Abstract
Feline chronic enteropathy (CE) is a common gastrointestinal disorder in cats and mainly comprises inflammatory bowel disease (IBD) and small cell lymphoma (SCL). Differentiation between IBD and SCL can be diagnostically challenging. We characterized the fecal metabolome of 14 healthy cats and 22 cats with naturally occurring CE (11 cats with IBD and 11 cats with SCL). Principal component analysis and heat map analysis showed distinct clustering between cats with CE and healthy controls. Random forest classification revealed good group prediction for healthy cats and cats with CE, with an overall out-of-bag error rate of 16.7%. Univariate analysis indicated that levels of 84 compounds in cats with CE differed from those in healthy cats. Polyunsaturated fatty acids held discriminatory power in differentiating IBD from SCL. Metabolomic profiles of cats with CE resembled those in people with CE with significant alterations of metabolites related to tryptophan, arachidonic acid, and glutathione pathways.
Similar content being viewed by others
Introduction
Feline chronic enteropathy (CE) is a spontaneously arising disorder in cats that is especially common in the elderly cat population. It is defined as the chronic presence (i.e., presence for longer than 3 weeks) of signs of gastrointestinal disease such as weight loss, vomiting and/or diarrhea in the absence of infectious intestinal or extraintestinal causes1. The most common types of CE in cats are inflammatory bowel disease (IBD) and alimentary small cell lymphoma (SCL)2,3,4,5. Diagnosis and differentiation require invasive and expensive procedures, including the collection and histopathologic examination of intestinal tissue biopsies. Therapeutic strategies generally consist of treatment with immunosuppressive drugs such as corticosteroids for cases of IBD and or cytotoxic drugs such as chlorambucil for cases with SCL2,6,7. Hence, the discovery of less invasive biomarkers for the diagnosis and differentiation of CE in cats and the identification of new therapeutic targets would be highly desirable.
Studies in human subjects have revealed global metabolic changes in people with IBD and the potential to use metabolomic profiling for the diagnosis and differentiation of IBD and IBD subtypes such as Crohn’s disease and ulcerative colitis8,9,10,11,12,13. Common metabolic perturbations described included pathways affecting tryptophan and other amino acids, fatty acids, bile acids, sphingolipids, and biogenic amines8,9,10,11,12,13.
Although metabolomic profiles have been reported in plasma14,15 serum16 and urine17 of healthy cats, information from untargeted metabolomic studies in cats with CE are lacking.
Feline alimentary SCL shows some histological parallels to monomorphic epitheliotropic intestinal T-cell lymphoma (MEITL) in people (i.e., formally known as enteropathy associated T-cell lymphoma Type 2 (EATL Type 2)), such as a monomorphic infiltration of the intestinal mucosa with small to medium lymphocytes of predominantly T-cell origin, epitheliotropism, and a predilection for the small intestine4,18,19. However, while there are histopathological parallels, the clinical course of feline SCL and EATL Type 2 is very different. While alimentary SCL in cats is slowly progressing with a median survival time of 1.5 to 3.5 years20,21, MEITL in people is a clinically aggressive disease and generally associated with a poor prognosis18. Recently, other authors suggested that the cat might be a suitable model for indolent digestive T-cell lymphoproliferative disorder22,23. This rare disorder in people is characterized by a superficial monoclonal intestinal T-cell infiltrate and the disease is generally indolent or slowly progressive19,22,24,25,26,27.
We hypothesized that cats with CE have metabolomic perturbations and that metabolomic profiling can distinguish cats with IBD from cats with SCL. We further hypothesized that metabolic perturbances observed in cats with CE would be similar to those observed in humans with IBD.
Results
A total of 36 cats were enrolled into this study, 14 healthy cats and 22 cats with chronic enteropathy (11 with IBD and 11 with SCL). A fecal sample was collected from all cats. Demographic characteristics are shown in Table 1.
Age (p = 0.353) and sex distribution (p > 0.999) were comparable between control cats and cats with CE (Table 1). Cats with CE had a significantly lower body weight (p = 0.007) and body condition score28 (p < 0.001).
Cats with SCL were significantly older (median age: 12 years, range: 9–15 years) than cats with IBD (median age: 7 years, range: 2–16; p = 0.028). Cats with IBD and cats with SCL did not differ significantly regarding sex, body weight, or body condition score (BCS). Cats with CE had a median feline chronic enteropathy activity index (FCEAI)29 of 6 (range: 2–11). However, the FCEAI did not differ between cats with IBD (median: 6, range: 3–11) and cats with SCL (median: 5, range: 2–10; p = 0.176).
A total of 856 named metabolites were detected. Nonparametric univariate analysis using a FDR of 5% revealed a total of 84 metabolites that differed significantly between control cats and cats with CE (Table 2).
Principal component analysis (PCA), hierarchical clustering and a heatmap showing compounds that differed significantly between control cats and cats with CE indicated clustering of the two cohorts (Fig. 1a,b). Random forest classification revealed a group prediction, with a 16.7% out of bag (OOB) error rate (Table 3). The random forest importance plot identified 7 metabolites key in classifying the data with sphingomyelin (d18:1/14:0, d16:1/16:0), 3-(3-hydroxyphenyl)propionate, beta-cryptoxanthin, myristoleate (14:1n5), N1-methyl-4-pyridone-3-carboxamide, 2-oxindole-3-acetate, and 5-hydroxyindoleacetate, having the most influence on classification (Fig. 2).
Multivariate analysis of the fecal metabolome of healthy cats and cats with chronic enteropathy. (a) Heat map showing metabolites that were significantly different between healthy cats and cats with inflammatory bowel disease (IBD) and alimentary small cell lymphoma (SCL). Groups are represented by the colored bars at the top of the figure as red (healthy, n = 14), green (IBD, n = 11), and blue (SCL, n = 11). Clusters can be identified between healthy cats and cats with chronic enteropathy (CE) but not between the disease subgroups IBD and SCL. (b) PCA score plots of metabolites in feces from healthy cats (green) and cats with chronic enteropathy (CE, red). (c) PCA score plots of metabolites in feces from healthy cats (red), cats with inflammatory bowel disease (blue), and cats with alimentary small cell lymphoma (green). A cluster can be identified for healthy cats but no specific clusters can be seen for the subgroups of IBD and SCL. Data was mean centered and divided by the standard deviation of each variable (autoscaled).
Random Forest variable importance plot and scatter plots of the top seven metabolites with the highest importance for the model accuracy. (a) Random Forest variable importance plot. Random Forest is a supervised, machine learning algorithm used for regression (prediction) and classification analysis of large data sets. This Random Forest algorithm was based on the comparison of cats with chronic enteropathy (FCE) and healthy control cats. The model identifies features with the highest predictive accuracy for health status. The variable importance plots shows the significant features identified by Random Forest. The features are ranked by the mean decrease in classification accuracy when they are permuted. A higher value indicates the importance of that metabolite in predicting the group (healthy vs. chronic enteropathy). Data was mean centered and divided by the standard deviation of each variable (autoscaled). (b) Scatterplots of the top seven metabolites from (a).
Amongst biochemicals found to be increased in feces from cats with CE were various amino acids and their metabolites (e.g., aspartate, cysteine sulfinic acid, phenylalanine, leucine, and valine), fatty acids (e.g., arachidonate and eicosanoids), metabolites within vitamin E metabolism, and simple sphingolipids (e.g., ceramide, sphingomyelins, and sphingosines). Compounds significantly less abundant in cats with CE than in healthy controls included those involved in the tryptophan (i.e., indole-derivates) and vitamin A metabolism (i.e., carotendiols), and sterols.
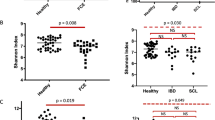
Upon analysis of the CE subgroups, IBD and SCL, multivariate analysis revealed no visible clustering between cats with IBD and cats with SCL (Fig. 1a,c). Random forest analysis revealed poor group prediction with an OOB of 47.2% and class error rates of 82% and 55% for cats with IBD and cats with SCL, respectively. However, univariate analysis revealed 18 metabolites to differ significantly between healthy cats, cats with IBD, and cats with SCL. Post hoc analysis showed that 3 polyunsaturated fatty acids (i.e., eicosapentaenoate, heneicosapentaenoate, and stearidonate) within the eicosanoid family differed between cats with IBD and cats with SCL (Table 4).
Discussion
Our study revealed global metabolic changes in cats with chronic enteropathy as compared with healthy controls, with many metabolic pathways affected. Both hierarchical cluster analysis and PCA demonstrated clustering among cats diagnosed with chronic enteropathy and healthy control cats. Random forest analysis revealed a good class prediction of 80.6%. Univariate analysis showed a total of 84 metabolites to differ significantly between the two groups after controlling for a 5% FDR. Similar studies in humans with inflammatory bowel disease have also shown significant metabolomic differences between affected patients and healthy controls10,11,12,30. Although IBD in humans and CE in cats share only a few characteristics, the use of metabolomics data in humans with chronic enteropathies would support this type of approach in further assessing cats with various forms of CE. In addition, metabolic consequences of feline CE and human IBD appear to be very similar. Metabolites or metabolite families commonly found to be affected by IBD in humans are amino acids8,10,11,12,13,30, bile acids10, fatty acids10,11, and metabolites of the tryptophan pathway10,31.
We found multiple amino acids to be increased in the feces from cats with CE, indicating malabsorption, likely as consequence of mucosal inflammation, neoplastic infiltration, or both. Interestingly, 2-hydroxybutyrate/2-hydroxyisobutyrate and gamma-glutamylglutamine, two metabolites related to the glutathione-metabolism, were found to be significantly more abundant in feces from cats with CE based on both univariate and multivariate analysis. Gamma-glutamyl amino acids are precursors for the formation of glutathione in the gamma-glutamyl cycle (glutathione salvage pathway)32 (Fig. 3). Reactive oxygen species have been implicated to contribute to tissue injury in patients with Crohn’s disease and ulcerative colits33,34. Glutathione is the major intracellular antioxidant and thus a critical part of the defense mechanism against oxidative stress in inflammatory conditions such as CE (Fig. 3)35,36. Consequently, glutathione precursors are in high demand during catabolic conditions and corresponding higher loads of oxidative stress30. Increased fecal concentrations of gamma-glutamyl glutamine in cats with CE might indicate increased loss and correspond to a decreased mucosal availability and glutathione synthesis.
Our study showed arachidonate to be more abundant in feces from cats with CE. Arachidonate is a well described mediator of inflammation, including intestinal inflammation, and a precursor for prostaglandins, which are essential immune signaling molecules37. In addition, arachidonate has been found to increase the expression of intercellular adhesion molecule 1 (ICAM-1)38. Adhesion molecules such as ICAM-1 and mucosal addressin cell adhesion molecule (MAdCAM) are involved in recruiting leucocytes to the site of inflammation and have been identified as therapeutic targets for human patients with IBD39. Increased fecal arachidonate may reflect mucosal upregulation during inflammation and subsequent leakage into the fecal stream. On the other hand, omega-3 polyunsaturated fatty acids, including eicosapentaenoate, have been found to be significantly more abundant in the SCL subgroup of cats with CE compared to healthy controls.
Eicosapentanoate and other omega-3 fatty acids have anti-inflammatory properties and inhibit conversion of arachidonate into the pro-inflammatory thromboxane-2 and prostaglandin-2 families40. Increased fecal levels of omega-3 fatty acids might indicate an increased loss or malabsorption and thus may correspond to the increased arachidonate concentration found in this study.
We also found several metabolites of the indole family and pathway to be altered in cats with CE. Amongst a wide range of structurally divergent exo- and endogenic chemicals41, indole derivates are important ligands for the aryl hydrocarbon receptor (AhR)42. AhR signaling participates integrally in intestinal mucosal homeostasis by acting on innate and adaptive immune cells as well as on epithelial renewal and mucosal barrier function43. Tryptophan plays a central role in AhR activation because it is transformed into indole and indole derivates by the gut microbiota. Studies in murine and porcine models of colitis have found significantly reduced disease activities after oral supplementation with indole-3-propionate or L-tryptophan by activating anti-inflammatory pathways mediated by IL-10 and IL-2242,44. We found indole derivates, such as 2-oxindole-3-acetate and 5-hydroxyindoleacetate, to be significantly less abundant in feces from cats with CE. Although, tryptophan did not significantly differ between CE and control cats, it showed a trend (p = 0.0855, q = 0.2385) towards a higher fecal excretion in cats with CE42,45,46 (Appendix C, Tables 5.5, 5.6). These findings might indicate disrupted transformation of tryptophan into indole derivates, possibly linked to intestinal dysbiosis in this cohort of cats with CE.
Finally, our study showed increased concentrations of several simple sphingolipids in feces from cats with CE. The role of sphingolipids in IBD is complex and incompletely understood. It appears, however, that a functional equilibrium between simple sphingolipids (e.g., sphingomyelin, sphingosine, ceramide) and complex sphingolipids (e.g., gangliosides GM3 and GD3) is essential in maintaining intestinal homeostasis47. Proinflammatory sphingolipid patterns have previously been described, with increased concentrations of sphingomyelin and ceramide in feces of animal models of colitis48,49 as well as in the ileal mucus of human patients with Crohn’s disease50 indicating that sphingomyelin and ceramide accompany and possibly aggravate chronic intestinal inflammation.
Fecal extracts obtained from cats with IBD and SCL revealed similar global changes in metabolic profiles. However, changes observed in cats with IBD were less pronounced than those observed in cats with SCL. This suggests that metabolic consequences are more severe in SCL than in IBD. In particular, higher concentrations of polyunsaturated fatty acids of the eicosanoid family were found in SCL.
This study had several limitations. A clinical diagnosis of IBD was based on the presence of chronic gastrointestinal signs of at least 3 weeks duration, the absence of known enteropathogens or other causes of signs of gastrointestinal disease, and the histopathologic confirmation of intestinal inflammation51. Hence, we did not exclude food-responsive enteropathy in all participating cats. However, in response to dietary interventions human IBD patients can show complete or partial resolution of clinical signs without being reclassified as having food-responsive enteropathy52,53,54,55. Fecal cultures for specific pathogens were not performed. However, gastrointestinal disease caused by bacteria usually cause acute diarrhea. In addition, all cats showed either lymphoplasmacytic enteritis or lymphoma on histopathologic examination. Cats with neutrophilic enteritis, a hallmark of gastrointestinal bacterial infections, were excluded from the study. Differentiating IBD from SCL can impose significant challenges with clinical signs and histopathologic changes commonly overlapping3. Moreover, mucosal changes may have an irregularly distribution, inflammatory and neoplastic lesions often coincide and inflammatory lesions might even progress to lymphoma over time3,4. Therefore, using currently available measures, the classification into groups of IBD and SCL will always be associated with a degree of ambiguity. However, in all cats intestinal biopsies were available and results of immunohistochemistry and clonality testing were utilized, where indicated, to provide the highest accuracy in category assignment.
Inter-individual variation of metabolomic profiles have been described in humans, dogs, and cats and might be related to a variety of exogenous and endogenous factors such as the environment, diet, gut microbiota, xenobiotics, and the genome14,17,56. This study intended to identify clinically relevant perturbations of the fecal metabolome of cats with CE compared to healthy subjects. Demographic characteristics like age, sex, and breed were controlled for in this study. However, we did not control for environmental factors, such as diet and housing, since this likely would have impacted the clinical relevance of our results. This concept is supported by recent studies showed that standardization in is a major source of poor reproducibility preclinical trials57,58. Although, we cannot exclude that these factors confounded our results, most are in line with findings across different species with spontaneous or induced IBD and thus likely reflect true changes of the fecal metabolome. In addition, most cats in both groups were housed indoors and fed a variety of different commercial diets. Another limitation is the limited number of animals in this study. However, untargeted “omics” analyses usually provide only relative changes between subjects or groups and are generally conducted using a smaller sample size. Therefore, all untargeted analyses have to be followed by targeted assays to confirm results of those fishing expeditions on a larger number of subjects. However, this was beyond the scope of this study.
In summary, our study revealed global metabolic changes in cats with CE compared to healthy controls. Many metabolic pathways were affected such as amino acids, fatty acids (e.g., arachidonic acid and eicosanoids), sphingolipids, and metabolites of the tryptophan pathway (i.e., indole-derivates). Metabolic profiles resembled patterns found in humans and other animal models with IBD and thus metabolic consequences of feline CE and human IBD might be similar. Future studies of the mucosal and serum metabolome of cats with CE should be conducted to further elucidate the origin of metabolic perturbations and allow further insights into pathogenesis. In addition, targeted analysis of the compounds found to be altered in cats with CE is indicated to confirm results and to investigate their value as non-invasive biomarkers.
Materials and methods
Study approval and enrollment
This prospective study was conducted at the Veterinary Medical Teaching Hospital at Texas A&M University between May 2015 and September 2017. The study protocol was approved by the Texas A&M University Institutional Animal Care and Use Committee (IACUC 2015-0276 CA and IACUC 2014-0369 CA). All experiments were performed in accordance with relevant guidelines and regulations. Cat owners provided written informed consent prior to study enrollment. Cats with clinical signs of chronic enteropathy (n = 22) and control cats (n = 14) were recruited from the hospital population at the Small Animal Hospital of Texas A&M University in College Station, Texas or the Veterinary Specialty Hospital in San Diego, California59.
The health status of cats in the group considered healthy was verified by an owner questionnaire on general and gastrointestinal health59. The questionnaire covered the following areas: attitude/activity, appetite, drinking, urination, chronic illnesses, weight loss, vomiting, diarrhea, and treatment with antibiotics, antacids, anti-inflammatory drugs, or steroids59. In addition, a physical examination and blood testing was available and performed by a single board-certified internist (SM) in 11 of 14 cats. The body condition score was assessed using a previously established a nine-point condition scoring system28. Blood was collected from a peripheral vein or the jugular vein and the following tests were performed: complete blood count, serum chemistry profile, total T4, cobalamin, folate, feline pancreatic lipase immunoreactivity (fPLI), and feline trypsin-like immunoreactivity (fTLI). Cats with gastrointestinal signs (weight loss, hyporexia, vomiting > 2×/ month, diarrhea) within 6 months prior to enrollment were excluded. In addition, cats with systemic diseases, chronic illnesses or clinically significant laboratory abnormalities were excluded from the study59. Finally, cats that had received any antibiotics, antacids, anti-inflammatory drugs, or corticosteroids within the past 6 months were excluded59.
Cats with clinical signs of chronic enteropathy (weight loss, hyporexia, vomiting, diarrhea) of at least 3 weeks duration were eligible for enrollment into the group of cats with chronic enteropathy59. Extra-gastrointestinal disease as well and, where indicated, infectious intestinal diseases were excluded based on a complete blood count, serum chemistry profile, total T4 and fecal flotation59. All cats in this group underwent gastro-duodenoscopy and ileo-colonoscopy for diagnostic purposes. Histopathologic examination of H&E stained endoscopic formalin-fixed, paraffin-embedded (FFPE) tissue sections was performed by board-certified pathologists (MA or JSE) blinded to the clinical status of the cats59.
Cases with a histopathological diagnosis of SCL or cases where the pathologist was suspicious of an underlying SCL underwent additional diagnostic testing with immunohistochemistry and PCR for antigen receptor rearrangement testing for diagnostic confirmation. A final diagnosis of IBD or SCL was reached upon integration of results from histopathology, immunohistochemistry, and PARR based on the current EuroClonality/BIOMED-2 guidelines for interpretation and reporting of Ig/TCR clonality testing in suspected lymphoproliferations59,60,61,62. Cats that had received antibiotics within 4 weeks or corticosteroids within the past 2 weeks prior to fecal sampling were excluded from the study59.
Sample collection
Fecal samples were collected after spontaneous defecation or digitally while the cat was under anesthesia and prepared for endoscopy. Fecal samples were refrigerated or frozen immediately after collection and shipped to the Gastrointestinal Laboratory at Texas A&M University on ice packs or dry ice within 24 h. Upon arrival, fecal samples were immediately divided into alliquots and stored at − 80 °C until analysis59.
Metabolite extraction
Untargeted fecal metabolomic analysis was performed by Metabolon, Inc. (Durham, NC) as previously described63,64,65. Metabolites were extracted from lyophilized and homogenized samples using methanol extraction. Extracts were analyzed by an ACQUITY ultra-performance liquid chromatographer (Waters, Milford, CA) and a ThermoFisher Scientific Q-Exactive high resolution mass spectrometer interfaced with a heated electrospray ionization (HESI-II) source and Orbitrap mass analyzer (ThermoFisher Scientific, Waltham, Massachusetts) operated at 35,000 mass resolution. The scan range covered 70–1000 m/z. Metabolite identification was performed by automated comparison of the ion features in the experimental samples to a reference library. Chemical standard entries included retention time, molecular weight (m/z), preferred adducts, and in-source fragments as well as associated MS spectra, and were curated by visual inspection for quality control using software developed at Metabolon. Peaks were quantified using the area-under-the-curve. Compounds were corrected for inter-day variation by registering the medians to equal 1.00 and normalizing each data point proportionally. Missing values were imputed with the observed minimum for each compound.
Statistical analyses
Patient demographics were compared by the Mann–Whitney or Fisher’s exact test as appropriate. Differences in the abundance of fecal metabolites between control cats and cats with CE were evaluated using a Mann–Whitney test. A post hoc analysis to assess differences in the abundance of metabolites between the subgroups (i.e., controls, IBD, SCL) was performed by Dunn’s test. Statistical significance was set at p < 0.05. Results were adjusted by False Discovery Rate (FDR) and reported as the q-value where appropriate. Univariate analysis was performed using Prism 7.0b (Graph Pad Software, La Jolla, CA) and JMP Pro 14.1.0 (SAS Institute Inc., Cary, NC).
Multivariate analysis was performed using MetaboAnalyst66. Data was mean centered and divided by the standard deviation of each variable. PCA and hierarchical clustering was performed and a heatmap was created as a visual aid for the dendrogram. Random forest regression analysis was used to evaluate the classification performance of metabolomics.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors.
Research involving animal rights
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Jergens, A. E. et al. A scoring index for disease activity in canine inflammatory bowel disease. J. Vet. Intern. Med. 17, 291–297. https://doi.org/10.1111/j.1939-1676.2003.tb02450.x (2003).
Jergens, A. E. Feline idiopathic inflammatory bowel disease: What we know and what remains to be unraveled. J. Feline Med. Surg. 14, 445–458. https://doi.org/10.1177/1098612X12451548 (2012).
Sabattini, S. et al. Differentiating feline inflammatory bowel disease from alimentary lymphoma in duodenal endoscopic biopsies. J. Small Anim. Pract. 57, 396–401. https://doi.org/10.1111/jsap.12494 (2016).
Moore, P. F., Rodriguez-Bertos, A. & Kass, P. H. Feline gastrointestinal lymphoma: Mucosal architecture, immunophenotype, and molecular clonality. Vet. Pathol. 49, 658–668. https://doi.org/10.1177/0300985811404712 (2012).
Kiupel, M. et al. Diagnostic algorithm to differentiate lymphoma from inflammation in feline small intestinal biopsy samples. Vet. Pathol. 48, 212–222. https://doi.org/10.1177/0300985810389479 (2011).
Stein, T. J., Pellin, M., Steinberg, H. & Chun, R. Treatment of feline gastrointestinal small-cell lymphoma with chlorambucil and glucocorticoids. J. Am. Anim. Hosp. Assoc. 46, 413–417. https://doi.org/10.5326/0460413 (2010).
Kiselow, M. A. et al. Outcome of cats with low-grade lymphocytic lymphoma: 41 cases (1995–2005). J. Am. Vet. Med. Assoc. 232, 405–410. https://doi.org/10.2460/javma.232.3.405 (2008).
Santoru, M. L. et al. Cross sectional evaluation of the gut-microbiome metabolome axis in an Italian cohort of IBD patients. Sci. Rep. 7, 9523. https://doi.org/10.1038/s41598-017-10034-5 (2017).
Kolho, K. L., Pessia, A., Jaakkola, T., de Vos, W. M. & Velagapudi, V. Faecal and serum metabolomics in paediatric inflammatory bowel disease. J. Crohns Colitis. 11, 321–334. https://doi.org/10.1093/ecco-jcc/jjw158 (2017).
Jansson, J. et al. Metabolomics reveals metabolic biomarkers of Crohn’s disease. PLoS ONE 4, e6386. https://doi.org/10.1371/journal.pone.0006386 (2009).
Bjerrum, J. T. et al. Metabonomics of human fecal extracts characterize ulcerative colitis Crohn’s disease and healthy individuals. Metabolomics 11, 122–133. https://doi.org/10.1007/s11306-014-0677-3 (2015).
Le Gall, G. et al. Metabolomics of fecal extracts detects altered metabolic activity of gut microbiota in ulcerative colitis and irritable bowel syndrome. J. Proteome Res. 10, 4208–4218. https://doi.org/10.1021/pr2003598 (2011).
Marchesi, J. R. et al. Rapid and noninvasive metabonomic characterization of inflammatory bowel disease. J. Proteome Res. 6, 546–551. https://doi.org/10.1021/pr060470d (2007).
Colyer, A., Gilham, M. S., Kamlage, B., Rein, D. & Allaway, D. Identification of intra- and inter-individual metabolite variation in plasma metabolite profiles of cats and dogs. Br. J. Nutr. 106(Suppl 1), S146-149. https://doi.org/10.1017/S000711451100081X (2011).
Hall, J. A., Jackson, M. I., Vondran, J. C., Vanchina, M. A. & Jewell, D. E. Comparison of circulating metabolite concentrations in dogs and cats when allowed to freely choose macronutrient intake. Biol. Open https://doi.org/10.1242/bio.036228 (2018).
Allaway, D. et al. Metabolic profiling reveals effects of age, sexual development and neutering in plasma of young male cats. PLoS ONE 11, e0168144. https://doi.org/10.1371/journal.pone.0168144 (2016).
Rivera-Velez, S. M. & Villarino, N. F. Feline urine metabolomic signature: Characterization of low-molecular-weight substances in urine from domestic cats. J. Feline Med. Surg. 20, 155–163. https://doi.org/10.1177/1098612X17701010 (2018).
Delabie, J. et al. Enteropathy-associated T-cell lymphoma: Clinical and histological findings from the international peripheral T-cell lymphoma project. Blood 118, 148–155. https://doi.org/10.1182/blood-2011-02-335216 (2011).
Swerdlow, S. H. et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood 127, 2375–2390. https://doi.org/10.1182/blood-2016-01-643569 (2016).
Lingard, A. E. et al. Low-grade alimentary lymphoma: clinicopathological findings and response to treatment in 17 cases. J. Feline Med. Surg. 11, 692–700. https://doi.org/10.1016/j.jfms.2009.05.021 (2009).
Pope, K. V., Tun, A. E., McNeill, C. J., Brown, D. C. & Krick, E. L. Outcome and toxicity assessment of feline small cell lymphoma: 56 cases (2000–2010). Vet. Med. Sci. 1, 51–62. https://doi.org/10.1002/vms3.9 (2015).
Paulin, M. V. et al. Feline low-grade alimentary lymphoma: An emerging entity and a potential animal model for human disease. BMC Vet. Res. 14, 306. https://doi.org/10.1186/s12917-018-1635-5 (2018).
Freiche, V. et al. Feline low-grade intestinal T cell lymphoma: a unique natural model of human indolent T cell lymphoproliferative disorder of the gastrointestinal tract. Report No. 1530–0307, (Nature Publishing Group, 2021).
Nijeboer, P. et al. Treatment response in enteropathy associated T-cell lymphoma; survival in a large multicenter cohort. Am. J. Hematol. 90, 493–498. https://doi.org/10.1002/ajh.23992 (2015).
Ondrejka, S. & Jagadeesh, D. Enteropathy-associated T-cell lymphoma. Curr. Hematol. Malig. Rep. 11, 504–513. https://doi.org/10.1007/s11899-016-0357-7 (2016).
Said, J. T-cell LPD of the GIT: First do no harm. Blood 122, 3548–3550. https://doi.org/10.1182/blood-2013-09-526368 (2013).
Matnani, R. et al. Indolent T- and NK-cell lymphoproliferative disorders of the gastrointestinal tract: A review and update. Hematol. Oncol. 35, 3–16. https://doi.org/10.1002/hon.2317 (2017).
Bjornvad, C. R. et al. Evaluation of a nine-point body condition scoring system in physically inactive pet cats. Am. J. Vet. Res. 72, 433–437. https://doi.org/10.2460/ajvr.72.4.433 (2011).
Jergens, A. E. et al. A clinical index for disease activity in cats with chronic enteropathy. J. Vet. Intern. Med. 24, 1027–1033. https://doi.org/10.1111/j.1939-1676.2010.0549.x (2010).
Bjerrum, J. T. et al. Metabonomics in ulcerative colitis: Diagnostics, biomarker identification, and insight into the pathophysiology. J. Proteome Res. 9, 954–962. https://doi.org/10.1021/pr9008223 (2010).
Nikolaus, S. et al. Increased tryptophan metabolism is associated with activity of inflammatory bowel diseases. Gastroenterology 153, 1504. https://doi.org/10.1053/j.gastro.2017.08.028 (2017).
Priolo, C. et al. Impairment of gamma-glutamyl transferase 1 activity in the metabolic pathogenesis of chromophobe renal cell carcinoma. Proc. Natl. Acad. Sci. U S A 115, E6274–E6282. https://doi.org/10.1073/pnas.1710849115 (2018).
Keshavarzian, A. et al. Excessive production of reactive oxygen metabolites by inflamed colon: analysis by chemiluminescence probe. Gastroenterology 103, 177–185. https://doi.org/10.1016/0016-5085(92)91111-g (1992).
Simmonds, N. J. et al. Chemiluminescence assay of mucosal reactive oxygen metabolites in inflammatory bowel disease. Gastroenterology 103, 186–196. https://doi.org/10.1016/0016-5085(92)91112-h (1992).
Jones, D. P. Redox potential of GSH/GSSG couple: Assay and biological significance. Methods Enzymol. 348, 93–112. https://doi.org/10.1016/s0076-6879(02)48630-2 (2002).
Sido, B. et al. Impairment of intestinal glutathione synthesis in patients with inflammatory bowel disease. Gut 42, 485–492. https://doi.org/10.1136/gut.42.4.485 (1998).
Morita, H., Nakanishi, K., Dohi, T., Yasugi, E. & Oshima, M. Phospholipid turnover in the inflamed intestinal mucosa: arachidonic acid-rich phosphatidyl/plasmenyl-ethanolamine in the mucosa in inflammatory bowel disease. J. Gastroenterol. 34, 46–53. https://doi.org/10.1007/s005350050215 (1999).
Ramakers, J. D., Mensink, R. P., Schaart, G. & Plat, J. Arachidonic acid but not eicosapentaenoic acid (EPA) and oleic acid activates NF-kappaB and elevates ICAM-1 expression in Caco-2 cells. Lipids 42, 687–698 (2007).
Reinisch, W., Hung, K., Hassan-Zahraee, M. & Cataldi, F. Targeting endothelial ligands: ICAM-1/alicaforsen, MAdCAM-1. J. Crohns. Colitis. 12, S669–S677. https://doi.org/10.1093/ecco-jcc/jjy059 (2018).
Calder, P. C. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology?. Br. J. Clin. Pharmacol. 75, 645–662. https://doi.org/10.1111/j.1365-2125.2012.04374.x (2013).
Denison, M. S. & Nagy, S. R. Activation of the aryl hydrocarbon receptor by structurally diverse exogenous and endogenous chemicals. Annu. Rev. Pharmacol. Toxicol. 43, 309–334. https://doi.org/10.1146/annurev.pharmtox.43.100901.135828 (2003).
Alexeev, E. E. et al. Microbiota-derived indole metabolites promote human and murine intestinal homeostasis through regulation of interleukin-10 receptor. Am. J. Pathol. 188, 1183–1194. https://doi.org/10.1016/j.ajpath.2018.01.011 (2018).
Lamas, B., Natividad, J. M. & Sokol, H. Aryl hydrocarbon receptor and intestinal immunity. Mucosal. Immunol. 11, 1024–1038. https://doi.org/10.1038/s41385-018-0019-2 (2018).
Kim, C. J. et al. l-Tryptophan exhibits therapeutic function in a porcine model of dextran sodium sulfate (DSS)-induced colitis. J. Nutr. Biochem. 21, 468–475. https://doi.org/10.1016/j.jnutbio.2009.01.019 (2010).
Jacobs, J. P. et al. A disease-associated microbial and metabolomics state in relatives of pediatric inflammatory bowel disease patients. Cell. Mol. Gastroenterol. Hepatol. 2, 750–766. https://doi.org/10.1016/j.jcmgh.2016.06.004 (2016).
Lamas, B. et al. CARD9 impacts colitis by altering gut microbiota metabolism of tryptophan into aryl hydrocarbon receptor ligands. Nat. Med. 22, 598. https://doi.org/10.1038/nm.4102 (2016).
Abdel Hadi, L., Di Vito, C. & Riboni, L. Fostering inflammatory bowel disease: Sphingolipid strategies to join forces. Mediat. Inflamm. 2016, 3827684. https://doi.org/10.1155/2016/3827684 (2016).
Qi, Y. et al. PPARalpha-dependent exacerbation of experimental colitis by the hypolipidemic drug fenofibrate. Am. J. Physiol. Gastrointest. Liver Physiol. 307, G564-573. https://doi.org/10.1152/ajpgi.00153.2014 (2014).
Fischbeck, A. et al. Sphingomyelin induces cathepsin D-mediated apoptosis in intestinal epithelial cells and increases inflammation in DSS colitis. Gut 60, 55–65. https://doi.org/10.1136/gut.2009.201988 (2011).
Braun, A. et al. Alterations of phospholipid concentration and species composition of the intestinal mucus barrier in ulcerative colitis: A clue to pathogenesis. Inflamm. Bowel. Dis. 15, 1705–1720. https://doi.org/10.1002/ibd.20993 (2009).
Jergens, A. E. & Simpson, K. W. Inflammatory bowel disease in veterinary medicine. Front. Biosci. (Elite Ed) 4, 1404–1419. https://doi.org/10.2741/470 (2012).
Riordan, A. M. et al. Treatment of active Crohn’s disease by exclusion diet: East Anglian multicentre controlled trial. Lancet 342, 1131–1134. https://doi.org/10.1016/0140-6736(93)92121-9 (1993).
Bartel, G. et al. Ingested matter affects intestinal lesions in Crohn’s disease. Inflamm. Bowel Dis. 14, 374–382. https://doi.org/10.1002/ibd.20295 (2008).
Jones, V. A. et al. Crohn’s disease: Maintenance of remission by diet. Lancet 2, 177–180. https://doi.org/10.1016/s0140-6736(85)91497-7 (1985).
Chiba, M. et al. Lifestyle-related disease in Crohn’s disease: Relapse prevention by a semi-vegetarian diet. World J. Gastroenterol. 16, 2484–2495. https://doi.org/10.3748/wjg.v16.i20.2484 (2010).
Vuckovic, D. Improving metabolome coverage and data quality: Advancing metabolomics and lipidomics for biomarker discovery. Chem. Commun. (Camb) 54, 6728–6749. https://doi.org/10.1039/c8cc02592d (2018).
Voelkl, B., Vogt, L., Sena, E. S. & Wurbel, H. Reproducibility of preclinical animal research improves with heterogeneity of study samples. PLoS Biol. 16, e2003693. https://doi.org/10.1371/journal.pbio.2003693 (2018).
Karp, N. A. Reproducible preclinical research-Is embracing variability the answer?. PLoS Biol. 16, e2005413. https://doi.org/10.1371/journal.pbio.2005413 (2018).
Marsilio, S. et al. Characterization of the fecal microbiome in cats with inflammatory bowel disease or alimentary small cell lymphoma. Sci. Rep. 9, 19208. https://doi.org/10.1038/s41598-019-55691-w (2019).
Keller, S. M., Vernau, W. & Moore, P. F. Clonality testing in veterinary medicine: A review with diagnostic guidelines. Vet. Pathol. 53, 711–725. https://doi.org/10.1177/0300985815626576 (2016).
van Dongen, J. J. et al. Design and standardization of PCR primers and protocols for detection of clonal immunoglobulin and T-cell receptor gene recombinations in suspect lymphoproliferations: Report of the BIOMED-2 Concerted Action BMH4-CT98-3936. Leukemia 17, 2257–2317. https://doi.org/10.1038/sj.leu.2403202 (2003).
Langerak, A. W. et al. EuroClonality/BIOMED-2 guidelines for interpretation and reporting of Ig/TCR clonality testing in suspected lymphoproliferations. Leukemia 26, 2159–2171. https://doi.org/10.1038/leu.2012.246 (2012).
Jump, R. L., Kraft, D., Hurless, K., Polinkovsky, A. & Donskey, C. J. Impact of tigecycline versus other antibiotics on the fecal metabolome and on colonization resistance to clostridium difficile in mice. Pathog. Immun. 2, 1–20. https://doi.org/10.20411/pai.v2i1.159 (2017).
DeHaven, C. D., Evans, A. M., Dai, H. P. & Lawton, K. A. Organization of GC/MS and LC/MS metabolomics data into chemical libraries. J. Cheminform 2, 9. https://doi.org/10.1186/1758-2946-2-9 (2010).
Evans, A. M., DeHaven, C. D., Barrett, T., Mitchell, M. & Milgram, E. Integrated, nontargeted ultrahigh performance liquid chromatography/electrospray ionization tandem mass spectrometry platform for the identification and relative quantification of the small-molecule complement of biological systems. Anal. Chem. 81, 6656–6667. https://doi.org/10.1021/ac901536h (2009).
Xia, J. & Wishart, D. S. Web-based inference of biological patterns, functions and pathways from metabolomic data using MetaboAnalyst. Nat. Protoc. 6, 743–760. https://doi.org/10.1038/nprot.2011.319 (2011).
Acknowledgements
Financial support for this study was provided by the WINN Feline Foundation (MT16-018). The authors thank Jacqueline Sedillo for her help with handling samples and processing data.
Author information
Authors and Affiliations
Contributions
S.M. designed the study, examined a subset of cats, collected and aliquoted the feces, performed data collection and analysis, prepared all figures and wrote the manuscript. B.C. and S.H. examined a subset of cats, collected feces, and gave input into manuscript writing and review. M.R.A. and J.S.E. reviewed the tissue biopsy sections and assisted in the disease classification of the cases. B.S. assisted with data analysis, assisted with creating data Tables 2 and 4 and created Fig. 3. R.P. assisted with data analysis and gave input into manuscript writing and review. J.A.L., J.M.S., J.S.S. assisted with the data analysis and gave input into manuscript writing and review. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors Drs. Marsilio, Pilla, Lidbury, Suchodolski, and Steiner and the author Sarawichitr were or are currently employed by the Gastrointestinal Laboratory at Texas A&M University, which offers laboratory tests, including histopathology services, on a fee-for-service basis. Dr. Ackermann is affiliated with the Gastrointestinal Laboratory at Texas A&M University, which offers laboratory tests, including histopathology services, on a fee-for-service basis. Dr. Estep is employed at Texas Veterinary pathology, LLC, which offers histopathology services on a fee-for-service basis. Drs. Chow and Hill have nothing to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Marsilio, S., Chow, B., Hill, S.L. et al. Untargeted metabolomic analysis in cats with naturally occurring inflammatory bowel disease and alimentary small cell lymphoma. Sci Rep 11, 9198 (2021). https://doi.org/10.1038/s41598-021-88707-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-88707-5
- Springer Nature Limited
This article is cited by
-
Unbiased serum metabolomic analysis in cats with naturally occurring chronic enteropathies before and after medical intervention
Scientific Reports (2024)
-
The effects of a hydrolyzed protein diet on the plasma, fecal and urine metabolome in cats with chronic enteropathy
Scientific Reports (2023)
-
Microbiome function underpins the efficacy of a fiber-supplemented dietary intervention in dogs with chronic large bowel diarrhea
BMC Veterinary Research (2022)