Abstract
Vitamin D insufficiency/deficiency has been linked to an increased risk of preeclampsia. Impaired placental amino acid transport is suggested to contribute to abnormal fetal intrauterine growth in pregnancies complicated by preeclampsia. However, if vitamin D-regulated amino acid transporter is involved in the pathophysiologic mechanism of preeclampsia has not been clarified yet. The aberrant expression of key isoform of L-type amino acid transporter LAT1 was determined by western blot and immunohistochemistry in the placenta from normotensive and preeclamptic pregnancies. The role for vitamin D on placental LAT1 expression was investigated through the exposure of HTR-8/SVneo human trophoblast cells to the biologically active 1,25(OH)2D3 and the oxidative stress-inducer cobalt chloride (CoCl2). Our results showed that placental LAT1 expression was reduced in women with preeclampsia compared to normotensive pregnancies, which was associated with decreased expression of vitamin D receptor (VDR). 1,25(OH)2D3 significantly upregulated LAT1 expression in placental trophoblasts, and also prevented the decrease of mTOR activity under CoCl2-induced oxidative stress. siRNA targeting VDR significantly attenuated 1,25(OH)2D3-stimulated LAT1 expression and mTOR signaling activity. Moreover, treatment of rapamycin specifically inhibited the activity of mTOR signaling and resulted in decrease of LAT1 expression. In conclusion, LAT1 expression was downregulated in the placenta from women with preeclampsia. 1,25(OH)2D3/VDR could stimulate LAT1 expression, which was likely mediated by mTOR signaling in placental trophoblasts. Regulation on placental amino acid transport may be one of the mechanisms by which vitamin D affects fetal growth in preeclampsia.
Similar content being viewed by others
Introduction
Preeclampsia (PE) is a pregnancy-specific hypertensive disorder which is associated with substantial morbidity and mortality in mothers and their fetuses worldwide1. It also increases the future risk of metabolic and cardiovascular disease later in life2. The pathogenesis of preeclampsia remains not fully elucidated, but the impaired remodeling of uterine spiral artery is believed at the root etiology of this disease3. This causes reduced blood flow to the placenta and leads to an exposure of the developing fetus to an insufficient oxygen and nutrient supply, which contributes to fetal growth restriction in preeclampsia.
Placental trophoblastic transport of amino acids is vital for the growing fetus in protein synthesis, metabolic and biosynthetic processes. Decreased trophoblastic transfer of amino acids is believed to contribute to fetal growth restriction during pregnancy4,5. The L-type amino acid transporter (LAT) is a primary transport system present in the placenta, which is required for the Na+-independent antiport of essential amino acids. The isoform LAT1, as a major accumulative transporter in L-type system, is abundant on the apical surface of syncytiotrophoblast and active in the uptake of specific amino acids from maternal circulation into the placenta6. It has been found that trophoblastic LAT1 expression is increased in women with type 2 diabetes mellitus and associated with birth weight and neonatal fat mass7, but decreased in the placentas from small-for gestational-age (SGA) fetus8. In addition, inhibition of LAT1 transporters is found to be involved in the mouse implantation and placentation by affecting trophoblast differentiation and invasion9,10. Placental transport of amino acids is intensively regulated by mammalian target of rapamycin (mTOR) signaling via modulating translocation or global expression of amino acid transporters in trophoblast cells11. Jansson et al. demonstrated that mTOR mediated amino acid uptake by regulating transporter cell surface abundance in primary human trophoblast cells12. We previously reported that suppression of mTOR signaling down-regulated the expressions of LAT1 and LAT2 in placental trophoblasts13. So far, there are few published data regarding the expression and regulatory mechanism of LAT1 in the placenta from preeclampsia.
Emerging evidence have shown that insufficient or deficient vitamin D status in pregnancy increases the risk of several pregnancy complications including preeclampsia, fetal growth restriction, and preterm birth14. On the contrary, supplementation of vitamin D during pregnancy has been suggested to reduce the risk of preeclampsia15. Although the role for vitamin D in placental trophoblasts has not been fully established, it has been clearly demonstrated that vitamin D exerted anti-inflammatory and anti-oxidative stress properties in placental trophoblasts. In our previous studies, we found that the biologically active 1,25(OH)2D3 significantly suppressed the cyclooxygenase-2 activity and the downstream prostaglandin E2 production in placental trophoblasts in response to hypoxic stimulation16, it also attenuated the oxidative stress-induced microparticle shedding from placental trophoblasts17. Vitamin D is also implicated to involve in trophoblast proliferation and migration18, activation of autophagy19, alleviation of insulin resistance20 in placental trophoblasts. However, there was little data except one study from Southampton Women’s survey showed that maternal 25(OH)D3 and vitamin D binding protein (VDBP) levels were positively related to expression of specific placental amino acid transporters, indicating that vitamin D may be involved in the regulation of placental amino acid transport21.
To further study the beneficial properties of vitamin D in placental trophoblasts, we aimed to investigate if vitamin D-regulated amino acid transporter was involved in the pathophysiology of preeclampsia in the present study. To test this, the expression of LAT1 was determined in human placentas from preeclampsia. Then, the role for vitamin D in placental LAT1 expression and underlying mechanisms was investigated through the exposure of HTR-8/SVneo human placental trophoblast cells to 1,25(OH)2D3 and the hypoxia mimic CoCl2.
Results
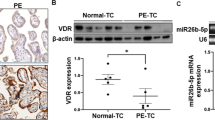
Reduced placental VDR expression is associated with decreased expression of LAT1 in preeclampsia
To explore the involvement of LAT1 in preeclampsia, we first observed the expression of LAT1 in placenta from women with preeclampsia. As shown in Fig. 1A, expressions of LAT1 and VDR were mainly located in the cytoplasm of placental syncytiotrophoblasts. The LAT1 expression was significantly reduced in the placenta from preeclamptic pregnancies compared with that from normotensive pregnancies (Fig. 1B,C). The VDR expression was also decreased in preeclamptic placentas (Fig. 1B,D). Since placental hypoxia is at the root pathology of preeclampsia, we further determined the effects of hypoxia on LAT1 expression in cultured human HTR-8/SVneo trophoblast cells. Our results showed that the expression of LAT1 was dose-dependently decreased when trophoblast cells were cultured with an increasing concentration of CoCl2 compared to untreated cells (Fig. 2A). Similar to the effects of CoCl2 on LAT1, the VDR expression was also reduced in a dose-dependent manner in trophoblast cells treated with CoCl2 (Fig. 2A).
Expression of LAT1 and VDR in normal and preeclamptic placentas. (A) Representative immunostaining images of LAT1 and VDR expressions in tissue sections from normal and preeclamptic (PE) placentas. Bar = 100 micron. (B) Representative blots of LAT1 and VDR expression in placenta from normotensive and PE pregnancies. (C,D) The bar graphs show the relative density of protein expression for LAT1 and VDR after normalization with β-actin expression in each sample. n = 5, #P < 0.05, PE vs Normal.
Effects of 1,25(OH)2D3 and CoCl2 on LAT1 and VDR expression in placental trophoblasts. (A) Protein expression for LAT1 and VDR in HTR-8/SVneo trophoblast cells treated with different concentrations of CoCl2. The bar graphs show relative protein expression for LAT1 and VDR after normalized against β-actin in each sample from three independent experiments. CoCl2 induced a dose-dependent decrease in both LAT1 and VDR expression in trophoblasts. *P < 0.05, **P < 0.01, ***P < 0.001, CoCl2 treated vs control cells. (B) Protein expression for LAT1 and VDR in HTR-8/SVneo trophoblast cells treated with different concentrations of 1,25(OH)2D3. The bar graphs show relative protein expression for LAT1 and VDR after normalization with β-actin in each sample from four independent experiments. In contrast to CoCl2, 1,25(OH)2D3 stimulated LAT1 and VDR expression in a dose-dependent manner in trophoblast cells. #P < 0.05, ##P < 0.01, 1,25(OH)2D3 treated vs control cells.
1,25(OH)2D3 Stimulates LAT1 expression and prevents hypoxia-induced decrease of LAT1 expression in placental trophoblasts
The role for vitamin D on LAT1 expression in placental trophoblasts was investigated using cultured HTR-8/SVneo trophoblast cells which were cultured with 1,25(OH)2D3 in the presence or absence of CoCl2. The results showed that 1,25(OH)2D3 treatment could stimulate both LAT1 and VDR expression in a dose-dependent way in trophoblast cells (Fig. 2B). Moreover, the LAT1 expression was significantly reduced in the cells cultured with 250 μM of CoCl2, and such a CoCl2-induced decrease of LAT1 was significantly prevented when the cells were treated with 100 nM of 1,25(OH)2D3 (Fig. 3A). The LAT1 expression was also examined by immunofluorecent staining in trophoblast cells. Consistent with the Western blot data, 1,25(OH)2D3 could attenuate CoCl2-induced decrease of LAT1 expression in trophoblast cells (Fig. 3B).
Effects of 1,25(OH)2D3 on CoCl2-induced LAT1 expression in placental trophoblasts. (A) Protein expression of LAT1 detected by Western blot in HTR-8/SVneo trophoblast cells treated with CoCl2 in the presence or absence of 1,25(OH)2D3. The decreased LAT1 expression induced by CoCl2 was attenuated when cells were treated with 1,25(OH)2D3. The bar graphs show relative protein expression after being normalized by β-actin in each sample from six independent experiments. **P < 0.01, CoCl2 treated vs control; #P < 0.05, 1,25(OH)2D3 + CoCl2 vs CoCl2 alone. (B) Representative imaging of immunofluorescent staining of LAT1 in trophoblasts treated by CoCl2 with or without 1,25(OH)2D3. Consistent with Western blot results, 1,25(OH)2D3 could prevent CoCl2-induced decreased LAT1 expression in trophoblast cells. (C) Protein expression of VDR and LAT1 expression in trophoblasts treated with VDR siRNA in the presence or absence of 1,25(OH)2D3. Inhibition of VDR expression attenuates 1,25(OH)2D3-stimulated LAT1 expression in trophoblast cells. The bar graphs show relative VDR and LAT1 expression after being normalized by β-actin in each sample from six independent experiments, *P < 0.05, **P < 0.01, treated vs control; ##P < 0.01, ###P < 0.001, 1,25(OH)2D3 + VDR siRNA vs 1,25(OH)2D3 alone.
Then, VDR siRNA silencing was performed to knockdown VDR expression to elucidate the specificity of vitamin D-stimulated LAT1 in trophoblast cells. Our results showed that VDR expression was significantly reduced in the cells transfected with VDR siRNA compared with control cells, and 1,25(OH)2D3 was unable to upregulate VDR expression when the cells were treated with VDR siRNA (Fig. 3C). The LAT1 expression was markedly downregulated in the cells transfected with VDR siRNA, and 1,25(OH)2D3 could not upregulate LAT1 expression when the VDR expression was knocked down in trophoblast cells (Fig. 3C).
1,25(OH)2D3 Upregulates mTOR signaling via VDR in placental trophoblasts
In order to investigate whether mTOR signaling is involved in the vitamin D-regulated LAT1 expression, we determined the effect of 1,25(OH)2D3 on the activity of mTOR signaling using the mTOR downstream effector, 70 kD ribosomal protein S6 kinase 1 (p70S6K1). As shown in Fig. 4A, the expression of phosphorylated p70S6K1 (P-p70S6K1) but not total p70S6K1 was downregulated in the cells cultured with CoCl2, and such a CoCl2-induced decrease of P-p70S6K1 was attenuated by the treatment of 1,25(OH)2D3. Moreover, siRNA silence targeting VDR significantly suppressed the expressions of P-p70S6K1 and total p70S6K1 in trophoblast cells (Fig. 4B).
Effects of 1,25(OH)2D3 on altered mTOR activity induced by CoCl2 in placental trophoblasts. (A) Protein expression for p70S6K1 and P-p70S6K1 in HTR-8/SVneo trophoblast cells treated with CoCl2 in the presence or absence of 1,25(OH)2D3. p70S6K1 is one downstream indicator of mTOR signaling activity, 1,25(OH)2D3 prevented CoCl2-induced decrease of P-p70S6K1 expression in trophoblasts. The bar graphs show relative expression of p70S6K1 and P-p70S6K1 after being normalized by β-actin in each sample from six independent experiments. **P < 0.01, CoCl2 treated vs control; #P < 0.05, 1,25(OH)2D3 + CoCl2 vs CoCl2 alone. (B) Protein expression for p70S6K1 and P-p70S6K1 in HTR-8/SVneo trophoblast cells treated with VDR siRNA in the presence or absence of 1,25(OH)2D3. Inhibition of VDR expression markedly suppressed P-p70S6K1 and total p70S6K1 expression in trophoblasts. The bar graphs show relative expression of p70S6K1 and P-p70S6K1 after being normalized by β-actin in each sample. Data are from four independent experiments. *P < 0.05, VDR siRNA vs control; #P < 0.05, ##P < 0.01, treated vs 1,25(OH)2D3 alone.
Suppression of mTOR pathway downregulates LAT1 expression in placental trophoblasts
We used the specific inhibitor of mTOR signaling, rapamycin, to determine the critical role for mTOR signaling in the regulation of LAT1 expression in trophoblast cells. Treatment of rapamycin significantly inhibited P-p70S6K1 expression in trophoblast cells, suggesting a suppressed activity of mTOR signaling caused by rapamycin. Importantly, LAT1 expression was clearly decreased when the mTOR signaling was inhibited by rapamycin in the presence or absence of CoCl2 (Fig. 5). The P-p70S6K1 and LAT1 expressions were also suppressed in the cells cultured with CoCl2 alone.
Effects of mTOR inhibitor on LAT1 expression in placental trophoblasts. (A) Protein expression of LAT1 in HTR-8/SVneo trophoblast cells treated with mTOR inhibitor rapamycin in the presence or absence of CoCl2. Rapamycin markedly inhibit P-p70S6K1 expression in trophoblast cells. The LAT1 expression was significantly downregulated in cells treated with rapamycin. (B) The bar graphs show relative expression of LAT1 after normalization by β-actin in each sample from six independent experiments. **P < 0.01, ***P < 0.001, treated vs control.
Material and methods
Placenta tissue collection
Human placentas were collected immediately after delivery at the Second Affiliated Hospital of Harbin Medical University. A total of 10 placentas were used in the study, 5 from normal and 5 from preeclamptic pregnancies. Uncomplicated pregnancy is defined as pregnancy with a blood pressure < 140/90 mmHg, and absence of proteinuria, obstetrical and medical complications. Preeclampsia was defined as systolic blood pressure ≥ 140 mmHg or diastolic blood pressure ≥ 90 mmHg with at least two separate readings and coexistence of proteinuria (> 1+) at dipstick or ≥ 300 mg protein/day in urine. Patients complicated with HELLP syndrome, diabetes, and/or renal disease were excluded.
Study approval
Human placenta collection was approved by the Ethical Committee for the Use of Human Samples of Harbin Medical University (82001577). All the participants signed a written informed consent for study enrollment. All the experiments were performed in accordance with the relevant guidelines and regulations of ethics committee of Harbin Medical University.
Human trophoblast cell culture and treatment
The HTR-8/SVneo human trophoblast cell line was obtained from BeNa Cultrue Collection (Beijing, China). The cells were routinely maintained in Dulbecco’s Modified Eagle Media: Nutrient Mixture F-12 (DMEM/F12) medium supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin and incubated under standard conditions of 5% CO2 at 37 °C. 1,25(OH)2D3 was used as bioactive vitamin D, and cobalt chloride (CoCl2) was used as an inducer of hypoxia that causes oxidative stress in placental trophoblasts. HTR-8/SVneo cells were seeded in six-well plates (4 × 105 cells/well) and treated with CoCl2 at concentrations of 10, 50, 100, 250, and 500 μM or with 1,25(OH)2D3 at 10, 50, 100, 250, and 500 nM for 24 h. In the experiment to test the role for vitamin D on hypoxia-induced LAT1 expression, CoCl2 at a concentration of 250 μM and 1,25(OH)2D3 at a concentration of 100 nM were used. At the end of each experiment, total cellular protein or RNA was extracted and used to determine protein expression or mRNA expression.
VDR siRNA transfection
VDR Knockdown was carried out by transfection of VDR siRNA in HTR-8/SVneo cells using Lipofectamine 2000 transfection reagent (Invitrogen, CA, USA) according to the manufacture’s instruction. Briefly, 18 h after seeding, cells were starved with serum-free DMEM/F12 for 2 h and then incubated with Opti-MEM medium for 6 h, which contains 50 nM of VDR siRNAs (GenePharma, Suzhou, China) mixed with Lipofectamine transfection reagent. Cellular protein was collected 48 h after transfection, and protein expression for VDR and LAT1 was then determined by Western blot.
Immunohistochemical staining
Fresh placental tissue was fixed with 10% formalin and embedded in paraffin. Expression of LAT1 and VDR was examined by immunohistochemistry (IHC) staining of paraffin-embedded tissue sections. A standard immunohistochemistry staining procedure was performed as previously described22. Stained slides with the same antibody were all processed at the same time. Stained slides were reviewed under a microscope, and images were captured with a digital scanning microscopy imaging system (PreciPoint, Germany).
Protein expression by western blot
Placental and trophoblastic protein expressions for LAT1, VDR, p70S6K1, and phospho-p70S6K1 were examined by western blot. Antibody against human LAT1 (sc-134994) was purchased from Santa Cruz Biotechnology (CA, USA), antibodies against human VDR (AF6159), p70S6K1 (AF6226), and phospho-p70S6K1 (AF3228) were all obtained from Affinity Biosciences (Jiangsu, China). An aliquot of 10 μg of tissue of cellular protein was subject to electrophoresis. The bound antibody was visualized with an enhanced chemiluminescencent detection kit (Yeasen, Shanghai, China). The bands for LAT1, VDR, and p70S6K1 were detected at 55KD, 48KD and 70KD, respectively. The band density was analyzed by ImageJ software (National Institutes of Health, USA). β-actin expression was determined and used to normalize relative protein expression in each sample.
Immunofluorescent staining
HTR-8/SVneo cells were cultured on glass coverslips in 24-well plates (1 × 105 cells/well) and fixed with ice-cold methanol and permeabilized with Triton X-100. Cells were incubated with primary anti-human LAT1 antibody followed by matched secondary antibody. After staining, coverslips were mounted on glass slides with 30% glycerol with 4,6-diamido-2-phenylindole (DAPI) and reviewed under a fluorescent microscope (Nikon corporation, Tokyo, Japan). Images were captured with a digital camera linked to a computer with imaging software (PreciPoint, Germany).
Data presentation and statistics
All data are presented as mean ± SEM. Statistical analysis was performed with unpaired t test or One-way ANOVA using GraphPad Prism 8 software. A Tukey test was used as post hoc test. A value of P < 0.05 was considered statistically significant.
Discussion
Emerging studies have shown that low maternal vitamin D levels in pregnancy was associated with increased risk of preeclampsia and preterm birth, suggesting that vitamin D deficiency is a risk factor of preeclampsia23,24. However, the underlying mechanisms remain unclear. In the current study, we demonstrate that placental LAT1 expression is reduced in women with preeclampsia, and 1,25(OH)2D3 stimulates LAT1 expression through VDR in cultured placental trophoblasts. Since altered placental transport of amino acids has been implicated in the fetal growth restriction in preeclampsia4, our findings suggest that the role for vitamin D in placental amino acid transporter is one possible mechanism underlying the linkage between maternal vitamin D levels and fetal growth in preeclampsia.
The fetal growth is largely dependent on placental transport of nutrients. The studies mentioned above and other studies have indicated that an altered placental transport of amino acids is intimately associated with fetal growth in pregnancy4,5,25, but controversial data are present regarding the modifications of placental amino acid transport in pregnancy disorders. For example, using IHC, Aiko et al. found that L-type amino acid transporter 4F2hc and LAT1 was increased in placental syncytiotrophoblast from pregnancies complicated by preeclampsia or intrauterine growth restriction (IUGR), suggesting an adaptive response to help maintaining growth in both of pregnancy complications26. Using real-time PCR, Malina et al. reported that mRNA levels for placental A-type amino acid transporters is not different between preeclampsia and normal pregnancies27. Interestingly, Shibata et al. found that A-type amino acid transporter activity was not reduced in the placentas of SGA infants from preeclamptic pregnancies, but significantly reduced in the placenta from SGA pregnancies without preeclampsia28, suggesting that growth restriction in the two disorders may be largely different. A recent study using multiple approaches including bioinformatic analysis, molecular biology, and mathematical diagramming demonstrated that amino acid transporter y+LAT1 was significantly increased in placentas associated with PE, but decreased in IUGR placentas29. This opposite but significant changes between preeclampsia and IUGR may suggest a different function of y+LAT1 in these two diseases. y+LAT1 is responsible for the transport of Na+-independent cationic amino acids, such as lysine, ornithine and arginine30. High expression of placental y+LAT1 could result lowered maternal levels of lysine, ornithine and arginine during pregnancy, which may elicit similar symptoms as reported in women complicated with lysinuric protein intolerance. Nevertheless, the pregnancy with lysinuric protein intolerance has been associated with an increased risk of serious complications, including preeclampsia31.
In the present study, the findings regarding the reduced placental LAT1 expression might be associated with fetal growth restriction observed in preeclampsia. LAT1 is a major Na+ independent transporter for indispensable amino acids, such as lysine, leucine, and histidine et al. Low expression of placental LAT1 associated decreased transport of amino acids across the placenta might cause high maternal plasma amino acid concentrations, which is in agreement with the previous findings as reported by Powers et al.32. The differentially expression patterns of placental LAT1 in preeclampsia in the above studies might result from the differing methods, sample size, or criteria of preeclampsia sample selection.
Information for the regulation of vitamin D on amino acid transport in placental trophoblasts is very scarce. Only Jansson et al. reported that bioactive 1,25-dihydroxy vitamin D3 markedly increased mRNA expression of the A-type isoform SNAT2 and the activity of A-type transporters in primary human placental trophoblasts, but had no effect on L-type and did not affect mTOR signaling33. On the contrary, our results showed that 1,25-dihydroxy vitamin D3 could significantly stimulate the protein expression of L-type transporter LAT1 under normoxic condition in cultured placental trophoblasts, and more importantly attenuate the decrease of LAT1 caused by CoCl2-induced hypoxia. We further demonstrated that knockdown of VDR by siRNA silencing prevented 1,25-dihydroxy vitamin D3-stimulated LAT1 expression in placental trophoblasts. Our findings suggest that vitamin D-stimulated LAT1 may make up for the shortage of decreased placental LAT1 in preeclampsia, which helps to maintain fetal growth. Those opposite findings between our and Jasson’s study indicate that the role for vitamin D-regulated LAT1 in preeclampsia should be further investigated.
Regarding the role for vitamin D in mTOR signaling, we found a similar trend as the vitamin D-regulated LAT1 that 1,25-dihydroxy vitamin D3 could prevent CoCl2-induced decrease of mTOR activity. However, 1,25-dihydroxy vitamin D3 had no effect on mTOR signaling in placental trophoblasts under normoxic condition, which was consistent with the results in Jasson’s study33. The VDR knockdown produced by siRNA silencing blocked the effects of vitamin D on CoCl2-induced mTOR activity. Since the mTOR signaling is directly implicated in the expression of LAT1 which is also confirmed in the present study, we propose that vitamin D-stimulated LAT1 might be mediated by mTOR signaling in placental trophoblasts. There was a study revealed that the role for vitamin D in mTOR signaling involved occupancy of vitamin D response elements (VDREs) of the gene for DNA-damage-inducible transcript 4 (DDIT4), an inhibitor of mTOR signaling34. They found that DDIT4 was a direct target for 1,25-dihydroxy vitamin D3 and could be induced by 1,25-dihydroxy vitamin D3 treatment in osteoblasts, which might be resulted from the competition for the binding to DDIT4 gene promoter by VDR and VDRE binding protein34.
In the past decade, several novel vitamin D metabolites including 20(OH)D3, 22(OH)D3, 20,22(OH)2D3 and 20,23(OH)2D3 were recognized in placenta, adrenal glands, and epidermal keratinocytes, suggesting 1,25(OH)2D3 is not solely bioactive form of vitamin D in vivo35,36. And their action may not through the genomic site of the VDR37. However, the biological functions of these vitamin D hydroxyderivatives in placenta and preeclampsia are still unknown (Supplementary Figures).
In summary, our study shows that placental LAT1 expression is reduced in preeclamptic pregnancies and vitamin D stimulated-LAT1 expression may be mediated by mTOR signaling in placental trophoblasts. Our results provide evidence for the vitamin D supplementation during pregnancy may be beneficial for reducing the risk of fetal growth restriction observed in preeclampsia probably by increasing essential amino acid transport across the placenta.
Data availability
The datasets are available from the corresponding author on reasonable request.
References
Phipps, E. et al. Preeclampsia: Updates in pathogenesis, definitions, and guidelines. Clin. J. Am. Soc. Nephrol. 11, 1102–1113 (2016).
Wu, P. et al. Preeclampsia and future cardiovascular health: A systematic review and meta-analysis. Circ. Cardiovasc. Qual. Outcomes. https://doi.org/10.1161/CIRCOUTCOMES.116.003497 (2017).
Redman, C. W. & Sargent, I. L. Latest advances in understanding preeclampsia. Science 308, 1592–1594 (2005).
Brett, K. E. et al. Maternal-fetal nutrient transport in pregnancy pathologies: The role of the placenta. Int. J. Mol. Sci. 15, 16153–16185 (2014).
Desforges, M. & Sibley, C. P. Placental nutrient supply and fetal growth. Int. J. Dev. Biol. 54, 377–390 (2010).
Zaugg, J. et al. Small molecule inhibitors provide insights into the relevance of LAT1 and LAT2 in materno-foetal amino acid transport. J. Cell Mol. Med. 24, 12681–12693 (2020).
Castillo-Castrejon, M. et al. Effect of type 2 diabetes mellitus on placental expression and activity of nutrient transporters and their association with birth weight and neonatal adiposity. Mol. Cell Endocrinol. 532, 111319 (2021).
Chen, Z. et al. PPARgamma stimulates expression of L-type amino acid and taurine transporters in human placentas: The evidence of PPARgamma regulating fetal growth. Sci. Rep. 5, 12650 (2015).
Wang, X. et al. Positive regulation of placentation by L-amino acid transporter-1 (lat1) in pregnant mice. Int. J. Clin. Exp. Pathol. 10, 9551–9558 (2017).
Chrostowski, M. K. et al. Role of the L-amino acid transporter-1 (LAT-1) in mouse trophoblast cell invasion. Placenta 31, 528–534 (2010).
Roos, S. et al. Regulation of placental amino acid transporter activity by mammalian target of rapamycin. Am. J. Physiol. Cell Physiol. 296, C142–C150 (2009).
Rosario, F. J. et al. Mammalian target of rapamycin signalling modulates amino acid uptake by regulating transporter cell surface abundance in primary human trophoblast cells. J. Physiol. 591, 609–625 (2013).
Xu, J. et al. Downregulation of placental amino acid transporter expression and mTORC1 signaling activity contributes to fetal growth retardation in diabetic rats. Int. J. Mol. Sci. 21, 1849 (2020).
Kiely, M. E. et al. Vitamin D in pregnancy: Where we are and where we should go. J. Steroid Biochem. Mol. Biol. 201, 105669 (2020).
Khaing, W. et al. Calcium and vitamin D supplementation for prevention of preeclampsia: A systematic review and network meta-analysis. Nutrients 9, 1141 (2017).
Cao, Y. et al. Vitamin D stimulates miR-26b-5p to inhibit placental COX-2 expression in preeclampsia. Sci. Rep. 11, 11168 (2021).
Xu, J. et al. Vitamin D reduces oxidative stress-induced procaspase-3/ROCK1 activation and MP release by placental trophoblasts. J. Clin. Endocrinol. Metab. 102, 2100–2110 (2017).
Westwood, M. et al. Vitamin D attenuates sphingosine-1-phosphate (S1P)-mediated inhibition of extravillous trophoblast migration. Placenta 60, 1–8 (2017).
Pi, Y. et al. Vitamin D alleviates hypoxia/reoxygenation-induced injury of human trophoblast HTR-8 cells by activating autophagy. Placenta 111, 10–18 (2021).
Li, G. et al. 1,25(OH)2D3 protects trophoblasts against insulin resistance and inflammation via suppressing mTOR signaling. Reprod. Sci. 26, 223–232 (2019).
Cleal, J. K. et al. Placental amino acid transport may be regulated by maternal vitamin D and vitamin D-binding protein: Results from the Southampton Women’s Survey. Br. J. Nutr. 113, 1903–1910 (2015).
Xu, J. et al. Reduced CD200 expression is associated with altered Th1/Th2 cytokine production in placental trophoblasts from preeclampsia. Am. J. Reprod. Immunol. 79, e12763 (2018).
Mirzakhani, H. et al. Early pregnancy vitamin D status and risk of preeclampsia. J. Clin. Investig. 126, 4702–4715 (2016).
Agarwal, S. et al. Vitamin D and its impact on maternal-fetal outcomes in pregnancy: A critical review. Crit. Rev. Food Sci. Nutr. 58, 755–769 (2018).
Bell, A. W. & Ehrhardt, R. A. Regulation of placental nutrient transport and implications for fetal growth. Nutr. Res. Rev. 15, 211–230 (2002).
Aiko, Y. et al. Differential levels of amino acid transporters system L and ASCT2, and the mTOR protein in placenta of preeclampsia and IUGR. BMC Pregn. Childbirth 14, 181 (2014).
Malina, A. et al. Placental system A transporter mRNA is not different in preeclampsia, normal pregnancy, or pregnancies with small-for-gestational-age infants. Hypertens. Pregn. 24, 65–74 (2005).
Shibata, E. et al. Placental system A amino acid transport is reduced in pregnancies with small for gestational age (SGA) infants but not in preeclampsia with SGA infants. Placenta 29, 879–882 (2008).
Huang, X. et al. Identification of placental nutrient transporters associated with intrauterine growth restriction and pre-eclampsia. BMC Genomics 19, 173 (2018).
Fotiadis, D. et al. The SLC3 and SLC7 families of amino acid transporters. Mol. Aspects Med. 34, 139–158 (2013).
Mikolajek-Bedner, W. et al. Pregnancy delivery and puerperium in a patient with lysinuric protein intolerance—A case report. Ginekol. Pol. 84, 654–656 (2013).
von Versen-Hoeynck, F. M. & Powers, R. W. Maternal-fetal metabolism in normal pregnancy and preeclampsia. Front. Biosci. 12, 2457–2470 (2007).
Chen, Y. Y. et al. 1,25-Dihydroxy vitamin D3 stimulates system A amino acid transport in primary human trophoblast cells. Mol. Cell Endocrinol. 442, 90–97 (2017).
Lisse, T. S. et al. Gene targeting by the vitamin D response element binding protein reveals a role for vitamin D in osteoblast mTOR signaling. FASEB J. 25, 937–947 (2011).
Slominski, A. T. et al. In vivo evidence for a novel pathway of vitamin D(3) metabolism initiated by P450scc and modified by CYP27B1. FASEB J. 26, 3901–3915 (2012).
Slominski, A. T. et al. Detection of novel CYP11A1-derived secosteroids in the human epidermis and serum and pig adrenal gland. Sci. Rep. 5, 14875 (2015).
Slominski, A. T. et al. Characterization of a new pathway that activates lumisterol in vivo to biologically active hydroxylumisterols. Sci. Rep. 7, 11434 (2017).
Acknowledgements
This work was supported by National Natural Science Foundation of China (82001577), University Nursing Program for Young Scholars with Creative Talents in Heilongjiang Province (UNPYSCT-2018054), China Postdoctoral Fund (2018M640304), Heilongjiang Postdoctoral Foundation (LBH-Z18108).
Author information
Authors and Affiliations
Contributions
J.X. and H.Z. conceived and designed the experiments; X.J. and Y.C. performed the experiments; X.J. and L.Y. analysed the data; X.L. and Y.H. contributed reagents/materials/analysis tools; X.Y. collected the placentas and clinical information of study subjects; J.X. wrote the manuscript. C.L. and H.Z. revised the manuscript. All authors reviewed and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jia, X., Cao, Y., Ye, L. et al. Vitamin D stimulates placental L-type amino acid transporter 1 (LAT1) in preeclampsia. Sci Rep 12, 4651 (2022). https://doi.org/10.1038/s41598-022-08641-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-08641-y
- Springer Nature Limited