. Lower levels of LDL-C have been shown to reduce cardiovascular (CV) events in multiple clinical trials and human mendelian genetic studies2,3. However, a significant residual CV risk remains in patients under intensive statin therapy with controlled LDL-C levels4,5. Previous epidemiological studies demonstrated that a low level of high-density lipoprotein cholesterol (HDL-C) is a risk factor of ASCVD6,7. In a meta-analysis of 20 randomized clinical trials of statins, a significant inverse association between on-treatment HDL-C levels and the CV risk was observed in the placebo group as well as in the statin groups8. But almost all subsequent clinical trials with pharmacological therapies to raise HDL-C in statin-treated patients failed to improve clinical outcomes despite substantial increases in HDL-C levels9. Human mendelian genetic study also found genetic polymorphisms that raise plasma HDL-C levels were not associated with a lower risk of acute myocardial infarction (MI)10. Low HDL-C is a common lipid phenotype in Asia. Population studies in the Asia–Pacific region showed that the prevalence of low HDL-C was 33.1% in Asians which was significantly higher than that in non-Asians11. However, few studies have evaluated the clinical importance of HDL-C levels in Asian patients treated with statins. The Taiwanese Secondary Prevention for patients with AtheRosCLErotic disease (T-SPARCLE) registry is a large-scale observational study to recruit and follow up patients with ASCVD who have been receiving secondary prevention therapies in Taiwan12. Using the T-SPARCLE registry data, we evaluated whether on-treatment HDL-C levels carry any prognostic significance in patients with ASCVD under statin treatment. The specific aims of this study were to (1) evaluate the prevalence and determining factors of low HDL-C in patients with ASCVD treated with statins and (2) assess whether the low on-treatment HDL-C is an important predictive factor of adverse clinical outcomes in these patients.
Methods
Patient population
We used the data of patients who were under statin treatment at enrollment in the T-SPARCLE registry to perform the study. The T-SPARCLE registry is a prospective, multicenter, observational study that included adult ASCVD patients from 14 major hospitals across Taiwan. The study protocol has been described previously12. In this study, the inclusion criteria were patients who were (1) age > 18 years; (2) with stable symptomatic ASCVD, including coronary artery disease, cerebrovascular disease or peripheral artery disease; (3) taking a statin; (4) willing to follow a National Cholesterol Education Program (NCEP) therapeutic lifestyle change or similar cholesterol-lowering diet; and (5) willing to provide informed consent. The exclusion criteria were patients with (1) other serious heart disease; (2) ≥ New York Heart Association functional class III heart failure; (3) life-threatening malignancy; (4) treatment with immunosuppressive agents; (5) other atherosclerotic vascular diseases with unknown disease type; (6) two or more statins treatment at enrollment; (7) chronic dialysis or (8) any condition or situation which, in the opinion of the investigators, might be not suitable for this registry study. The patients’ demographic data, major vascular risk factors, previous disease history, and medications were collected according to a predetermined protocol. The body mass index (BMI) was calculated as the body weight divided by the square of the body height (kg/m2). Hypertension, diabetes, heart failure, and ASCVD were diagnosed following conventional definitions and confirmed by the physicians that recruited the study participants. Laboratory test results, including creatinine levels, total cholesterol (TC), triglyceride (TG), LDL-C and HDL-C were obtained after enrollment. Low HDL-C was defined as < 40 mg/dL for male and < 50 mg/dL for female. Serum creatinine was used to calculate estimated glomerular filtration rate (eGFR) by using the Modification of Diet in Renal Disease equation. Chronic kidney disease (CKD) was defined as patients with eGFR < 60 mL/min/1.73m2. The definition of statin intensity was based on the recommendation of the 2013 American College of Cardiology/American Heart Association cholesterol guideline13. Written informed consent was obtained from each patient included in the study. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki and was approved by the Taiwan Joint Institutional Review Board for each participating hospital (JIRB number 09-S-015).
Follow-up
The eligible patients who fulfilled the enrollment criteria were followed up in the original hospitals that recruited the patients. The follow-up information of the patients was collected every year after enrollment. In this study, the primary outcome was a composite endpoint of CV death, nonfatal stroke, nonfatal MI, or cardiac arrest with resuscitation. The follow-up time in the analysis was from the enrollment to the occurrence of first primary outcome event or till the last follow-up visit if there was no primary outcome occurred.
Statistical analysis
Continuous variables were presented as means ± standard deviations (SDs) and categorical variables were presented as numbers and percentages. The intergroup comparisons were made by using chi-square test with or without Yates’ correction for categorical variables and unpaired Student’s t test or Mann–Whitney rank sum test for continuous variables. A 2-tailed p value < 0.05 was considered significant. The influence of HDL-C on primary outcome was analyzed by using Cox proportional hazards models. The HDL-C levels were analyzed in the models either as a categorical variable or as a continuous variable. Multivariate Cox proportional hazard analysis was used to identify independent predictors of the primary outcome while adjusting multiple clinical factors, including age, sex, BMI, eGFR, vascular risk factors, smoking status, MI, ischemic stroke/transient ischemic attack, coronary/peripheral intervention and concomitant medications. When HDL-C was treated as a continuous variable, restricted cubic splines was used to produce a curve of hazard ratio (HR) between HDL-C and risk of primary outcome. Three knots at 0.25, 0.50, and 0.75 were used in the splines and the HR between two points of a continuous variable was estimated. We used SAS statistical package (version 9.4 for Windows; SAS Institute, Cary, NC, USA) for all analyses.
Results
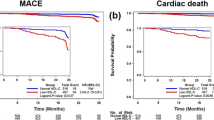
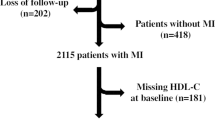
Overall, 7866 ASCVD patients were screened from January 2010 to October 2014. After exclusion, 6050 patients were enrolled and 3731 of them (mean age 65.6 ± 11.3 years, 75.6% men) who were taking a statin at enrollment formed the basis of this analysis (Fig. 1). Among them, 1629 (43.7%) patients had low HDL-C under statin treatment. Table 1 shows the clinical characteristics of patients with and without low HDL-C under statin treatment. Patients with low HDL-C were younger and had more female and higher BMI. More of them had a history of diabetes mellitus, coronary or peripheral intervention, coronary artery disease and MI. The TG level was also higher in patients with low HDL-C. Multivariate logistic analysis found that younger age, female, diabetes, smoking, use of beta-blocker, increased BMI and CKD with eGFR ≤ 30 ml/min/1.73m2 were the independent predicting factors of low HDL-C level (Table 2). Statin intensity was not a predicting factor of low HDL-C. The mean total follow-up time was 2.7 year. During this period, 110 (3.0%) patients developed the primary outcome events, including 22 patients with CV death, 37 with nonfatal stroke, 44 with nonfatal MI and 7 with cardiac arrest with resuscitation. Compared with those without events, the patients who developed outcome events were older and had lower BMI, more heart failure, diabetes and CKD (Table 3). The HDL-C levels were similar between patients with and without outcome events (44.0 ± 13.1 vs. 44.9 ± 12.9 mg/dL, p = 0.47). In the multivariate Cox proportional hazard analysis after adjusting for all clinical factors, we found age, smoking, without beta-blocker use, heart failure and CKD with eGFR ≤ 30 ml/min/1.73m2 were independent predictors of the primary outcome. Low HDL-C was not significantly associated with an increased risk of the primary outcome (normal vs. low HDL-C, HR, 0.83, 95% confidence interval [CI], 0.56–1.23) (Table 4). Statin intensity had no association with clinical outcome. When the level of HDL-C was analyzed as a continuous variable by using restricted cubic splines, a significantly negative association of HDL-C with the risk of primary outcome was observed in women with low HDL-C levels (HDL-C < 49 mg/dL), but not in men (HDL-C < 42 mg/dL) (Fig. 2). On the contrary, elevated HDL-C levels were continuously associated with a decreased risk of outcome events in men (HDL-C > 42 mg/dL) and the protective effect of elevated HDL-C was much more evident in men than in women (Fig. 2).
Discussion
The major findings of this study were: (1) low HDL-C was a common lipid phenotype in patients with ASCVD treated with statins, (2) statin intensity was not associated with low HDL-C, (3) low HDL-C was associated with an increased risk of adverse clinical outcomes in female patients with ASCVD under statin treatment, and (4) a more protective effect of increased HDL-C was observed in men than in women.
In this Asian cohort, we found low HDL-C was a common lipid phenotype (43.7%) in patients under statin treatment. In the Treating to New Targets (TNT) study, 9770 patients with stable coronary artery disease were randomized to receive 10 mg or 80 mg/day atorvastatin therapy. At 3 months after treatment, 57.8% patients had HDL-C < 48 mg/dL14. In the 3574 patients with acute MI in the Korea Acute Myocardial Infarction Registry, more than 75% of these patients received statin treatment and 45.5% patients had HDL-C < 40 mg/dL at 12-month follow-up after discharge15. Although HDL-C raising ability was different among statins16,17, we found there was no relation between the presence of low HDL-C level and statin intensity. In recent decades, there were great changes of lipid phenotype between Asian and Western countries. Population-based studies demonstrated that there was an increased TC in Asian countries, but declined in most Western countries18. The increase of TC in Japan and South Korea was mainly due to an increase of HDL-C. But in China, a rise in non-HDL-C was the reason for increased TC. In most Western countries, there was mainly a decrease of non-HDL-C and an increase of HDL-C18. The differences in genetic background, economic development, dietary habit changes, and prescription rate of lipid lowering drugs among the different regions cause the variations of lipid phenotype. All these factors also cause complexity of the relationship between HDL-C and CV outcome.
The prognostic role of HDL-C in statin-treated patients is still controversial. The post-hoc analysis of the TNT study showed that an increase of 1 mg/mL in HDL-C reduced the risk of major CV events by 1.1% (p = 0.003). However, there was no significant difference in CV risk between the highest and the lowest quintile of HDL-C in high-intensity statin treated patients14. By analyzing HDL-C in categories, both the Pravastatin or Atorvastatin Evaluation and Infection Therapy–Thrombolysis in Myocardial Infarction 22 (PROVE IT–TIMI 22) trial and Justification for the Use of Statins in Prevention: an Intervention Trial Evaluating Rosuvastatin (JUPITER) trials found the on-treatment HDL-C level had no significant predictive value for subsequent CV risk in patients with statin therapy19,20. In our study, by treating HDL-C as a continuous variable, restricted cubic spline curves showed that decreased HDL-C levels were associated with increased risk of primary outcome in female patients. The protective effects of elevated HDL-C was more prominent in men than in women. Gender has great influence on the relation of HDL-C to the risk of ASCVD. In the Los Angeles Atherosclerosis Study (LAAS), after adjusting potential confounders in the multivariate models, the progression of carotid intima-media thickness was inversely associated with serum levels of HDL-C in men. For women, the protective effect of HDL-C level diminished in the age of menopause21. In the 1380 females from the Multi-Ethnic Study of Atherosclerosis (MESA) study, the higher HDL-C was not even protective but associated with a greater risk for predicting the presence of carotid plaque in postmenopausal women22. One of the explanations for the different HDL-C effect of female is related to estrogen reduction in menopause. Gradual decrease of the antioxidant effect of estrogen over the menopausal transition increases formation of reactive oxygen species and changes the HDL-C composition resulting in less anti-inflammatory and antioxidant properties of HDL-C23. Another problem of the current measurement of HDL-C, the cholesterol carried by HDL particles, may not reflect the true effect of HDL on CV risk. Other HDL-related biomarkers, such as HDL function or HDL particle number, may carry more important clinical significance than HDL-C levels. In the JUPITER trial, the HDL particle number and HDL cholesterol efflux capacity had a statistically significant and stronger negative association with CV disease than HDL-C levels among rosuvastatin-treated individuals24.
The major limitation of our study is its observational study design. The confounding factors that could not be identified or measured potentially may bias the clinical outcomes. For example, we did not know the severity of coronary stenosis in patients with coronary artery disease. The data of left ventricular ejection fraction were also unknown. These unmeasured confounding factors could influence the clinical outcomes and bias the study results. Second, information bias including the drug compliance and clinical endpoint identification during the clinical follow up was another study limitation. Third, the follow-up time was still short. The long term effect of low HDL-C in these patients was unknown. Finally, the status of menopause in female patients was not known in our study. We found gender played an important role and low HDL-C was associated with increased risk of adverse clinical outcomes only in female patients. However, the influence of menopause could not be analyzed in our study.
In conclusion, this observational study indicates that low on-treatment HDL-C is a common lipid phenotype in statin-treated patients in Taiwan. It is associated with increased risk of adverse clinical outcomes in female patients with ASCVD. The protective effect of increased HDL-C is more evident in men than in women.
References
Ference, B. A. et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur. Heart. J. 38, 2459–2472 (2017).
Cholesterol Treatment Trialists’ (CTT) Collaboration et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet 376, 1670–1681 (2010).
Ference, B. A. et al. Effect of long-term exposure to lower low-density lipoprotein cholesterol beginning early in life on the risk of coronary heart disease: a Mendelian randomization analysis. J. Am. Coll. Cardiol. 60, 2631–2639 (2012).
Reith, C. & Armitage, J. Management of residual risk after statin therapy. Atherosclerosis 245, 161–170 (2016).
Cardiometabolic Risk Working Group: Executive Committee et al. Identification and management of cardiometabolic risk in Canada: a position paper by the cardiometabolic risk working group (executive summary). Can. J. Cardiol. 27, 124–131 (2011).
Jacobs, D. R. Jr., Mebane, I. L., Bangdiwala, S. I., Criqui, M. H. & Tyroler, H. A. High density lipoprotein cholesterol as a predictor of cardiovascular disease mortality in men and women: the follow-up study of the Lipid Research Clinics Prevalence Study. Am. J. Epidemiol. 131, 32–47 (1990).
Emerging Risk Factors Collaboration et al. Major lipids, apolipoproteins, and riskof vascular disease. JAMA 302, 1993–2000 (2009).
Jafri, H., Alsheikh-Ali, A. A. & Karas, R. H. Meta-analysis: statin therapy does not alter the association between low levels of high-density lipoprotein cholesterol and increased cardiovascular risk. Ann. Int. Med. 153, 800–808 (2010).
Choi, H. Y., Hafiane, A., Schwertani, A. & Genest, J. High-density lipoproteins: biology, epidemiology, and clinical management. Can. J. Cardiol. 33, 325–333 (2017).
Voight, B. F. et al. Plasma HDL cholesterol and risk of myocardial infarction: a mendelian randomisation study. Lancet 380, 572–580 (2012).
Huxley, R. R. et al. Asia Pacific Cohort Studies Collaboration and the Obesity in Asia Collaboration. Isolated low levels of high-density lipoprotein cholesterol are associated with an increased risk of coronary heart disease: an individual participant data meta-analysis of 23 studies in the Asia-Pacific region. Circulation 124, 2056–2064 (2011).
Ho, L. T. et al. Taiwanese Secondary Prevention for patients with AtheRosCLErotic disease (T-SPARCLE) Registry Investigators. Determinants for achieving the LDL-C target of lipid control for secondary prevention of cardiovascular events in Taiwan. PLoS ONE 10, e0116513 (2015).
Stone, N. J. et al. American College of Cardiology/American Heart Association Task Force on Practice Guidelines.2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J. Am. Coll. Cardiol. 63, 2889–2934 (2014).
Barter, P. et al. Treating to new targets investigators. HDL cholesterol, very low levels of LDL cholesterol, and cardiovascular events. N. Engl. J. Med. 357, 1301–1310 (2007).
Lee, C. H. et al. other Korea Acute Myocardial Infarction Registry (KAMIR) Investigators. Roles of high-density lipoprotein cholesterol in patients with acute myocardial infarction. Med. (Baltimore) 95, e3319 (2016).
Jones, P. H. et al. STELLAR Study Group. Comparison of the efficacy and safety of rosuvastatin versus atorvastatin, simvastatin, and pravastatin across doses (STELLAR Trial). Am. J. Cardiol. 92, 152–160 (2003).
Barter, P. J., Brandrup-Wognsen, G., Palmer, M. K. & Nicholls, S. J. Effect of statins on HDL-C: a complex process unrelated to changes in LDL-C: analysis of the VOYAGER Database. J. Lipid. Res. 51, 1546–1553 (2010).
NCD Risk Factor Collaboration (NCD-RisC). National trends in total cholesterol obscure heterogeneous changes in HDL and non-HDL cholesterol and total-to-HDL cholesterol ratio: a pooled analysis of 458 population-based studies in Asian and Western countries. Int. J. Epidemiol. 49, 173–192 (2020).
Ray, K. K. et al. Prognostic utility of apoB/AI, total cholesterol/HDL, non-HDL cholesterol, or hs-CRP as predictors of clinical risk in patients receiving statin therapy after acute coronary syndromes: results from PROVE IT-TIMI 22. Arterioscler. Thromb. Vasc. Biol. 29, 424–430 (2009).
Ridker, P. M. et al. JUPITER Trial Study Group. HDL cholesterol and residual risk of first cardiovascular events after treatment with potent statin therapy: an analysis from the JUPITER trial. Lancet 376, 333–339 (2010).
Fan, A. Z. & Dwyer, J. H. Sex differences in the relation of HDL cholesterol to progression of carotid intima-media thickness: the Los Angeles Atherosclerosis Study. Atherosclerosis 195, e191-196 (2007).
El Khoudary, S. R. et al. HDL (high-density lipoprotein) metrics and atherosclerotic risk in women. Arterioscler. Thromb. Vasc. Biol. 38, 2236–2244 (2018).
Zago, V. et al. Impaired high density lipoprotein antioxidant activity in healthy postmenopausal women. Atherosclerosis 177, 203–210 (2004).
Khera, A. V. et al. Cholesterol efflux capacity, high-density lipoprotein particle number, and incident cardiovascular events: an analysis from the JUPITER Trial (Justification for the Use of Statins in Prevention: An Intervention Trial Evaluating Rosuvastatin). Circulation 135, 2494–2504 (2017).
Funding
The study was supported by the Taiwan Society of Lipids and Atherosclerosis, and Taiwan Ministry of Science and Technology Grants 102-2325-B-002-097, 104-2325-B-002-036, 105-2325-B-002-035 and 106-2321-B-002-029.
Author information
Authors and Affiliations
Consortia
Contributions
Conceptualization, Y.H.L., W.K.T., J.W.C. and C.C.W.; Data collection, Y.H.L., W.K.T., W.H.Y., F.J.L., Y.W.W., I.C.H., T.H.L.,W.H.H.S., H.I.Y., J.W.C., C.C.W.; Funding acquisition, C.C.W.; Methodology, Y.H.L., W.K.T., W.H.Y., F.J.L., Y.W.W., I.C.H., T.H.L.,W.H.H.S., H.I.Y., J.W.C., C.C.W.; Supervision, J.W.C. and C.C.W.; Writing original draft, Y.H.L. All authors have read and agree to the published version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declared that there is nothing to disclose regarding conflict of interest with respect to this manuscript. Part of the data in this work was presented as a poster at the American Heart Association Scientific Sessions, November 10–12, 2018, in Chicago, Illinois, USA.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, YH., Tseng, WK., Yin, WH. et al. Prognostic effect of high-density lipoprotein cholesterol level in patients with atherosclerotic cardiovascular disease under statin treatment. Sci Rep 10, 21835 (2020). https://doi.org/10.1038/s41598-020-78828-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-78828-8
- Springer Nature Limited