Abstract
The population is rapidly aging worldwide, and there is an age-related decline in muscle mass. Therefore, it is important to examine the prevalence and associated factors of low appendicular skeletal muscle mass index (ASMI) in older adults. The objectives of this cross-sectional study were (i) to determine the prevalence of low ASMI (ASM/height2) and (ii) to identify factors associated with low ASMI. This study included 1211 community-dwelling adults aged ≥ 65 years. Low ASMI was defined as < 7.0 kg/m2 in males and < 5.7 kg/m2 in females (bioelectrical impedance analysis). Gender-specific cut-off values of calf circumference for low ASMI were determined. The prevalence of low ASMI in the overall cohort was 59.9%, i.e., 57.0% among males and 61.8% among females, with no significant difference between genders (P = 0.1068). The prevalence of low ASMI was 81.3% in individuals at risk of malnutrition compared to 20.6% in their counterparts with normal nutritional status (P < 0.0001). Participants with low ASMI were older, had lower physical activity scores, and greater likelihood of hospitalization in prior 6 months compared with normal ASMI (all P < 0.0001). Low ASMI was associated with risk of malnutrition (odds ratio: 3.58 for medium risk, odds ratio: 12.50 for high risk), older age, smoking, drinking, smaller calf circumference, and lower bone mass (all P ≤ 0.0328). Cut-off values of calf circumference for low ASMI for males was 33.4 cm and for females was 32.2 cm. In conclusion, we found that low ASMI was highly prevalent among community-dwelling older adults at risk of malnutrition. Other significant factors associated with low ASMI were age, smoking, drinking, calf circumference, and bone mass. Screening community-dwelling older adults for risk of malnutrition can prevent or delay onset of low ASMI.
Similar content being viewed by others
Introduction
Aging is associated with physiological decline in skeletal muscle mass, which can escalate the tolls on overall health and function, and may ultimately increase risk of death1,2,3. Across Asia, low muscle mass is reported to occur commonly in community-dwelling older adults4,5,6,7,8, ranging from 20 to 63%. It is also known that women have lower muscle mass than men9. Since age-related muscle decline occurs in both women and men, women remain to have lower muscle mass than men in older age10,11,12,13. Thus, gender-specific measurements and cut-offs are important for more accurate identifications of low muscle mass, especially for surrogate marker of muscle mass such as calf circumference.
Factors predisposing older adults to poor muscle health are low socio-economic status, underlying chronic diseases, poor nutritional intake, and adverse lifestyle (Fig. 1). Socio-economic deprivation includes low income and low education level14,15. Chronic diseases which can increase the risk of muscle decline include diabetes and chronic obstructive pulmonary disease16,17. In addition, acute illness or injury can initiate a cascade ultimately leading to poor muscle health18. In older adults, anorexia of aging is associated with low muscle mass and strength19. Lifestyle-related risks such as physical inactivity20, smoking21, and drinking14 are also associated with poor muscle health.
Older adults with evidence of low muscle mass are at high risk of adverse health outcomes such as reduced mobility, impaired ability to perform activities of daily living and lower quality of life, fall-related injuries, infections, hospitalization and need for long-term care3,4 (Fig. 1). A vicious cycle may then develop resulting in more loss of muscle mass, further disability, poorer health, ultimately leading to increased mortality risk2,3. However, there is recognized heterogeneity in the literature findings on the impact of muscle mass on functional ability in older adults22,23.
In a recent narrative review, low muscle mass or sarcopenia was consistently predictive of higher healthcare expenditure in community, perioperative, and general hospital settings24. In a study of older people in Taiwan, men and women with a lower skeletal muscle mass index (SMMI) had more hospitalizations, longer lengths of stay, and higher healthcare costs25. The annual difference in hospitalization costs between individuals with highest and lowest risk for low SMMI was equivalent to about United States dollars (USD) $1200 per patient year25. A United States study estimated direct healthcare costs for people with sarcopenia at 1.5% of the total annual healthcare expenditure, i.e., costs equivalent to USD $26.3 billion in 202026. A more recent estimate of US costs for hospitalization in individuals with sarcopenia was USD $40.4 billion27.
Singapore’s population is growing older, similarly, but even faster than populations worldwide28. The global population of people ≥ 65 years has been projected to double from 703 million in 2019 to 1.5 billion by 205028. Of this total, approximately 1.0 billion older adults will be living in Asia. In Singapore, the percentage of older adults is expected to double over the next decade from 12.4% in 2019 to 22.5% in 203028. Thus, the objectives of this cross-sectional study were: (1) to determine the prevalence of low appendicular skeletal muscle mass index (ASMI; ASM/height2), and (2) to identify factors associated with low ASMI. The findings from this study may help to identify community-dwelling older adults at risk of low ASMI, in order to prevent and address the adverse consequences.
Methods
Study design and participants
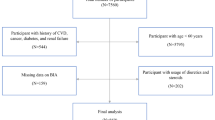
Strengthening Health In ELDerly through nutrition (SHIELD) was a cross-sectional study of community-dwelling older adults in Singapore. Participants were recruited between August 2017 and March 2019. All procedures involving human subjects were approved by the Centralized Institutional Review Board in Singapore (reference number: 2017/2271). The present study was conducted according to the ethical standards laid down in the Declaration of Helsinki. Written informed consent was obtained from all participants. The trial was registered at clinicaltrials.gov as NCT03240952. The Nutritional Health for the Elderly Reference Centre (NHERC) is the name of the overarching project. One component of NHERC is the clinical trial, which is known as the SHIELD study.
A total of 1211 individuals consisting of 400 with normal nutritional status and 811 at risk of malnutrition took part in this study. The data for this study was derived from baseline measurements of two unique cohorts of the SHIELD study29,30. Participants were recruited from the public, community centers, polyclinics, and hospitals through public talks, flyers, posters, and referrals from healthcare professionals.
Assessments
All study participants were asked to attend one visit at baseline, where the following data were collected.
Socio-demographic data such as age, gender, ethnicity, marital status, education, number of prescribed drugs, smoker status, and alcohol consumption were collected during the visit. Charlson Comorbidity Index was used to determine the comorbidity level31,32, and Malnutrition Universal Screening Test (MUST) was used to determine the risk of malnutrition33.
Physical Activity Scale for the Elderly (PASE)34,35 was used to determine physical activity level and Modified Barthel Index (MBI)36 was used to measure functional independence for ten activities of daily living.
For anthropometry and body composition measurements, standing height was measured without shoes by using a stadiometer to the nearest millimeter (Avamech B1000), and body weight and composition were measured to the nearest 0.1 kg using a bioelectrical impedance analysis (BIA) machine (Tanita MC-780). BIA was used to estimate muscle mass, fat mass, and bone mass. Mid upper arm circumference was measured at mid-point of the acromion and olecranon, and calf circumference was measured at the largest part of the calf. Both were measured to the nearest 0.1 cm.
Serum 25-hydroxyvitamin D levels were measured using immunochemistry analyzer COBAS e801 and vitamin D cut-off values were based on the definition described by Holick37.
Healthcare utilization data on hospitalization and length of stay was collected using medical records and questionnaires if the former is unavailable.
Statistical analysis
Baseline characteristics of the study participants were reported as means and standard deviation (SD) for continuous variables, and numbers and percentages for categorical variables. For continuous variables, normality of the data was assessed using the Shapiro-Wilks test (P < 0.001) and graphical methods. Two-sample t test, Wilcoxon rank sum test, fisher exact test, and chi-square test were used to compare the characteristics between genders, nutritional status, and ASMI status. Low ASMI was determined based on BIA cut-offs recommended by the Asian Working Group for Sarcopenia (AWGS)1, i.e., male < 7.0 kg/m2 and female < 5.7 kg/m2.
Multiple linear regression model was used to examine the associations between ASMI and all potential variables based on the literature. The adjusted R-square values measuring goodness of model fit were ≥ 0.6 for all the three models (overall cohort, males, and females). Multicollinearity between the predictors were tested using variance inflation factor and tolerance. The highest variance inflation factor (VIF) was bone mass with 3.6 VIF and the lowest tolerance was 0.3, which confirmed that there was no multicollinearity. Multiple logistic regression model was used to examine the associations between low ASMI and potential factors. Odds ratio and 95% confidence interval (CI) were estimated from logistic regression models.
Logistic regression and Receiver Operating Characteristics (ROC) method were used to determine the cut-off values for calf circumference associated with low ASMI in males and females. Kappa statistics was used to test the agreement between the cut-off values for low ASMI in the present study and the cut-off values for screening for sarcopenia recommended by the AWGS1, i.e., male < 34 cm and female < 33 cm.
SAS version 9.4 (SAS Institute, Cary, NC, USA) was used for all statistical analyses. P < 0.0500 was considered statistically significant.
Institutional review board statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Centralized Institutional Review Board in Singapore of SingHealth (reference number: 2017/2271 and date of approval: 23 May 2017).
Informed consent statement
Informed consent was obtained from all subjects involved in the study.
Results
Participants’ characteristics
Table 1 shows the characteristics of the overall cohort, males, and females. A total of 1211 older adults participated in this study. Mean (SD) age of participants was 73.20 (6.82) years and body mass index (BMI) was 20.43 (3.69) kg/m2. Most participants were Chinese (85.5%) and over 94% had a Charlson Comorbidity score of 0. More than half of the participants (57.0%) had either vitamin D deficiency or insufficiency. The prevalence of low ASMI in the overall cohort was nearly 60%, with no significant difference between genders (57.0% males vs. 61.8% females, P = 0.1068). Supplementary Table 1 shows the characteristics of older adults with normal nutritional status and at risk of malnutrition. The prevalence of low ASMI was 81.3% in older adults at risk of malnutrition and 20.6% for those with normal nutritional status (P < 0.0001).
Characteristics by ASMI status
Table 2 shows the characteristics of the overall cohort, males, and females by their ASMI status. In the overall cohort, compared to those with normal ASMI, participants with low ASMI were older (74.30 years vs. 71.06 years), lighter (46.05 kg vs. 59.28 kg), with lower mid upper arm circumference (23.05 cm vs. 27.07 cm), calf circumference (30.47 cm vs. 34.94 cm), and bone mass (1.93 kg vs. 2.37 kg) (all P < 0.0001). There was a significant association between ASMI status and vitamin D status (P = 0.0472). Participants with low ASMI were likely to have vitamin D deficiency compared to those with normal ASMI (13.02% vs. 18.52%; P = 0.0144, data not shown). In addition, participants with low ASMI were almost twice as likely to have been admitted to the hospital within the prior 6 months (9.62% vs. 5.74%; P = 0.0190) and had significantly lower PASE scores (103.3 vs. 121.5; P < 0.0001). There was a significant difference in education level between the older adults with normal ASMI and low ASMI (P < 0.0001), with the latter having a higher percentage with no formal education (35.07% vs. 20.13%). Supplementary Table 2 shows the characteristics of older adults by their ASMI and nutritional status.
Factors associated with ASMI
As shown in Table 3, in the overall cohort, factors associated with ASMI included age, gender, calf circumference, bone mass, and nutritional status. For every one-year increase in age while holding other factors constant, ASMI was significantly lower by − 0.011 (95% CI − 0.017, − 0.005). Females had significantly lower ASMI than males (− 0.792, 95% CI − 0.891, − 0.694). Compared to older adults with normal nutritional status, ASMI was significantly lower among those at medium risk (− 0.373, 95% CI − 0.460, − 0.286) and high risk (− 0.608, 95% CI − 0.713, − 0.503) of malnutrition. On the other hand, calf circumference and bone mass were positively associated with ASMI (both P < 0.0001). In addition, for both males and females, age and risk of malnutrition were negatively associated with ASMI, whereas calf circumference and bone mass were positively associated with ASMI (all P ≤ 0.0139).
Factors associated with low ASMI
Multiple logistic regression model showed that factors associated with low ASMI included age, smoking, drinking, calf circumference, bone mass, and nutritional status (Table 4). With every one-year increase in age, the odds of having low ASMI increased by 6% (odds ratio: 1.06, 95% CI 1.02, 1.09). Compared to non-smoker, daily or occasional smokers had significantly higher odds of having low ASMI (odds ratio: 3.76, 95% CI 1.55, 9.16). The odds of having low ASMI for older adults who drank at least once a month was 2.51 (95% CI 1.34, 4.67), compared to non-drinkers. Calf circumference and bone mass were associated with lower odds of having low ASMI (both P < 0.0001). Compared to older adults with normal nutritional status, the odds ratio of having low ASMI was 3.58 (95% CI 2.41, 5.30) among those at medium risk of malnutrition, and 12.50 (95% CI 7.02, 22.25) among those at high risk of malnutrition. In addition, for both males and females, calf circumference and bone mass were associated with lower odds of having low ASMI (all P < 0.0001). On the other hand, malnutrition risk was associated with higher odds of having low ASMI in both genders (both P < 0.0001), and age for females only (P = 0.0425).
Calf circumference cut-offs
The cut-off values of calf circumference for low ASMI for males was 33.4 cm (sensitivity = 82%, specificity = 82%) and for females was 32.2 cm (sensitivity = 82%, specificity = 74%). As shown in Fig. 2, the area under the ROC curve for males was 0.8988, and for females was 0.8580. The Kappa statistics showed strong agreement between the cut-off values of calf circumference for low ASMI derived from this study and the cut-off values for screening for sarcopenia as recommended by the AWGS (males = 0.88, females = 0.87; both P < 0.0001).
Discussion
To our knowledge, this is a first-of-kind study that determined the prevalence of low ASMI and its associated factors in community-dwelling, ambulant older adults. The findings of our study revealed that low ASMI was especially common among this cohort. We further identified factors that were associated with increased risk for low ASMI, i.e., age, malnutrition or its risk, smoking, alcohol consumption, low calf circumference, and low bone mass. These findings can be used to promote public health programs that can help older adults restore and maintain physical function, thus sustaining independent living in the community.
Gender
Men are known to have more muscle mass than women, both in terms of absolute amount as well as in relation to overall composition9. This is predominantly due to the difference in muscle mass in the upper body. However, both genders experienced the same decline in muscle mass with aging, which is noticeable after the fifth decade9. The literature has also reported gender-specific differences in mid-upper arm circumference38, calf circumference39, and ASMI1,2. These differences between genders are also reflected in the present study.
Older age
Older age was a key factor in predicting low ASMI. Our findings showed that there was a 6% increase in the odds of having low ASMI for every year older above 65. While age is a non-modifiable risk factor, it serves as an important marker for low ASMI.
This association between age and low ASMI is also reflected in the association between age and sarcopenia40,41. This is because low muscle mass is one of the diagnostic criteria for sarcopenia. In fact, researchers associated the highest likelihood of sarcopenia in the oldest old5,42, with older adults aged ≥ 70 years reported to lose up to 15% of muscle mass every decade43. In addition, participants aged ≥ 80 years were six times more likely to be at risk of sarcopenia as compared to those aged 60–80 years40. This finding is reflected in results from a Singapore study, which showed that more than one in two older adults above 80 years old will be at risk of losing at least one instrumental activity of daily living and adversely affect the ability to live independently44.
Risk of malnutrition
In our study, low ASMI was significantly and prominently correlated with poor nutritional status. The odds of having low ASMI for older adults at high risk of malnutrition was 12.5 compared to their nourished counterparts, after adjusting for potential confounders. In fact, low muscle mass is now considered a defining characteristic of malnutrition45. The association between low muscle mass and malnutrition has been reported in the literature46,47,48. According to Landi et al., malnutrition needs to be addressed clinically as a muscle-related disorder, and clinicians should integrate nutrition assessment with muscle mass measurements for optimal evaluation of these two interrelated entities48. In parallel, Deutz et al. advised clinicians to place muscle mass at the core of nutrition assessment and management strategies47.
Our findings of low muscle mass are also in line with a recent study, which reported that older adults with malnutrition or at risk of malnutrition were 13.6 times more likely to be at risk of sarcopenia, compared to those with normal nutritional status40. In another study of community-dwelling older adults in Singapore, risk of malnutrition was strongly associated with sarcopenia (odds ratio = 9.9)49. Similarly, a cohort study found that malnutrition was associated with an approximately four-fold higher risk of sarcopenia developing during a four-year follow-up50. Community-dwelling older Chinese adults with malnutrition experienced adverse outcomes such as sarcopenia, frailty, and mortality at 14-year follow-up51.
These observations suggest that assessing the risk for malnutrition in community-dwelling older adults could be a useful strategy for early detection of individuals at risk of low ASMI. Validated tools such as MUST and Mini Nutritional Assessment - Short Form can be used to assess the risk of malnutrition52,53.
Bone mass
Our findings of a direct correlation between low ASMI and low bone mass in older people are also in line with other studies in this area54,55. Specifically, in a Chinese population, leg lean mass was the strongest factor for prediction of femur bone mass54. Reduction in muscle strength over time was found to be the most significant independent factor associated with bone loss for Korean men and women56. Taken together, all these findings are in line with the established link between skeletal muscle and bone health.
Lifestyle-related risk factors
In the present study, we found that people with low ASMI had lower physical activity scores than those with normal ASMI. In addition, smoking and drinking were associated with low ASMI. Previous research reported that lifestyle-related risks for low muscle mass include physical inactivity20, smoking21, and drinking14. Low physical activity leads to muscle decline due to disuse atrophy57,58. Smoking may impair muscle protein synthesis, while chronic alcoholic myopathy can occur in up to 50% of alcohol misusers leading to reduction of muscle mass and strength59. All three of these factors are potentially modifiable, so risk can be lessened by increasing physical activity, smoking cessation, and reducing alcohol consumption.
Calf circumference cut-off values for low ASMI
Previous research showed that low muscle mass was significantly associated with low calf circumference in older people60,61,62. In the present study, we used ROC curve analysis to determine calf circumference cut-off values for low ASMI, i.e., 33.4 cm for males and 32.2 cm for females. A previous study found that calf circumference was positively correlated with appendicular skeletal muscle mass and skeletal muscle index, with cut-off values of calf circumference for predicting low muscle mass as < 34 cm in men and < 33 cm in women in a large study in Japan (n = 526)60. Similarly, a large study of older adults in Korea (n = 657 adults aged 70–84 years) found optimal cut-off values of calf circumference for low muscle mass as 35 cm for males and 33 cm for females61. Importantly, low calf circumference based on these cut-off values was associated with poor physical function61. Overall, these findings are in line with the calf circumference cut-offs for screening for sarcopenia from AWGS (< 34 cm in men and < 33 cm in women)1, thus supporting calf measurement as a screening tool for both low ASMI and sarcopenia in the community.
In the community, initial screening for low muscle mass can be done by measuring calf circumference60,61,62. We have established the cut-off values for low ASMI in community-dwelling older adults in Singapore, with strong agreement with the AWGS cut-off values for screening for sarcopenia (Kappa statistics for males = 0.88, for females = 0.87). As the measurement of calf circumference is low cost, easy to perform, and non-invasive, it can potentially be an ideal screening tool both in the community and clinical settings.
Strengths and limitations
Our study has multiple strengths in design and outcomes, including (1) a large population of 1211 community-dwelling older adults, (2) extensive data with a full picture of interacting factors, (3) observation of significant associations between risk of malnutrition and low ASMI after adjusting for potential confounders, and (4) establishment of gender-specific calf circumference cut-off values for low ASMI. As for limitations, this is a cross-sectional study, so it shows associations between low ASMI and certain health and demographic factors, but it does not prove causal relationship. Since our population size was substantial, and these associations were statistically significant, we propose that healthcare professionals use these findings to identify aging individuals at risk of low ASMI. In addition, the results of this study in the multiethnic Singapore population may be applicable to other Asian populations.
Conclusions
In conclusion, results of our study showed a high prevalence of low ASMI in community-dwelling older adults in Singapore, particularly among those at risk of malnutrition. The odds of having low ASMI was 12.5 for older adults at high risk of malnutrition compared to their nourished counterparts. For community-dwelling older adults, early identification of low muscle mass offers opportunities to prevent or delay its worsening, and its associated adverse consequences. We advise identification of malnutrition risk as a targeted core strategy to screen for low muscle mass in older adults.
Other risk factors included smaller calf circumference, older age, and lower bone mass, which were also significantly associated with risk of low ASMI. We have also determined gender-specific cut-off values of calf circumference for low ASMI in this population group and may thus serve as an easy-to-measure screening tool for low ASMI. Furthermore, low ASMI was associated with modifiable risk factors such as smoking and drinking, which suggests that current targeted community health promotion programs can improve muscle health, in addition to other established benefits.
Early identification of risk for low ASMI in older adults can guide appropriate interventions, which can in turn reduce associated health complications, lower healthcare costs, improve function and quality of life, and thus supporting healthier, more independent and meaningful lives for as long as possible.
Data availability
The data presented in this study are available within the paper and its Supplementary Dataset.
References
Chen, L.-K. et al. Asian Working Group for Sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J. Am. Med. Dir. Assoc. 21, 300–307. https://doi.org/10.1016/j.jamda.2019.12.012 (2020).
Cruz-Jentoft, A. J. et al. Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48, 16–31. https://doi.org/10.1093/ageing/afy169 (2019).
Prado, C. M. et al. Implications of low muscle mass across the continuum of care: a narrative review. Ann. Med. 50, 675–693. https://doi.org/10.1080/07853890.2018.1511918 (2018).
Kim, M., Kim, J. & Won, C. W. Association between involuntary weight loss with low muscle mass and health-related quality of life in community-dwelling older adults: Nationwide surveys (KNHANES 2008–2011). Exp. Gerontol. 106, 39–45. https://doi.org/10.1016/j.exger.2018.02.027 (2018).
Pang, B. W. J. et al. Prevalence and associated factors of sarcopenia in Singaporean adults - The Yishun Study. J. Am. Med. Dir. Assoc. 22(885), e881-885.e810. https://doi.org/10.1016/j.jamda.2020.05.029 (2021).
Tey, S. L. et al. Factors associated with muscle mass in community-dwelling older people in Singapore: findings from the SHIELD study. PLoS ONE 14, e0223222. https://doi.org/10.1371/journal.pone.0223222 (2019).
Xu, H.-Q. et al. Sarcopenia-related features and factors associated with low muscle mass, weak muscle strength, and reduced function in Chinese rural residents: a cross-sectional study. Arch. Osteoporos. 14, 2. https://doi.org/10.1007/s11657-018-0545-2 (2019).
Zhang, Y. et al. Prevalence and risk factors governing the loss of muscle function in elderly sarcopenia patients: a longitudinal study in China with 4 years of follow-up. J. Nutr. Health Aging 24, 518–524. https://doi.org/10.1007/s12603-020-1361-0 (2020).
Janssen, I., Heymsfield, S. B., Wang, Z. M. & Ross, R. Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. J. Appl. Physiol. 89, 81–88. https://doi.org/10.1152/jappl.2000.89.1.81 (2000).
Auyeung, T. W., Lee, S. W., Leung, J., Kwok, T. & Woo, J. Age-associated decline of muscle mass, grip strength and gait speed: a 4-year longitudinal study of 3018 community-dwelling older Chinese. Geriatr. Gerontol. Int. 14(Suppl 1), 76–84. https://doi.org/10.1111/ggi.12213 (2014).
Boutari, C. & Mantzoros, C. S. Decreasing lean body mass with age: challenges and opportunities for novel therapies. Endocrinol. Metab. (Seoul) 32, 422–425. https://doi.org/10.3803/EnM.2017.32.4.422 (2017).
Goodpaster, B. H. et al. The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J. Gerontol. A Biol. Sci. Med. Sci. 61, 1059–1064. https://doi.org/10.1093/gerona/61.10.1059 (2006).
Kim, S.-K. et al. Changes in body composition according to age and sex among young non-diabetic Korean adults: The Kangbuk Samsung Health Study. Endocrinol Metab 32, 442–450. https://doi.org/10.3803/EnM.2017.32.4.442 (2017).
Tyrovolas, S. et al. Factors associated with skeletal muscle mass, sarcopenia, and sarcopenic obesity in older adults: a multi-continent study. J Cachexia Sarcopenia Muscle 7, 312–321. https://doi.org/10.1002/jcsm.12076 (2016).
Swan, L., Warters, A. & O’Sullivan, M. Socioeconomic inequality and risk of sarcopenia in community-dwelling older adults. Clin. Interv. Aging 16, 1119–1129. https://doi.org/10.2147/CIA.S310774 (2021).
Kim, K.-S. et al. Type 2 diabetes is associated with low muscle mass in older adults. Geriatr. Gerontol. Int. 14, 115–121. https://doi.org/10.1111/ggi.12189 (2014).
Barreiro, E. & Jaitovich, A. Muscle atrophy in chronic obstructive pulmonary disease: molecular basis and potential therapeutic targets. J. Thorac. Dis. 10, S1415–S1424. https://doi.org/10.21037/jtd.2018.04.168 (2018).
Welch, C., Hassan-Smith, Z., Greig, C., Lord, J. & Jackson, T. Acute sarcopenia secondary to hospitalisation—an emerging condition affecting older adults. Aging Dis. 9, 151–164. https://doi.org/10.14336/AD.2017.0315 (2018).
Ilhan, B., Bahat, G., Erdogan, T., Kilic, C. & Karan, M. A. Anorexia Is independently associated with decreased muscle mass and strength in community dwelling older adults. J. Nutr. Health Aging 23, 202–206. https://doi.org/10.1007/s12603-018-1119-0 (2019).
Kwon, Y.-J., Kim, H. S., Jung, D.-H. & Kim, J.-K. Cluster analysis of nutritional factors associated with low muscle mass index in middle-aged and older adults. Clin. Nutr. 39, 3369–3376. https://doi.org/10.1016/j.clnu.2020.02.024 (2020).
Bai, H.-J. et al. Age-related decline in skeletal muscle mass and function among elderly men and women in Shanghai, China: a cross sectional study. Asia Pac. J. Clin. Nutr. 25, 326–332. https://doi.org/10.6133/apjcn.2016.25.2.14 (2016).
Franzon, K., Zethelius, B., Cederholm, T. & Kilander, L. The impact of muscle function, muscle mass and sarcopenia on independent ageing in very old Swedish men. BMC Geriatr. 19, 153. https://doi.org/10.1186/s12877-019-1142-y (2019).
Wakayama, S. et al. Skeletal muscle mass and higher-level functional capacity in female community-dwelling older adults. Int. J. Environ. Res. Public Health 18, 6692. https://doi.org/10.3390/ijerph18136692 (2021).
Norman, K. & Otten, L. Financial impact of sarcopenia or low muscle mass—a short review. Clin. Nutr. 38, 1489–1495. https://doi.org/10.1016/j.clnu.2018.09.026 (2019).
Lo, Y.-T.C. et al. Medical costs of a low skeletal muscle mass are modulated by dietary diversity and physical activity in community-dwelling older Taiwanese: a longitudinal study. Int. J. Behav. Nutr. Phys. Act. 14, 31. https://doi.org/10.1186/s12966-017-0487-x (2017).
Janssen, I., Shepard, D. S., Katzmarzyk, P. T. & Roubenoff, R. The healthcare costs of sarcopenia in the United States. J. Am. Geriatr. Soc. 52, 80–85. https://doi.org/10.1111/j.1532-5415.2004.52014.x (2004).
Goates, S. et al. Economic impact of hospitalizations in US adults with sarcopenia. J. Frailty Aging 8, 93–99. https://doi.org/10.14283/jfa.2019.10 (2019).
United Nations. World Population Ageing 2019: Highlights. (Department of Economic and Social Affairs, Population Division, 2019).
Cheong, M. et al. Nutritional biomarkers and associated factors in community-dwelling older adults: Findings from the SHIELD study. Nutrients 12, 3329. https://doi.org/10.3390/nu12113329 (2020).
Chew, S. T. H. et al. Impact of specialized oral nutritional supplement on clinical, nutritional, and functional outcomes: a randomized, placebo-controlled trial in community-dwelling older adults at risk of malnutrition. Clin. Nutr. 40, 1879–1892. https://doi.org/10.1016/j.clnu.2020.10.015 (2021).
Charlson, M., Szatrowski, T. P., Peterson, J. & Gold, J. Validation of a combined comorbidity index. J. Clin. Epidemiol. 47, 1245–1251. https://doi.org/10.1016/0895-4356(94)90129-5 (1994).
Charlson, M. E., Pompei, P., Ales, K. L. & MacKenzie, C. R. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J. Chronic Dis. 40, 373–383. https://doi.org/10.1016/0021-9681(87)90171-8 (1987).
Stratton, R. J. et al. Malnutrition in hospital outpatients and inpatients: prevalence, concurrent validity and ease of use of the “Malnutrition Universal Screening Tool” ('MUST’) for adults. Br. J. Nutr. 92, 799–808. https://doi.org/10.1079/bjn20041258 (2004).
Washburn, R. A., McAuley, E., Katula, J., Mihalko, S. L. & Boileau, R. A. The Physical Activity Scale for the Elderly (PASE): evidence for validity. J. Clin. Epidemiol. 52, 643–651. https://doi.org/10.1016/S0895-4356(99)00049-9 (1999).
Washburn, R. A., Smith, K. W., Jette, A. M. & Janney, C. A. The Physical Activity Scale for the Elderly (PASE): development and evaluation. J. Clin. Epidemiol. 46, 153–162. https://doi.org/10.1016/0895-4356(93)90053-4 (1993).
Shah, S., Vanclay, F. & Cooper, B. Improving the sensitivity of the Barthel Index for stroke rehabilitation. J. Clin. Epidemiol. 42, 703–709. https://doi.org/10.1016/0895-4356(89)90065-6 (1989).
Holick, M. F. Vitamin D deficiency. N. Engl. J. Med. 357, 266–281. https://doi.org/10.1056/NEJMra070553 (2007).
Hu, F. J. et al. Mid-upper arm circumference as an alternative screening instrument to appendicular skeletal muscle mass index for diagnosing sarcopenia. Clin. Interv. Aging 16, 1095–1104. https://doi.org/10.2147/CIA.S311081 (2021).
Hwang, A.-C., Liu, L.-K., Lee, W.-J., Peng, L.-N. & Chen, L.-K. Calf circumference as a screening instrument for appendicular muscle mass measurement. J. Am. Med. Dir. Assoc. 19, 182–184. https://doi.org/10.1016/j.jamda.2017.11.016 (2018).
Sato, P. H. R., Ferreira, A. A. & Rosado, E. L. The prevalence and risk factors for sarcopenia in older adults and long-living older adults. Arch. Gerontol. Geriatr. 89, 104089. https://doi.org/10.1016/j.archger.2020.104089 (2020).
Therakomen, V., Petchlorlian, A. & Lakananurak, N. Prevalence and risk factors of primary sarcopenia in community-dwelling outpatient elderly: a cross-sectional study. Sci. Rep. 10, 19551. https://doi.org/10.1038/s41598-020-75250-y (2020).
Martone, A. M. et al. Treating sarcopenia in older and oldest old. Curr. Pharm. Des. 21, 1715–1722. https://doi.org/10.2174/1381612821666150130122032 (2015).
Grimby, G. & Saltin, B. The ageing muscle. Clin. Physiol. 3, 209–218. https://doi.org/10.1111/j.1475-097x.1983.tb00704.x (1983).
Chan, A. et al. Transitions in Health, Employment, Social Engagement And Intergenerational Transfers In Singapore Study (THE SIGNS Study) – I: Descriptive Statistics and Analysis of Key Aspects of Successful Ageing: 1–224. Duke-NUS Medical School Centre for Ageing Research and Education, Singapore. https://doi.org/10.25722/w8ye-r177 (2018).
Cederholm, T. et al. GLIM criteria for the diagnosis of malnutrition: a consensus report from the global clinical nutrition community. Clin. Nutr. 38, 1–9. https://doi.org/10.1016/j.clnu.2018.08.002 (2019).
Argilés, J. M., Campos, N., Lopez-Pedrosa, J. M., Rueda, R. & Rodriguez-Mañas, L. Skeletal muscle regulates metabolism via interorgan crosstalk: Roles in health and disease. J. Am. Med. Dir. Assoc. 17, 789–796. https://doi.org/10.1016/j.jamda.2016.04.019 (2016).
Deutz, N. E. P. et al. The underappreciated role of low muscle mass in the management of malnutrition. J. Am. Med. Dir. Assoc. 20, 22–27. https://doi.org/10.1016/j.jamda.2018.11.021 (2019).
Landi, F. et al. Muscle loss: the new malnutrition challenge in clinical practice. Clin. Nutr. 38, 2113–2120. https://doi.org/10.1016/j.clnu.2018.11.021 (2019).
Lu, Y. et al. Systemic and metabolic signature of sarcopenia in community-dwelling older adults. J. Gerontol. A Biol. Sci. Med. Sci. 75, 309–317. https://doi.org/10.1093/gerona/glz001 (2020).
Beaudart, C. et al. Malnutrition as a strong predictor of the onset of sarcopenia. Nutrients 11, 2883. https://doi.org/10.3390/nu11122883 (2019).
Yeung, S. S. Y., Chan, R. S. M., Kwok, T., Lee, J. S. W. & Woo, J. Malnutrition according to GLIM criteria and adverse outcomes in community-dwelling Chinese older adults: a prospective analysis. J. Am. Med. Dir. Assoc. 22, 1953-1959.e1954. https://doi.org/10.1016/j.jamda.2020.09.029 (2021).
Skipper, A. et al. Adult malnutrition (undernutrition) screening: An evidence analysis center systematic review. J. Acad. Nutr. Diet. 120, 669–708. https://doi.org/10.1016/j.jand.2019.09.010 (2020).
Skipper, A. et al. Position of the Academy of Nutrition and Dietetics: malnutrition (undernutrition) screening tools for all adults. J. Acad. Nutr. Diet. 120, 709–713. https://doi.org/10.1016/j.jand.2019.09.011 (2020).
Cheng, Q. et al. A cross-sectional study of loss of muscle mass corresponding to sarcopenia in healthy Chinese men and women: reference values, prevalence, and association with bone mass. J. Bone Miner. Metab. 32, 78–88. https://doi.org/10.1007/s00774-013-0468-3 (2014).
Kim, B. J., Ahn, S. H., Kim, H. M., Lee, S. H. & Koh, J. M. Low skeletal muscle mass associates with low femoral neck strength, especially in older Korean women: the Fourth Korea National Health and Nutrition Examination Survey (KNHANES IV). Osteoporos. Int. 26, 737–747. https://doi.org/10.1007/s00198-014-2959-z (2015).
Kim, K. M. et al. Longitudinal changes in muscle mass and strength, and bone mass in older adults: Gender-specific associations between muscle and bone losses. J. Gerontol. A Biol. Sci. Med. Sci. 73, 1062–1069. https://doi.org/10.1093/gerona/glx188 (2018).
Bell, K. E., von Allmen, M. T., Devries, M. C. & Phillips, S. M. Muscle disuse as a pivotal problem in sarcopenia-related muscle loss and dysfunction. J. Frailty Aging 5, 33–41. https://doi.org/10.14283/jfa.2016.78 (2016).
Oikawa, S. Y., Holloway, T. M. & Phillips, S. M. The impact of step reduction on muscle health in aging: Protein and exercise as countermeasures. Front. Nutr. 6, 75. https://doi.org/10.3389/fnut.2019.00075 (2019).
Rom, O., Kaisari, S., Aizenbud, D. & Reznick, A. Lifestyle and sarcopenia–etiology, prevention, and treatment. Rambam Maimonides Med. J. 3, 1–12 (2012).
Kawakami, R. et al. Calf circumference as a surrogate marker of muscle mass for diagnosing sarcopenia in Japanese men and women. Geriatr. Gerontol. Int. 15, 969–976. https://doi.org/10.1111/ggi.12377 (2015).
Kim, S. et al. Calf circumference as a simple screening marker for diagnosing sarcopenia in older Korean adults: the Korean Frailty and Aging Cohort Study (KFACS). J. Korean Med. Sci. 33, e151. https://doi.org/10.3346/jkms.2018.33.e151 (2018).
Maeda, K., Koga, T., Nasu, T., Takaki, M. & Akagi, J. Predictive accuracy of calf circumference measurements to detect decreased skeletal muscle mass and European Society for Clinical Nutrition and Metabolism-defined malnutrition in hospitalized older patients. Ann. Nutr. Metab. 71, 10–15. https://doi.org/10.1159/000478707 (2017).
Acknowledgements
We would like to thank the participants for their commitment and enthusiasm in participating in this study. This study would not have been possible without all the invaluable contributions, dedication and commitment from all the co-investigators, study teams of Abbott Nutrition, Changi General Hospital, and SingHealth Polyclinics. We also thank Cecilia Hofmann, medical writer, C. Hofmann & Associates (Western Springs, Illinois, USA) for her editorial support.
Funding
This study received financial support from the Economic Development Board of Singapore (Grant number: COY-15-IDS-LL/160011), Abbott Nutrition Research and Development, and Changi General Hospital. The Economic Development Board of Singapore had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results. Abbott Nutrition Research and Development and Changi General Hospital were involved in the study design, data collection and analysis, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
S.L.T., D.T.T.H., G.B., C.H.H., Y.L.L., M.C., W.L.C., N.C.T., and S.T.H.C. conceptualized the study. Y.B., G.B., and S.L.T. analyzed the data. S.L.T. curated the data. S.L.T. and S.T.H.C. drafted the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
S.L.T., D.T.T.H., G.B., and Y.L.L. are employees of Abbott. Y.B. is an employee of Cognizant Technologies Solution Pvt. Ltd., a Contract Research Organization, which provides statistical services to Abbott Nutrition and has no competing interests. S.T.H.C. reports receiving honoraria for speaking engagement from Abbott. All other authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tey, S.L., Huynh, D.T.T., Berde, Y. et al. Prevalence of low muscle mass and associated factors in community-dwelling older adults in Singapore. Sci Rep 11, 23071 (2021). https://doi.org/10.1038/s41598-021-02274-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-02274-3
- Springer Nature Limited
This article is cited by
-
Does high-frequency resistance exercise offer additional benefits to older adults? learnings from a randomized controlled trial
BMC Sports Science, Medicine and Rehabilitation (2024)
-
Prevalence and some related factors of low muscle mass in older adults: findings from the Tehran lipid and glucose study
Journal of Diabetes & Metabolic Disorders (2024)
-
Prevalence and associated factors of sarcopenia in community-dwelling older adults at risk of malnutrition
BMC Geriatrics (2022)