Abstract
Planning and implementation of schistosomiasis control activities requires an understanding of the prevalence, intensity of infection and geographical distribution of the disease in different epidemiological settings. Although, Tanzania is known to be highly endemic to schistosomiasis, there is paucity of data on the geographical distribution of schistosomiasis in potential large water bodies in the country. Thus, the present study was conducted to determine the prevalence, infection intensities and geographical distribution of schistosomiasis along villages located on the shoreline of Lake Nyasa, southern Tanzania. A cross-sectional study was conducted among 1560 children aged 1–13 years old living in villages located along the shoreline of Lake Nyasa. A single urine and stool sample was obtained from each participating child and screened for S. mansoni using Kato Katz (KK) technique to detect eggs and using point-of-care circulating Cathodic Antigen (POC-CCA) test to detect antigen in urine. Urine filtration technique was used to screen for S. haematobium eggs in urine samples. Villages/primary school were mapped using geographical information system and prevalence map was generated using ArcView GIS software. The overall prevalence of S. mansoni based on KK technique and POC-CCA test was 15.1% (95%CI: 13.4–16.9) and 21.8% (95%CI: 18.5–25.3) respectively. The prevalence S. haematobium was 0.83% (95%CI: 0.5–1.4) and that of haematuria was 0.9%. The arithmetic mean egg intensities for S. haematobium and S. mansoni were 18.5 mean eggs/10 ml (95%CI: 5.9–57.6) of urine and 34.7 mean epg (95%CI: 27.7–41.7) respectively. Villages located on the southern end of the lake had significantly high prevalence of S. mansoni than those located on the northern part (χ2 = 178.7838, P = 0.001). Cases of S. haematobium were detected only in three villages. Both S. mansoni and S. haematobium infections occur in villages located along the shoreline of Lake Nyasa at varying prevalence. These finding provide insights that can provide guidance in planning and implementation of MDA approach and other recommended measures such as improvement in sanitation, provision of clean water and behaviour changes through public health education.
Similar content being viewed by others
Introduction
The sub-Saharan African region, carries 90% of the over 250 million cases of schistosomiasis occurring worldwide1,2. In this region, after Nigeria, Tanzania is second country having the highest cases of schistosomiasis and approximately 51.5%0 of the Tanzanian population is either exposed or live in areas with high risk of exposure3,4. The country is endemic to both Schistosoma mansoni and Schistosoma haematobium, these infections are common in communities characterised with limited access to water, sanitation, hygienic practices and health services4. Schistosoma mansoni infection is associated with hepatosplenic disease characterised with hepatomegaly, splenomegaly, progressive periportal fibrosis (PPF) which can lead to portal hypertension and its related sequelae, mainly ascites, liver surface irregularities, oesophageal varices and haematemesis5,6,7. The main consequences of S. haematobium infection are haematuria, dysuria, nutritional deficiencies, urinary bladder lesions, hydronephrosis, urinary bladder squamous cell carcinoma and in children, growth retardation8. Preventive chemotherapy using mass drug administration (MDA) of praziquantel targeting primary school aged children is the main strategy for controlling schistosomiasis in Tanzania9.
An important aspect for establishing preventive chemotherapy strategy for schistosomiasis is to understand the geographical distribution of the disease and the infection level in endemic communities living in different geographical settings10,11,12,13. It remains important to identify areas where infections have continued to be a public health problem despite repeated rounds of MDA, this will allow development of a focused integrated control measures14,15. In many of the schistosomiasis endemic countries, this has never been the case, for example in Tanzania, there is inadequate attention given to research on the geographical distribution of schistosomiasis in other areas outside the historically known and highly researched areas16,17,18,19. This paucity of data affects the designing, implementation, monitoring and evaluation of control interventions. In Tanzania, the general knowledge indicate that transmission of schistosomiasis occurs within large freshwater water bodies such as lakes, dams, irrigation schemes and in seasonal/temporal water bodies4. The north-western region located along the Lake Victoria basin and the southern part of the Lake Victoria are known to be highly endemic to both S. mansoni and S. haematobium infections4. However, there is limited information on the geographical distribution of schistosomiasis in the two other large water bodies in Tanzania, Lake Tanganyika in the western region4,20,21 and Lake Nyasa in the southern highlands4,22. We focused on the Lake Nyasa, in the southern part of the country, on the international border between Malawi, Mozambique and Tanzania. Available information from Malawi, indicate that villages surrounding Lake Malawi are highly endemic to S. haematobium with the prevalence ranging from 21 to 72.7%23, whereas inland villages have moderate prevalence ranging from 10.2 to 26.4%23. The prevalence of S. mansoni infection is low in the lakeshore communities (3.9%)23. In contrast, the endemicity levels of schistosomiasis disease/infection in villages surrounding Lake Nyasa on the Tanzanian side is not known. The lack of epidemiological data on the Tanzanian side of the lake may present a unique challenge for the control efforts on each side of the border/lake. In addition, this has implication in the design and implementation of control measures especially MDA at this time when resources are very scarce and without data on geographical distribution of schistosomiasis, resources might be allocated to areas where they are not needed or underestimated24,25. Thus, accurate and up-to-date information on schistosomiasis infection/disease is essential for informing the design and implementation of a cost-effective control interventions and monitoring the impact of MDA at village, district, regional and national levels24. In addition, describing the geographical distribution and prevalence of infection in areas bordering countries opens cross-border cooperation and collaboration to fight against neglected tropical diseases23. The mapping exercises always target the most at risk population such as school aged children to demonstrate the true prevalence and burden of the infection16. Several studies have demonstrated that school aged children harbour the highest prevalence and intensity of schistosome infections1. Recent data shows that pre-school aged children are also infected with schistosomes at early ages and carries heavy intensity of infection26. Thus, inclusion of the highly risk two groups in the mapping surveys will demonstrate a clear picture of the infection and transmission status of the disease. In that context, the present cross-sectional study was conducted to determine the prevalence, infection intensities and geographical distribution of schistosomiasis (both S. haematobium and S. mansoni) infection among pre-school aged children and school aged children living in villages surrounding Lake Nyasa, in Nyasa District, Southern Tanzania. Understanding the prevalence and geographical distribution not only help in identifying high risk communities but also help in planning and implementation of mass drug administration based on level of infection24.
Methods
Study area
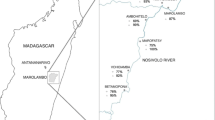
Nyasa district is among the six (6) districts of Ruvuma region and the district lies between latitudes 10,015′N and 1103′S and longitudes 34,024′W and 350,228′E. The district has a total area of 3811 km2 and located between 1200 and 2000 M above the se level. The district, border Lake Nyasa to the West. Lake Nyasa/lake Malawi is located on the southern part of the East African Rift valley and is an international border of Malawi, Mozambique and Tanzania. The districts border Mozambique to the south and Malawi to the West (separated by Lake Nyasa). According to the 2012 national census, the district was occupied by 146,160 inhabitants27.
In terms of topography, the Livingstone mountains forms the narrow stretches of lowland to Lake Nyasa and the mountains ranges are the main sources of seasonal rivers and streams flowing down the Lake. The district is characterised by uni-modal rainfall pattern starting from November or December and ends in April/May. On average, the districts receive an approximately 1224 mm of rainfall per year and temperature ranges from 29 to 31 °C and drops between 19 and 23 °C during June and August (the cold season). The main economic activities of the inhabitants are mainly subsistence farming growing cassava, maize and sweet potatoes and fishing. Fishing is the main economic activity practiced in villages located along the shorelines of the Lake Nyasa. The study was conducted in villages located within a radius of 5-km from the shoreline of Lake Nyasa basin and they included Hongi, Chiulu, Kwambe, Litimba, Liwundi, Lundo, Lundu, Mbuli, Tumbi and Mbaba-bay. Schistosomiasis control in the district mainly focus on MDA targeting primary school aged children and is organized and implemented by the district health department under the National Neglected Tropical Diseases Control Programme. No any other schistosomiasis intervention measure are implemented in the district under the district NTD control programme.
Study design, inclusion and exclusion criteria
This was a cross-sectional study conducted from September to October, 2019 among pre-school (aged 1–5 years) and school aged children (aged 6–13 years from class 1–5) in selected villages surrounding Lake Nyasa. Based on previous knowledge among school children of inverse relationship between prevalence of S.mansoni and proximity to the lake10,25 and the knowledge that the prevalence of S.haematobium increases with increase in distance to inland from the lake shore in north-western Tanzania4, village located within a radius of 5 km within the lake basin were selected for the study. The study included children aged 1–13 years, living in the study villages, having no history of participating in previous rounds of MDA in the past 6 months prior to the study, who were either male or female, had signed parent/guardians informed consent.
Sample size and sampling strategies
Based on the previous evidence on the distribution of S. haematobium and S. mansoni along the Lake Malawi (Lake Nyasa in Tanzania) basin23, village located along the shoreline of the lake were purposively sample based on their proximity to the lake. The WHO recommended sample size and sampling strategies for evaluating prevalence and intensity of infection for the purpose of assessing the need for control measure was used28,29. This cross-sectional survey(s) involved a random selection of 150–250 pre-school and school aged children from the list of the registered students in school attendance book on the day of sample collection. The sampling technique used in this study has been described elsewhere11,30.
Data collection
Collection of participants information
A pre-tested questionnaire was used to collect children’s demographic information (age, sex) and height. The questionnaire also collected information relating to participation in mass drug administration. The questionnaire for children aged 3–6 years was administered to their caregivers (parents/guardians). The data collection form was translated to Kiswahili and back-translated to English before data entry.
Parasitological screening for Schistosoma mansoni using Kato Katz technique
Duplicate Kato Katz (KK) thick smears were prepared from a single stool samples collected from each participating child. A template of 41.7 mg was used to make the thick smears. The prepared smears were examined by two independent laboratory technicians trained on KK technique. For quality assurance, 20% of all the positives and negative KK thick smears were re-examined by a third laboratory technician blinded of the results of the other two technicians.
Examination of Schistosoma mansoni circulating cathodic antigen
A single urine sample was obtained from 574 pre-school and school aged randomly selected children who participated in the study due to limited number of the POINT OF CARE CIRCULATING CATHODIC ANTIGEN TESTS (POC-CCA) test kits. The POC-CCA test active infection by detecting the CCA produced by live worms in the blood vessels and excreted in urine of infected individuals31 (http://www.rapid-diagnostics.com/). The test was performed by laboratory technicians trained using the manufacturers manual.
Parasitological screening for Schistosoma haematobium infection
A single urine sample was collected from all pre-school and school aged children participated in the study. Urine collection was done between 10:00 AM and 2:00 PM at the agreed site in the community and at the school environment. Collected urine sample were grossly examined for macro haematuria and using a urine dipstick/urinalysis reagent strips (MISSION, Expert, USA) was used to determine micro-haematuria. A filtration technique was used for screening of urine samples and a light microscopy was used to examined urine filters for presence of S. haematobium eggs32. Each sample was examined independently by two medical laboratory technicians and at the end of each fieldwork day, 20% of all the positive and negative samples were re-examined as described above.
Geographical distribution of infection
To determine the geographical distribution of the infection prevalence, positions of all the villages/10 primary schools participating in the study were mapped using a Tropical Data Kit Tools using the GPS software installed in a mobile phone or tablets33. All the collected coordinates were imported in ARVVIEW Software (https://www.esri.com/en) and allowed the maps of the district to be generated (Figs. 3 and 4).
Data analysis
Data were double entered in a MICROSFOT EXCEL SHEET, cleaned and exported to STATA VERSION 15 (StataCorp, 2017). The focus of the analysis was to determine the prevalence of S. haematobium and S. mansoni based as diagnosed using Kato Katz technique, Urine filtration technique POINT OF CARE CIRCULATING CATHODIC ANTIGEN TESTS (POC-CCA) rapid test. Continuous (age, eggs intensities) variables were summarised using mean ± standard deviation (SD). Frequencies/proportions/categorical were compared using Chi-square (χ2) or Fisher’s exact tests and continuous variables were compared using t-test. For S. mansoni eggs, arithmetic mean eggs counts were obtained from the counts of four KK smears and multiplied by 24 to obtain the individuals’ eggs per gram of feaces. The mean egg counts for S. mansoni between sex and age groups were compared using either t-test (two groups) or ANOVA (for more than two groups). Intensity of infection was categorized according to WHO criteria, 1–99 epg, 100–399 epg and ≥ 400 defined as low, moderate and heavy intensities of infection34. For S. haematobium infection, the Geometrical Mean eggs output was estimated based only on infected children because the number of infected children was too small. Infection intensities were classified into two categories as per WHO recommendation35 (i) light infection (< 50 eggs/10 ml of urine) and (ii) heavy infections (≤ 50 eggs/10mls of urine).
Ethics approval and consent to participate
Ethical approval for this study was provided by the National Ethical Committee, The National Institute for Medical Research, Tanzania (cert. NIMR/HQ/R.8a/Vol.1X/3061) and the methods used to collect the presented data followed the recommended standard operating procedures and the study was conducted according to Helsinki recommendation. The study received further permission from regional and districts authorities of Ruvuma and Nyasa district. The village authorities were also informed before data collection was done. Parents and guardians of pre-school children and school aged children received information through the village government communication channels and the school. Written informed assent and consent form were received from children and parents/guardians before participation in the study. An assent form was developed for children aged 9–13 years and they were informed about the study procedures. Confidentiality was maintained throughout the study. All children diagnosed with either S. haematobium or S. mansoni were treated with PZQ (40 mg/kg) according to WHO recommendation.
Results
A total of 1560 pre-school (34.9%) and school aged children (65.1%) aged 1–13 years participated in the study. Of these children, 46.3% (722/1560) and 53.7% 838/1560) were female and male respectively. Table 1 shows distribution of sex and age among the study participants.
Prevalence of haematuria, Schistosoma haematobium and infection intensity
The overall prevalence of haematuria (both macro and micro haematuria) was 0.9% (14/1560) with no sex difference (χ2 = 0.6346, P = 0.43). Based on the urine filtration technique, 0.83% (13/1560) (95%CI: 0.48–1.4) of the children screened had S. haematobium infection. Almost 79% (11/13) of the children detected with eggs of S. haematobium in their urine samples were from the age group 6–10 years. In relation to S. haematobium infection intensity, 61.5% (8/13) and 38.5% (5/13) of the children had light and heavy infection intensity respectively. The overall geometrical mean egg count was 18.53 mean egg counts/10 ml of urine (95%CI: 5.9–57.6).
Prevalence and intensity of Schistosoma mansoni based on Kato Katz technique
The overall prevalence of Schistosoma mansoni was 15.1% (95%: 13.4–16.9), with no gender difference but with significant difference among age groups (χ2 = 10.3642, P = 0.001). The age group 6–10 years and 11–13 years had the highest prevalence of S. mansoni infection at 16.7% and 21.9% respectively (Fig. 1). The overall arithmetic mean eggs intensity of S. mansoni was 34.7 eggs per gram of feaces (34.7 ± 141.06 epg, 95%CI: 27.7–41.7), with no significant difference between sex (t = 1.1567, P = 0.24) but with significance difference between age groups (F = 4.99, P = 0.01). The age groups 6–10 years (41.88 ± 168.4 epg) and 11–13 years (53.9 ± 138 epg) had the highest mean eggs intensities. Based on the WHO criteria for infection intensity, 40%, 42.1% and 17.8% of the children had mild, moderate and heavy infection intensities.
Prevalence of Schistosoma mansoni infection based on Point-of-Care Circulating Cathodic antigen rapid test
A total of 574 randomly school children from all the selected study villages were screened for S. mansoni infection using Point-of-Care Circulating Cathodic Antigen test. Of the screened children, 244 (42.5%) were children under the age of five years. The overall prevalence of S. mansoni based on the POC-CCA test was 21.8% (95%CI: 18.5–25.3) with neither age (χ2 = 0.2181, P = 0.8) nor sex (χ2 = 2.0679, P = 0.2). Almost 79% of the children diagnosed with S. mansoni were detected by both POC-CCA and Kato Katz technique (Fig. 2).
Distribution of Schistosoma mansoni and Schistosoma haematobium in the selected villages along Lake Nyasa
Using both KK technique and POC-CCA, Schistosoma mansoni was the most frequently diagnosed in all the ten villages selected for the study. Using the KK technique, the prevalence of S. mansoni ranged from 2.8 to 41.1% in Zambia (Mbaba-bay area). Villages which were located on the southern end of the Lake Nyasa had significantly higher prevalence of S. mansoni based on both test than villages which were located on the north side of the lake (χ2 = 178.7838, P = 0.001) (Figs. 3 and 4).
For urogenital schistosomiasis using urine filtration technique, out of the ten villages included in the study, S. haematobium was detect in three (3) villages only, which included Lundu (1%), Tumbi (1%) and Zambia-Mbaba-bay (7%) (Fig. 3).
Discussion
Although Tanzania bears a disproportionately high burden of schistosomiasis in the African region, the mapping of schistosomiasis prevalence remains incomplete in many areas of the country. To our knowledge, this is the first cross-sectional study which provides evidence of existence and endemicity of urogenital and intestinal schistosomiasis in communities living along the shorelines of Lake Nyasa, southern Tanzania. The prevalence of schistosomiasis infection increased with increase in age of the children. There was a variation in prevalence of S. haematobium and S. mansoni between villages, with villages located on the southern part of the lake having high prevalence of S. mansoni infection (diagnosed by POC-CCA and KK technique) compared to village located on the northern part of the lake. The prevalence of S. haematobium was very low and cases were detected only in three villages located on north and southern part of the lake. Our findings on the existence of S. haematobium and S. mansoni infections along the shorelines of Lake Nyasa are consistent with previous research conducted in communities living along shorelines of the Lake Malawi in Malawi23.
Prevalence and intensities of S. mansoni and S. haematobium infection
The overall prevalence of S. mansoni based on Kato Katz technique was lower compared to the prevalence of > 50% recorded among pre-school and school aged children along the shoreline of the Lake Victoria in north-western Tanzania10,26,36. However, the observed prevalence was higher compared to studies conducted along the same lake shore communities in Malawi where they recorded a prevalence 3.9% based on Kato Katz technique23. Using Point of Care Circulating Cathodic Antigen test, the overall prevalence of schistosomiasis was 21.8% which was lower compared to 80%26 recorded in pre-school children in Ukerewe island and 71.3%13 recorded among school aged children along the Lake Victoria in north-western Tanzania13 and on islands of Lake Victoria in Uganda13,37. The variation in prevalence of S. mansoni from one epidemiological setting to another can be explained by the focal distribution of S. mansoni, abundance and competence of the intermediate snail hosts, proximity of the villages/communities to transmission sites such as the lake, effectiveness of the control intervention such as MDA and level of environmental contamination with human feaces10,11,12,25.
The prevalence and intensity of S. mansoni increased with increase in the age group of the children, the youngest had lower prevalence and intensity of infection compared to those aged above 6 years. This is a common observed in S. mansoni endemic communities and the age prevalence curve shows that S. mansoni prevalence and infection intensities usually peaks at the age groups 6–19 years, thereafter, declines with increased age38. However, it is worth noting that in high transmission setting like around the Lake Victoria basin in north-western region of Tanzania, the prevalence and intensities of infection are similar to all age groups6,39. The present study noted that in this setting, S. mansoni infection starts at very young ages (< 5 years), meaning that the age group is exposed at early life and this calls for inclusion of the age group in treatment programme. This observation is consistent to what has been reported in schistosomiasis endemic areas in sub-Saharan Africa26,40,41.
In contrast, the prevalence of S. haematobium in the screened pre-school and school-aged children was lower compared to reports of previous studies conducted in Zanzibar (5.4%)42 and north-Western Tanzania (34.8%)43. The observed prevalence of S. haematobium in the present study remained lower than the prevalence of 56.2% to 94% of S. haematobium observed among schools aged children in lakeshore communities of Lake Malawi23 and in the inland communities in Malawi (15.3–57.1%)23. The contrasting findings on the S. haematobium infection prevalence in communities living along the shorelines of the lake between the two countries could partly be explained by the focal distribution of the S. haematobium intermediate hosts in a defined geographical area, while in Malawi, high transmission appears to occur along the lakeshore communities compared to inland communities23, in Tanzanian side of the same lake, S.haematobium transmission is very low but the area maintain high transmission of S. mansoni. Perhaps, transmission of S. haematobium in the area surrounding Lake Nyasa occurs more on the inland areas compared to lakeshore areas. The present study did not include the inland communities located out of the lake shorelines and malacological surveys were not done. The inclusion of these areas could have given a different picture of S. haematobium and S. mansoni transmission levels in the district. This call for a follow-up study.
On the other hand, all children identified to be infected with S. haematobium were aged > 6 years and majority had low infection intensities. Supporting evidence from previous studies which have reported that children aged > 6 years remains at high risk of S. haematobium than the youngest age groups43. This can be explained by increased duration and frequencies of human-water contact activities among the members of the age group which exposes them to infection43. The absence of S. haematobium infection in children under five in the present study can also be explained by a low transmission of S. haematobium in the study setting or limitation of parasitological techniques to identify children with low infection intensities, which is common observation in children under-fives42. In sub-Saharan Africa, S. haematobium infection has been noted to start at young age44,45,46, for instance in Zimbabwe, S. haematobium infection starts at young ages (< 5 years) and these children carries high infection intensities47. Probably, high transmission of the parasite occurs in water bodies which are accessible by children under five years and this exposes them to infection48. Previous studies noted that distance from household to an open water sources increases the risk of S. haematobium infection49. A recent knowledge indicates that the presence of the actual habitat close to the household(s) was a predictor of S. haematobium infection rather than distance43.
Variation in prevalence of S. mansoni and S. haematobium by villages
A noted variation in prevalence of S. haematobium and S. mansoni infection prevalence was observed between the villages involved in the study. Schistosoma mansoni infection was present in all the selected villages at varying prevalence, with the villages located on the southern part of the lake having the highest prevalence compared to villages located on the northern part of the lake. Consistent observation has been reported in S. mansoni endemic areas, with a variation occurring just within a single village10,11,12,25. In contrast, in lake Malawi, the prevalence of S. mansoni was very low in villages located along the shorelines23. An inverse relationship between proximity to the lake and prevalence of S. mansoni prevalence and infection intensity has been noted by previous studies12,25, with villages located away from the lake shoreline having the lowest prevalence25. For S. haematobium, cases were only identified in only three (3) villages which were located on the northern part of the lake. The prevalence was very low compared to what was observed in villages located along the shoreline of lake Malawi23, which indicated that transmission of S. haematobium was high in villages located along the lake shoreline compared to inland villages23. In addition, in Malawi, villages located on the southern part of the lake had higher prevalence of S. haematobium mainly in shallow water of the lake23. Overall, the observed difference in prevalence of S. mansoni and S. haematobium between villages indicate that, in these villages, transmission of S. mansoni is high and occurs along the lake shoreline, while the transmission of S. haematobium is low along the shoreline’s villages. However, this observation needs follow-up studies to understand in detail the ecology of transmission of S. mansoni and S. haematobium infections with a focus to understand the transmission patterns, involved intermediate hosts and their distributions and geographical variations among villages located at different points from the lake shoreline. In lake Malawi, where transmission of S. haematobium is very high, the main intermediate host is Bulinus nyassanus and in the inland villages both Bulinus nyassanuss and Bulinus globosus are involved23. In the area, the transmission of S. haematobium is limited to areas where the B. nyassannus occurs, mainly in shallow water close to the shore23. For S. mansoni, the main intermediate host reported was Biomphalaria pfeifferi and was noted to be predominant in the inland villages compared to lakeshore villages23, explaining the low prevalence of the infection in lakeshore communities. However, this information is not available on the Tanzanian side of lake Nyasa and calls for extensive malacological studies in this area.
Implication for mass drug administration
The prevalence and distribution of S. mansoni and S. haematobium infection in the study villages has implication on the planning and implementation of mass drug administration. The WHO health organization recommends MDA against schistosomiasis when the prevalence exceeds 10%34. Considering this recommendation, most of the villages requires MDA against S. mansoni infection at least one round every year with exception of only two villages which will require one rounds of MDA after every one year34. For S. haematobium infection, if resources are enough, a test and treat approach is recommended to reduce the wastage of drugs. The fact that S. mansoni and S. haematobium are co-endemic in some villages, MDA against one of them especially S. mansoni will take care of the S. haematobium infection. Our study has noted that children aged 5 years and below were also infected by S. mansoni infection and the age group is normally excluded from MDA programme44,45. It is worthwhile to note that children under-fives carry heavy infection intensity similar to primary school aged children and adult individuals in endemic areas26,40,47. Available evidence indicated that in endemic areas children under-five bears significant hepatosplenic and urogenital morbidities associated with S. mansoni and S. haematobium infection44,50. These evidences indicate the need for inclusion of children under-fives for preventive chemotherapy for schistosomiasis and studies have demonstrated that the currently available PZQ drug has less side effects in the age group47. Thus, planning and implementation of next MDA rounds should consider inclusion of this age groups.
Although the current study has shown for the first time the geographical distribution of S. haematobium and S. mansoni in villages located along the Lake Nyasa in southern Tanzania, it is worthwhile to note that the study included only selected ten (10) villages located along the shorelines. Village located outside the lake shoreline and those located far south of the lake were not included. This may limit the generalizability of the findings to other areas outside the district. In addition, the study did not include ultrasound studies to understand the level of hepatosplenic and urogenital morbidities associated with S. mansoni and S. haematobium in pre-school and school aged children. Future studies should cover these gaps.
Conclusion
Our findings demonstrate that villages in closest proximity to lake Nyasa in southern Tanzania had the highest S. mansoni prevalence while the prevalence of S. haematobium was very low (below 10%). These findings provide guide for planning and implementation of mass drug administration against schistosomiasis in Nyasa district. Furthermore, the findings have further demonstrated the need for paediatric praziquantel formulation or any feasible intervention to address schistosomiasis in pre-school children. Lastly, the findings call for further studies to understand the ecology of schistosomiasis transmission (malacological surveys) in villages surrounding lake shore and inland areas of Nyasa district.
Data availability
Data supporting the write-up and conclusion of this article are provided with the articles. Raw data are available for sharing upon reasonable request through the institutional review board/ethical review committee.
Abbreviations
- PPF:
-
Periportal fibrosis
- MDA:
-
Mass drug administration
- SAC:
-
School aged children
- PSAC:
-
Pre-school aged children
- PZQ:
-
Praziquantel
- KK:
-
Kato Katz technique
- POC-CCA:
-
Point-of-care circulating cathodic antigen
- GPS:
-
Geographical positioning system
- NIMR:
-
National Institute for Medical Research
- NTD:
-
Neglected tropical diseases
References
Hotez, P. J. & Kamath, A. Neglected tropical diseases in sub-saharan Africa: Review of their prevalence, distribution, and disease burden. PLoS Negl. Trop. Dis. 3, e412. https://doi.org/10.1371/journal.pntd.0000412 (2009).
Steinmann, P., Keiser, J., Bos, R., Tanner, M. & Utzinger, J. Schistosomiasis and water resources development: Systematic review, meta-analysis, and estimates of people at risk. Lancet Infect. Dis. 6, https://doi.org/10.1016/S1473-3099(06)70521-7 (2006).
Rollinson, D. et al. Time to set the agenda for schistosomiasis elimination. Acta Trop. 128, 423–440. https://doi.org/10.1016/j.actatropica.2012.04.013 (2013).
Mazigo, H. D. et al. Epidemiology and control of human schistosomiasis in Tanzania. Parasit. Vectors 5, 274. https://doi.org/10.1186/1756-3305-5-274 (2012).
Chofle, A. A. et al. Oesophageal varices, schistosomiasis, and mortality among patients admitted with haematemesis in Mwanza, Tanzania: A prospective cohort study. BMC Infect. Dis. 14, 303. https://doi.org/10.1186/1471-2334-14-303 (2014).
Mazigo, H. D. et al. Periportal fibrosis, liver and spleen sizes among S. mansoni mono or co-infected individuals with human immunodeficiency virus-1 in fishing villages along Lake Victoria shores, North-Western, Tanzania. Parasit. Vectors 8, 260, https://doi.org/10.1186/s13071-015-0876-4 (2015).
Malenganisho, W. L. et al. Schistosoma mansoni morbidity among adults in two villages along Lake Victoria shores in Mwanza District, Tanzania. Trans. R. Soc. Trop. Med. Hyg. 102, https://doi.org/10.1016/j.trstmh.2008.03.006 (2008).
van der Werf, M. J. et al. Quantification of clinical morbidity associated with schistosome infection in sub-Saharan Africa. Acta Trop. 86, 125–139 (2003).
WHO. Schistosomiasis: Progress Report 2001–2011, Strategic Plan 2012–2020. 2013. https://doi.org/10.1016/j.parint.2011.10.006 (World Health Organization, Geneva, 2012).
Mugono, M. et al. Intestinal schistosomiasis and geohelminths of Ukara Island, North-Western Tanzania: Prevalence, intensity of infection and associated risk factors among school children. Parasit. Vectors 7, 612. https://doi.org/10.1186/s13071-014-0612-5 (2014).
Odiere, M. R. et al. High prevalence of schistosomiasis in Mbita and its adjacent islands of Lake Victoria, western Kenya. Parasit. Vectors 5, 278. https://doi.org/10.1186/1756-3305-5-278 (2012).
Odiere, M. R. et al. Geographical distribution of schistosomiasis and soil-transmitted helminths among school children in informal settlements in Kisumu City, Western Kenya. Parasitology 138, 1569–1577. https://doi.org/10.1017/S003118201100059X (2011).
Standley, C. J. et al. Performance of circulating cathodic antigen (CCA) urine-dipsticks for rapid detection of intestinal schistosomiasis in schoolchildren from shoreline communities of Lake Victoria. Parasit. Vectors 3, 7. https://doi.org/10.1186/1756-3305-3-7 (2010).
Kittur, N. et al. Defining persistent hotspots: Areas that fail to decrease meaningfully in prevalence after multiple years of mass drug administration with praziquantel for control of schistosomiasis. Am. J. Trop. Med. Hyg. https://doi.org/10.4269/ajtmh.17-0368 (2017).
Pennance, T. et al. Urogenital schistosomiasis transmission on Unguja Island, Zanzibar: Characterisation of persistent hot-spots. Parasit. Vectors 9, 646. https://doi.org/10.1186/s13071-016-1847-0 (2016).
Clements, A. C. et al. Bayesian spatial analysis and disease mapping: Tools to enhance planning and implementation of a schistosomiasis control programme in Tanzania. Trop. Med. Intern. Health 11, 490–503. https://doi.org/10.1111/j.1365-3156.2006.01594.x (2006).
Brooker, S. et al. Predicting the distribution of urinary schistosomiasis in Tanzania using satellite sensor data. Trop. Med. Int. Health 6, 998–1007 (2001).
Webbe, G. The transmission of Schistosoma haematobium in an area of Lake Province, Tanganyika. Bull. World Health Organ. 27, 59–85 (1962).
Webbe, G. Known transmission patterns of S. haematobium in Tanganyika and the possible influence of irrigation on incidence of infection. East Afr. Med. J. 40, 235–239 (1963).
Bakuza, J. S., Denwood, M. J., Nkwengulila, G. & Mable, B. K. Estimating the prevalence and intensity of Schistosoma mansoni infection among rural communities in Western Tanzania: The influence of sampling strategy and statistical approach. PLoS Negl. Trop. Dis. 11, e0005937. https://doi.org/10.1371/journal.pntd.0005937 (2017).
Steiner, F. et al. Acute schistosomiasis in European students returning from fieldwork at Lake Tanganyika, Tanzania. J. Travel Med. 20, 380–383. https://doi.org/10.1111/jtm.12069 (2013).
Webbe, G. & Jordan, P. Recent advances in knowledge of schistosomiasis in East Africa. Trans. R. Soc. Trop. Med. Hyg. 60, 279–312 (1966).
Madsen, H. et al. Schistosomiasis in Lake Malawi villages. EcoHealth 8, 163–176. https://doi.org/10.1007/s10393-011-0687-9 (2011).
Cha, S. et al. Epidemiological findings and policy implications from the nationwide schistosomiasis and intestinal helminthiasis survey in Sudan. Parasit. Vectors 12, 429. https://doi.org/10.1186/s13071-019-3689-z (2019).
Handzel, T. et al. Geographic distribution of schistosomiasis and soil-transmitted helminths in Western Kenya: Implications for anthelminthic mass treatment. Am. J. Trop. Med. Hyg. 69, 318–323 (2003).
Ruganuza, D. M., Mazigo, H. D., Waihenya, R., Morona, D. & Mkoji, G. M. Schistosoma mansoni among pre-school children in Musozi village, Ukerewe Island, North-Western-Tanzania: prevalence and associated risk factors. Parasit. Vectors 8, 377. https://doi.org/10.1186/s13071-015-0997-9 (2015).
Tanzania National Bureau of Statistics. Tanzania Populations Census 2012. (Tanzania Government, 2012).
Montresor, A., Crompton, D.W., Hall, A., Bundy, D.A.P. & L. S. Guidelines for the Evaluation of Soil‐Transmitted Helminthiasis and Schistosomiasis at Community Level. (WHO/CTD, 1998).
Lwanga, S.K. & Lemeshow, S. Sample Size Determination in Health Studies: A Practical Manual 1–22 (World Health Organization, Geneva, 1991).
Mintsa Nguema, R. et al. Baseline mapping of schistosomiasis and soil transmitted helminthiasis in the Northern and Eastern health regions of Gabon, Central Africa: Recommendations for preventive chemotherapy. Trop. Med. Infect. Dis. 3, 119 (2018).
Van Dam, G. et al. Diagnosis of schistosomiasis by reagent strip test for detection of circulating cathodic antigen. J. Clin. Microbiol. 42, 5458–5461 (2004).
WHO. Basic Laboratory Methods in Medical Parasitology. (World Health Organization, 1991).
Hartung, C., Lerer, A., Anokwa, Y., Tseng, C. & Brunette, W. Open data kit: Tools to build information services for developing regions. in Proceedings of the 4th ACM/IEEE International Conference on Information and Communication Technologies and Development. December 13–16, 2010, London, United Kingdom 1–12 (2010).
World Health Organization. Prevention and control of schistosomiasis and soil-transmitted helminthiasis. World Health Organ. Tech. Rep. Ser. 912, i (2002).
Montresor, A., Crompton, D.W.T., Hall, A., Bundy, D.A.P. & Savioli, L. Guidelines for the Evaluation of Soil-Transmitted Helminthiasis and Schistosomiasis at Community Level. (World Health Organization, Geneva, 1998).
Mazigo, H. D. et al. Co-infections with Plasmodium falciparum, Schistosoma mansoni and intestinal helminths among schoolchildren in endemic areas of northwestern Tanzania. Parasit. Vectors 3, 44. https://doi.org/10.1186/1756-3305-3-44 (2010).
Standley, C. J., Adriko, M., Besigye, F., Kabatereine, N. B. & Stothard, R. J. Confirmed local endemicity and putative high transmission of Schistosoma mansoni in the Sesse Islands, Lake Victoria, Uganda. Parasit. Vectors 4, 29. https://doi.org/10.1186/1756-3305-4-29 (2011).
Barakat, R., Farghaly, A., El Masry, A. G., El-Sayed, M. K. & Hussein, M. H. The epidemiology of schistosomiasis in Egypt: Patterns of Schistosoma mansoni infection and morbidity in Kafer El-Sheikh. Am. J. Trop. Med. Hyg. 62, 21–27 (2000).
Malenganisho, W. L. et al. Schistosoma mansoni morbidity among adults in two villages along Lake Victoria shores in Mwanza District, Tanzania. Trans. R. Soc. Trop. Med. Hyg. 102, 532–541. https://doi.org/10.1016/j.trstmh.2008.03.006 (2008).
Odogwu, S. E. et al. Schistosoma mansoni in infants (aged < 3 years) along the Ugandan shoreline of Lake Victoria. Ann. Trop. Med. Parasitol. 100, 315–326. https://doi.org/10.1179/136485906X105552 (2006).
Kabetereine, N.B. et al. Adult resistance to schistosomiasis mansoni: Age-dependence of re-infection remains in communities with diverse exposure patterns. . Parasitology 118, 101–105 (1999).
Knopp, S. et al. Urogenital schistosomiasis elimination in Zanzibar: accuracy of urine filtration and haematuria reagent strips for diagnosing light intensity Schistosomahaematobium infections. Parasit. Vectors 11, 552. https://doi.org/10.1186/s13071-018-3136-6 (2018).
Angelo, T. et al. Geographical and behavioral risks associated with Schistosomahaematobium infection in an area of complex transmission. Parasit. Vectors 11, 481. https://doi.org/10.1186/s13071-018-3064-5 (2018).
Stothard, J.R., Sousa-Figueiredo, J. C., Betson, M., Bustinduy A & J., R.-R. Schistosomiasis in African infants and preschool children: Let them now be treated. Trends Parasitol. 29, 197–205 (2013).
Mduluza, T. & Mutapi, F. Putting the treatment of paediatric schistosomiasis into context. Infect. Dis. Poverty 6, 85. https://doi.org/10.1186/s40249-017-0300-8 (2017).
Osakunor, D. N. M., Woolhouse, M. E. J. & Mutapi, F. Paediatric schistosomiasis: What we know and what we need to know. PLoS Negl. Trop. Dis. 12, e0006144. https://doi.org/10.1371/journal.pntd.0006144 (2018).
Mutapi, F. et al. Schistosomahaematobium treatment in 1–5 year old children: Safety and efficacy of the antihelminthic drug praziquantel. PLoS Negl. Trop. Dis. 5, e1143. https://doi.org/10.1371/journal.pntd.0001143 (2011).
Mutsaka-Makuvaza, M. J. et al. Urogenital schistosomiasis and risk factors of infection in mothers and preschool children in an endemic district in Zimbabwe. Parasit. Vectors 12, 427. https://doi.org/10.1186/s13071-019-3667-5 (2019).
Amazigo, U. O., Anago-Amanze, C. I. & Okeibunor, J. C. Urinary schistosomiasis among school children in Nigeria: Consequences of indigenous beliefs and water contact activities. J. Biosoc. Sci. 29, 9–18. https://doi.org/10.1017/s0021932097000096 (1997).
Nalugwa, A., Nuwaha, F., Tukahebwa, E. M. & Olsen, A. Schistosoma mansoni-associated morbidity among preschool-aged children along the shores of lake Victoria in Uganda. Trop. Med. Infect. Dis. 2, https://doi.org/10.3390/tropicalmed2040058 (2017).
Acknowledgements
The author thanks the project team from the National Institute for Medical Research and the Catholic University of Health Sciences for their tireless effort to generate epidemiological data to help in planning and implementation control activities. Authors also thank the community members, children, guardians/parents and the district neglected tropical diseases control coordinators for supporting the data collection process.
Funding
This work was supported by the National Institute for Health Research (NIHR) (16/136/33) using UK aid from the UK Government to support global health research. The views expressed in this publication are those of the author(s) and not necessarily those of the NIHR or the UK Department of Health and Social Care. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the supporting offices.
Author information
Authors and Affiliations
Contributions
H.D.M., U.M. and P.K. conceptualised the study and developed the protocol. C.U. and A.N. participated in training of the field data collection team and participated in data collection. H.D.M. and U.M., participated in data analysis and interpretation and wrote the first draft of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mazigo, H.D., Uisso, C., Kazyoba, P. et al. Prevalence, infection intensity and geographical distribution of schistosomiasis among pre-school and school aged children in villages surrounding Lake Nyasa, Tanzania. Sci Rep 11, 295 (2021). https://doi.org/10.1038/s41598-020-80317-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-80317-x
- Springer Nature Limited
This article is cited by
-
Molecular diagnosis of urogenital schistosomiasis in pre-school children, school-aged children and women of reproductive age at community level in central Senegal
Parasites & Vectors (2023)
-
Detecting Schistosoma haematobium infection by microscopy and polymerase chain reaction (PCR) in school children in three senatorial districts of Cross River State, Nigeria
Journal of Parasitic Diseases (2022)