Abstract
Elucidating correlations between wild pig (Sus scrofa) behavior and landscape attributes can aid in the advancement of management strategies for controlling populations. Using GPS data from 49 wild pigs in the southeastern U.S., we used hidden Markov models to define movement path characteristics and assign behaviors (e.g., resting, foraging, travelling). We then explored the connection between these behaviors and resource selection for both sexes between two distinct seasons based on forage availability (i.e., low forage, high forage). Females demonstrated a crepuscular activity pattern in the high-forage season and a variable pattern in the low-forage season, while males exhibited nocturnal activity patterns across both seasons. Wild pigs selected for bottomland hardwoods and dense canopy cover in all behavioral states in both seasons. Males selected for diversity in vegetation types while foraging in the low-forage season compared to the high-forage season and demonstrated an increased use of linear anthropogenic features across seasons while traveling. Wild pigs can establish populations and home ranges in an array of landscapes, but our results demonstrate male and female pigs exhibit clear differences in movement behavior and there are key resources associated with common behaviors that can be targeted to improve the efficiency of management programs.
Similar content being viewed by others
Introduction
Understanding how animals move throughout landscapes and interact with heterogeneously distributed resources is critical for management of invasive species because this knowledge provides insight regarding how populations persist and expand, and is thus one of the central goals of ecological research1,2. Habitat characteristics that meet specific needs for different behavioral states (e.g., resting vs. foraging) of an animal are usually spatially segregated; therefore, investigation of movement patterns and habitat selection at a fine spatial scale can be used to illustrate the asynchrony of the behavioral strategies employed over time3. The observed movement patterns that make up an animal’s home range are determined by single movement steps that provide information on the interactions between the individual’s external environment and behavioral state4,5. Therefore, this interaction represents an animal’s response to the environment6. For example, in heterogeneous landscapes an animal can respond to variable stimuli such as food availability, cover, and water that can change the trajectory of their movement path6. These responses are ultimately the result of a continual decision-making trade-offs every animal has to make about the wide range of competing demands to survive and reproduce3. Understanding these underlying fine-scale interactions with resources allows managers to predict movements of animals in different landscapes to optimize management planning7,8.
Despite the relevance of these fine-scale behavioral questions to conservation and management goals, behavior-specific resource selection is understudied in most species due to the lack of behavioral context associated with animal location data9. Animal behaviors, and the driving factors behind these behaviors, are difficult to quantify in the absence of proper data resolution and analytical tools10. However, continued advancements in global positioning system (GPS) tracking technologies and behavioral analysis techniques provide the ability to estimate behavioral states using movement path characteristics such as turning angles and step-lengths11,12,13. In particular, hidden Markov models (HMM) allow for the exploration of patterns in movement path characteristics created by underlying behavioral states and estimation of the probabilities of transitioning among the identifiable states10,14,15. Thus, the application of HMMs to animal relocation data can uncover physiological or behavioral states of tracked individuals, which in turn can be used in a resource selection analysis to infer resource selection associated with identified behaviors.
In the case of an adaptable generalist like invasive wild pigs (Sus scrofa), innovative management is critical given the rapid increase in size and distribution of populations throughout their introduced range. In addition, management is important to mitigate the extensive impacts of this species on anthropogenic and natural systems16,17. The correlation between behavior and landscape patterns can inform how unexpected populations emerge in new places and continue to expand their range (e.g., travelling via movement corridors and identifying suitable resources to reside), as well as help identify areas that may act as hotspots for disease transmission (e.g., areas associated with close contact behaviors such as resting and foraging). These are major concerns for wildlife managers since wild pigs have the potential to alter ecosystems across broad spatial scales and have extreme economic impacts6,16,17,18. Like most wild animals, the movement behavior of wild pigs is largely driven by spatio-temporal variation in the distribution of resources throughout the landscape19,20. Wild pigs move deliberately, choosing different resource patches depending on their current needs (rest, forage, mates, etc.) relative to what is available. Their movements also depend on whether or not the tradeoff for accessing these resources is energetically reasonable21,22,23. When targeting these resources for specific behaviors or needs, wild pig movements tend to be methodical, as they often consistently use the same trails and interact with the same areas on the landscape24,25. These patterns tend to change at a broad scale with food availability and dietary shifts throughout the year26; however, there is little to no information regarding how wild pigs change fine-scale resource selection and activity patterns associated with specific behaviors as a result of changing landscape characteristics or food availability. Identifying fine-scale behavioral resource selection and activity patterns of wild pigs can inform more effective and efficient selection and development of site-specific management techniques.
In this study, we estimated population-level resource selection patterns (Second Order)27 of wild pigs across two distinct periods (hereafter ‘seasons’) based on food availability (high- and low-forage availability) in the Southeast U.S. We then used HMM’s to distinguish and define movement patterns into associated behavioral states (e.g., resting, foraging, traveling) of wild pigs. Lastly, we evaluated the relationship between behavioral states and resource selection. We tested the hypothesis that wild pigs exhibit differential resource selection patterns depending on their behavioral state (Third Order)27 and availability of forage resources. We expected females and males to demonstrate different activity patterns throughout the day (i.e., diel patterns: crepuscular, nocturnal, diurnal) due to differences in reproductive responsibilities (e.g., mate seeking, gestation, farrowing) (Table 1). For example, females may have a more variable activity pattern that coincides with farrowing since they reduce movements during the period when piglets have limited mobility28 (Table 1). In addition, we expected associated resource selection with movement behaviors to shift throughout the year based on food availability. Overall, given their association with riparian areas19,29,30, we expected behavioral states that aligned with restricted movements (i.e., resting and foraging) to be associated with forested areas proximal to water (i.e., bottomland hardwoods) and areas with greater canopy cover, especially in the warmer and mast (e.g., acorns) producing months (Table 1). In contrast, given the heterogeneous distribution of riparian areas throughout our study site we expected wild pigs would more extensively use upland pines and linear features such as roads while traveling (Table 1). During low-forage months, we expected wild pigs to be more opportunistic foragers leading to more variable patterns of resource selection while foraging (Table 1).
Methods
Study area
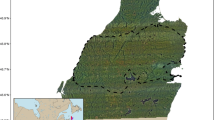
Our work was conducted on the Savannah River Site (SRS), a ~ 800 km2 site managed by the U.S. Department of Energy (DOE) on the Georgia-South Carolina border (Fig. 1). Although established for industrial activities, facilities and infrastructure comprise a small proportion of the landscape, with most of the landscape being managed by the United States Forest Service (USFS) for timber production and wildlife conservation. The SRS was comprised of approximately 50% upland pine including loblolly pine (Pinus taeda), longleaf pine (Pinus palustris), and slash pine (Pinus elliottii), 25% was bottomland hardwood forest, 10% shrub/herbaceous-dominated areas, 8% upland hardwoods, and the rest was mixed forest, developed, and barren land. Wild pigs have been managed on the SRS since the early 1950s, when an active live-trap-and-removal program was initiated to mitigate damages caused by wild pigs31. This program is managed by USFS and currently removes ~ 1300–1800 pigs annually32. Despite this control, there are several thousand wild pigs inhabiting the SRS that are distributed throughout the site33. Since the SRS was previously used to manufacture nuclear materials and manage nuclear waste34, there is limited public access across the site. The diversity of habitat types of the SRS combined with the limited public access, diversity of other wildlife species present, and high wild pig densities (i.e., > 4000–5000 individuals; ~ 5–6.25 individuals/km2)33 make the site an ideal location to study movement patterns and resource selection of this species.
Overall study area with distinct vegetative communities and the 1.2 km2 polygon representing the specified area used to develop available locations for second order resource selection functions of male and female wild pigs (Sus scrofa) during two distinct seasons (i.e., low-forage and high forage) between Janurary 2014–December 2019 in South Carolina, USA.
Field methods
We captured wild pigs throughout the SRS from January 2014–December 2019 using baited-corral traps equipped with a combination of remote-operated and trip-wire mechanisms. We monitored traps using remote cameras (Reconyx PC900, Holmen, WI, USA) to identify dominant sows to receive GPS collars, as well as all breeding-aged males. We used a dart rifle (X-Caliber, Pneu-Dart Inc., Pennsylvania, USA) to anesthetize captured pigs using a combination of butorphanol [0.077 mg/kg], azaperone [0.026 mg/kg], medetomidine [0.031 mg/kg] (BAM; 0.031 ml/kg; Wildlife Pharmaceuticals Inc., Colorado, USA; Ellis et al. 2019) and Ketamine (2.2 mg/kg; Wildlife Pharmaceuticals Inc., Colorado, USA) or Xylazine (2.2 mg/kg; Wildlife Pharmaceuticals Inc., Colorado, USA) and Telazol (4.4 mg/kg; MWI Veterinary Supply, Idaho, USA). While under anesthesia, we recorded sex and assessed age through examination of tooth eruption36. We fit the largest subadult or adult female in each sounder (i.e., social unit) and breeding-aged males (i.e., > 1 year) with an iridium GPS collar (Telonics Gen4 GPS/Iridium System; Sensor weight = 260 g; Total Collar Weight = ~ 500–880 g, Telonics, Inc., Mesa, Arizona or VECTRONIC GPS PLUS Globalstar-3; Total Collar Weight = ~ 830 g, VECTRONIC Aerospace, Coralville, Iowa). Anesthetized wild pigs were allowed to recover at the capture site after being reversed with a combination of Atipamezole (25 mg/ml; Wildlife Pharmaceuticals Inc.) and Naltrexone (50 mg/ml; Wildlife Pharmaceuticals Inc., Colorado, USA). Collars were programmed to record GPS locations at 30-min or one-hour intervals and equipped with a mortality sensor that became activated after 12 h with no movement by the animal. To avoid pseudo-replication, we only tracked one individual from each social group and all solitary boars. All experimental protocols were approved by and conducted in compliance with the University of Georgia’s Animal Care and Use Committee (Protocols: A2012 08-004, A2015 05-004, and A2018 08-013), and where applicable, with Animal Research: Reporting In Vivo Experiments (ARRIVE) guidelines.
To estimate location error of GPS transmitters, we left a subset of three collars out for 10 days in fixed locations, 5 days in open vegetation and 5 days in forest vegetation. We used these data to calculate the average error among fixes for each habitat type, and to inform initial parameters for behavioral states.
Identification of movement states
We used HMMs to model the movement characteristics and associated behavioral states of wild pigs for two distinct seasons based on food availability. We considered January through April to represent a low-food availability time period based on dietary preferences of wild pigs19, which also generally represents the peak trapping season in the Southeastern U.S. May through December was considered a high-food availability time period when ample amounts of fruits and plants are available throughout the Spring and Summer months, followed by acorns and other mast in Fall and early Winter. Initially, we used all data collected at 30-min intervals and compared HMM outputs for 30-min locations to outputs of models for the same individuals when subset to 1-h locations, and there were no substantial differences. Therefore, we subset data for wild pigs with a 30-min GPS fix rate to 1-h intervals to maintain an equivalent temporal resolution within our dataset. We also removed any duplicate locations (e.g., same date-time stamp) and locations associated with non-pig movements (e.g., locations after mortality). From collars we were able to retrieve and download, less than 0.01% of locations were 2-Dimensional fixes (i.e., locations collected with three satellites). Therefore, we included all locations regardless of dimensional fix within our dataset to be consistent across all individuals. We also removed the first 48 h of GPS fixes to account for any potential bias associated with residual anesthetic effects.
We used step-lengths and turning angles as our observational input data in HMMs to differentiate among behaviors. We compared HMM results from 25 different sets of randomly chosen starting values for step-lengths and turning angle distribution parameters for each behavioral state to ensure we were capturing global maximums of the likelihood function12 (Supplementary Table S1). In addition, using an array of starting values from parameter distributions ensures that models were numerically stable12. We tested HMMs with two and three movement states based on model parsimony13, but also took into consideration the biological relevance of identified states because model selection criteria sometimes tend to favor models with a greater number of states than makes biological sense37.
Sex has been found to be an important predictor of wild pig home range size, with males typically having a larger home range and greater movement rates than females38. Also, wild pigs have demonstrated seasonal differences in home range size and habitat selection based on resource availability19,23,39. Therefore, we expected sex-specific and seasonal-specific differences in the movement parameters (e.g., step-lengths and turning angles) associated with each behavioral state. We also expected differences in transition probabilities among states throughout the diel period, which ultimately adds to the insight of the model when using it to decode states. We ran two and three movement state HMMs separately for males and females in both the low- and high-forage seasons and tested for an additive effect of time of day on the probability of transitioning among states. Therefore, we ran a total of eight HMMs (Table 2). We selected the most parsimonious model for both seasons for females and males separately using Akaike Information Criterion (AIC)40. Next, we decoded the most likely sequence of states to have produced each location in the movement path of each wild pig given the most parsimonious model using the Viterbi algorithm15. All computations were done using the moveHMM package12 in the statistical computing software R 3.6.141. We partitioned GPS locations into appropriate behavioral states and quantified resource selection for both sexes in each season and behavioral state at the third order (i.e., home range) spatial scale27.
Resource selection analyses
Habitat covariates
We generated individual raster layers for five types of land cover from the 2016 National Land Cover Database (NLCD) raster layer (30 × 30 m-resolution)42 for resource selection analyses: (1) upland pines, (2) bottomland hardwoods, (3) shrub and herbaceous, (4) upland hardwoods, and (5) developed (i.e., buildings/structures). We also characterized the distribution of streams and roads within our study area from existing SRS geospatial layers. We classified primary roads as those that were paved and routinely used for travel by SRS employees, whereas secondary roads were unpaved gravel and/or logging roads. We used the Euclidean distance tool in ArcGIS 10.7.1 (Environmental System Research Institute, Inc., Redlands, CA, USA) to calculate the distance to each of the covariates for used and available locations to provide a less ambiguous approach compared to a classification or categorical-approach43 (i.e., A location would receive a “0” for the vegetation type it is observed in). Lastly, we used the NLCD 2016 USFS tree canopy cover raster (30 × 30 m-resolution) to estimate the percent canopy cover.
Second order
We selected a 481 km2 area within the SRS to represent the study area for this analysis. We generated a minimum convex polygon (MCP) around all GPS locations and buffered it by 1.2 km to account for any long distance movements (Fig. 1)17,35. We quantified habitat availability for the population at the second order by systematically sampling the study area (every 3rd pixel, i.e., 90 m; available locations) to ensure the entire area was represented yet maintain a dataset that was computationally manageable, compared to random sampling which may involve uncertainty and not effectively represent the overall landscape44. We compared these locations to locations classified as ‘used’ generated by systematically sampling (every 3rd pixel, i.e., 90 m; used locations) within a 95% fixed kernel home range for each individual. Uniformly sampling locations across home ranges allows a comprehensive representation of the resources within a home range to compare to the available locations within the study area. We used the adehabitat package with the reference bandwidth (href) smoothing parameter45 in the statistical computing software R 3.6.141 to generate and sample all home ranges. We created individual home ranges for both seasons to compare seasonal shifts in home range distribution. We evaluated used locations specific to each individual home range against the same set of available locations throughout the study area for all individuals. We calculated Pearson’s correlation coefficients to test for collinearity between each of our covariates and excluded covariates with a Pearson’s |r|> 0.63. Covariates that were highly correlated included distance to primary roads and distance to buildings/structures (r = 0.75); therefore, we retained the covariate of distance to primary roads only for modeling purposes given this covariate was more represented in the areas wild pigs were captured. We then fit a global (i.e., including all covariates) generalized linear model (GLM) with binomial response distribution (logistic regression) and logit link to the used-available data individually for both sexes in both the low-forage and high-forage seasons46,47. This resulted in four comprehensive models representative of second order resource selection for females and males in the low-forage season and high-forage seasons (Table 2). We standardized all variables prior to model development [(xi − \(\overline{\text{x}}\))/s] (Supplementary Table S2). We then back-transformed, exponentiated, and raised all distance variable coefficients to the one-hundredth power to represent 100 m increments and canopy cover to the tenth power to represent 10 percent increments for interpretation using predictive odds ratios. We did not use a model selection technique to rank candidate models because a global model included the full set of covariates that were of interest for hypothesis testing and, therefore, allowed a direct comparison between coefficient estimates across sexes and seasons48. All GLM models were computed using the glm function in R version 3.6.141,49. We assessed how well the second order model explained the data using area under the receiver-operating characteristic curve (AUC50,51,52), which we computed using the pROC package in R version 3.6.141,53. A value of 0.5 indicates the model provides predictions that are no better than random predictions, but values greater than 0.7 indicate a better model fit with more accurate predictions51.
Third order
To assess fine-scale resource selection of wild pigs, we used a resource selection function (RSF) framework47 to compare resource selection of wild pigs across the three behavioral states associated with the movement path characteristics identified from the HMM (i.e., resting, foraging, and traveling). We quantified habitat availability for individuals at the third order by comparing GPS locations (i.e., used locations) to systematically sampled locations (every 3rd pixel, i.e., 90 m; available locations) within home ranges across each of the aforementioned covariates (see above). The sampling framework provided inference on the similarities and differences of wild pig resource selection in three prominent behavioral states that can be extracted to the population level. We used a generalized linear mixed model (GLMM) with binomial response distribution (i.e., used vs. available, logistic regression)46, logit link, and a random intercept to account for variation among individuals 54. We standardized all variables prior to model development [(xi − \(\overline{\text{x}}\))/s]. We then back-transformed, exponentiated, and raised all distance variable coefficients to the one-hundredth power to represent 100 m increments and canopy cover to the tenth power to represent 10 percent increments for interpretation using predictive odds ratios. All GLMM models were computed using the lme4 package in R version 3.6.141,49.
We calculated Pearson’s correlation coefficients to test for collinearity between each of our covariates3. We created a global model including all covariates for each sex in each behavioral state in each season (i.e., 2 sexes × 3 behavioral states × 2 seasons = 12 RSFs) (Table 2). As with our second-order analyses, we did not use a model selection technique, and used AUC to assess how well the model explained the data50,51,52.
Disclaimer
This manuscript was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process, or service by trade name, trademark, manufacturer, or otherwise does not constitute or imply its endorsement, recommendation, or favoring by the United States Government or any agency thereof. The views and opinions of the authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.
Results
Identification of movement states
We used a sample of 49 wild pigs tracked between January 2014 and December 2019, resulting in 117,150 validated and cleaned GPS locations (Table 3). In the low-forage season (January–April), we tracked 37 wild pigs (21 females, 16 males), resulting in 47,983 GPS locations, and in the high-forage season (May–December) we tracked 41 wild pigs (20 females, 21 males), resulting in 69,177 GPS locations (Table 3). From these data, we estimated movement path characteristics (e.g., behavioral states) for 29,433 and 42,277 locations for females during the low- and high-forage seasons, respectively. For males, we had 18,550 locations during the low-forage season and 26,900 during the high-forage season to inform our analyses (Table 3). We determined average collar error in forested vegetation to be 22.3 m and in open vegetation to be 11.9 m.
We concluded a three-state HMM with a Gamma distribution for step-length, a wrapped Cauchy distribution for turning angle, and an added covariate of hour in the diel period fit the data of both sexes in both seasons best and provided the most reasonable biological interpretation (Supplementary Table S3). From the three-state HMMs, we identified three general types of movements associated with common behavioral states: (1) a state with short step-lengths and high degrees of turning concentrated around π radians; (2) a state with short to intermediate step-lengths and high degrees of turning concentrated around π radians; and (3) a state with long step-lengths and more straightforward movements with turning concentrated around 0 radians, which likely represents resting, foraging, and traveling behaviors, respectively (Table 4; Figs. 2, 3).
Step-length parameter distributions from three-state hidden Markov models (HMMs) for wild pigs (Sus scrofa) in the Southeast USA by sex and season: (a) females in low-forage months (January–April); (b) males in low-forage months; (c) females in high-forage months (May–December); (d) males in high-forage months.
Turn angle parameter distributions from three-state hidden Markov models (HMMs) for wild pigs (Sus scrofa) in the Southeast USA by sex and season: (a) females in low-forage months (January–April); (b) males in low-forage months; (c) females in high-forage months (May–December); (d) males in high-forage months.
Male and female wild pigs exhibited clear differences in movement behavior. Specifically, average step-lengths differed between sexes, and males and females exhibited differences in partitioning of behavioral states across the diel period (Fig. 4). Males typically traveled farther than females in hour segments (Table 3) and demonstrated evident nocturnal activity by traveling mainly throughout the nighttime hours and resting during most of the day (Fig. 4). Males also maintained a consistent movement pattern across seasons. In contrast, females exhibited their longest step-lengths in the evening hours around dusk in the low-forage season and had a variable behavioral pattern throughout the remainder of the day. However, in high-forage months females had a crepuscular activity pattern with peak traveling and foraging movements around dawn and dusk (Fig. 4). Step-lengths for both sexes were longer during the resting and foraging behaviors in the high-forage season compared to the low-forage season (Table 4).
Proportion of steps per hour for each behavioral state of wild pigs (Sus scrofa) on the Savannah River Site in South Carolina by sex and season: (a) females in low-forage months (January–April); (b) males in low-forage months; (c) females in high-forage months (May–December); (d) males in high-forage months. The dark gray bars represent average nighttime hours while the light gray bar represents the average daytime hours.
Resource selection
Second order
Female wild pigs selected all vegetation types (i.e., upland pines, upland hardwoods, bottomland hardwoods, shrub/herbaceous) across our study area in their home-range placement at the second order in both the low and high-forage seasons (Fig. 5, Supplementary Table S4), likely reflecting the ubiquitous establishment of wild pigs across the Savannah River Site33. Females also selected locations closer to streams and avoided areas near roads. In contrast, males in the low-forage season selected home ranges in or near upland pines, shrub/herbaceous vegetation, and bottomland hardwoods (Fig. 5). In addition, males selected areas close to streams and primary roads. During the high-forage season, males selected resources similarly to the low-forage season, with the main difference of primary roads no longer being an important driver of home range placement (Fig. 5). AUC values in the low-forage season models for females and males were 0.62, 0.66 and in the high-forage season as 0.64, 0.59, respectively.
Predictive odds with 95% confidence intervals for second order selection (Johnson 1980) of female and male wild pigs (Sus scrofa) on the Savannah River Site in South Carolina during two distinct seasons based on forage availability, (a) low-forage availability (January–April) and (b) high-forage availability (May–December), for every 100 m increase for distance variables and every 10% increase for canopy cover. In cases where the confidence interval crosses 1, the variable is considered not significant.
Third order
During the resting state, female wild pigs in the low-forage season strongly selected areas in or close to bottomland hardwoods and shrub/herbaceous habitats (Fig. 6, Supplementary Table S5). For example, there was a 23% decrease in use for every 100 m farther away from bottomland hardwoods, and a there was a 10% decrease in use for every 100 m farther away from shrub and herbaceous habitats. During the high-forage season, female wild pigs selected resting areas similarly to the low-forage season with the addition of a strong selection for upland hardwoods (Supplementary Table S5). Also, the resting model for females in both seasons indicated they avoided areas near secondary roads and streams (Fig. 6). Similarly, males selected resting areas in or close to bottomland hardwoods, upland hardwoods, and shrub/herbaceous communities in both seasons. However, males differed between seasons in selecting to rest near streams during the low-forage season but not during the high-forage season. For example, males demonstrated a 5.4% decrease in use for every 100 m farther away from a stream during the low-forage season (Fig. 6).
Predictive odds with 95% confidence intervals of third-order selection of male and female wild pigs (Sus scrofa) on the Savannah River Site in South Carolina during two distinct seasons based on forage availability [i.e., low-forage season (January–April) and the high-forage season (May–December)]. It demonstrates selection or avoidance of vegetation types, streams, and characteristics of development (e.g., roads) for every 100 m increase and canopy cover for every 10% increase by state where states represent resting, foraging, and traveling behaviors, respectively: (a) Females in low-forage months (January–April); (b) males in low-forage months; (c) females in high-forage months (May–December); (d) males in high-forage months. In cases where the confidence interval crosses 1, the variable is considered not significant.
Throughout the foraging state, females differed in relative probability of selection for specific vegetation types and landscape characteristics between the low- and high-forage seasons (Fig. 6). For example, females selected areas near primary roads and bottomland hardwoods during the low-forage season, yet during the high-forage season they selected areas near upland hardwoods, upland pines, bottomland hardwoods, and areas near primary roads. Males demonstrated more diversity in selection while foraging in the low-forage season including shrub/herbaceous, bottomland hardwoods, and both secondary and primary roads; however, during the high-forage season, males concentrated foraging in areas near or in bottomland hardwood vegetation (Fig. 6). During the high-forage season, males exhibited a 23% decrease in use for every 100 m farther from bottomland hardwoods. In addition, the selection for areas with a high percentage of canopy cover was consistent between sexes and seasons within the foraging behavioral state (Fig. 6).
When traveling, resource selection was similar between seasons for females and males. Females selected primary roads and bottomland hardwoods when traveling in both seasons, with the addition of upland hardwoods in the high-forage season (Fig. 6). Males selected shrub/herbaceous vegetation, primary and secondary roads, and bottomland hardwoods while traveling in both seasons (Fig. 6). For example, in the high-forage season, males displayed a 16% decrease in use for every 100 m farther from secondary roads while traveling (Fig. 6).
The AUC values (overall fit for resting, foraging, and traveling behavioral states in low- and high-forage seasons) were 0.81, 0.79, 0.76 and 0.73, 0.75, 0.73 for females and 0.77, 0.80, 0.70 and 0.77, 0.80, 0.74 for males, respectively.
Discussion
Wild pigs are a major agricultural and environmental pest in their invasive range, and managing impacts is often expensive and difficult to implement17. Therefore, acquiring and analyzing movement data at a fine scale provides important insight on when and where damage or disease transmission is likely to occur. This information provides the ability to improve the efficiency and effectiveness of current management strategies. Therefore, using an extensive dataset of wild pig GPS data across a heterogeneous landscape in the Southeastern U.S., here we demonstrate the differential resource selection tactics employed by wild pigs at both broad (i.e., home range placement) and fine (i.e., within-home-range, behavior-specific) spatial scales for males and females across two distinct seasons. Movement path characteristics of wild pigs in our study were influenced by a combination of local and landscape-level habitat attributes such as bottomland and upland hardwoods, streams, secondary roads, and shrub/herbaceous vegetation communities. While males and females tended to select areas to establish home ranges (population scale) similarly, we found notable differences in the fine-scale use of habitats within home ranges between sexes and seasons. However, both males and females selected bottomland hardwood habitats and areas with extensive canopy cover extensively. Further, through the use of step-lengths and turn angles to define behavioral-based resource selection patterns, we found that females and males differed in daily movement patterns. In addition, we found that wild pigs exhibited differential selection of landscape attributes among behavioral states.
Based on the results of our HMM analyses, we distinguished three biologically relevant behavioral states generally based on patterns in the movement characteristics of wild pigs (i.e., resting, foraging, traveling). Previous studies have identified similar patterns for other species11,37,55; however, behavioral states associated with movement characteristics may be assigned differently depending on prior knowledge of different animal species and fix rate at which GPS data were collected. Specifically, the interpretation of a behavioral state associated with short to intermediate step-lengths (what we defined as foraging) may differ among species. For example, this category of behavior was defined as “locally active at the kill site” for wolves56, “moderately active” for Florida panthers57, and “encamped” for American black bears55. However, for caribou, this intermediate behavioral state was assigned as “foraging” and was associated with a foraging behavior for black bears as well11,55. Although wild pigs exhibit several behaviors that correspond to short and intermediate step-lengths and tight turn angles (e.g., resting, wallowing, rubbing, tusking, foraging, etc.), for management purposes of wild pigs classifying behaviors into resting, foraging, and traveling encapsulated the most common and consistent motivations of space use (e.g., forage, cover, thermoregulation)19 as demonstrated by Blasetti et al.58 using captive adult wild pigs that spent approximately 58.9% of their time resting, 14% of their time foraging, and 27.1% of their time traveling. Also, wild pigs have demonstrated variable activity patterns that can shift throughout the year29,59,60. Classifying these dominant behaviors and understanding that other similar movement-type behaviors are encompassed as well allows the development of knowledge about where to target certain management strategies or further research.
Both females and males decreased movements or traveling behavior in the mid-day, most likely due to the association with high temperatures in the southeast during the high-forage season19,38, and males maintained a consistent nocturnal activity pattern between seasons. However, females exhibited seasonal differences in movement patterns that were likely related to reproductive stages of the reproductive cycle throughout the year, as the timing of farrowing is related to the seasonal availability of forage28,61. In the low-forage season, which corresponded with peak farrowing in our study area28 (Chinn, unpublished data), females demonstrated a sharp increase in traveling at dusk, an increase in foraging throughout daytime hours, a slight increase in resting mid-day, and a distinct increase in resting throughout nighttime hours. However, during the high-forage season when farrowing rates are lower and juvenile pigs are more mobile, females demonstrated a more crepuscular activity pattern compared to the low-forage season. Pre-parturition and parturition-associated behaviors in some wildlife species, such as wild pigs, are associated with reduced movements and home range sizes26,38. Irregular and/or reduced movements can continue after parturition causing an unusual activity pattern in females61, as we found throughout the low-forage season. While reproduction can make it more difficult to assign behaviors and demonstrate consistent patterns in movements for females, this demonstration of a change in activity patterns across seasons is consistent with previous literature and reveals the rigor of the methods used in this study. Males and females have different reproductive tendencies and responsibilities as a polygamous species62 in which males breed multiple females and provide no parental care. Therefore, behavioral differences between sexes likely reflect different reproductive obligations61 and should be a focus for further research, as well as a consideration when designing management plans. Also, the overlap in model parameters between the resting and foraging states for males throughout both seasons and females in the low-forage season indicates that these two states may not be distinct throughout parts of the year. Additional information on animal movement through the use of accelerometers or direct observation, for example, would help to differentiate states with similar distributions of step-lengths and turning angles13,63.
Although wild pigs are an invasive habitat generalist, our approach of evaluating population-scale resource selection in contrast to fine-scale behavioral resource selection revealed wild pigs exhibit differential selection of habitats relative to spatial scale. In areas where wild pigs are abundant, they often occur throughout the landscape, which was reflected in our second order (i.e., home range placement) analysis as wild pigs established home ranges in areas proximal to streams containing broad availability of most vegetation types present on the landscape. However, although wild pigs are well documented to select for areas near streams19,32,64, here we demonstrate this selection is scale dependent, as neither males or females exhibited focused activity within their home ranges around streams across behavioral states. This difference in selection between spatial scales should be considered when targeting an invasive species for management purposes. The second order models for males and females did not demonstrate much strength in the AUC evaluation (< 0.7); therefore, indicating these models do not fit the data exceptionally well. However, we believe this is due to extensive variation in habitat selection among individuals stemming from the fact that wild pigs are a habitat generalist at the population scale.
Wild pigs can demonstrate multiple behaviors in similar vegetation types65, but there are certain habitat characteristics and vegetation types that facilitate specific behaviors (e.g., relocation using roads)6. Although wild pigs are ecological generalists, they exhibit spatio-temporal differences in resource selection that reflect underlying biological needs (e.g., thermoregulation)19,39. Dense cover and areas proximal to water (i.e., bottomland hardwoods) are two key vegetation characteristics that provide resources that pigs require19, and we found that females and males selected for bottomland hardwoods and areas with high percentages of canopy cover in every behavioral state during the low-forage season. In addition, wild pigs forage on subterranean foods such as roots and tubers when other sources are scarce19,66,67; therefore, selecting bottomland hardwoods and areas with extensive canopy cover typically coincide with these forage types and provide access to water and cover.
While foraging, males selected for a variety of vegetation types and structures throughout the low-forage season. For example, at the home-range scale males demonstrated a change in selection for primary roads between seasons. In the low-forage season, males selected for areas closer to primary roads in all three behavioral states. Also, males selected for secondary roads in the foraging and traveling states at the home-range scale. The selection for areas near or along both primary and secondary roads while foraging is likely due to the decrease in resources in adjacent natural areas and the consistent availability of grasses along open roadsides during the low-forage season67,68. These results coincide with the increase in use of urbanized and anthropogenic areas when natural forage is scarce69,70. However, the result of wild pigs utilizing roads could shift in other areas that are associated with hunting or shooting pigs on roads. Wild pigs on the SRS are rarely persecuted (i.e., dog hunting, etc.) on roads; therefore, we expect roads are not associated with negative interactions with humans. Lastly, during the resting state females demonstrated selection for shrub and herbaceous vegetation, which was characterized by a mixture of areas in early successional stages and grasslands that both typically occurred together near linear features such as secondary roads, power lines, and streams, while males selected for this vegetation type in every behavioral state. Areas dominated by this vegetation type most likely provided forage, cover, and easy access to linear features when transitioning to traveling in the low-forage season. Therefore, interactions between wild pig behavior and the attributes of vegetation demonstrated in shrub and herbaceous communities in this study allows for the design of a more informed management plan.
During the high-forage season, at the home-range scale males selected for areas closer to secondary roads while traveling but avoided these areas when foraging and resting. Selecting for anthropogenic and natural linear features can help increase an animal’s pace (step-length) and directional movement, which can assist in traversing the landscape quickly when dispersing, searching for a mate, or transitioning between resting and foraging behaviors19,71,72. Also, males selected primarily for bottomland hardwoods while foraging in the high-forage season, and females selected for upland and bottomland hardwoods during all behavioral states, likely reflecting the availability of food, water, and cover in these habitats19. Selection for bottomland hardwoods is most likely associated with mast producing hardwoods (e.g., oak acorns) and productive plants in the understory throughout summer months, as well as dense cover and proximity to water. Lastly, throughout the high-forage season, males and females avoided streams at the home-range scale, which is likely due to the extensive stream system throughout the SRS and the ability to access dense cover away from streams during times of extreme temperatures. Other studies have demonstrated the insignificance of streams at the home-range scale throughout certain times of the year when water is generally present throughout the landscape72. Unlike the second order models, the AUC values of all third order resource selection models were greater than 0.7 indicating good model fits with meaningful predictions.
Wild pigs exhibit substantive behavioral plasticity making them the perfect invasive species17. They can adjust their life history strategies such as daily activity patterns to decrease interaction with humans in populated areas. In addition, wild pigs can adjust their diet throughout the year and in a variety of climatic conditions to benefit their long-term survival depending on local environmental conditions19,69,73,74. Although our study was limited to the SRS in the Southeastern U.S., wild pigs demonstrate consistent selection patterns for vegetation types associated with certain resources (i.e., water, mast, etc.)19,26,75,76,77. Therefore, our findings are likely applicable in similar areas throughout this species’ native and introduced range. Further research, though, should focus on wild pig behavioral state resource selection in other geographic regions to elucidate spatio-temporal differences in wild pig behavior across areas of differing climate and resource base. In addition, due to rapid growth in body weights and associated limitations of collecting long-term GPS data on free-ranging wild pigs, not all individuals within our dataset were represented across both seasons. We recognize comparing different individuals across seasons could influence the overall results but given our robust sample size, any differences due to individual variation likely would be minor and not alter the ultimate management implications of this work.
While our general findings are consistent with previous literature on wild pig habitat selection, through the investigation of fine-scale movement patterns coupled with behavioral-based resource selection we were able to demonstrate pigs exhibit clear differences in temporal patterns of activity and selection of habitats among behavioral states. Thus, delineating GPS observational data into unique behavioral states provides unique insights into the relative importance of environmental attributes critical to the invasion of an ecosystem or management of a species that may otherwise be obscured through more coarse-scale resource selection approaches3.
Accounting for behavior when studying habitat selection can provide more useful and accurate information for managers dealing with an invasive species. Specifically, for wild pigs, understanding the driving forces of resource selection at a fine scale can inform when, where, and how to deploy traps, toxicants, attractants, etc. to ensure visitations occur quickly and consistently19,78, as well as areas to focus mitigation efforts from wild pig damage. In addition, understanding how wild pigs use the landscape can provide an advantage for managers and/or disease biologists when trying to predict areas of high risk for disease transmission. Our results indicated vegetation class and other landscape features all determined habitat use by wild pigs when resting, foraging, and traveling. Therefore, targeting specific vegetation types, features, and times throughout the diel period could provide an advantage for managers when strategically employing specific management techniques in areas where wild pigs would be most vulnerable. For example, to increase efficiency and effectiveness of management techniques such as trapping and toxicant deployment, targeting wild pigs in habitat types they select for during the foraging and/or traveling behavioral states could greatly increase the number of pigs removed during these management processes78.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author upon request.
References
Wiens, J. A., Stenseth, N. C., Van Horne, B. & Ims, R. A. Ecological mechanisms and landscape ecology. Oikos 66, 369–380 (1993).
Ellner, S. P. et al. Habitat structure and population persistence in an experimental community. Nature 412, 538–543 (2001).
Roever, C. L., Beyer, H. L., Chase, M. J. & van Aarde, R. J. The pitfalls of ignoring behaviour when quantifying habitat selection. Divers. Distrib. 20, 322–333 (2014).
Moorcroft, P. R., Moorcroft, P. & Lewis, M. A. Mechanistic Home Range Analysis (Princeton University Press, 2006).
Boerger, L., Dalziel, B. D. & Fryxell, J. M. Are there general mechanisms of animal home range behaviour? A review and prospects for future research. Ecol. Lett. 11, 637–650 (2008).
Forester, J. D. et al. State-Space Models link elk movement patterns to landscape characteristics in Yellowstone National Park. Ecol. Monogr. 77, 285–299 (2007).
Northrup, J. M., Anderson, C. R., Hooten, M. B. & Wittemyer, G. Movement reveals scale dependence in habitat selection of a large ungulate. Ecol. Appl. 26, 2746–2757 (2016).
Northrup, J. M., Anderson, C. R. & Wittemyer, G. Environmental dynamics and anthropogenic development alter philopatry and space-use in a North American cervid. Divers. Distrib. 22, 547–557 (2016).
Beyer, H. L. et al. The interpretation of habitat preference metrics under use—availability designs. Philos. Trans. R. Soc. B Biol. Sci. 365, 2245–2254 (2010).
Patterson, T. A., Basson, M., Bravington, M. V. & Gunn, J. S. Classifying movement behaviour in relation to environmental conditions using hidden Markov models. J. Anim. Ecol. 78, 1113–1123 (2009).
Franke, A., Caelli, T. & Hudson, R. J. Analysis of movements and behavior of caribou (Rangifer tarandus) using hidden Markov models. Ecol. Model. 173, 259–270 (2004).
Michelot, T., Langrock, R., Patterson, T. moveHMM: An R package for the analysis of animal movement data. 20 (2016).
Leos-Barajas, V. et al. Multi-scale modeling of animal movement and general behavior data using hidden markov models with hierarchical structures. JABES 22, 232–248 (2017).
Schick, R. S. et al. Understanding movement data and movement processes: Current and emerging directions. Ecol. Lett. 11, 1338–1350 (2008).
Zucchini, W., MacDonald, I. L., Langrock, R., MacDonald, I. L. & Langrock, R. Hidden Markov Models for Time Series: An Introduction Using R 2nd edn. (Chapman and Hall/CRC, 2016).
Beasley, J. C., Ditchkoff, S. S., Mayer, J. J., Smith, M. D. & Vercauteren, K. C. Research priorities for managing invasive wild pigs in North America. J. Wildl. Manag. 82, 674–681 (2018).
VerCauteren, K.C., Mayer, J.J., Beasley, J.C., Ditchkoff, S.S., Roloff, G.J., Strickland, B.K. Introduction, invasive wild pigs in North America: Ecology, impacts, and management. 1–5 (2020).
Barrios-Garcia, M. & Ballari, S. Impact of wild boar (sus scrofa) in its introduced and native range: A review. Biol. Invasions 14, 2283–2300 (2012).
Gray, S. M., Roloff, G. J., Montgomery, R. A., Beasley, J. C. & Pepin, K. M. Wild Pig Spatial Ecology and Behavior, Invasive Wild Pigs in North America: Ecology, Impacts, and Management 33–56 (CRC Press, 2020).
Lewis, J. S. et al. Biotic and abiotic factors predicting the global distribution and population density of an invasive large mammal. Sci. Rep. 7, 44152 (2017).
Fortin, D. et al. Wolves influence elk movements: Behavior shapes a trophic cascade in Yellowstone National Park. Ecology 86, 1320–1330 (2005).
Forester, J. D., Im, H. K. & Rathouz, P. J. Accounting for animal movement in estimation of resource selection functions: Sampling and data analysis. Ecology 90, 3554–3565 (2009).
Wilber, M. Q. et al. Predicting functional responses in agro-ecosystems from animal movement data to improve management of invasive pests. Ecol. Appl. 30, e02015 (2020).
Hanson, R. P. & Karstad, L. Feral swine in the southeastern United States. J. Wildl. Manag. 23, 64 (1959).
Oliveira-Santos, L. G. R., Forester, J. D., Piovezan, U., Tomas, W. M. & Fernandez, F. A. S. Incorporating animal spatial memory in step selection functions. J. Anim. Ecol. 85, 516–524 (2016).
Mayer, J.J. Wild pig behavior, wild pigs: Biology, damage, control techniques and management. 408 (2009).
Johnson, D. H. The comparison of usage and availability measurements for evaluating resource preference. Ecology 61, 65–71 (1980).
Comer, C.E., Mayer, J.J. Wild pigs: Biology, damage, control techniques and management. 408 (2009).
Singer, F. J., Otto, D. K., Tipton, A. R. & Hable, C. P. Home ranges, movements, and habitat use of european wild boar in Tennessee. J. Wildl. Manag. 45, 343–353 (1981).
Gaston, W., Armstrong, J., Arjo, W., Stribling, H.L. Home range and habitat use of feral hogs (Sus scrofa) on Lowndes County WMA, Alabama. In National Conference on Feral Hogs (2008).
Mayer, J.J., Beasley, J.C., Boughton, R., Ditchkoff, S.S. Wild Pigs in the southeast, Invasive Wild Pigs in North America: Ecology, Impacts, and Management (2020).
Beasley, J. C., Grazia, T. E., Johns, P. E. & Mayer, J. J. Habitats associated with vehicle collisions with wild pigs. wilr 40, 654–660 (2014).
Keiter, D. A. et al. Effects of scale of movement, detection probability, and true population density on common methods of estimating population density. Sci. Rep. 7, 9446 (2017).
White, D.L. & Gaines, K.F. The savannah river site: Site description, land use and management history. 8–17 (2000).
Ellis, C. K. et al. Comparison of the efficacy of four drug combinations for immobilization of wild pigs. Eur. J. Wildl. Res. 65, 78 (2019).
Mayer, J.J., Smyser, T.J., Piaggio, A.J., & Zervanos, S.M. Wild pig taxonomy, morphology, genetics, and physiology, Invasive Wild Pigs in North America: Ecology, Impacts, and Management. 7–32 (2020).
Pohle, J., Langrock, R., van Beest, F. M. & Schmidt, N. M. Selecting the number of states in hidden markov models: Pragmatic solutions illustrated using animal movement. JABES 22, 270–293 (2017).
Kay, S.L., Fischer, J.W., Monaghan, A.J., Beasley, J.C., Boughton, R., Campbell, T.A., et al. Quantifying drivers of wild pig movement across multiple spatial and temporal scales. Mov. Ecol. 5 (2017).
Keuling, O., Stier, N. & Roth, M. Annual and seasonal space use of different age classes of female wild boar Sus scrofa L.. Eur. J. Wildl. Res. 54, 403–412 (2009).
Burnham, K. P. & Anderson, D. R. Model Selection and Multimodel Inference: A Practical Information-Theoretic Approach, Second (Springer, 2002).
R Core Team. R: A Language and Environment for Statistical Computing. (R Foundation for Statistical Computing, Vienna, 2019).
Jin, S. et al. Overall methodology design for the United States national land cover database 2016 products. Remote Sens. 11, 2–32 (2019).
Conner, L. M., Smith, M. D. & Burger, L. W. A comparison of distance-based and classification-base analyses of habitat use. Ecology 84, 526–531 (2003).
Benson, J. F. Improving rigour and efficiency of use-availability habitat selection analyses with systematic estimation of availability. Methods Ecol. Evol. 4, 244–251 (2013).
Calenge, C. The package adehabitat for the R software: A tool for the analysis of space and habitat use by animals. Ecol. Model. 197, 516–519 (2006).
Johnson, C. J., Nielsen, S. E., Merrill, E. H., McDonald, T. L. & Boyce, M. S. Resource selection functions based on use-availability data: Theoretical motivation and evaluation methods. J. Wildl. Manag. 70, 347–357 (2006).
Manly, B. F. J., McDonald, L. L., Thomas, D. L., McDonald, T. L. & Erickson, W. P. Resource Selection Functions from Logistic Regression, Resource Selection by Animals: Statistical Analysis and Design for Field Studies 83–110 (Kluwer Academic Publishers, 2002).
Kohl, M. T., Krausman, P. R., Kunkel, K. & Williams, D. M. Bison versus cattle: Are they ecologically synonymous?. Rangeland Ecol. Manag. 66, 721–731 (2013).
Bates, D., Mächler, M., Bolker, B., & Walker, S. Fitting linear mixed-effects models using lme4. arrXiv:14065823 [stat] (2014).
Fielding, A. H. & Bell, J. F. A review of methods for the assessment of prediction errors in conservation presence/absence models. Environ. Conserv. 24, 38–49 (1997).
Zipkin, E. F., Grant, E. H. C. & Fagan, W. F. Evaluating the predictive abilities of community occupancy models using AUC while accounting for imperfect detection. Ecol. Appl. 22, 1962–1972 (2012).
Latif, Q. S., Saab, V. A., Dudley, J. G., Markus, A. & Mellen-McLean, K. Development and evaluation of habitat suitability models for nesting white-headed woodpecker (Dryobates albolarvatus) in burned forest. PLoS ONE 15, e0233043 (2020).
Robin, X. et al. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 12, 77 (2011).
Gillies, C. S. et al. Application of random effects to the study of resource selection by animals. J. Anim. Ecol. 75, 887–898 (2006).
Karelus, D. L. et al. Incorporating movement patterns to discern habitat selection: Black bears as a case study. wilr 46, 76–88 (2019).
Franke, A., Caelli, T., Kuzyk, G. & Hudson, R. J. Prediction of wolf (Canis lupus) kill-sites using hidden Markov models. Ecol. Model. 197, 237–246 (2006).
van de Kerk, M. et al. Hidden semi-Markov models reveal multiphasic movement of the endangered Florida panther. J. Anim. Ecol. 84, 576–585 (2015).
Blasetti, A., Boitani, L., Riviello, M. C. & Visalberghi, E. Activity budgets and use of enclosed space by wild boars (Sus scrofa) in captivity. Zoo Biol. 7, 69–79 (1988).
Campbell, T. A. & Long, D. B. Activity patterns of wild boars (Sus scrofa) in southern Texas. Southwestern Nat. 55, 564–567 (2010).
Ilse LM and Hellgren EC, Resource Partitioning in Sympatric Populations of Collared Peccaries and Feral Hogs in Southern Texas, Journal of Mammalogy 76:784–799.
Snow, N. P., Miller, R. S., Beasley, J. C. & Pepin, K. M. Wild Pig Population Dynamics, Invasive Wild Pigs in North America: Ecology, Impacts, and Management 57–82 (CRC Press, 2020).
Matiuti, M., Bogdan, A.T., Crainiceanu, E., Matiuti, C. Research regarding the hybrids resulted from the domestic pig and the wild boar. Sci. Pap. 4 (2010).
Ditmer, M. A. et al. Moose at their bioclimatic edge alter their behavior based on weather, landscape, and predators. Curr. Zool. 64, 419–432 (2018).
Dexter, N. The influence of pasture distribution and temperature on habitat selection by feral pigs in a semi-arid environment. Wildl. Res. 25, 547–559 (1998).
Abrahms, B. et al. Lessons from integrating behaviour and resource selection: activity-specific responses of African wild dogs to roads. Anim. Conserv. 19, 247–255 (2016).
Ditchkoff, S. S. & Mayer, J. J. Wild Pig Food Habits, Wild Pigs: Biology, Damage, Control Techniques and Management 105–143 (Savannah River Nuclear Solutions LLC, 2009).
Ballari, S. A. & Barrios-García, M. N. A review of wild boar Sus scrofa diet and factors affecting food selection in native and introduced ranges. Mammal Rev. 44, 124–134 (2014).
Lewis, J. S., VerCauteren, K. C., Denkhaus, R. M. & Mayer, J. J. Wild Pig Populations Along the Urban Gradient, Invasive Wild Pigs in North America: Ecology, Impacts, and Management 439–464 (CRC Press, 2020).
Podgórski, T. et al. Spatiotemporal behavioral plasticity of wild boar (Sus scrofa) under contrasting conditions of human pressure: Primeval forest and metropolitan area. J. Mammal 94, 109–119 (2013).
Castillo-Contreras, R. et al. Urban wild boars prefer fragmented areas with food resources near natural corridors. Sci. Total Environ. 615, 282–288 (2018).
Brown, G. P., Phillips, B. L., Webb, J. K. & Shine, R. Toad on the road: use of roads as dispersal corridors by cane toads (Bufo marinus) at an invasion front in tropical Australia. Biol. Cons. 133, 88–94 (2006).
Thurfjell, H. et al. Habitat use and spatial patterns of wild boar Sus scrofa (L.): agricultural fields and edges. Eur. J. Wildl. Res. 55, 517–523 (2009).
Senior, A. M., Grueber, C. E., Machovsky-Capuska, G., Simpson, S. J. & Raubenheimer, D. Macronutritional consequences of food generalism in an invasive mammal, the wild boar. Mammal. Biol. 81, 523–526 (2016).
Lyons, P. C., Okuda, K., Hamilton, M. T., Hinton, T. G. & Beasley, J. C. Rewilding of Fukushima’s human evacuation zone. Front. Ecol. Environ. 18, 127–134 (2020).
Graves, H. B. Behavior and ecology of wild and feral swine (Sus Scrofa). J. Anim. Sci. 58, 482–492 (1984).
Dardaillon, M. Seasonal variations in habitat selection and spatial distribution of wild boar (Sus Scrofa) in the Camargue, Southern France. Behav. Proc. 13, 251–268 (1986).
Meriggi, A. & Sacchi, O. Habitat requirements of wild boars in the northern Apennines (N Italy): A multi-level approach. Ital. J. Zool. 68, 47–55 (2001).
Pepin, K. M., Snow, N. P. & VerCauteren, K. C. Optimal bait density for delivery of acute toxicants to vertebrate pests. J. Pest. Sci. 93, 723–735 (2020).
Acknowledgements
We thank the numerous field technicians, graduate students, and other research personnel including S. Chinn, P. Schlichting, and D. Keiter who assisted with this research, as well as the USFS for their assistance in the capture of wild pigs used in this study. Thanks also to M. Kohl for his insight on data analysis techniques. This material is based upon work supported by the U.S. Department of Energy under Award No. DE-EM0004391 to the UGA Research Foundation and the US Department of Agriculture’s Animal and Plant Health Inspection Service.
Funding
This material is based upon work supported by the US Department of Energy under Award No. DE-EM0004391 to the UGA Research Foundation and the US Department of Agriculture’s Animal and Plant Health Inspection Service.
Author information
Authors and Affiliations
Contributions
L.M.C. and J.C.B. conceived the study. L.M.C. wrote the paper with input from J.C.B., K.M.P., and K.C.V. L.M.C. carried out fieldwork and analyzed the data with input from K.M.P. J.C.B. and K.C.V. secured funding. All author(s) edited and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Clontz, L.M., Pepin, K.M., VerCauteren, K.C. et al. Behavioral state resource selection in invasive wild pigs in the Southeastern United States. Sci Rep 11, 6924 (2021). https://doi.org/10.1038/s41598-021-86363-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-86363-3
- Springer Nature Limited
This article is cited by
-
Movement behavior in a dominant ungulate underlies successful adjustment to a rapidly changing landscape following megafire
Movement Ecology (2024)
-
Fishers (Pekania pennanti) are forest structure specialists when resting and generalists when moving: behavior influences resource selection in a northern Rocky Mountain fisher population
Movement Ecology (2024)
-
What drives wild pig (Sus scrofa) movement in bottomland and upland forests?
Movement Ecology (2024)
-
Flexible hidden Markov models for behaviour-dependent habitat selection
Movement Ecology (2023)
-
Individual-level patterns of resource selection do not predict hotspots of contact
Movement Ecology (2023)