Abstract
High-altitude pulmonary edema (HAPE) is a common acute altitude sickness. This study was designed to investigate the effect of MIR17HG polymorphisms on HAPE risk in the Chinese population. The Agena MassARRAY platform was used to genotype six single-nucleotide polymorphisms (SNPs) in the MIR17HG gene in 244 HAPE patients and 243 non-HAPE controls. The odds ratio (OR) and 95% confidence interval were used to evaluate the association between each MIR17HG polymorphisms and the risk of HAPE under a polygenetic model. Statistical analysis was performed using the χ2 test. Multifactor dimensionality reduction (MDR) analysis was used to analyze the impacts of SNP–SNP interactions on the risk of HAPE. According to the allele model, the HAPE risk of people with the rs7318578 A allele of MIR17HG was lower than that of people with the C allele (OR 0.74, p = 0.036).Logistic regression analysis of four models for all selected MIR17HG SNPs showed significant differences in the frequencies of rs7318578 (OR 0.74, p = 0.037) and rs17735387 (OR 1.51, p = 0.036) between cases and controls. The results of the sex stratification analysis showed that among males, rs17735387 in the MIR17HG gene is associated with an increased risk of HAPE. MDR analysis showed that the best combination model was a three-locus model incorporating rs72640334, rs7318578, and rs7336610. This study revealed the correlations between rs7318578 and rs17735387 on the MIR17HG gene and the risk of HAPE in the Chinese population, providing a theoretical basis for the early screening, prevention, and diagnosis of HAPE in high-risk populations.
Similar content being viewed by others
Introduction
Among plateau-related diseases, acute mountain sickness (AMS) is the most common. There are regional differences in the incidence of AMS. Generally, the higher the altitude is, the higher the incidence of AMS and the more severe the clinical symptoms. AMS includes two types: mild and severe. High-altitude pulmonary edema (HAPE) is a severe form of AMS1. This diseaseis a serious life-threatening noncardiogenic pulmonary edema caused by a high-altitude hypoxic environment. HAPE is a special and common disease that develops after a short period of exposure to altitudes above 2250 m and is characterized by acute onset, rapid progress and serious consequences1,2. People living in the plains usually have HAPE within 2 to 5 days after entering the plateau. Specifically, when people are rapidly exposed to high-altitude environments, low oxygen air tension can cause acute pulmonary artery vasoconstriction, which in turn leads to an increase in high pulmonary artery pressure. The resulting pressure on the pulmonary microvascular system can cause excessive fluid to leak into the lungs, and ultimately lead to the occurrence of HAPE3. It can be seen that the high-altitude and low-oxygen environment easily leads to the occurrence of HAPE. Increasing evidence has shown that genetic factors, such as IL1R24, NR3C25, NR3C16, and NOS37, also play key roles in the occurrence of HAPE.
The pathogenesis of HAPE is more complex, and it is unclear. It is generally believed that the key to its occurrence is the pulmonary vasoconstriction and increased pulmonary arterial pressure caused by hypoxia at high altitude. Studies have found that lncRNA polymorphisms were associated with genetic susceptibility to pulmonary hypertension8. Therefore, we believe that lncRNA polymorphisms may also be associated with genetic susceptibility to high altitude pulmonary edema.
MIR17HG is derived from the mir-17-92a-1 cluster host gene and is a class of pri-miRNAs located in the 800 base pair region of human chromosome 13. Studies have confirmed that MIR17HG plays important roles in various growth and developmental processes, such as cell proliferation and differentiation and angiogenesis9,10,11. Wei et al.12 evaluated the expression of MIR17HG is down-regulated in NSCLC, while the over-expression of MIR17HG can lead to up-regulation of miR-142-3p and down-regulation of Bach-1 expression, thereby inhibiting the invasion and migration ability of non-small cell lung cancer (NSCLC) cells through methylation-specific PCR (MSP) and Transwell invasion and migration tests. By conducting an experiment with a mouse model, Rogers et al.13 showed that the expression of MIR17HG is related to bronchopulmonary dysplasia. Liao et al.14 analyzed asthma-related information from an online database and showed that MIR17HG plays an important role in asthma and may become a potential target and prognostic biomarker for asthma treatment. These findings indicate that MIR17HG plays an important role in lung diseases. A number of studies have shown that MIR17HG polymorphisms are associated with various disease risks15, and multiple reports have shown that gene polymorphisms are associated with HAPE risk16. However, there is no report on the relationship between MIR17HG polymorphisms and HAPE risk.. This study aims to explore the correlations between MIR17HG polymorphisms and the risk of HAPE in the Chinese population through a case–control strategy to provide a theoretical basis for the early screening, prevention and diagnosis of HAPE in high-risk groups.
Results
Basic characteristics of the participants
As Table 1 shows, 487 participants were recruited. Among them, 244 participants had HAPE (19 females, 225 males), while 243 participants had no symptoms of HAPE (19 females, 224 males). The mean ages in the HAPE group and healthy group were 32.41 ± 10.81 and 32.70 ± 9.52 years, respectively. No significant differences were found in sex or age between the case and control groups (both p > 0.05). The selected SNPs were functionally annotated using HaploRegv4.1 software. The results showed that these six SNPs (rs75267932, rs72640334, rs7336610, rs7318578, rs17735387 and rs1428) in the MIR17HG gene are associated with promoter histone marks, enhancer histone marks, DNase, motif changes, and GRASP QTLs, suggesting that they may play important roles in related biological functions in patients.
Associations of genetic polymorphisms with HAPE risk
Table 2 shows the basic information of the six SNPs in the MIR17HG gene identified in this study. All SNPs were found to be at Hardy–Weinberg equilibrium (HWE) based on exact tests. In the allele model, we observed a lower risk of HAPE (odds ratio (OR) 0.74, 95% confidence interval (95% CI) 0.56–0.98, p = 0.036) associated with the A allele of MIR17HG SNP rs7318578 than with the C allele. Logistic regression analysis of the four models for all of the selected MIR17HG SNPs between cases and controls showed significant differences for rs7318578 and rs17735387 in the HAPE population. For rs7318578, under the log-additive model, we found that people carrying the A allele had a decreased risk of HAPE compared with C allele carriers, which with the A allele playing a protective role (OR 0.74, 95% CI 0.56–0.98, p = 0.037; Table 3). Individuals carrying the rs17735387 A allele had a 51% higher risk of HAPE than those with the G allele (OR 1.51, 95% CI 1.03–2.21, p = 0.036; Table 3).
Stratification analysis of the associations of SNPs with HAPE risk
To further clarify the associations between MIR17HG polymorphisms and HAPE risk, we conducted age- and sex-stratified analyses. In the age stratification analysis, we found no association of any of the six polymorphic loci (rs75267932, rs72640334, rs7336610, rs7318578, rs17735387, and rs1428) in the MIR17HG gene with the risk of HAPE (Supplementary Table 2). The results of the sex stratification analysis showed that among men, individuals with the rs17735387 genotype AG had an increased risk of HAPE compared with GG individuals (OR 1.55, 95% CI 1.03–2.32, p = 0.036; Table 4).
SNP–SNP interactions
As shown in Fig. 1A, B a dendrogram and a Fruchterman Reingold algorithm were used to describe the interactions among these SNPs. We evaluated all three-way to five-way models (Table 5). We chose a three-loci model as the best model considering the small sample size. The overall best model included rs72640334, rs7318578, and rs7336610, with a testing accuracy of 56.58%, a maximum cross-validation consistency (CVC) of 10/10 and p value of < 0.001 obtained by the χ2 test. Compared with the low-risk genotype combination, the generated combination of high-risk genotypes was associated with an increased HAPE risk (OR 2.43, 95% CI: 1.61–3.68, p < 0.001; Table 5). The MDR approach uses entropy to evaluate the correlation between variable combinations and attribute interactions (IG)17. The strongest interaction effect was found between rs7318578 and rs7336610, with an IG value of 0.59%.
MDR analysis results. MDR-3.0.2 software: http://sourceforge.net/projects/mdr/. Dendogram (A) and Fruchterman Rheingold plot (B) of the interactions of MIR17HG SNPs rs75267932, rs72640334, rs7336610, rs7318578, rs17735387 and rs1428 for HAPE risk. Shorter connections among nodes indicate stronger redundant interactions. Negative percent entropy indicates redundancy.
Haplotype analysis with the risk of HAPE
In addition, we conducted linkage disequilibrium (LD) and haplotype analysis of MIR17HG polymorphisms. The constructed LD map is shown in Fig. 2. The LD block consists of four SNPs, rs7336610, rs7318578, rs17735387 and rs1428, in the MIR17HG gene. The frequency distributions of the haplotypes in the case group and the control group are shown in Table 6. However, there was no significant association between haplotype and HAPE risk.
LD plots of six SNPs in the MIR17HG gene. Haploview-4.2 software: https://www.broadinstitute.org/haploview/downloads#JAR. The squares in the graph represent the strength of linkage disequilibrium (r2) between every two SNPs. The darker the color, the greater the r2 value. The inverted black triangles represent haplotype blocks defined by the confidence interval method. Agena MassARRAY Assay Design 3.0 software: https://seqpws1.agenabio.com/AssayDesignerSuite.html#design:new=Genotyping. Agena Typer 4.0 software: (Agena, San Diego, CA, USA) https://seqpws1.agenabio.com/AssayDesignerSuite.html#design:new=Genotyping. The Agena Typer 4.0 software for genotyping was purchased from the MassARRAY chip company (Agena, San Diego, CA, USA).
Meta-analysis
Correlation analysis was carried out on the genotyping data of six SNPs on MIR17HG of HAPE that has been reported. Meta analysis showed that rs7318578 on the MIR17HG gene reduced the risk of HAPE under the allele model (OR 0.77, 95% CI 0.61–0.98, p = 0.037) (Supplementary Table 3), which is consistent with our previous research results.
Discussion
HAPE is a very harmful disease to humans. This study explored the impact of the MIR17HG polymorphisms on the risk of HAPE in the Chinese population. In our study, we found that MIR17HG rs7318578 minimized the risk and that rs17735387 increased the risk.
MIR17HG is a noncoding RNA that affects protein synthesis by regulating gene expression, and its sole function is to produce six miRNAs (miR-17, miR-18a, miR-19a, miR-20a, miR-19b-1, and miR-92-1)18,19. Yin et al.20 showed that miR-17 is involved in the regulation of EGFR-TKI resistance and can be used as a predictive biomarker for NSCLC EGFR-TKI resistance. Li et al.21 showed that the expression of miR-18a affects the occurrence of lung fibroblast fibrosis. In addition, Ren et al.22 showed that the downregulation of miR-19a-3p expression is related to the reduction of sepsis-induced lung injury. These findings indicate that MIR17HG plays an important role in the occurrence and development of a variety of lung diseases. Multiple reports have revealed that MIR17HG polymorphisms are associated with various disease risks23,24, however, there is no report about the polymorphisms of MIR17HG and the risk of HAPE.. Our research revealed that people carrying the rs7318578 A allele had a reduced risk of HAPE compared with C allele carriers, and that individuals carrying the rs17735387 A allele had a 51% increased risk of HAPE compared with G-allele carriers. Meta-analysis also showed that rs7318578 is related to the reduction of HAPE risk. Our study further revealed that the MIR17HG polymorphisms are associated with the risk of HAPE. The incidence of HAPE differs between the sexes. The results of the sex stratification analysis showed that among males, individuals with the rs17735387 AG genotype had an increased risk of HAPE compared with GG individuals, which indicated that the rs17735387 polymorphism of the MIR17HG gene affects the risk of HAPE. This result has implications for the screening of biomarkers of HAPE risk in male populations.
Studies have confirmed that p53 and HIF-1α can inhibit the expression of MIR17HG25. In addition, studies have shown that VEGF-induced MIR17HG expression is involved in the regulation of angiogenesis26. Chamorro-Jorganes et al.27 showed that the downstream effector of HIF-1α, VEGF, affects the occurrence of HAPE by regulating vascular permeability. However, there is no report on the association between MIR17HG and HAPE, and we hypothesize that there is a correlation between the two. The promoter of the miR-17-92 cluster host gene MIR17HG is highly conserved, and the promoter region has multiple conserved transcription factor binding sites28.SNPs might impact biological processes involved in the production or functional effects of the miR-17-92 cluster host gene. We found that MIR17HG rs7318578 minimized the risk of HAPE and that rs17735387 increased the risk. These results provide some empirical support for our hypothesis, however, to clarify the associations between MIR17HG and HAPE, we conducted an MDR analysis. MDR is a recently developed method for analyzing interactions. It can simultaneously detect and describe the effects of multifactor interactions on diseases and has been widely used in scientific research29,30. This study clarified the influences of MIR17HG polymorphism site interactions on the risk of HAPE through MDR. The results showed that rs7318578 and rs7336610 had a strong synergistic effect on HAPE sensitivity and that rs7336610 and rs1428 had a strong antagonistic effect on HAPE sensitivity (Fig. 1B). The best model combination for HAPE sensitivity prediction was rs72640334, rs7318578, and rs7336610. All of these results upport our hypothesis, which MIR17HG polymorphisms are related to HAPE.
This study has limitations. First, the sample size was small. Second, although the correlation between six SNPs in MIR17HG and the risk of HAPE in the Chinese population was explored in this study, the MIR17HG gene includes many other sites. In our future work, we will explore the correlations between other sites on MIR17HG and the risk of HAPE. Third, due to a lack of cell biology methods, we did not explore the molecular mechanisms underlying the influences of MIR17HG polymorphisms on the occurrence of HAPE. In the future, we will explore the specific role of MIR17HG in HAPE from these perspectives.
In conclusion, this study clarified that MIR17HG polymorphisms are associated with the risk of HAPE in the Chinese population, which lays a foundation for further research on the mechanism of MIR17HG action on HAPE and provides a scientific basis for early screening and prevention of HAPE in high-risk population.
Methods
Participants
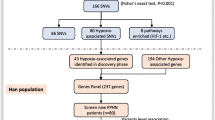
Based on the case–control strategy for exploring polymorphisms in the MIR17HG gene, we recruited 487 participants from the Affiliated Hospital of Qinghai University. All participants lived in low-altitude areas below 2500 m and reached high-altitude areas above 3000 m due to work or travel. By examining the symptoms (dyspnea), signs (cyanosis at rest) and imaging results (such as X-ray radiography and computed tomography (CT) of the patient chest) for all participants, and adhering to with the diagnostic criteria of HAPE31, we found that 244 participants had HAPE, forming the case group, while 243 participants had no symptoms of HAPE, forming the control group. There were 225 males and 19 females in the case group, and 224 and 19 females in the control group.
Genotyping
To ensure the successful statistical analysis of genotypes in the research population, SNPs with a minor allele frequency of at least 5% in the Han Chinese from Beijing (CHB) population were initially screened using the Thousand Genomes Project database (http://www.internationalgenome.org/). Haploview software (version 4.2) (https://www.broadinstitute.org/haploview/downloads#JAR) was then used to conduct LD haplotype analysis32. Subsequently, function prediction analysis (https://www.ncbi.nlm.nih.gov/snp/) of tag single nucleotide polymorphisms (tagSNP) was performed online with the dbSNP database and showed that the tagSNPs are introns that affect the activity of splice sites. Following further analysis, six SNPs in the MIR17HG gene (rs75267932, rs72640334, rs7336610, rs7318578, rs17735387 and rs1428) were selected and explored for their correlations with the risk of HAPE in the Chinese population. Agena MassARRAY Assay Design 3.0 software (https://seqpws1.agenabio.com/AssayDesignerSuite.html) was used to design the primers (Supplementary Table 1). Five milliliters of peripheral venous blood was collected according to standard procedures in an EDTA-containing anticoagulation tube and stored in an ultralow-temperature freezer for subsequent DNA extraction. The extraction of DNA from whole blood samples was performed using the GoldMag–Mini Purification Kit (GoldMag Co, Ltd, Xi’an, China). Quality monitoring, including concentration and purity assessment, of the extracted DNA was conducted using a spectrophotometer. Finally, genotyping of the six polymorphisms of the MIR17HG gene was performed using the Agena MassARRAY RS1000 system according to the manufacturer’s. The Agena Typer 4.0 software (Agena, San Diego, CA, USA) was used to analyze the experimental results and obtain the genotyping data following a previously described method4,33.
Bioinformatics analysis
The HaploReg v4.1 (https://pubs.broadinstitute.org/mammals/haploreg/haploreg.php) database is often used to annotate noncoding genomes on haplotype blocks, including disease-related SNPs. In this study, HaploReg v4.1 was used to predict the potential functional role of candidate SNPs.
Meta-analysis
This study obtained genotyping data of six SNPs (rs75267932, rs72640334, rs7336610, rs7318578, rs17735387, rs1428) on the MIR17HG of the control group from the 1000 Genome Projects (http://www.internationalgenome.org/). The genotyping data of the six SNPs in the case group was obtained through the interpolation procedure. The data obtained above was analyzed by STATA 11.0 software (Stata Corp., College Station, TX, USA)34.
Statistical analysis
The analysis of the general characteristics of the subjects, the chi-square test was used to compare the categorical variable between the case group and the control group. The t-test was used to compare the continuous variable (age) between the case and control groups. The goodness of fit chi-square test was used to calculate the HWE of each polymorphism in the control group to test the representativeness and randomness of the study population. The chi-square test was used to detect the distribution differences of genotype and allele frequency of each polymorphism between the case group and the control group. Logistic regression was used to calculate OR values and 95% CIs to evaluate the strength of the association between each genotype and HAPE by PLINK software. All statistical tests were bilateral, with a significance threshold of 0.05. In this study, the minimum sample size and proportions of the experimental group and the control group were determined by G * Power 3.1.9.2 software35. The analysis results indicated that the total sample size should be greater than 388 and that the sample size of each of the control group and case group should be greater than 194. The total sample size in this study was 487, with 243 subjects in the control group and 244 in the case group thus, the sample sizes of this study met the statistical requirements. In addition, the statistical power of this study to detect an effect at the specified level of significance was 99.98%. Multifactor dimensionality reduction (MDR) analysis was performed using MDR_3.0.2 software (http://sourceforge.net/projects/mdr/) to determine a high-order interaction model for the risk of HAPE36.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of the Affiliated Hospital of Qinghai University and was conducted in compliance with the Declaration of Helsinki. The purpose of this study was well conveyed to all participants, and written informed consent was obtained from all participants prior to biological material collection in this study. All subsequent study analyses were conducted in accordance with the approved guidelines and regulations.
Consent for publication
Written informed consent was obtained from the patients for publication of this report.
Data availability
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- HAPE:
-
High-altitude pulmonary edema
- AMS:
-
Acute mountain sickness
- SNP:
-
Single nucleotide polymorphism
- HWE:
-
Hardy–Weinberg equilibrium
- OR:
-
Odds ratio
- 95% CI:
-
95% confidence intervals
- CT:
-
Computed tomography
- MAF:
-
Minor allele frequency
- MM:
-
Multiple myeloma
- MIR17HG :
-
MiR-17-92 cluster host gene
- LD:
-
Linkage disequilibrium
References
Basnyat, B. & Murdoch, D. R. High-altitude illness. Lancet 361, 1967–1974. https://doi.org/10.1016/S0140-6736(03)13591-X (2003).
Maggiorini, M. High altitude-induced pulmonary oedema. Cardiovasc. Res. 72, 41–50. https://doi.org/10.1016/j.cardiores.2006.07.004 (2006).
Kylhammar, D. & Rådegran, G. The principal pathways involved in the in vivo modulation of hypoxic pulmonary vasoconstriction, pulmonary arterial remodelling and pulmonary hypertension. Acta Physiol. (Oxf.) 219, 728–756. https://doi.org/10.1111/apha.12749 (2017).
Jin, T. et al. Association between the IL1R2 rs2072472 polymorphism and high-altitude pulmonary edema risk. Mol. Genet. Genomic Med. 7, e542. https://doi.org/10.1002/mgg3.542 (2019).
Yang, Y. et al. NR3C2 gene is associated with susceptibility to high-altitude pulmonary edema in Han Chinese. Wilderness Environ. Med. 29, 488–492. https://doi.org/10.1016/j.wem.2018.07.006 (2018).
Yang, Y. et al. NR3C1 gene polymorphisms are associated with high-altitude pulmonary edema in Han Chinese. J. Physiol. Anthropol. 38, 4. https://doi.org/10.1186/s40101-019-0194-1 (2019).
Wang, P., Ha, A. Y., Kidd, K. K., Koehle, M. S. & Rupert, J. L. A variant of the endothelial nitric oxide synthase gene (NOS3) associated with AMS susceptibility is less common in the Quechua, a high altitude Native population. High Alt. Med. Biol. 11, 27–30. https://doi.org/10.1089/ham.2009.1054 (2010).
Zhuo, Y. et al. Functional polymorphism of lncRNA MALAT1 contributes to pulmonary arterial hypertension susceptibility in Chinese people. Clin. Chem. Lab. Med. 55, 38–46. https://doi.org/10.1515/cclm-2016-0056 (2017).
Lu, Y., Thomson, J. M., Wong, H. Y., Hammond, S. M. & Hogan, B. L. Transgenic over-expression of the microRNA miR-17-92 cluster promotes proliferation and inhibits differentiation of lung epithelial progenitor cells. Dev. Biol. 310, 442–453. https://doi.org/10.1016/j.ydbio.2007.08.007 (2007).
Zhou, M., Ma, J., Chen, S., Chen, X. & Yu, X. MicroRNA-17-92 cluster regulates osteoblast proliferation and differentiation. Endocrine 45, 302–310. https://doi.org/10.1007/s12020-013-9986-y (2014).
Dews, M. et al. The myc-miR-17~92 axis blunts TGF{beta} signaling and production of multiple TGF{beta}-dependent antiangiogenic factors. Cancer Res. 70, 8233–8246. https://doi.org/10.1158/0008-5472.CAN-10-2412 (2010).
Wei, S., Liu, J., Li, X. & Liu, X. LncRNA MIR17HG inhibits non-small cell lung cancer by upregulating miR-142-3p to downregulate Bach-1. BMC Pulm. Med. 20, 78. https://doi.org/10.1186/s12890-020-1112-3 (2020).
Rogers, L. K. et al. Attenuation of miR-17∼92 cluster in bronchopulmonary dysplasia. Ann. Am. Thorac. Soc. 12, 1506–1513. https://doi.org/10.1513/AnnalsATS.201501-058OC (2015).
Liao, Y. et al. Construction of asthma related competing endogenous RNA network revealed novel long non-coding RNAs and potential new drugs. Respir. Res. 21, 14. https://doi.org/10.1186/s12931-019-1257-x (2020).
Abdel-Gawad, A. R. et al. Association of microRNA 17 host gene variant (rs4284505) with susceptibility and severity of systemic lupus erythematosus. Immun. Inflamm. Dis. 8, 595–604. https://doi.org/10.1002/iid3.344 (2020).
Sharma, K. et al. High-altitude pulmonary edema is aggravated by risk loci and associated transcription factors in HIF-prolyl hydroxylases. Hum. Mol. Genet. 30, 1734–1749. https://doi.org/10.1093/hmg/ddab139 (2021).
Moore, J. H. et al. A flexible computational framework for detecting, characterizing, and interpreting statistical patterns of epistasis in genetic studies of human disease susceptibility. J. Theor. Biol. 241, 252–261. https://doi.org/10.1016/j.jtbi.2005.11.036 (2006).
Zhu, H., Han, C. & Wu, T. MiR-17-92 cluster promotes hepatocarcinogenesis. Carcinogenesis 36, 1213–1222. https://doi.org/10.1093/carcin/bgv112 (2015).
He, L. et al. A microRNA polycistron as a potential human oncogene. Nature 435, 828–833. https://doi.org/10.1038/nature03552 (2005).
Yin, J. et al. let-7 and miR-17 promote self-renewal and drive gefitinib resistance in non-small cell lung cancer. Oncol. Rep. 42, 495–508. https://doi.org/10.3892/or.2019.7197 (2019).
Li, X. et al. lncRNA PFAL promotes lung fibrosis through CTGF by competitively binding miR-18a. FASEB J. 32, 5285–5297. https://doi.org/10.1096/fj.201800055R (2018).
Ren, H., Mu, W. & Xu, Q. miR-19a-3p inhibition alleviates sepsis-induced lung injury via enhancing USP13 expression. Acta Biochim. Pol. 68, 201–206. https://doi.org/10.18388/abp.2020_5505 (2021).
Wu, H. et al. Correlation of microRNA 17-92 cluster host gene (MIR17HG) polymorphisms with susceptibility and prognosis for multiple myeloma. Clin. Lymphoma Myeloma Leuk. 19, e359–e366. https://doi.org/10.1016/j.clml.2019.03.018 (2019).
Chen, P. et al. Association between polymorphisms of MIR17HG and risk of colorectal cancer in the Chinese Han population. Mol. Genet. Genomic Med. 7, e667. https://doi.org/10.1002/mgg3.667 (2019).
Yan, H. L. et al. Repression of the miR-17-92 cluster by p53 has an important function in hypoxia-induced apoptosis. EMBO J. 28, 2719–2732. https://doi.org/10.1038/emboj.2009.214 (2009).
Tsai, S. H. et al. Roles of the hypoximir microRNA-424/322 in acute hypoxia and hypoxia-induced pulmonary vascular leakage. FASEB J. 33, 12565–12575. https://doi.org/10.1096/fj.201900564RR (2019).
Chamorro-Jorganes, A. et al. VEGF-induced expression of miR-17-92 cluster in endothelial cells is mediated by ERK/ELK1 activation and regulates angiogenesis. Circ. Res. 118, 38–47. https://doi.org/10.1161/circresaha.115.307408 (2016).
Ji, M. et al. The miR-17-92 microRNA cluster is regulated by multiple mechanisms in B-cell malignancies. Am. J. Pathol. 179, 1645–1656. https://doi.org/10.1016/j.ajpath.2011.06.008 (2011).
Wu, Y. et al. A multifactor dimensionality reduction-logistic regression model of gene polymorphisms and an environmental interaction analysis in cancer research. Asian Pac. J. Cancer Prev.: APJCP 12, 2887–2892 (2011).
Bhat, M. A. & Gandhi, G. CYP2D6 (C2850T, G1846A, C100T) polymorphisms, haplotypes and MDR analysis in predicting coronary artery disease risk in north-west Indian population: A case–control study. Gene 663, 17–24. https://doi.org/10.1016/j.gene.2018.04.008 (2018).
Gabry, A. L., Ledoux, X., Mozziconacci, M. & Martin, C. High-altitude pulmonary edema at moderate altitude (< 2,400 m; 7,870 feet): A series of 52 patients. Chest 123, 49–53. https://doi.org/10.1378/chest.123.1.49 (2003).
Liu, C. X. et al. Association of TBX5 gene polymorphism with ventricular septal defect in the Chinese Han population. Chin. Med. J. 122, 30–34 (2009).
He, X. et al. Long Non-coding RNA LINC-PINT and LINC00599 polymorphisms are associated with high-altitude pulmonary edema in Chinese. Arch. Bronconeumol. https://doi.org/10.1016/j.arbres.2019.09.021 (2019).
Dhillon, R. K. et al. Additional information from chromosomal microarray analysis (CMA) over conventional karyotyping when diagnosing chromosomal abnormalities in miscarriage: A systematic review and meta-analysis. BJOG 121, 11–21. https://doi.org/10.1111/1471-0528.12382 (2014).
Faul, F., Erdfelder, E., Lang, A. G. & Buchner, A. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 39, 175–191. https://doi.org/10.3758/bf03193146 (2007).
Leem, S. & Park, T. An empirical fuzzy multifactor dimensionality reduction method for detecting gene-gene interactions. BMC Genomics 18, 115. https://doi.org/10.1186/s12864-017-3496-x (2017).
Acknowledgements
We thank all participants for taking part in this study.
Funding
This work was financially supported by the National Natural Science Foundation of China (No. 81460284, No. 81460051, No. 31560039), the Natural Science Foundation of Qinghai Province (No. 2014-ZJ-944Q), and the key R & D and Transformation Program of Qinghai Province of China(2022-SF-139).
Author information
Authors and Affiliations
Contributions
Z.Y.: conceived and designed the experiments. S.L.: performed the experiments. W.H.: analyzed the data. J.Y. and Y.Y.: analyzed the data and prepared the figures and/or tables. W.R.: contributed reagents/materials/analysis tools. L.Q.: prepared the figures and/or tables. Z.Y.: drafted the work or revised it critically for important content. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Si, L., Wang, H., Jiang, Y. et al. MIR17HG polymorphisms contribute to high-altitude pulmonary edema susceptibility in the Chinese population. Sci Rep 12, 4346 (2022). https://doi.org/10.1038/s41598-022-06944-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-06944-8
- Springer Nature Limited