Abstract
Lymph node (LN) metastasis is known as one of the most important prognostic factors for early gastric cancer (EGC) patients. Patients without LNM normally have better prognosis. However, there is no evaluation criteria to accurately assess the possibility of LN metastasis. Therefore, this study aims to establish an effective nomogram for prognosis prediction. In this study, 285 EGC patients from January 2010 to December 2015 were enrolled. Pearson’s Chi-Square (χ2) test (including continuity correction when appropriate) and logistics regression analyses was used to identify the risk factors for LN metastasis. The independent risk factors identified were then incorporated in a nomogram model. The predictive accuracy and discriminative ability of the nomogram were evaluated by receiver operating characteristic curve (ROC) and calibration curve. LN metastasis occurred in 59 (20.7%) EGC patients. And most of these patients were submucosal cancers (48/59). Chi-square test indicated lymphovascular emboli, carbohydrate antigen 19-9 (CA19-9), ulcer, tumor size, tumor infiltration and histological grade were the risk factors, and multivariate logistics analyses confirmed all these six factors were independent risk factors of LN metastasis, which were selected to construct the nomogram. The nomogram proved well calibrated and had good discriminative ability (C-index value: 0.842). The proposed nomogram could result in more-accurate risk prediction for EGC patients.
Similar content being viewed by others
Introduction
Gastric Cancer (GC) is known as one of the most frequently diagnosed cancer and leading cause of cancer deaths in China1,2. Early gastric cancer (EGC) is defined as invasive gastric cancer that invades no more deeper than the submucosa, regardless of lymph node (LN) metastasis3,4. An overall survival rate (OS) in EGC was considered to be greater than 90% after standardized D2 lymphadenectomy treatment5, with LN metastasis being the most significant prognostic factor6,7. However, just about 20% of patients exhibited LN metastasis, most of the patients received excessive surgical treatment8.
Nowadays, endoscopic resection (ER) has been considered to be an alternative option besides surgery, which could improve the quality of life for patients9,10. ER was composed of endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD), with a comparable long-term outcome to surgery10,11. However, ER can’t supply treatment of lymph node dissection, so it’s essential to evaluate the status of LN metastasis before and after surgery. According to the Japanese guidelines for ESD and EMR for EGC12, the absolute indication of ESD/EMR is as follows: differentiated-type adenocarcinoma without ulcerative findings, of which the depth of invasion is clinically diagnosed as T1a and the diameter is ≤ 20 mm. Only the patients who fulfill the absolute indication are recommended for ER treatment. However, because the absolute indication is excessively strict, the indication for ESD was expanded for the patients with no or lower risks of lymph node metastasis13. Therefore, a quantitative prediction model of LN metastasis based on individual information is urgently needed, which is crucial to weigh the benefits and risks of treatment.
A nomogram, also called a nomograph, is a graphical calculating tool, which has been developed in most types of cancer at present14,15,16. However, there are few reports on nomogram of LN metastasis in patients with EGC. In the present study, we aim to establish a nomogram for predicting lymph node metastasis in EGC patients.
Patients and methods
Patients
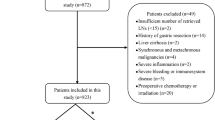
285 EGC patients in total who underwent surgical treatment in Sun Yat-sen University Cancer Center from January 2010 to December 2015 were selected into this retrospective study. The inclusion criteria were as follows: (i) diagnosed as early gastric cancer and (ii) underwent gastrectomy and D2 lymphadenectomy, (iii) achieved radical (R0) resection. The exclusion criteria were as follows: (i) distant metastasis, (ii) multiple cancers, (iii) stump cancer or recurrent cancer, (iv) accepted preoperative treatment, and (v) died in the perioperative period. In addition, those patients with indications of postoperative chemotherapy have accepted chemotherapy, either S-1 or XELOX regimen.
Follow-up assessments including clinical and laboratory examinations, for the first 2 years were conducted every quarterly. During the 3rd to 5th years, assessments were conducted twice a year, and every year after that until death. The primary endpoint was 5-year OS. OS was calculated from the date of surgery until death or to the last follow-up contact, which was used as a measure of prognosis.
Data collection
We further collected the clinic-pathological data of all included patients. The clinical information included age, gender, surgery type, carcinoembryonic antigen (CEA), carbohydrate antigen 19-9 (CA19-9) and carbohydrate antigen 72-4 (CA72-4) before surgery. The pathological information included depth of tumor infiltration, tumor size, tumor location, Lauren type, ulcer, lymphovascular emboli and histological grade. The depth was measured at the deepest point of penetrated carcinoma cells and tumor size was defined as the maximum diameter17. The maximum diameter of the lesions was recorded as the tumor size. The surgery type includes distal, proximal and total resection. The tumor location was separated into three parts (upper third, middle third and lower third part of stomach). Tumor histological grade was divided into two groups: the undifferentiated group, which includes undifferentiated and poorly adenocarcinomas, and the differentiated group, which includes moderately and well differentiated adenocarcinomas18.
Statistical analyses
Statistical analyses to identify risk factors were conducted by the SPSS (version 26.0). The continuous variables were shown as the mean ± standard deviation (SD). The categorical variables were analyzed by Pearson’s Chi-Square (χ2) test (including continuity correction when appropriate). The independent risk factors of LN metastasis were assessed by multivariate logistic regression analyses. Moreover, the OS were calculated by the Kaplan–Meier method and compared using the log-rank test. Multivariate analyses were used Cox proportional hazards regression models. The primary end point was 5-year OS.
Then, a nomogram was formulated using the package of rms19 in R studio (version 1.3.10), based on the results of the multivariate logistic regression model. The performance of the nomogram was assessed by calibration curve as indicator of internal calibration, and ROC as a measure of discriminative ability20. Meanwhile, the Hosmer–Lemeshow test was used to evaluate goodness of fit of the nomogram.
P values (two sided) < 0.05 were considered statistical significance in all statistical analyses.
Ethical approval statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of our institutional research committee and with the 1964 Helsinki declaration and its later amendments. This article does not contain any studies with animals performed by any of the authors. Due to the retrospective nature of the study, this research was approved by the Ethics Committee of the Sun Yat-sen University Cancer Center, and inform consent was granted a waiver.
Results
Clinical-pathological factors and non-parametric test
In total, 285 EGC patients had undergone treatment at our institution from January 2010 to December 2015, who were selected into present study. LN metastasis was confirmed pathologically in 59 (20.7%) of those 285 cases. The average number of retrieved lymph nodes was 32.6 ± 14.5(range 2–98). The median age of those EGC patients was 57 ± 11.5(range, 22–82) years. Non-parametric test indicated that LN metastasis was associated with lymphovascular emboli, CA19-9, ulcer, tumor size, tumor infiltration and histological grade (all P < 0.05). Patients with lymphovascular emboli, larger size > 2 cm, or ulcer have a higher possibility of LN metastasis (all p < 0.001). Tumors with submucosal invasion was associated with higher LNM metastasis than those with intra-mucosa invasion (p = 0.001). Undifferentiated carcinomas were related to lower lymph node metastases (p = 0.005). Patients with CA19-9 ≤ 35U/ml have a lower probability of LN metastasis than those with CA19-9 > 35U/ml (p = 0.028). But LN metastasis was not correlated with gender, age, CA72-4, CEA, tumor location, surgery type, or Lauren type (Table 1).
Multivariate analyses and risk nomogram for LN metastasis
We furthermore summarized both univariate and multivariate logistic analyses of LN metastasis (Table 2). And multivariate analyses confirmed lymphovascular emboli (p < 0.001, OR 8.313, 95%CI 3.295–20.973), CA19-9 (p = 0.025, OR 6.921, 95%CI 1.316 to 36.401), ulcer (p = 0.001, OR 4.861, 95%CI 1.931 to 12.238), tumor size (p = 0.001, OR 4.717, 95%CI 1.898 to 11.724), tumor infiltration (p = 0.018, OR 2.669, 95%CI 1.176 to 6.061) and histological grade (p = 0.035, OR 2.295, 95%CI 1.058 to 4.976) were the risk factors, indicating that all these six factors were independent risk factors of LN metastasis. Therefore, these six clinical-pathological factors were selected to construct a predictive nomogram for LN metastasis (Fig. 1). For each EGC patient, each of these clinicopathological features were assigned points, and the total points was calculated according to the nomogram. The total points corresponded to a probability of predicted LN metastasis. The p value of Hosmer–Lemeshow test was 0.287, indicating a good fit of the nomogram. Then a ROC curve was constructed to evaluate the predictive accuracy of the nomogram, which owned an AUC of 0.842 (95% CI 0.795 to 0.882), indicating a good discriminative ability (Fig. 2A). In addition, we developed a calibration curve of the model, which showed that the nomogram performed well with additional 1000 bootstraps (Fig. 2B).
A nomogram predicting the probability of metastatic lymph node involvement for patients with early gastric cancer. The scores of each variable are added to obtain the total score, and then a vertical line is subtracted from the row of total-points to estimate the probability of lymph node metastasis.
Validation of nomogram for predicting lymph node metastasis in early gastric cancer patients. (A) A receiver operating characteristics (ROC) curve of the multivariate logistic regression model. The AUC value was 0.842 (95%CI: 0.795 to 0.882), indicating a good discriminative ability (B). Calibration plot. The reference line represents perfect equality of the predicted probability and the actual incidence of lymph node metastasis.
Comparison of overall survival
Of the total 285 EGC patients, the median follow-up time was 46.5 (range 2 -112) mouths. In our study, the 1-year, 3-year and 5-year OS in EGC were 97.9%, 92.6% and 90.6%, respectively, which were calculated by Kaplan–Meier method. Additionally, the 5-year OS of those with LNM and without LNM were 93.4% and 79.5%. The primary end point of this study is 5-year OS. Kaplan–Meier method (Log-rank test) indicated that OS was significantly associated with age (p = 0.003), CEA (p < 0.001), CA19-9 (p = 0.033), CA72-4 (p = 0.009), LN metastasis (p < 0.001), lymphovascular emboli (p = 0.008), surgery type (p = 0.013), pN stage (p < 0.001) and TNM stage (p < 0.001). But it was no relationship with gender (p = 0.177), tumor size (p = 0.959), tumor location (0.246), tumor infiltration (p = 0.324), histological grade (p = 0.168), ulcer (p = 0.302), or Lauren type (p = 0.556). Kaplan–Meier plots are shown in Fig. 3 and Fig. 4. In addition, multivariate Cox regression analyses indicated that only surgery type, CEA and pN stage were independent prognostic predictors (Table 3).
The Kaplan–Meier analyses results of the clinical-pathological features, which proved to be significantly correlated with overall survival. The p values of these features are as follows: (A) age, 0.003; (B) CA19-9, 0.033; (C) CA72-4, 0.009; (D) CEA, < 0.001; (E) LN metastasis, < 0.001; (F) lymphovascular emboli, 0.009; (G) pN stage, < 0.001; (H) TNM stage, < 0.001; (I) surgery type, 0.013.
The Kaplan–Meier analyses results of the clinical-pathological features, which showed no significantly correlated with overall survival. The p values of these features are as follows: (A) Lauren type, 0.556; (B) tumor location, 0.246; (C) gender, 0.177; (D) tumor infiltration, 0.087; (E) ulcer, 0.302; (F) tumor size, 0.959; (G) histological grade, 0.168.
Discussion
Since long, gastrectomy with D2 lymphadenectomy was considered as the standard and optimal treatment for EGC patients21,22,23. The rate of LN metastasis in patients with EGC ranges from 5 to 22%, which means that approximately 70% to 80% of EGC patients undergo overtreatment with D2 lymphadenectomy24,25. The development of less invasive treatments, including endoscopic mucosa resection (EMR) and endoscopic submucosa dissection (ESD), have had an important impact on the treatment strategies revolution in the last few decades9,10,26. The EGC patients meeting suitable conditions (low probability of LN metastasis), were recommended to receive less invasive treatments. Therefore, we retrospectively analyzed 285 EGC patients based on clinical-pathological features to identify the risk factors for LN metastasis and construct a nomogram to guide treatment.
In this retrospective study, the incidence rate of LN metastasis was 20.7% in EGC patients. The incidence rate of LN metastasis for the patients with one or more risk features, such as lymphovascular emboli, presence of ulcer, tumor size > 2 cm, submucosa infiltration, undifferentiated type, or CA19-9 > 35 U/ml, was higher than those patients without these clinical-pathological features. Multivariate analyses showed that these six features were independent risk factors for LN metastasis, and the presence of lymphovascular emboli was considered the most important factor. Previous studies have confirmed these features were independent risk factors. Lymphovascular emboli is the characteristic of lymphovascular invasion, which is considered a rate-limiting step in the lymph node metastatic process27. And therefore, lymphovascular emboli is significantly associated with LN metastasis28. CA19-9 > 35U/ml has been reported as a reliable risk factor for predicting LN metastasis previously29. Larger size (≥ 20 mm), presence of ulcer, tumor submucosal infiltration and undifferentiated type were related to regional lymph node metastasis29,30,31,32,33,34,35,36. The results of our study were consistent with the studies mentioned above.
And then, we established a nomogram with these six variables related to LN metastasis, which results in more intuitive and accurate in predicting the risk of LN metastasis. The high AUC value (o.842) and calibration curve indicates a good discriminative ability and universal clinical applicability of our nomogram.
LN metastasis has been well considered as one of the most important prognostic factors in both early gastric cancer and advanced gastric cancer23,37. For those EGC patients with low probability of LN metastasis, ESD or EMR was the optimal treatment38. However, there is still a lack of criteria to evaluate the probability of lymph node metastasis. Our study provides a helpful method to solve this problem. For example, a differentiated mucosal patient without lymphovascular or other features, has a low risk of LN metastasis (less than 5%), and is suitable for EMR. On the contrary, an undifferentiated submucosal patient with lymphovascular emboli, larger tumor size (> 2 cm) and ulcer, has a probability of more than 90% for LN metastasis regardless of other features. This patient is recommended to undergo gastrectomy with D2 lymphadenectomy instead of endoscopic treatment. Thus, we think this quantified and visual predictive model allows clinicians to make more objective decisions on treatment options based on the possibility of LN metastasis.
This study had some potential limitations. Firstly, the final selected sample size is small, so we consider improving the nomogram by expanding the sample size in the future. Secondly, this was a single-center retrospective study. In order to further improve the clinical application environment of the nomogram, additional external validation on different populations is required. Finally, P53, HER-2 or other genetic information was not included in the nomogram, which may provide more individualized evidence of our nomogram. In the future, we intend to incorporate more variables into the nomogram to improve its accuracy, including the information of imaging examination and molecular detection. In spite of the above limitations, we believe that our rosette map can be helpful for clinical work. A strength of our nomogram is that it was built from the variables which could be easily obtained before ESD or EMR treatment. It therefore means that this nomogram can be used to preliminarily determine the risk of lymph node metastasis of a patient before surgery, and hence to prescribe a preliminary treatment.
Conclusion
In conclusion, we established a nomogram to help clinicians predict the risk of lymph node metastasis in the patients with early gastric cancer. This tool could help clinicians and patients quantify the potential incidence of lymph node metastasis. Surgery plus lymph node dissection is recommended for certain patients with a high risk of LN metastasis, and endoscopic dissection is suitable for those with a low risk. In the future, we will conduct further research to improve clinical application value of the nomogram.
References
Zheng, R. et al. Estimates of cancer incidence and mortality in China 2013. Chin. J. Cancer https://doi.org/10.1186/s40880-017-0234-3 (2017).
Chen, Y. S. et al. Long-term survival trends of gastric cancer patients between 1972 and 2011 in Qidong. Chin. J. Cancer 34(12), 602–607. https://doi.org/10.1186/s40880-015-0058-y (2015).
Kajitani, T. The general rules for the gastric cancer study in surgery and pathology. Part I. Clinical classification. Jpn. J. Surg. 11(2), 127–139. https://doi.org/10.1007/bf02468883 (1981).
Kajitani, T. The general rules for the gastric cancer study in surgery and pathology. Part II. Histological classification of gastric cancer. Jpn. J. Surg. 11(2), 140–145 (1981).
Wang, Z. et al. Long-term outcomes after D2 gastrectomy for early gastric cancer: Survival analysis of a single-center experience in China. Asian Pac. J. Cancer Prev. 15(17), 7219–7222. https://doi.org/10.7314/apjcp.2014.15.17.7219 (2014).
Kim, J. P. et al. Significant prognostic factors by multivariate analysis of 3926 gastric cancer patients. World J. Surg. 18(6), 872–877. https://doi.org/10.1007/BF00299091 (1994).
Folli, S. et al. Early gastric cancer: Prognostic factors in 223 patients. Br. J. Surg. 82(7), 952–956. https://doi.org/10.1002/bjs.1800820732 (1995).
Kawata, N. et al. Risk factors for lymph node metastasis and long-term outcomes of patients with early gastric cancer after non-curative endoscopic submucosal dissection. Surg. Endosc. 31(4), 1607–1616. https://doi.org/10.1007/s00464-016-5148-7 (2017).
Choi, I. J. et al. Long-term outcome comparison of endoscopic resection and surgery in early gastric cancer meeting the absolute indication for endoscopic resection. Gastrointest. Endosc. 81(2), 333–341. https://doi.org/10.1016/j.gie.2014.07.047 (2015).
Yoshifuku, Y. et al. Long-term prognosis after endoscopic submucosal dissection for early gastric cancer in super-elderly patients. Surg. Endosc. 30(10), 4321–4329. https://doi.org/10.1007/s00464-016-4751-y (2016).
Pyo, J. H. et al. Long-term outcome of endoscopic resection vs. surgery for early gastric cancer: A non-inferiority-matched cohort study. Am. J. Gastroenterol. 111(2), 240–249. https://doi.org/10.1038/ajg.2015.427 (2016).
Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2014 (ver 4). Gastric Cancer 20(1), 1–19. https://doi.org/10.1007/s10120-016-0622-4 (2017).
Hirasawa, T. et al. Incidence of lymph node metastasis and the feasibility of endoscopic resection for undifferentiated-type early gastric cancer. Gastric Cancer 12(3), 148–152. https://doi.org/10.1007/s10120-009-0515-x (2009).
Bochner, B. H., Kattan, M. W. & Vora, K. C. Postoperative nomogram predicting risk of recurrence after radical cystectomy for bladder cancer. J. Clin. Oncol. 24(24), 3967–3972. https://doi.org/10.1200/JCO.2005.05.3884 (2006).
Wierda, W. G. et al. Prognostic nomogram and index for overall survival in previously untreated patients with chronic lymphocytic leukemia. Blood 109(11), 4679–4685. https://doi.org/10.1182/blood-2005-12-051458 (2007).
Wang, Y. et al. Prognostic nomogram for intrahepatic cholangiocarcinoma after partial hepatectomy. J. Clin. Oncol. 31(9), 1188–1195. https://doi.org/10.1200/JCO.2012.41.5984 (2013).
Ren, G. et al. Prediction of risk factors for lymph node metastasis in early gastric cancer. World J. Gastroenterol. 19(20), 3096–3107. https://doi.org/10.3748/wjg.v19.i20.3096 (2013).
Li, X. et al. The characteristics, prognosis, and risk factors of lymph node metastasis in early gastric cancer. Gastroenterol. Res. Pract. 2018, 6945743–6945747. https://doi.org/10.1155/2018/6945743 (2018).
Harrell, F. E. Regression Modeling Strategies (Springer, 2001).
Vickers, A. J. & Cronin, A. M. Traditional statistical methods for evaluating prediction models are uninformative as to clinical value: Towards a decision analytic framework. Semin. Oncol. 37(1), 31–38. https://doi.org/10.1053/j.seminoncol.2009.12.004 (2010).
Borie, F. et al. Appropriate gastric resection with lymph node dissection for early gastric cancer. Ann. Surg. Oncol. 11(5), 512–517. https://doi.org/10.1245/ASO.2004.06.025 (2004).
Nam, S. Y. et al. Effect of repeated endoscopic screening on the incidence and treatment of gastric cancer in health screenees. Eur. J. Gastroenterol. Hepatol. 21(8), 855–860. https://doi.org/10.1097/MEG.0b013e328318ed42 (2009).
Kitano, S. et al. A multicenter study on oncologic outcome of laparoscopic gastrectomy for early cancer in Japan. Ann. Surg. 245(1), 68–72. https://doi.org/10.1097/01.sla.0000225364.03133.f8 (2007).
Yang, H. Sentinel node biopsy in early gastric cancer: constant exertion for clinical application. Ann. Surg. Oncol. 19(2), 353–354. https://doi.org/10.1245/s10434-011-2126-0 (2012).
An, J. Y. et al. Assessment of open versus laparoscopy-assisted gastrectomy in lymph node-positive early gastric cancer: A retrospective cohort analysis. J. Surg. Oncol. 102(1), 77–81. https://doi.org/10.1002/jso.21554 (2010).
Oh, S. et al. Outcomes of minimally invasive surgery for early gastric cancer are comparable with those for open surgery: Analysis of 1,013 minimally invasive surgeries at a single institution. Surg. Endosc. 28(3), 789–795. https://doi.org/10.1007/s00464-013-3256-1 (2014).
Alpaugh, M. L. et al. Cooperative role of E-cadherin and sialyl-Lewis X/A-deficient MUC1 in the passive dissemination of tumor emboli in inflammatory breast carcinoma. Oncogene 21(22), 3631–3643. https://doi.org/10.1038/sj.onc.1205389 (2002).
Park, W. Y. et al. Pathologic definition and number of lymphovascular emboli: Impact on lymph node metastasis in endoscopically resected early gastric cancer. Hum. Pathol. 44(10), 2132–2138. https://doi.org/10.1016/j.humpath.2013.04.006 (2013).
Feng, F. et al. Diagnostic and prognostic value of CEA, CA19-9, AFP and CA125 for early gastric cancer. BMC Cancer 17(1), 737. https://doi.org/10.1186/s12885-017-3738-y (2017).
Sano, T., Kobori, O. & Muto, T. Lymph node metastasis from early gastric cancer: Endoscopic resection of tumour. Br. J. Surg. 79(3), 241–244. https://doi.org/10.1002/bjs.1800790319 (1992).
Maehara, Y. Predictors of lymph node metastasis in early gastric cancer. Br. J. Surg. 79, 245–247 (1992).
Sekiguchi, M. et al. Risk stratification and predictive risk-scoring model for lymph node metastasis in early gastric cancer. J. Gastroenterol. 51(10), 961–970. https://doi.org/10.1007/s00535-016-1180-6 (2016).
Jung, D. H. et al. Risk-stratification model based on lymph node metastasis after noncurative endoscopic resection for early gastric cancer. Ann. Surg. Oncol. 24(6), 1643–1649. https://doi.org/10.1245/s10434-017-5791-9 (2017).
Fang, C. et al. Risk factors of lymph node metastasis in early gastric carcinomas diagnosed by WHO criteria in 379 Chinese patients. J. Dig. Dis. 17(8), 526–537. https://doi.org/10.1111/1751-2980.12385 (2016).
Park, J. W. et al. Predictive factors for lymph node metastasis in early gastric cancer with lymphatic invasion after endoscopic resection. Surg. Endosc. 31(11), 4419–4424. https://doi.org/10.1007/s00464-017-5490-4 (2017).
Bausys, R. et al. Risk factors for lymph node metastasis in early gastric cancer patients: Report from Eastern Europe country-Lithuania. BMC Surg. 17(1), 108. https://doi.org/10.1186/s12893-017-0304-0 (2017).
Haist, T. et al. Prognostic risk factors of early gastric cancer: A western experience. Langenbecks Arch. Surg. 401(5), 667–676. https://doi.org/10.1007/s00423-016-1395-2 (2016).
Smyth, E. C. et al. Gastric cancer. Lancet 396(10251), 635–648. https://doi.org/10.1016/S0140-6736(20)31288-5 (2020).
Author information
Authors and Affiliations
Contributions
All authors participated in data collection. H.B.Q. and Y.B.C. contributed to the conception and design of the study; M.Q.Z., C.D., and L.X. performed the literature search, data extraction, quality assessment, and statistical analyses; M.Q.Z., C.D., L.X. and H.B.Q. composed the first draft of the manuscript; S.C.F., Y.D.L., J.R.G., Y.L., Y.B.C., and Z.W.Z. checked and corrected the draft. All authors contributed to the writing or editing of the manuscript and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, M., Ding, C., Xu, L. et al. A nomogram to predict risk of lymph node metastasis in early gastric cancer. Sci Rep 11, 22873 (2021). https://doi.org/10.1038/s41598-021-02305-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-02305-z
- Springer Nature Limited
This article is cited by
-
Development of a nomogram for prediction of central lymph node metastasis of papillary thyroid microcarcinoma
BMC Cancer (2024)
-
An artificial intelligence system for comprehensive pathologic outcome prediction in early gastric cancer through endoscopic image analysis (with video)
Gastric Cancer (2024)
-
Development and validation of a risk prediction model for frailty in patients with diabetes
BMC Geriatrics (2023)