Abstract
Huangqin decoction (HQD) is a Traditional Chinese Medicine formula for ulcerative colitis. However, the pharmacology and molecular mechanism of HQD on ulcerative colitis is still unclear. Combined microarray analysis, network pharmacology, and molecular docking for revealing the therapeutic targets and molecular mechanism of HQD against ulcerative colitis. TCMSP, DrugBank, Swiss Target Prediction were utilized to search the active components and effective targets of HQD. Ulcerative colitis effective targets were obtained by microarray data from the GEO database (GSE107499). Co-targets between HQD and ulcerative colitis are obtained by Draw Venn Diagram. PPI (Protein–protein interaction) network was constructed by the STRING database. To obtain the core target, topological analysis is exploited by Cytoscape 3.7.2. GO and KEGG enrichment pathway analysis was performed to Metascape platform, and molecular docking through Autodock Vina 1.1.2 finished. 161 active components with 486 effective targets of HQD were screened. 1542 ulcerative colitis effective targets were obtained with |Log2FC|> 1 and adjusted P-value < 0.05. The Venn analysis was contained 79 co-targets. Enrichment analysis showed that HQD played a role in TNF signaling pathway, IL-17 signaling pathway, Th17 cell differentiation, etc. IL6, TNF, IL1B, PTGS2, ESR1, and PPARG with the highest degree from PPI network were successfully docked with 19 core components of HQD, respectively. According to ZINC15 database, quercetin (ZINC4175638), baicalein (ZINC3871633), and wogonin (ZINC899093) recognized as key compounds of HQD on ulcerative colitis. PTGS2, ESR1, and PPARG are potential therapeutic targets of HQD. HQD can act on multiple targets through multi-pathway, to carry out its therapeutic role in ulcerative colitis.
Similar content being viewed by others
Introduction
Ulcerative Colitis (UC) is one subtype of inflammatory bowel disease (IBD) of unknown aetiology that affects the colorectum, often leads to bloody diarrhoea and abdominal pain1. Multiple pathogenesis, such as environmental factors, genetically susceptibility and gut microbiome dysfunction, have been implicated2. The mucosal inflammation initiating in the rectum and extending proximally in the colon, and limited to the mucosal layer, contrast to Crohn’s disease (CD) is involving all layers of the bowel wall1. Owing to intestinal homeostasis dysfunction and intestinal barrier disruption, may result in long-term relapsing and remitting mucosal inflammation3. Patients with ulcerative colitis experience unpredictable flares-up may present fever, tenesmus, anemia, and possibly intestinal perforation, which is an afflicted condition of an individual4,5. Despite 5-ASA, rectal corticosteroids, and biological drugs are the main treatment of ulcerative colitis, approximately 15% of patients may require restorative proctocolectomy with ileal pouch-anal anastomosis (IPAA) for medically refractory disease or to treat colonic neoplasia6,7. However, with the incidence and prevalence of ulcerative colitis is increasing worldwide, governments should be informed of the management and economic burden of this disease. Therefore, it is urgent to seek more therapeutic armamentariums with fewer adverse side effects for ulcerative colitis.
Traditional Chinese Medicine (TCM) has almost 5000 years of history, which creates many treatments for disease. Huangqin decoction (HQD), a traditional herbal formula from “Shang Han Lun” written by Zhang Zhongjing in Han Dynasty, which is used for “heat-dampness” ZHENG (resulting in fever, abdominal pain, and hematochezia) for over 1800 years. HQD consists of four herbs: Scutellariae Radix (SR, Huangqin), Paeoniae Radix Alba (PRA, Baishao), Jujubae Fructus (JF, Dazao), Glycyrrhiza glabra L. (GL, Gancao). Recently, some fundamental analysis has proved that HQD may regulate the gut microbiota, suppress Ras-PI3K-Akt-HIF-1α and NF-κB pathways8, target ESR1 and EGFR to relieve endothelial dysfunction9 to treat ulcerative colitis. However, pharmacology and molecular mechanisms remain unclear.
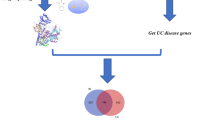
In this study, we combined network pharmacology and bioinformatics to reveal the core target and pathways that are participated in the pathogenesis of HQD against ulcerative colitis and screen for molecular targets of HQD for the treatment of ulcerative colitis. The workflow is shown in Fig. 1.
Materials and methods
Pharmacokinetic evaluation of active components
Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform (TCMSP, https://tcmspw.com/tcmsp.php10 were used to filter out active components, according to absorption, distribution, metabolism, excretion (ADME) criteria (OB ≥ 30% and DL ≥ 0.18)11. Confirmation of active components SMILES structures from PubChem (https://PubChem.ncbi.nlm.nih.gov) 12. Effective targets were selected from DrugBank (https://go.drugbank.com) and Swiss Target Prediction (http://www.swisstargetprediction.ch/). 13. Interaction of over 100 was recognized as an active component.
Screening for DEGs of ulcerative colitis and analysis
Expression profiling data from GSE107499 (GPL15207 [PrimeView] Affymetrix Human Gene Expression Array) was obtained from the GEO database (http://www.ncbi.nlm.nih.gov/geo/) with 44 healthy individuals and 75 ulcerative colitis colonic mucosa samples. Based on the GEO2R web tool (http://www.ncbi.nlm.nih.gov/geo/geo2r/), differentially expressed genes (DEGs) with the cut-off criteria of |Log2FC|> 1 and adjusted P-value < 0.05 and visualized by volcano plot.
Protein–protein interaction (PPI) network
Standardize gene names from Uniprot Knowledgebase (UniprotKB, http://www.uniprot.org), selecting “Homo sapiens”. Draw the Venn diagram (http://bioinformatics.psb.ugent.be/webtools/Venn/) for co-targets between HQD and DEGs and shown as a heatmap. Built on the co-targets result, the PPI network was conducted by Search Tool for the Retrieval of Interacting Genes (STRING, https://www.string-db.org/14, then visualized by Cytoscape3.7.215. The core target was topological analyzed using the CytoNCA plugin, limited core targets over 1 time medians of degree, betweenness, and closeness, respectively.
GO and KEGG pathway enrichment analysis
Metascape (http://metascape.org/16 serves as a gene annotation and analysis resource platform, limited “H.sapiens” and P < 0.01 for GO and KEGG pathway enrichment analysis17, selected top 10 results in bubble chart colored by − log10 (P values).
Molecular docking
3D protein structure of core target was downloaded from RCSB Protein Data Bank (RCSB PDB, http://www.pdb.org/), using Pymol software to separate modified, ligand and remove water. 3D structure of core component was downloaded from PubChem, after adding hydrogen and calculate charges, then using Autodock Vina 1.1.2 software to dock with core targets, limited binding energy ≤ − 5.0 kcal·mol-1 as stable connection site.
Results
Identification of active components
A total of 161 active components were obtained from the TCMSP database: 35 in Scutellariae Radix (SR, Huangqin), 13 in Paeoniae Radix Alba (PRA, Baishao), 29 in Jujubae Fructus (JF, Dazao), 92 in Glycyrrhiza glabra L. (GL, Gancao) (Fig. 2 and Supplement Table S1). Among 161 active components, 468 effective targets were selected by the Swiss Target Prediction and DrugBank database. Twenty core components as interactions over 100 were listed in Table 1.
Active components-targets of HQD performed by Cytoscape3.7.2. Green nodes are SR components, pink nodes are PRA components, red nodes are JF components, orange nodes are GL components, blue nodes are drug targets. MOL000358 is in SP, PRA, JF; MOL000492 is in PRA and JF; MOL000359 is in SR, PRA, and GL; MOL000499 is in SR and JF; MOL000211 is in PRA, JF, and GL; MOL000098 is in JF and GL. The size represents the degree.
Identification of DEGs and co-targets
According to the GEO2R web tool, a total of 1542 DEGs were identified, which consist of 1024 upregulated and 518 downregulated genes (Fig. 3 and Supplement Table S2). Venn analysis between 1542 DEGs and 468 HQD targets, had got 79 co-targets (Fig. 4A). Hierarchical cluster analysis of 79 co-targets between ulcerative colitis mucosal samples and normal controls in GSE107499 (Fig. 4B).
79 co-targets. (A) Venn analysis. The green is ulcerative colitis (1542 targets), blue is Paeoniae Radix Alba (84 targets), pink is Jujubae Fructus (147 targets), yellow is Glycyrrhiza glabra L. (130 targets), orange is Scutellariae Radix (385 targets). (B) Heatmap. Red dots are upregulated expression. Green dots are downregulated expression.
PPI network and core target
In the PPI network (Fig. 5), after hiding 3 disconnected nodes (RORA, PIM2, RPS6KA2), 76 targets were chosen for topological analysis. According to the median degree (23), betweenness (342.25), and closeness (0.26) obtained by topological analysis, 6 core targets were further selected, including Interleukin-6 (IL6), Tumor necrosis factor (TNF), Interleukin-1 beta (IL1B), Prostaglandin G/H synthase 2 (PTGS2), Estrogen receptor (ESR1), Peroxisome proliferator-activated receptor gamma (PPARG). (Table 2 and Supplement Table S3).
GO and KEGG pathway enrichment analysis
The GO and KEGG pathway enrichment analysis of 79 overlapping targets were performed relying on Metascape platform according to the P < 0.01. There were 818, 26, and 66 GO terms related to biological processes, cell components, and molecular functions, respectively (Fig. 6). Inflammatory response, collagen metabolic process, and leukocyte migration was the significantly enriched for BP. For Membrane raft, extracellular matrix, and side of membrane was the significantly enriched for CC. Oxidoreductase activity, endopeptidase activity, and protein tyrosine kinase activity was the significantly enriched for MF.
In addition, 184 pathways were contained in KEGG pathway enrichment analysis, mainly including TNF signaling pathway, IL-17 signaling pathway, Rheumatoid arthritis, Cytokine-cytokine receptor interaction, Th17 cell differentiation (Fig. 7).
Molecular docking
Molecular docking was applied to analyse the binding of the core targets (IL6, TNF, IL1B, PTGS2,ESR1, and PPARG) and all 19 active compounds of HQD (except for MOL001490, 3D conformer generation is disallowed on PubChem since too flexible), respectively. The binding energy score was all smaller than − 5.0 kcal·mol−1, and the results are listed in Table 3.
In detail, the active compound with the lowest binding energy to IL6 (RCSB PDB: 1ALU) and IL1B (RCSB PDB: 1I1B) was MOL000098 quercetin, TNF (RCSB PDB: 1TNF) and PPARG (RCSB PDB: 1PRG) was MOL002714 baicalein, PTGS2 (RCSB PDB: 5F19) was MOL000552 5,2′-Dihydroxy-6,7,8-trimethoxyflavone, and ESR1 (RCSB PDB: 3OS8) was MOL000173 wogonin. The binding energy scores of IL6-quercetin and IL1B-quercetin were − 7.2 and − 7.6 kcal·mol-1, TNF-baicalein and PPARG-baicalein were − 9.2 and − 9.3 kcal·mol-1, PTGS2-5,2′-Dihydroxy-6,7,8-trimethoxyflavone was − 9.5 kcal·mol-1, ESR1-wogonin was -8.5 kcal·mol-1, respectively (Fig. 8). These consequences state that the screened core targets indeed stably combine with HQD.
Furthermore, according to the ZINC15 database, quercetin (ZINC4175638), baicalein (ZINC3871633), and wogonin (ZINC899093) as the stable compound recognized as key compounds. From Table 3, the binding energy of 5F19 and 1PRG was lower than others, which indicated that PTGS2 and PPARG were the target of HQD on ulcerative colitis.
Discussion
Given its unclear pathogenesis, potentially progressive, and debilitating disease course, the therapeutic goals for ulcerative colitis have changed from treating symptoms to mucosal healing. However, conventional treatments are mainly restricted to control inflammation and clinical symptoms, and hard to meet the expectations of long-term management. Therefore, combination therapy is the urgent strategy for ulcerative colitis18. Profound knowledge of genetics, microbiota or cytokine imbalances in the bowel and its function in inflammation will sustain the outcome of possible therapeutic approaches if we act on those targets. In addition, the development of new ligands with therapeutic efficacy on the bowel would be a potential avenue in the search for treatments or prevention of ulcerative colitis. To date, with widely using TCM in different diseases, clinical effectiveness has been demonstrated. As for this concern, we made a combination of network pharmacology and bioinformatics might contribute to a further understanding of the disease aetiology and find novel potential therapeutic way.
In this study, 20 core components with 486 targets of HQD and 1542 DEGs (GSE107499) of ulcerative colitis were examined. 79 co-targets which indicated that the targets of HQD against ulcerative colitis may be through IL6, TNF, IL1B, PTGS2, ESR1,PPARG, which have a high degree in PPI network. IL6, TNF, IL1B have been proved that have a strong impact on the pathological process, as for docking test, we found PTGS2 and PPARG have the lower energy than others, and we focus on more details in PTGS2, ESR1, and PPARG.
ESR1, a nuclear hormone receptor, is involved in the regulation of eukaryotic gene expression and affects cellular proliferation and differentiation in target tissues by the steroid hormones and their receptors. ESR1 has mutual transrepression with NF-κB, e.g. decreases NF-κB DNA-binding activity and inhibits NF-κB-mediated transcription19. Also, it can involve activation of NOS3 and endothelial nitric oxide production20. Dysregulation of estrogen receptors in the intestinal mucosa of ulcerative colitis indicates that estrogen signaling may play a role in the local immune response and maintain epithelial homeostasis in a gender- and age-dependent manner21, even though there is no evidence in difference prevalence in male and female.
PPARG (PPARγ), a new coming experiment targeting the treatment of ulcerative colitis, is one of the isoforms of nuclear PPAR receptors that belong to the steroid receptor superfamily. It can be bound to DNA-specific PPAR response elements (PPRE) and modulates the transcription of its target genes, so that controls the peroxisomal beta-oxidation pathway of fatty acids. In addition to playing a role in the regulation of cardiovascular circadian rhythms adipocyte differentiation, and glucose homeostasis, PPARγ also acts as a critical regulator of gut homeostasis by suppressing NF-κB-mediated proinflammatory responses22,23,24.
Presence of PPARγ can be observed throughout the gastrointestinal tract: mesenteric adipocytes, macrophages, and epithelium, most prevalently in the more differentiated layer of epithelial cell, have the highest levels of PPARγ mRNA than other tissue25,26,27,28. However, disease onset of ulcerative colitis provokes inflammation of the colonic intestinal wall, showing significant downregulation of PPARγ, compared with remission mucosal tissue29,30,31. Increased transcriptional activity of PPARγ was linked to decreased intestinal inflammation in DSS-induced colitis mice32.
Besides, a present study has proved that PPARγ can be activation by COX-2 products (15d-PGJ2-G, a chemical metabolite of PGD2-G) to perform anti-inflammatory in ulcerative colitis33. PTGS2 (COX-2) is a key gene with a particular role in the inflammatory response and expresses in epithelial cells and mononuclear cells in IBD34,35. Prostaglandins are critical to the process of mucosal healing in the colonic tract36.
Moreover, core components of HQD act in a multi-component and multi-target way. Quercetin, acacetin, wogonin, norwogonin, baicalein, salvigenin, oroxylin a were key active components of HQD in the treatment of ulcerative colitis. Quercetin has an antioxidant effect, suppresses proinflammatory mediators' release and expression of inflammatory proteins37. Acacetin by inhibiting inflammation and regulating the intestinal microbiota in DSS-induced colitis mice38. Wogonin is quickly converted into baicalin and various flavones in metabolism and may act in the PI3K-Akt pathway, AMPK pathways, and inhibition of telomerase activity39. Baicalin has been confirmed to inhibit IL-33 and NF-κB p65 expression, increase IκB-α levels, regulate Th17/Treg balance and gut microbiota to act antiinflammation40,41. Oroxylin A as the main component extracted from Scutellariae radix can decrease the expression of inflammatory cytokines IL-6 and IL-1β, and attenuated IL-6/STAT3 signaling pathway in ulcerative colitis mice42. Norwogonin can increase Bax and decrease in Bcl-2 levels, induction of autophagy along with triggering G2/M phase cell cycle arrest to suppress the colon cancer43.
In GO and KEGG pathway enrichment analysis, HQD may through several cellular components, such as membrane raft, extracellular matrix, and receptor complex to active in molecular functions, including oxidoreductase activity, endopeptidase activity, and protein kinase activity to get involved in biological processes, such as TNF signaling pathway, IL-17 signaling pathway, Th17 cell differentiation, and NF-kappa B signaling pathway.
Recover intestinal mucosal homeostasis faces a particularly challenging change, because it is against infection, commensal organisms, and unbalance T cells differentiation44. Th17 is the most synthetic T cell, plays a vital role in gut between homeostasis and inflammation, and is thought to play a pathogenic role in IBD45,46. In a previous study, Th17 cell differentiation can be driven by lnc-ITSN1-2 and correlate with increased IBD risk, activity, and inflammatory cytokines47. NF-κB signaling is often dysregulated resulting in overzealous inflammation and distinctly into two pathways, canonical or non-canonical48. Different from rapid, acute inflammation is canonical NF-κB signaling pathway, non-canonical NF-κB signaling has become more evident in maintaining immune system homeostasis in the gut, which regulates T cells differentiation and function49,50. In sum, HQD mainly acts into multi-component, multi-target, multi-pathway in treating ulcerative colitis.
Limitations
Further pharmacological experiments are needed in order to validate the therapeutic mechanism of HQD on ulcerative colitis.
Conclusion
In this study, we combined bioinformatics analysis, network pharmacology, and molecular docking to reveal the mechanisms of HQD against ulcerative colitis. HQD may be through multi-components and multi-pathway in treating ulcerative colitis.
Data availability
The data used to support the findings of this study is available.
Abbreviations
- TCM:
-
Traditional Chinese Medicine
- UC:
-
Ulcerative colitis
- IBD:
-
Inflammatory bowel disease
- HQD:
-
Huangqin decoction
- SR:
-
Scutellariae Radix
- PRA:
-
Paeoniae Radix Alba
- JF:
-
Jujubae Fructus
- GL:
-
Glycyrrhiza glabra L.
- TCMSP:
-
Traditional Chinese Medicine Systems Pharmacology Database and Analysis Platform
- UniprotKB:
-
Uniprot knowledgebase
- GEO:
-
Gene expression omnibus
- DEGs:
-
Differentially expressed genes
- STRING:
-
Search Tool for the retrieval of interacting genes
- PPI:
-
Protein–protein interaction
- GO:
-
Gene Ontology
- KEGG:
-
The Kyoto encyclopedia of genes and genomes
- IL6:
-
Interleukin-6
- TNF:
-
Tumor necrosis factor
- IL1B:
-
Interleukin-1 beta
- PTGS2:
-
Prostaglandin G/H synthase 2
- ESR1:
-
Estrogen receptor
- PPARG:
-
Peroxisome proliferator-activated receptor gamma
References
Ungaro, R., Mehandru, S., Allen, P. B., Peyrin-Biroulet, L. & Colombel, J. F. Ulcerative colitis. Lancet 389, 1756–1770. https://doi.org/10.1016/s0140-6736(16)32126-2 (2017).
Hibi, T. & Ogata, H. Novel pathophysiological concepts of inflammatory bowel disease. J. Gastroenterol. 41, 10–16. https://doi.org/10.1007/s00535-005-1744-3 (2006).
Ordás, I., Eckmann, L., Talamini, M., Baumgart, D. C. & Sandborn, W. J. Ulcerative colitis. Lancet 380, 1606–1619. https://doi.org/10.1016/s0140-6736(12)60150-0 (2012).
Yu, Y. R. & Rodriguez, J. R. Clinical presentation of Crohn’s, ulcerative colitis, and indeterminate colitis: Symptoms, extraintestinal manifestations, and disease phenotypes. Semin. Pediatr. Surg. 26, 349–355. https://doi.org/10.1053/j.sempedsurg.2017.10.003 (2017).
Magro, F. et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis: Part 1: Definitions, diagnosis, extra-intestinal manifestations, pregnancy, cancer surveillance, surgery, and Ileo-anal pouch disorders. J. Crohn’s & Colitis 11L, 649–670. https://doi.org/10.1093/ecco-jcc/jjx008 (2017).
Ungaro, R., Colombel, J. F., Lissoos, T. & Peyrin-Biroulet, L. A treat-to-target update in ulcerative colitis: A systematic review. Am. J. Gastroenterol. 114, 874–883. https://doi.org/10.14309/ajg.0000000000000183 (2019).
Laine, L. et al. SCENIC international consensus statement on surveillance and management of dysplasia in inflammatory bowel disease. Gastroenterology 148, 639-651.e628. https://doi.org/10.1053/j.gastro.2015.01.031 (2015).
Yang, Y. et al. Gut microbiota drives the attenuation of dextran sulphate sodium-induced colitis by Huangqin decoction. Oncotarget 8, 48863–48874. https://doi.org/10.18632/oncotarget.16458 (2017).
Li, M. Y. et al. Effects of Huangqin Decoction on ulcerative colitis by targeting estrogen receptor alpha and ameliorating endothelial dysfunction based on system pharmacology. J. Ethnopharmacol. 271, 113886. https://doi.org/10.1016/j.jep.2021.113886 (2021).
Ru, J. et al. TCMSP: a database of systems pharmacology for drug discovery from herbal medicines. J. Cheminfo. 6, 13. https://doi.org/10.1186/1758-2946-6-13 (2014).
Wang, S., Wang, H. & Lu, Y. Tianfoshen oral liquid: A CFDA approved clinical traditional Chinese medicine, normalizes major cellular pathways disordered during colorectal carcinogenesis. Oncotarget 8, 14549–14569. https://doi.org/10.18632/oncotarget.14675 (2017).
Kim, S. et al. PubChem 2019 update: Improved access to chemical data. Nucleic Acids Res. 47, D1102-d1109. https://doi.org/10.1093/nar/gky1033 (2019).
Daina, A., Michielin, O. & Zoete, V. SwissTargetPrediction: updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Res. 47, W357-w364. https://doi.org/10.1093/nar/gkz382 (2019).
Szklarczyk, D. et al. The STRING database in 2017: quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res. 45, D362-d368. https://doi.org/10.1093/nar/gkw937 (2017).
Shannon, P. et al. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 13, 2498–2504. https://doi.org/10.1101/gr.1239303 (2003).
Zhou, Y. et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 10, 1523. https://doi.org/10.1038/s41467-019-09234-6 (2019).
Kanehisa, M., Furumichi, M., Sato, Y., Ishiguro-Watanabe, M. & Tanabe, M. KEGG: Integrating viruses and cellular organisms. Nucleic Acids Res. 49, D545-d551. https://doi.org/10.1093/nar/gkaa970 (2021).
Xiao, B. et al. Combination therapy for ulcerative colitis: orally targeted nanoparticles prevent mucosal damage and relieve inflammation. Theranostics 6, 2250–2266. https://doi.org/10.7150/thno.15710 (2016).
Stein, B. & Yang, M. X. Repression of the interleukin-6 promoter by estrogen receptor is mediated by NF-kappa B and C/EBP beta. Mol. Cell. Biol. 15, 4971–4979. https://doi.org/10.1128/mcb.15.9.4971 (1995).
Kim, K. H., Toomre, D. & Bender, J. R. Splice isoform estrogen receptors as integral transmembrane proteins. Mol. Biol. Cell 22, 4415–4423. https://doi.org/10.1091/mbc.E11-05-0416 (2011).
Jacenik, D. et al. Sex- and age-related estrogen signaling alteration in inflammatory bowel diseases: modulatory role of estrogen receptors. Int. J. Mol. Sci. https://doi.org/10.3390/ijms20133175 (2019).
Mukherjee, R., Jow, L., Croston, G. E. & Paterniti, J. R. Jr. Identification, characterization, and tissue distribution of human peroxisome proliferator-activated receptor (PPAR) isoforms PPARgamma2 versus PPARgamma1 and activation with retinoid X receptor agonists and antagonists. J. Biol. Chem. 272, 8071–8076. https://doi.org/10.1074/jbc.272.12.8071 (1997).
Yin, Y. et al. 3-phosphoinositide-dependent protein kinase-1 activates the peroxisome proliferator-activated receptor-gamma and promotes adipocyte differentiation. Mol. Endocrinol. 20, 268–278. https://doi.org/10.1210/me.2005-0197 (2006).
Park, S. H. et al. Endoplasmic reticulum stress-activated C/EBP homologous protein enhances nuclear factor-kappaB signals via repression of peroxisome proliferator-activated receptor gamma. J. Biol. Chem. 285, 35330–35339. https://doi.org/10.1074/jbc.M110.136259 (2010).
Fajas, L. et al. The organization, promoter analysis, and expression of the human PPARgamma gene. J. Biol. Chem. 272, 18779–18789. https://doi.org/10.1074/jbc.272.30.18779 (1997).
Saez, E. et al. Activators of the nuclear receptor PPARgamma enhance colon polyp formation. Nat. Med. 4, 1058–1061. https://doi.org/10.1038/2042 (1998).
Braissant, O. & Wahli, W. Differential expression of peroxisome proliferator-activated receptor-alpha, -beta, and -gamma during rat embryonic development. Endocrinology 139, 2748–2754. https://doi.org/10.1210/endo.139.6.6049 (1998).
Michalik, L. & Wahli, W. Peroxisome proliferator-activated receptors: three isotypes for a multitude of functions. Curr. Opin. Biotechnol. 10, 564–570. https://doi.org/10.1016/s0958-1669(99)00030-0 (1999).
Yamamoto-Furusho, J. K., Jacintez-Cazares, M., Furuzawa-Carballeda, J. & Fonseca-Camarillo, G. Peroxisome proliferator-activated receptors family is involved in the response to treatment and mild clinical course in patients with ulcerative colitis. Dis. Markers 2014, 932530. https://doi.org/10.1155/2014/932530 (2014).
Dubuquoy, L. et al. Impaired expression of peroxisome proliferator-activated receptor gamma in ulcerative colitis. Gastroenterology 124, 1265–1276. https://doi.org/10.1016/s0016-5085(03)00271-3 (2003).
Yamamoto-Furusho, J. K., Peñaloza-Coronel, A., Sánchez-Muñoz, F., Barreto-Zuñiga, R. & Dominguez-Lopez, A. Peroxisome proliferator-activated receptor-gamma (PPAR-γ) expression is downregulated in patients with active ulcerative colitis. Inflamm. Bowel Dis. 17, 680–681. https://doi.org/10.1002/ibd.21322 (2011).
Choo, J. et al. A novel peroxisome proliferator-activated receptor (PPAR)γ Agonist 2-Hydroxyethyl 5-chloro-4,5-didehydrojasmonate exerts anti-inflammatory effects in colitis. J. Biol. Chem. 290, 25609–25619. https://doi.org/10.1074/jbc.M115.673046 (2015).
Alhouayek, M., Buisseret, B., Paquot, A., Guillemot-Legris, O. & Muccioli, G. G. The endogenous bioactive lipid prostaglandin D(2)-glycerol ester reduces murine colitis via DP1 and PPARγ receptors. FASEB J. Official Pub. Federation Am. Soc. Exp. Biol. 32, 5000–5011. https://doi.org/10.1096/fj.201701205R (2018).
Farrokhyar, F., Swarbrick, E. T. & Irvine, E. J. A critical review of epidemiological studies in inflammatory bowel disease. Scand. J. Gastroenterol. 36, 2–15. https://doi.org/10.1080/00365520150218002 (2001).
Cox, D. G. et al. Haplotype of prostaglandin synthase 2/cyclooxygenase 2 is involved in the susceptibility to inflammatory bowel disease. World J. Gastroenterol. 11, 6003–6008. https://doi.org/10.3748/wjg.v11.i38.6003 (2005).
Wallace, J. L. Prostaglandin biology in inflammatory bowel disease. Gastroenterol. Clin. North Am. 30, 971–980. https://doi.org/10.1016/s0889-8553(05)70223-5 (2001).
Habtemariam, S. & Belai, A. Natural therapies of the inflammatory bowel disease: the case of rutin and its aglycone Quercetin. Mini Rev. Med. Chem. 18, 234–243. https://doi.org/10.2174/1389557517666170120152417 (2018).
Ren, J. et al. Acacetin ameliorates experimental colitis in mice via inhibiting macrophage inflammatory response and regulating the composition of gut microbiota. Front. Physiol. 11, 577237. https://doi.org/10.3389/fphys.2020.577237 (2020).
Huynh, D. L. et al. Anti-tumor activity of wogonin, an extract from Scutellaria baicalensis, through regulating different signaling pathways. Chin. J. Nat. Med. 15, 15–40. https://doi.org/10.1016/s1875-5364(17)30005-5 (2017).
Zhang, C. L. et al. Baicalin may alleviate inflammatory infiltration in dextran sodium sulfate-induced chronic ulcerative colitis via inhibiting IL-33 expression. Life Sci. 186, 125–132. https://doi.org/10.1016/j.lfs.2017.08.010 (2017).
Zhu, L. et al. Protective effect of baicalin on the regulation of Treg/Th17 balance, gut microbiota and short-chain fatty acids in rats with ulcerative colitis. Appl. Microbiol. Biotechnol. 104, 5449–5460. https://doi.org/10.1007/s00253-020-10527-w (2020).
Yang, X. et al. Oroxylin A inhibits colitis-associated carcinogenesis through modulating the IL-6/STAT3 signaling pathway. Inflamm. Bowel Dis. 19, 1990–2000. https://doi.org/10.1097/MIB.0b013e318293c5e0 (2013).
Wang, Z. et al. Norwogonin flavone suppresses the growth of human colon cancer cells via mitochondrial mediated apoptosis, autophagy induction and triggering G2/M phase cell cycle arrest. J. B.U.O.N. 25, 1449–1454 (2020).
Fujino, S. et al. Increased expression of interleukin 17 in inflammatory bowel disease. Gut 52, 65–70. https://doi.org/10.1136/gut.52.1.65 (2003).
Schirmer, M., Garner, A., Vlamakis, H. & Xavier, R. J. Microbial genes and pathways in inflammatory bowel disease. Nat. Rev. Microbiol. 17, 497–511. https://doi.org/10.1038/s41579-019-0213-6 (2019).
Ueno, A. et al. Th17 plasticity and its relevance to inflammatory bowel disease. J. Autoimmun. 87, 38–49. https://doi.org/10.1016/j.jaut.2017.12.004 (2018).
Nie, J. & Zhao, Q. Lnc-ITSN1-2, derived From RNA sequencing, correlates with increased disease risk, activity and promotes CD4(+) T cell activation, proliferation and Th1/Th17 cell differentiation by serving as a ceRNA for IL-23R via sponging miR-125a in inflammatory bowel disease. Front. Immunol. 11, 852. https://doi.org/10.3389/fimmu.2020.00852 (2020).
Morgan, D., Garg, M., Tergaonkar, V., Tan, S. Y. & Sethi, G. Pharmacological significance of the non-canonical NF-κB pathway in tumorigenesis: Biochimica et biophysica acta. Reviews on cancer 1874, 188449. https://doi.org/10.1016/j.bbcan.2020.188449 (2020).
Walsh, N. C. et al. LKB1 inhibition of NF-κB in B cells prevents T follicular helper cell differentiation and germinal center formation. EMBO Rep. 16, 753–768. https://doi.org/10.15252/embr.201439505 (2015).
Onder, L. et al. Alternative NF-κB signaling regulates mTEC differentiation from podoplanin-expressing precursors in the cortico-medullary junction. Eur. J. Immunol. 45, 2218–2231. https://doi.org/10.1002/eji.201545677 (2015).
Acknowledgements
We thank Tianjin University of Traditional Chinese Medicine for the experimental platform provided for this study.
Funding
This work was supported by the 4th National Academic Experience Training Program of Famous Chinese Medicine Experts (Li Gui-wei) (Letter from the State Administration of TCM [2017] 24).
Author information
Authors and Affiliations
Contributions
Y.W. and X.L. designed the study. Y.W. and G.L. collected data. Y.W. and performed the data analysis and wrote the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wu, Y., Liu, X. & Li, G. Integrated bioinformatics and network pharmacology to identify the therapeutic target and molecular mechanisms of Huangqin decoction on ulcerative Colitis. Sci Rep 12, 159 (2022). https://doi.org/10.1038/s41598-021-03980-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-03980-8
- Springer Nature Limited