Abstract
Transurethral resection of bladder tumor (TUR-BT) and radical cystectomy (RC) are standard treatment options for bladder cancer (BC). Neoadjuvant chemotherapy (NAC) prior to RC improves outcome of some patients but currently there are no valid biomarkers to identify patients who benefit from NAC. Presence of cancer stem cells (CSC) has been associated with poor outcome and resistance to chemotherapy in various cancers. Here we studied the expression of stem cell markers ALDH1, SOX2 and SSEA-4 with immunohistochemistry in tissue microarray material consisting of 195 BC patients treated with RC and 74 patients treated with TUR-BT followed by NAC and RC. Post-operative follow-up data of up to 22 years was used. Negative to weak cytoplasmic SOX2 staining was associated with lymphovascular invasion and non-organ confined disease. It was also associated with shortened cancer-specific survival, but the finding was not statistically significant. Contrary to previous reports, none of the other tested biomarkers were associated with cancer-specific mortality or clinicopathological characteristics. Neither were they associated with response to NAC. Despite the promising results of previously published studies, our results suggest that CSC markers ALDH1, SOX2 and SSEA-4 have little if any prognostic or predictive value in BC treated with RC.
Similar content being viewed by others
Introduction
Bladder cancer (BC) is the 10th most common cancer worldwide with estimated 549,000 new cases and 200,000 deaths annually1. The most important risk factors for BC include tobacco use, aging, male gender, and exposure to certain chemicals2.
Non-muscle invasive bladder cancer (NMIBC, stage pTa-T1 tumors) is routinely treated with transurethral resection of bladder tumor (TUR-BT) and various intravesical treatments. However, up to 78% of these tumors recur3. For muscle-invasive bladder cancer (MIBC, stage T2 or higher) radical cystectomy (RC) with pelvic lymphadenectomy and cisplatin-based neoadjuvant chemotherapy (NAC) are preferred when applicable. NAC improves 5-year progression free and overall survival in locally invasive disease by 8%2, but to date there are no immunohistochemical or other biomarkers to distinguish aggressive tumors from non-aggressive tumors, or chemotherapy responders from non-responders. Therefore, there is an unmet need of novel tools for patient stratification and selection.
Cancer stem cell theory dictates that similar to normal tissues, cancer tissue has a subpopulation of cancer stem cells (CSCs) or tumor-initiating cells that drive the proliferation of the tumor and initiate metastasis4,5,6,7. Several molecular biomarkers have been used to identify CSCs. However, many gene products are associated with CSC-like phenotype and none of them is universal for all cancer types. Sox2, a family member of sex determining region Y (SRY) -box genes, is a key regulator of pluripotency in stem cells8,9,10 and has been linked to poor survival in various malignancies including BC11,12,13,14. ALDH1, a member of aldehyde dehydrogenase subfamily, regulates pluripotency via retinoic acid pathway and has been implicated as a CSC biomarker15,16. ALDH1 has also been associated with poor outcome and clinicopathological features in BC17,18,19. Stage-specific embryonic antigen 4 (SSEA-4) is a ganglioside present on the cell surface of embryonic stem cells and synthesized by the enzyme ST3Gal II20,21. Increased expression of SSEA-4 has been linked to poor survival in various cancers including lung, breast, prostate and brain22,23,24,25 and SSEA-4 is also considered a potential drug target in glioblastoma multiforme23. BC cell line HTB-9 (5637) has been reported to express SSEA-426,27 but so far, the expression of SSEA-4 has not been studied in clinical BC specimens in more detail.
Importantly, there is data that the number of CSCs is enriched in cisplatin-resistant subclone of T24 bladder cancer cell line (DR-24T) compared to parental cells and these cells were more tumorigenic in mouse xenograft studies28. Prompted by aforementioned findings, we studied the expression of three CSC-related biomarkers, ALDH1, SOX2 and SSEA-4 in two different cohorts of BC patients treated with either RC or TUR-BT, followed by NAC and RC. To our surprise, none of tested markers were statistically significantly associated with disease progression in our patient cohorts. Neither were there significant correlations with patients’ NAC response implying that ALDH1, SOX2 and SSEA-4 play no major role in the progression of BC and/or multiple other factors determine the aggressiveness of the disease.
Results
Patient cohort
Patient characteristics of RC only cohort (n = 195) and NAC cohort (n = 74) are shown in Table 1. Patients included into NAC cohort underwent TUR-BT and received either 2–6 cycles of cisplatin-gemcitabine (68 of 74 patients) or carboplatin-gemcitabine (6 of 74 patients) prior to cystectomy.
Staining patterns
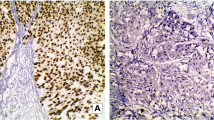
Immunohistochemical ALDH1, SOX2 and SSEA-4 stainings were successful from 173, 167, and 170 patients included into RC cohort, respectively. Representative examples of negative, weakly, and strongly stained carcinoma cells, as well as typical staining pattern on benign urothelium are shown in Fig. 1. None of the markers were associated with age, gender or smoking.
For ALDH1, there were clear differences in cytoplasmic staining intensity between individual patients’ tumors (from negative to strongly expressing cells) while the normal urothelium was generally negative (Fig. 1). H-score of ALDH1 was greater than 0 in 48 (28%) tumors and 4 or more in 22 (13%) tumors (Table 2).
SOX2 staining was found either cytoplasmic or nuclear in a proportion of tumors while the majority showed both the localizations (Fig. 1). This prompted us to analyze SOX2 staining patterns separately. Apparent nuclear SOX2 staining was detected in 119 (71%) tumors and cytoplasmic staining in 141 (84%) tumors (Table 2). In normal urothelium there was generally a strong cytoplasmic staining throughout the urothelium and some positively stained nuclei mostly concentrated at the basal or suprabasal layer of urothelium (Fig. 1).
Ganglioside SSEA-4 was generally absent in normal urothelium while many tumor cells showed positive staining in the cytoplasm or partially at the plasma membrane (Fig. 1). For SSEA-4, H-score was greater than 0 in 64 (38%) tumors and 4 or more in 33 (19%) tumors (Table 2).
Association with clinicopathological characteristics and cancer-specific survival
Based on previous published studies we first tested the prognostic value of ALDH1 in a RC cohort. H-score of 4 or more was not associated with carcinoma grade, stage (organ confinement) or lymphovascular invasion (LVI). Neither was there any significant difference in cancer-specific survival between the high and low expression groups (Table 3 and Fig. 2a).
For cytoplasmic SOX2, a low H-score (3 or lower) was associated with LVI and non-organ confined disease but not with histological grade (Table 3). In support, high cytoplasmic SOX2 expression was associated with improved cancer-specific survival although this failed to show statistically significant difference (Fig. 2c). Nuclear staining of SOX2 showed no association with any clinicopathological characteristic or survival (Table 3 and Fig. 2b).
For SSEA-4, H-score of 4 or higher was inversely associated with high grade, but not with LVI or cancer-specific survival (Table 3 and Fig. 2d). In addition, H-score of 4 or higher was marginally associated with organ-confined disease (Table 3).
Co-expression of ALDH1, SOX2 and SSEA-4 in bladder cancer
Potential co-expression of all the studied markers was further tested using data from all RC samples. 95% confidence interval for relative risk for high H-score (4 or more) was calculated between all markers. High ALDH1 staining intensity was marginally associated with nuclear SOX2. Furthermore, the nuclear SOX2 was associated with cytoplasmic SOX2 as expected. 95% Confidence intervals of risk ratios are shown in Table 4.
CSC markers as predictors of neoadjuvant chemotherapy
TMA samples from TUR-BT operations prior NAC were available from 65 patients for ALDH1 and 66 patients for SOX2 and SSEA-4 staining. Of these patients, 39, 36 and 40 respectively had also residual tumor in RC specimens after NAC. For any of the markers tested, H-score 4 or higher did not predict response to NAC (Table 5). To test the hypothesis that cancer stem cells are enriched in chemoresistant tumors and further after chemotherapy due to drug resistance, we compared the distributions of different H-scores of both chemosensitive and chemoresistant primary tumors to chemoresistant RC residual tumors. There was an enrichment of high H-score tumors for ALHD1 and SOX2 (N and C) after NAC but these differences failed to show statistically significant difference (Fig. 3).
Distributions of H-scores before (TUR-BT) and after (RC) neoadjuvant chemotherapy. The first p-value denotes the statistical difference between TUR-BT chemosensitive and RC chemoresistant tumors while the latter denotes the statistical difference between TUR-BT chemoresistant and RC chemoresistant tumors.
Discussion
In the present study, we evaluated the expression and prognostic and predictive value of stem cell markers ALDH1, SOX2 and SSEA-4 in two different MIBC cohorts treated either with RC only, or with RC after NAC. Based on previously published reports14,17,18,19,28 we originally hypothesized that BC patients whose carcinomas are enriched in CSCs would show less favorable clinicopathological characteristics, more frequent resistance to chemotherapy and shortened disease specific survival. Furthermore, if CSCs were in fact more resilient to chemotherapy, they were expected to enrich following NAC treatment. To our surprise, we found no strong evidence to support our initial hypotheses or previously published studies. The only exception was low cytoplasmic SOX2 staining, which was associated with LVI, non-organ confined disease and to some extent, shortened disease specific survival. To further confirm our findings, we repeated all the analyses using either staining intensity or proportion of positively stained cells alone as a read-out but found no major difference in the results (data not shown).
The association of ALDH1 with high clinicopathological grade, increased tumor size and poor differentiation has been found in multiple studies17,18,19,29. The same studies found an association between high ALDH1 expression and poor disease specific survival. For SOX2, a single study found an association between high expression and poor clinicopathological characteristics and survival in NMIBC14. Even though our results regarding the prognostic value of studied biomarkers were different from others’, the frequencies of both ALDH1 and SOX2 expression were congruent with previous studies. The reason for the different results of current and previous studies can only be speculated. However, it is important to note that SOX2 was previously evaluated in NMIBC cohort while ours consists of MIBC patients. Since NMIBCs more frequently harbor FGF3 mutations and MIBCs are enriched in TP53 and Rb mutations30 it is possible that these differences in molecular pathogenesis override the effects of CSC phenotype. While the patients’ mean age and smoking history in all the cohorts were relatively similar, it is also possible that differences in the expression of CSC markers and outcome are influenced by other risk factors and/or ethnic background of the patients. Equally, the differences in staining methods and antibody clones used may also explain the discordant results.
In many cancer types, such as oral squamous cell carcinoma and colorectal carcinoma both nuclear and cytoplasmic localizations of SOX2 have been previously reported while in lung squamous cell carcinoma and nasopharyngeal carcinoma nuclear localization appears to predominate31,32. The nuclear import and export of SOX2 is regulated by phosphorylation at Thr11833 and acetylation at Lys7534, respectively, and the phosphorylation of SOX2 is considered crucial for stem cell self-renewal or reprogramming33. Nevertheless, the data from different malignancies suggests that cytoplasmic and nuclear SOX2 may have diverse functions. Likewise, in our patient material both cytoplasmic and nuclear localizations were observed and associated with each other. Interestingly, low cytoplasmic staining was associated with LVI and non-organ confined disease, and to some extent, predicted less favorable survival after RC (Table 3 and Fig. 2C). Correspondingly, one would assume that high nuclear SOX2 staining indicates high SOX2 transcriptional activity and poor survival but this proved not to be the case and the more detailed action of SOX2 in different subcellular localizations of CSCs clearly warrants further studies.
To our knowledge, there is no previously published data about co-expression of ALDH1, SOX2 and SSEA-4 in BC, nor studies on enrichment of these markers post chemotherapy. Furthermore, ours is the first to investigate the role of SSEA-4 in BC patient material. Although all three are implicated in pluripotency of stem cells, they have vastly different cellular functions and signaling pathways8,9,10,15,16,20. Despite all of them being regarded as CSC markers and thus expected to reflect similar features in cancer cells, they were not significantly co-expressed in our study material. Further studies are needed to confirm this finding, but it appears clear that none of the CSC markers are universal and there may be multiple CSC subclones with different immunophenotypes within the tumors. Great individual variability in CSC number within tumors is commonly known and was predictably demonstrated in our results as well. Furthermore, there is still relatively little knowledge about the interactions of different CSC markers and the cellular mechanisms of their effects. CSCs are interacting with their surrounding non-malignant cells, and it can be speculated that some CSC biomarkers may display relevance to disease progression only within certain microenvironment. Taken together, a more detailed understanding of the underlying mechanisms of action of different CSC biomarkers could pave way for better biomarkers or sets of biomarkers in the future4,5,6.
Despite the pessimistic overall results, the strengths of this study include analysis of multiple CSC markers in parallel, a long follow-up time, low drop-out rate, and the inclusion of both neoadjuvant treated and chemotherapy naïve patients. The main weaknesses of our study are its retrospective nature, and the small sample size of NAC cohort. One could also question whether TMA material is representative regarding the overall expression of studied CSC markers within the tumor. However, we want to emphasize that up to three cores from each tumor were analyzed with highly similar results suggesting that the expression of studied markers is relatively uniform.
In conclusion, our results cast doubt on the reliability of ALDH1 and SOX2 as clinically relevant prognostic biomarkers in bladder cancer. We also conclude that SSEA-4 was neither prognostic nor predictive biomarker in our material, but this needs to be confirmed in the follow-up studies.
Materials and methods
Study population
The study population consisted of 195 RC patients treated at Turku University Hospital between 1985 and 2005 (RC cohort) and 74 patients who underwent TUR-BT prior to NAC and RC between 2007 and 2013 at either Turku University Hospital or Helsinki University Hospital (NAC cohort). All the patients included in the study had histologically confirmed MIBC (pT2 or higher) at TUR-BT and/or muscular invasion in imaging. The treatment was based on the European Association of Urology (EAU) guidelines at any given time. The clinical follow-up data was collected from hospital registries and survival data from the Finnish Cancer Registry. Informed consent was obtained from all subjects involved in the study.
Tissue microarrays
Diagnostic formalin-fixed and paraffin-embedded tissue blocks were collected from pathology archives of the Turku and Helsinki University Hospitals. The tumors were reviewed by two expert uro-pathologists and classified based on both the WHO 1973 and WHO/ISUP 2004 classifications. Three tissue cores of 1 mm in diameter were punched from representative tumor areas for each patient and transferred into recipient tissue microarray (TMA) blocks. In addition, one tissue core from morphologically benign urothelium for each patient was included into TMA whenever available in original tissue blocks. TMA was created from RC and TUR-BT specimens in both cohorts.
Immunohistochemistry
TMA sections were cut at four micrometer thickness, deparaffinized with xylene and rehydrated in graded series of alcohol. The sections were pretreated with Target Retrieval Solution (Dako) pH 6 (for SSEA-4) or pH 9 (for ALDH1 and SOX2), microwaved twice for 7 min and then washed three times with Tris–HCl buffer solution. Slides were next incubated in 3% hydrogen peroxide solution for 10 min, washed three times with Tris–HCl buffer solution and incubated for 10 min in Normal antibody diluent (Immunologic BD09-125). Mouse monoclonal IgG3 anti-human SSEA-4 antibody (clone MC-813-70, 1:1000, STEMCELL Technologies, Vancouver, Canada), mouse monoclonal IgG1 antibody against human ALDH1 aa. 7–128 (clone 44/ALDH, 1:200, BD Transduction Laboratories, NJ, USA) and mouse monoclonal IgG1 anti-human SOX2 (clone E-4, 1:500, Santa Cruz Biotechnology, Dallas, USA) were diluted in Normal antibody diluent and applied on the sections for 1 h. After washing three times with Tris–HCl buffer solution, the primary antibodies were detected with Brightvision + goat anti-mouse/rabbit HRP secondary antibodies (DPVB110HRP, Immunologic) for SSEA-4 and ALDH1A1, and EnVision + Dual Link HRP (Dako) for SOX2 for 20–30 min, followed by DAB substrate (Dako K3468) for 10 min. After washing three times with distilled water, the sections were counterstained with Mayer’s Hematoxylin Solution. BenchMark XT automated IHC/ISH slide staining system (Ventana Medical Systems, Inc.) was used for all the stainings and multiple staining conditions were tested for each antibody to achieve optimal staining results before staining the TMA material. Liver and testis were used as positive controls for ALDH1, benign urothelium for SOX2, and testis for SSEA-4. Lymphoid tissue was used as a negative control for all the antibodies.
All the slides were digitized using Pannoramic 250 scanner (3DHistech Ltd, Budapest, Hungary), viewed with CaseCenter software (3DHistech), and scored visually on a computer screen as follows: negative = 0, weak = 1, strong = 2. The most strongly stained carcinoma cells were first selected as a reference for high expression for each biomarker used. Carcinoma cells with no staining or equal to non-specific background were considered negative. Up to three carcinoma samples from each patient were analyzed and the cases with unsatisfactory tumor samples (e.g. tissue lost on the slide or no carcinoma cells in the TMA sample) were excluded from the final data analysis. The proportion of positively stained carcinoma cells was graded by approximation as follows: 0 for < 1% positive cells, 1 for 1–25% positive cells, 2 for 26–50% positive cells, 3 for 51–75% positive cells and 4 for 76–100% positive cells. Histological staining score (H-score) was calculated by multiplying the staining intensity score by the proportion score resulting in a number 0–8. In the final analysis, H-score of 4 or higher was considered high expression for each biomarker studied. Since morphologically benign tissue was not available from all the patients and these tissues may carry precancerous genomic alterations, the marker status in morphologically benign urothelium was not analyzed quantitatively.
Statistical analyses
The Kaplan–Meier method was used in survival analysis in Fig. 2. Pearson’s chi-squared test was used in Fig. 3 and Tables 3, 4 and 5. In the NAC cohort (Fig. 3), the cases lacking carcinoma tissue in cystectomy specimen were considered chemosensitive whereas the cases with viable residual/recidive carcinoma in cystectomy specimen were considered chemoresistant.
Institutional review board statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Research Ethics Board of the Hospital District of Southwest Finland (1.8.2006/301).
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Bray, F. et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA. Cancer J. Clin. 68, 394–424 (2018).
Witjes, J. A. et al. European Association of Urology Guidelines on Muscle-invasive and metastatic bladder cancer: Summary of the 2020 guidelines. Eur. Urol. 79, 1–10 (2021).
Sylvester, R. J. et al. Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: A combined analysis of 2596 patients from seven EORTC trials. Eur. Urol. 49, 466–477 (2006).
Beck, B. & Blanpain, C. Unravelling cancer stem cell potential. Nat. Rev. 13, 727–738 (2013).
Allegra, A. et al. The cancer stem cell hypothesis: A guide to potential molecular targets. Cancer Invest. 32, 470–495 (2014).
Ben-Porath, I. et al. An embryonic stem cell-like gene expression signature in poorly differentiated aggressive human tumors. Nat. Genet. 40, 499–507 (2008).
Valent, P. et al. Cancer stem cell definitions and terminology: The devil is in the details. Nat. Rev. Cancer 12, 767–775 (2012).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Sarkar, A. & Hochedlinger, K. The sox family of transcription factors: Versatile regulators of stem and progenitor cell fate. Cell Stem Cell 12, 15–30 (2013).
Arnold, K. et al. Sox2(+) adult stem and progenitor cells are important for tissue regeneration and survival of mice. Cell Stem Cell 9, 317–329 (2011).
Lundberg, I. V. et al. SOX2 expression is regulated by BRAF and contributes to poor patient prognosis in colorectal cancer. PLoS ONE 9, e101957 (2014).
Sholl, L. M., Barletta, J. A., Yeap, B. Y., Chirieac, L. R. & Hornick, J. L. Sox2 protein expression is an independent poor prognostic indicator in stage I lung adenocarcinoma. Am. J. Surg. Pathol. 34, 1193–1198 (2010).
Russo, M. V. et al. SOX2 boosts major tumor progression genes in prostate cancer and is a functional biomarker of lymph node metastasis. Oncotarget 7, 12372–12385 (2016).
Ruan, J. et al. Predictive value of Sox2 expression in transurethral resection specimens in patients with T1 bladder cancer. Med. Oncol. 30, 445–512 (2013).
Haselbeck, R. J., Hoffmann, I. & Duester, G. Distinct functions for Aldh1 and Raldh2 in the control of ligand production for embryonic retinoid signaling pathways. Dev. Genet. 25, 353–364 (1999).
Huang, E. H. et al. Aldehyde dehydrogenase 1 is a marker for normal and malignant human colonic stem cells (SC) and tracks SC overpopulation during colon tumorigenesis. Cancer Res. 69, 3382–3389 (2009).
Keymoosi, H., Gheytanchi, E., Asgari, M., Shariftabrizi, A. & Madjd, Z. ALDH1 in combination with CD44 as putative cancer stem cell markers are correlated with poor prognosis in urothelial carcinoma of the urinary bladder. Asian Pac. J. Cancer Prev. 15, 2013–2020 (2014).
Su, Y. et al. Aldehyde dehydrogenase 1 A1-positive cell population is enriched in tumor-initiating cells and associated with progression of bladder cancer. Cancer Epidemiol. Biomarkers Prev. 19, 327–337 (2010).
Senol, S. et al. Relation of stem cell markers ALDH1 and CD44 with clinicopathological factors in urothelial carcinomas of urinary bladder. Int. J. Clin. Exp. Med. 8, 4195–4203 (2015).
Kannagi, R. et al. Stage-specific embryonic antigens (SSEA-3 and -4) are epitopes of a unique globo-series ganglioside isolated from human teratocarcinoma cells. EMBO J. 2, 2355–2361 (1983).
Saito, S. et al. Human alpha2,3-sialyltransferase (ST3Gal II) is a stage-specific embryonic antigen-4 synthase. J. Biol. Chem. 278, 26474–26479 (2003).
Gottschling, S. et al. Stage-specific embryonic antigen-4 is expressed in basaloid lung cancer and associated with poor prognosis. Eur. Respir. J. 41, 656–663 (2013).
Lou, Y. W. et al. Stage-specific embryonic antigen-4 as a potential therapeutic target in glioblastoma multiforme and other cancers. Proc. Natl. Acad. Sci. USA 111, 2482–2487 (2014).
Aloia, A. et al. The sialyl-glycolipid stage-specific embryonic antigen 4 marks a subpopulation of chemotherapy-resistant breast cancer cells with mesenchymal features. Breast Cancer Res. 17, 146 (2015).
Hatano, K., Miyamoto, Y., Nonomura, N. & Kaneda, Y. Expression of gangliosides, GD1a, and sialyl paragloboside is regulated by NF-kappaB-dependent transcriptional control of alpha2,3-sialyltransferase I, II, and VI in human castration-resistant prostate cancer cells. Int. J. cancer. 129, 1838–1847 (2011).
Iskender, B., Izgi, K., Karaca, H. & Canatan, H. Myrtucommulone-A treatment decreases pluripotency- and multipotency-associated marker expression in bladder cancer cell line HTB-9. J. Nat. Med. 69, 543–554 (2015).
Sivasubramaniyan, K. et al. Expression of stage-specific embryonic antigen-4 (SSEA-4) defines spontaneous loss of epithelial phenotype in human solid tumor cells. Glycobiology 25, 902–917 (2015).
Zhang, Y. et al. Cancer stem-like cells contribute to cisplatin resistance and progression in bladder cancer. Cancer Lett. 322, 70–77 (2012).
Xu, N. et al. Aldehyde dehydrogenase 1 (ALDH1) expression is associated with a poor prognosis of bladder cancer. Cancer Epidemiol. 39, 375–381 (2015).
Nassar, A. H. et al. Mutational analysis of 472 urothelial carcinoma across grades and anatomic sites. Clin. Cancer Res. 25, 2458–2470 (2019).
Baillie, R. et al. Cancer stem cells in moderately differentiated oral tongue squamous cell carcinoma. J. Clin. Pathol. 69, 742–744 (2016).
van Schaijik, B., Davis, P. F., Wickremesekera, A. C., Tan, S. T. & Itinteang, T. Subcellular localisation of the stem cell markers OCT4, SOX2, NANOG, KLF4 and c-MYC in cancer: A review. J. Clin. Pathol. 71, 88–91 (2018).
Jeong, C.-H. et al. Phosphorylation of Sox2 cooperates in reprogramming to pluripotent stem cells. Stem Cells 28, 2141–2150 (2010).
Baltus, G. A. et al. Acetylation of Sox2 induces its nuclear export in embryonic stem cells. Stem Cells 27, 2175–2184 (2009).
Acknowledgements
Sinikka Collanus is thanked for her help with immunohistochemistry. Adj. Prof. Jari Sundstöm (Turku University Hospital) is thanked for providing antibody for ALDH1 staining.
Author information
Authors and Affiliations
Contributions
Conceptualization, M.B.; methodology, M.B., E.L., T.M., P.B. and P.T.; investigation, M.B., I.K.; statistical analysis, M.B. and E.L.; writing—original draft preparation, M.B.; writing—review and editing, P.B., T.M. and P.T.; visualization, M.B.; supervision, P.B. and P.T.; funding acquisition, P.B. and P.T.; All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Blomqvist, M., Koskinen, I., Löyttyniemi, E. et al. Prognostic and predictive value of ALDH1, SOX2 and SSEA-4 in bladder cancer. Sci Rep 11, 13684 (2021). https://doi.org/10.1038/s41598-021-93245-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-93245-1
- Springer Nature Limited