Abstract
Endothelial glycocalyx (EG) shedding is associated with septic shock and described following intravenous (IV) fluid administration. To investigate the possible impact of IV fluids on the pathobiology of septic shock we investigated associations between biomarkers of EG shedding and endothelial cell activation, and relationships with IV fluid volume. Serum samples were obtained on admission (T0) and at 24 h (T24) in patients undergoing haemodynamic resuscitation for suspected septic shock in the emergency department. Biomarkers of EG shedding—Syndecan-1 (Syn-1), Syndecan-4 (Syn-4), Hyaluronan, endothelial activation—Endothelin-1 (ET-1), Angiopoeitin-2 (Ang-2), Vascular Endothelial Growth Factor Receptor-1(VEGF-1) and leucocyte activation/inflammation—Resistin, Neutrophil Gelatinase Associated Lipocalin (NGAL) and a marker of cardiac stretch—Pro-Atrial Natriuretic Peptide (Pro-ANP) were compared to the total IV fluid volume administered using Tobit regression. Data on 86 patients (52 male) with a mean age of 60 (SD 18) years were included. The mean fluid volume administered to T24 was 4038 ml (SD 2507 ml). No significant association between fluid volume and Pro-ANP or any of the biomarkers were observed. Syn-1 and Syn-4 were significantly correlated with each other (Spearman Rho 0.43, p < 0.001) but not with Hyaluronan. Syn-1 and Syn-4 both correlated with VEGFR-1 (Rho 0.56 and 0.57 respectively, p < 0.001) whereas Hyaluronan correlated with ET-1 (Rho 0.43, p < 0.001) and Ang-2 (Rho 0.43, p < 0.001). There was no correlation between Pro-ANP and any of the EG biomarkers. Distinct patterns of association between biomarkers of EG shedding and endothelial cell activation were observed among patients undergoing resuscitation for sepsis. No relationship between IV fluid volume and Pro-ANP or any of the other biomarkers was observed.
Similar content being viewed by others
Introduction
Intravenous (IV) fluid resuscitation is the first line treatment for the haemodynamic resuscitation of patients with septic shock, with international consensus guidelines recommending initial resuscitation with 30 ml/kg of isotonic crystalloid over the first three hours1. There is emerging evidence of harm associated with excess fluid balance in sepsis patients2,3,4 and clinical trials in some populations have demonstrated better patient outcomes with a fluid-restricted approach5,6. Consequently, there is clinical uncertainty about the optimal dosing of IV fluid and the associated timing of vasopressor initiation7, and there is substantial variation in practice8.
One suggested mechanism for harm with IV fluids is shedding of the endothelial glycocalyx (EG) layer due to hypervolaemia and/or haemodilution9,10. The EG is a 0.5 μm thick gel-like layer of proteoglycans anchored to the luminal endothelial surface, with glycosaminoglycan (GAG) molecules incorporated throughout the structure. Syndecans are membrane-bound proteoglycans on the apical surface of endothelial cells to which several different GAGs such as Heparan Sulfate and Chondroitin Sulfate attach covalently to form the EG layer. In contrast, the GAG Hyaluronan is not attached to Syndecan but to cell surface CD44 or other GAG molecules11,12,13. In settings such as inflammation, cleavage of the Syndecan ectodomain by sheddases such as matrix metalloproteinases (MMPs) delivers the soluble component to the circulation14.
The EG is a key determinant of fluid homeostasis as well as maintaining inflammatory and haemostatic quiescence in health15. EG shedding occurs in response to both acute and chronic inflammation and elevation of blood Syndecan-1 (Syn-1) and Hyaluronan concentration has been described in patients with sepsis16,17. It has been suggested that EG shedding leads to fluid extravasation and propagation of the systemic inflammatory response in sepsis with resultant worsening organ dysfunction and shock9,18,19,20. However, recent preclinical studies have challenged whether EG shedding results in increased vascular permeability21,22. Additionally, it is unclear whether exogenous fluids have a direct effect on the EG, or whether this is mediated via MMPs activated by natriuretic peptides released in response to cardiac chamber dilatation9,23,24.
While it is well established that increased biomarkers of EG degradation are associated with illness severity and mortality in sepsis, data from patients undergoing fluid resuscitation have shown variable associations between IV fluid volume and concentrations of individual EG biomarkers17,25,26. Further, studies in healthy subjects and perioperative settings may not be relevant in settings such as sepsis where EG shedding is already established27. The question of the incremental effect of exogenously administered fluids on an already injured EG is unresolved.
While clinical uncertainty remains, it is important to test the hypothesis that IV fluid volume is detrimental in sepsis via the effect of EG shedding. The objective of this study was to examine the relationship between the volume of IV fluid administered during the first 24 h from commencement of treatment in the emergency department (ED) and soluble biomarkers of EG shedding, endothelial activation and inflammation in adult patients with suspected septic shock. The secondary objective was to explore associations between EG biomarkers and biomarkers of endothelial cell activation and inflammation after fluid resuscitation.
Methods
Participants and setting
We undertook secondary analysis of a convenience sample of adult (age 18 years or older) ED patients undergoing haemodynamic resuscitation for suspected septic shock recruited into two observational studies one in Perth, Australia between 2010 and 2015, the other in Boston, USA between 2012 and 2018. The methodologies of each study have previously been described28,29. The inclusion criteria were (1) the presence two or more systemic inflammatory response (SIRS) criteria and (2) a clinical decision to administer IV antibiotics for suspected infection and (3) a systolic blood pressure of < 90 mmHg despite at least 1 L of IV fluid along with either (a) blood lactate > 2 mmol/L or (b) a requirement for vasopressors to maintain perfusion targets. These criteria reflect an operational definition for septic shock. Recruitment took place according to the Sepsis 2 criteria which were based upon SIRS. The Sepsis 3 criteria introduced in 2016 placed greater importance on lactate and vasopressor requirement to define septic shock. All of these patients were undergoing resuscitation for hypotension/hypoperfusion related to infection in the ED and thus represent a clinically relevant population in terms of the research question.
Data collection
Blood samples were collected on recruitment in the ED (T0), and subsequently 24 h later (T24). Samples were processed within 2 h of collection, with serum collected by centrifugation at 3000 rpm for 10 min, followed by storage of 0.5 ml aliquots at − 80 °C. Clinical data included baseline demographics, vital signs, laboratory results, suspected source of infection, Sequential Organ Failure Assessment (SOFA) scores, Charlson Comorbidity Score (CCS), admission to the intensive care unit (ICU), length of hospital stay and in-hospital mortality. Fluid volumes were calculated as the total administered IV, prior to each time point (crystalloid, colloid, blood products), including prehospital fluids, but excluding drug infusions or flushes.
Biomarker analyses
Biomarker analyses were determined by enzyme-linked immunosorbent assay (ELISA). Selected EG biomarkers were Syndecan-1 (Syn-1), Syndecan-4 (Syn-4) and Hyaluronan. Selected endothelial cell activation biomarkers measured were Endothelin-1 (ET-1), Angiopoietin-2 (Ang-2) and Vascular Endothelial Growth Factor Receptor-1 (VEGFR-1). We also measured a biomarker of cardiac chamber stretch, Pro-Atrial Natriuretic Peptide (Pro-ANP), and the leucocyte activation biomarkers Resistin and Neutrophil Gelatinase-Associated Lipocalin (NGAL). These biomarkers were selected based upon previous work by our groups30,31,32. All analytes except ET-1 were assayed using R&D ‘DuoSet’ Enzyme Linked Immunosorbent Assays (ELISA), (R&D Systems Inc., MN, USA). Each kit was individually optimised prior to use, to achieve average intraplate coefficient of variations (CVs) as follows: Syn-1 5.14%, Syn-4 4.39%, Hyaluronan 8.59%, Ang-2 5.40%, VEGFR-1 8.31%, Pro-ANP 5.68%, Resistin 5.09% and NGAL 4.58%. ET-1 was assayed using a fully validated Quantikine ELISA kit (R&D Systems, MN, USA) in accordance with manufacturer’s instructions. All biomarker analyses were undertaken in the laboratory of the Centre for Clinical Research in Emergency Medicine in Perth, Australia.
Statistical analysis
We measured the relationship between total cumulative fluid volume and biomarkers at T0 and T24 and the change in value of each biomarker between T0 and T24, and with the cumulative total volume at T24. The relationship between pairs of biomarker concentrations at T24 was analysed by Spearman correlation. Because of censoring of some biomarker results both below and above the limit of assay detection, repeated measures Tobit regression was employed with an interaction of time and fluids to test for an association between cumulative fluid volume at T24 and the change over time in the biomarker. For biomarker variables with no censored values, random effects linear mixed models were used. Regression analyses are dependent on assumptions, but these are specific to each regression not to the data set as a whole. All biomarker concentrations were log transformed for multiple reasons. The first is that none of the biomarkers were normally distributed (for censored distributions this relates to the values between the limits) and while this is not an assumption of regression, but rather normality of the residuals, the latter generally occurs if the former is true. Secondly the log transformation can often solve the problem of heteroscedasticity if present. When the transformation did not successfully address these issues, bootstrapping was employed. This resampling technique produces standard errors and p values that are robust to the violation of assumptions.
Associations between the cumulative fluid volume and biomarker concentrations were examined for possible non-linearity using scatterplots with a Lowess fit as well as multivariable regression spline models. Where non-linearity was detected, piecewise Tobit or linear regressions were used to estimate multiple slopes in the one model. All regression models were adjusted for age, sex, baseline mean arterial pressure, baseline lactate, SOFA score, CCS, suspected source of infection (respiratory, urinary, other) and recruitment site. Where violations of assumptions were detected, regression analyses were boot-strapped to produce robust standard errors and p values. The analyses were performed using Stata v15 (College Station, TX, USA). Given the exploratory nature of the study p values were not adjust for multiple testing.
Ethics approval and consent to participate
The study was approved by the Human Research Ethics Committee of Royal Perth Hospital and the Institutional Review Board of Beth Israel Deaconess Medical Center. Informed consent was obtained from the participant or their next of kin, or under a waiver of consent for low-risk observational research in accordance with local laws.
Results
Of 86 adult participants recruited, 11 (13%) died within 30 days. The characteristics of the participants are described in Table 1.
The mean cumulative total fluid volume administered prior to T0 was 2330 ml (SD 1698 ml) and to T24 was 4073 ml (SD 2507 ml). A vasopressor infusion was commenced in 55 (64%) participants within 24 h. The predominant fluid administered was isotonic crystalloid. Additionally, fourteen patients received colloid (12 synthetic gelatin solution, 2 albumin) and one received blood. Two participants received more than 10L of IV fluid (12L and 14L respectively) within the first 24 h.
The median concentration of each biomarker at the two sampling time points are shown in Table 2. Syn-1 and Ang-2 significantly increased over time, whereas Syn-4, ET-1 and NGAL significantly decreased over time. There were no significant differences detected between time points for the remaining biomarkers. No significant difference in the EG shedding biomarkers (Syn-1, Syn-4 and Hyaluronan) was found at either time point between those with SOFA Renal < 3 vs > = 3, or across source of infection groups (respiratory, urinary, other). Both Syn-1 and Hyaluronan were significantly higher among those admitted to ICU at both time points, compared to those not admitted to ICU, but not between those who were ventilated and those who were not (Supplementary data file).
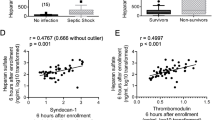
No significant relationship was observed between the biomarkers and the cumulative fluid volumes at T0 or at T24. No significant interaction between fluid volume at T24 and the change over time was found for any of the biomarkers. Figure 1 demonstrates the relationship between the value of each biomarker at T24 and the cumulative total fluid volume, adjusted for baseline confounding variables. Following the removal of two outlier values of fluid volume, a sensitivity analysis was conducted to determine whether these were masking a potential association between fluid volume and the measured biomarkers (Fig. 2). Again, there was no significant association found between any of the biomarkers and the fluid volume administered.
Sensitivity analysis (excluding two outlier cases) of relationship between volume of intravenous fluid and each biomarker at T24 adjusted for age, sex, mean arterial blood pressure, lactate, Charlson score, SOFA score, source of infection and recruitment site. Circles – data points, Dots – censored values.
Table 3 shows the results of Spearman correlations between each of the biomarkers at T24. Syn-1 and Syn-4 were significantly correlated with each other but not with Hyaluronan. Syn-1 and Syn-4 both correlated with VEGRF-1 whereas Hyaluronan correlated with ET-1 and Ang-2 Hyaluronan was also weakly correlated with the inflammatory biomarkers, Resistin and NGAL. Similar patterns were observed for biomarkers measured at T0 (data not shown). There was no correlation between Pro-ANP and any of the EG biomarkers.
Discussion
Statement of principal findings
In this observational study of adult patients with clinically suspected septic shock undergoing fluid resuscitation in two emergency departments, no relationship between volume of intravenous fluid and concentrations of biomarkers of EG shedding or endothelial activation was found. We did observe two distinct patterns of potential EG shedding; a significant association between Syn-1/Syn-4 and VEGFR-1; and between Hyaluronan and Ang-2, ET-1, Resistin and NGAL. There was no observed correlation between Syn1/Syn4 and Hyaluronan, nor between Pro-ANP and any of other EG biomarkers.
Strengths and weaknesses of the study
Strengths of the study include recruitment during the initial resuscitation phase of suspected septic shock. Limitations include its observational design which, despite controlling for variables that would be expected to influence the volume of fluid administered, residual confounding cannot be discounted. The large number of censored values for Hyaluronan (22/172 censored observations at T24) and VEGFR-1 (71/166 censored observations at T24) reduced the power to detect significant differences for these biomarkers. Measurements were limited to two time points at T0 and T24 and therefore would not capture the biomarker profiles during the first 6 h of resuscitation when the largest volumes of fluid are typically administered. The nature of real-world clinical research among acutely sick patients in the emergency setting is that many patients had treatment initiated prior to enrolment, even pre-hospital. The analyses chosen take account of this by describing the fluid administered prior to T0. The biomarkers selected were not a comprehensive suite of all molecules released by EG shedding and endothelial cell activity27. Moreover, soluble biomarkers are a surrogate measure of EG shedding and their utility as an accurate quantitative measure of this process has been questioned in a recent literature review27. The impact upon the expression of EG from vasopressors is unclear. In mitigation of this, the SOFA score does capture the use of vasopressors, and we adjusted for SOFA in the regression analyses. Due to the retrospective nature of this analysis, we were unable to perform direct estimation of EG thickness (for example by intravital sublingual microscopy) which would have provided a more comprehensive assessment33. Finally, the small number of participants precluded any exploration of potentially important differences in the effect of fluid types on the EG (e.g., balanced vs unbalanced crystalloids, synthetic colloids, or albumin)15 and patient subgroups. Given these factors, the lack of any demonstrated positive associations between fluid volume and EG shedding does not necessarily imply that such associations do not exist.
Strengths and weaknesses in relation to other studies
We did not observe any association between IV fluid volume, Pro-ANP or any biomarker of EG shedding, endothelial activation of inflammation. These clinical observations are in contrast to experimental and preclinical studies which have found evidence of EG shedding associated with administration of exogenous fluids, with some studies also showing a concurrent rise in Pro-ANP10,18,34. In patients with sepsis in the ED, Smart et al.described a positive association between fluid volume and hyaluronan during the first 3 h of resuscitation however no such relationship was seen for Syn-1 or Syn-417. Hippensteel et al.reported an association between the volume of IV fluid and the expression of Heparan Sulfate (measured by mass spectroscopy) after 6 h of resuscitation in patients with sepsis25. The timing of sampling in our study prevents direct comparison with these studies.
In a randomised trial comparing a fluid restricted resuscitation strategy versus a liberal fluid regimen for resuscitation among patients with septic shock, Soaraya et al.found Syn-1 levels at 6 h to be lower with the limited fluid regimen (geometric mean ratio of 0.82 (95% CI 0.66–1.02), p = 0.07)26. In a multicentre ICU study of 619 patients there was no relationship between fluid volume in the first day of admission and Syn-1 among patients with sepsis, although Syn-1 was associated with illness severity and mortality35. Similarly, in a prospective study of 175 patients with sepsis, Puskarich et al.found that high Syn-1 levels were associated with increased renal injury and mortality but there was no significant difference in the volume of IV fluids administered between two groups categorised according to the level of Syn-136. In contrast, Soraya et al. found that Syn-1 measured at the time of admission to the ICU was associated with subsequent fluid requirements, as well as with organ failure and mortality37. This raises the question of whether an association between elevated EG biomarkers and fluid volume simply represents larger fluid resuscitation requirements in patients with a leaky vasculature rather than fluid induced EG shedding. Further complicating the issue are recent preclinical experimental studies which have challenged whether there is any relationship between EG shedding and increased vascular permeability21,22,38.
The hypothesis of a relationship between fluid volume and EG degradation is supported by a study by Pouska et al. (n = 66) which demonstrated lasting changes in the perfusion boundary region (a surrogate measure of EG thickness assessed by intravital sublingual microscopy) following IV volume loading in sepsis patients39. There was a transient increased in PBR in elective surgical patients which resolved. A similar effect was seen in septic shock patients, however this persisted at least 120 min after the infusion. Whether this is a direct effect or mediated by the effect of ANP on MMPs is not clear9. Our results are consistent with those of Hippensteel et al.who found no significant relationship between ANP and Heparan sulfate, interleukin-6 levels (a marker of acute inflammation) or mortality25. This, however, may be explained by timing of sampling in different studies. Belavic et al. demonstrated an early rise in ANP along with Syn-1 after administration of IV fluids in surgical patients40. Possible explanations for these discordant results could be due to insufficient volume administered to generate cardiac stretch to increase expression. Alternatively, unlike euvolemic perioperative patients, sepsis patients may have absolute or relative hypovolaemia at baseline.
Of interest is the potential discordant patterns of EG shedding and associations with the endothelial activation biomarkers. Release of Hyaluronan occurs via incompletely understood mechanisms but includes cleavage of the CD44 molecule by MMP-15, as well as by Hyaluronidase and reactive oxygen species41. These distinct mechanisms may explain the unexpected finding of a lack of correlation between the Syndecans and Hyaluronan in this study. Replication of this discordance in further studies is an important first step.
There are other reasons for EG biomarkers to increase in circulation that is not specifically related to EG shedding27. The conditions of sepsis may upregulate endothelial Syn-1 and Syn-4 expression, which may contribute to circulating concentrations42. Further, serum Hyaluronan concentration may be influenced by interstitial washout of tissue Hyaluronan secondary to fluid loading, which may confound the temporal pattern of EG shedding of Hyaluronan. Others have found that Hyaluronan peaks within the first 3–6 h of treatment17,43, and therefore this would not be captured at the time points studied. The lack of increase in Hyaluronan at 24 h suggests that an earlier peak may have been missed. These differences may be due to various mechanisms of shedding, sources of EG biomarkers and temporal patterns and explain why relationships between individual EG biomarkers cannot be established at isolated time points.
There was an association between both Syndecan types and VEGFR-1. This molecule, also known as sFlt-1, has previously been found to be associated with organ failure and an accurate predictor of mortality among ED patients with sepsis44. In contrast Hyaluronan was not associated with VEGRF-1 but was associated with ET-1 and Ang-2. This finding supports recent data which suggests Ang-2 plays a role in Hyaluronan shedding41. It was also interesting to note the positive correlation between Hyaluronan and inflammatory biomarkers; fragmented Hyaluronan is known to be pro-inflammatory and may have driven this association45. When examining associations between biomarkers reported in this study, it is important to note that similar associations were identified at T0 and T24. Glycocalyx shedding and endothelial activation is likely on a continuum prior to, and during, treatment of septic shock, and that, despite attempts to adjust for confounding, this conceals any modification by the volume of resuscitation.
Syn-4 has been studied less than Syn-1 as a biomarker of EG shedding. In one study of patients with pneumonia, Syn-4 levels trended downwards over time, as observed in the present study, and had a negative correlation with illness severity46. Another study found no difference in Syn-4 concentrations between critically ill ICU patients and healthy controls47. The correlation between Syn-1 and Syn-4 is consistent with increased endothelial expression of these glycosaminoglycans in response to circulating cytokines in systemic inflammation in the setting of critical illness14. The discordant patterns between T0 and T24 may reflect downregulation of Syn-4 by Syn-1, as has been demonstrated in vitro48.
Meaning of the study and clinical implications
The clinical importance of this study relates to concerns about a possible detrimental effect of exogenously administered fluids, particularly in the setting of septic shock20. The question of the optimal approach to haemodynamic resuscitation in septic shock is important due to the high morbidity and mortality of this condition. A clinical trial in African children with sepsis and hypoperfusion found that fluid resuscitation increased mortality due to shock6,49. In a preclinical ovine sepsis model, resuscitation with intravenous fluids resulted in a larger subsequent dose of vasopressor to maintain a target blood pressure18. EG shedding is one of several mechanisms by which exogenous fluid resuscitation may be detrimental in septic shock. While emerging evidence favours a fluid-sparing or de-resuscitation fluid strategy among ICU patients with sepsis4, there is uncertainty about the optimal approach to initial haemodynamic resuscitation. Our findings do not support the hypothesis that IV fluid resuscitation exacerbates EG shedding in sepsis. Indeed, recent clinical observational studies have suggested that early intravenous fluid resuscitation is associated with reduced odds of mortality among ED patients with septic shock50,51. Thus, an overly restrictive fluid strategy in sepsis resuscitation due to fears of causing EG injury may be unwarranted. This is the subject of several active clinical trials52,53,54.
Unanswered questions and future research
In addition to the impact of IV fluids, further research is required to confirm the apparent finding of different patterns of EG shedding and the implications for clinical practice. The significant heterogeneity of sepsis may make a straightforward answer elusive. Indeed, it is possible that the inability to reproduce the findings of preclinical and healthy volunteer studies in the clinical setting likely reflects the fact that EG shedding is already established in critical illness. Novel therapies directed at restoring the shed glycocalyx is one avenue of current investigation55. While the results of clinical trials are awaited, a judicious approach to fluid administration which aims to protect the EG remains prudent15.
In conclusion, distinct patterns of association between biomarkers of EG shedding and endothelial cell activation were observed among patients undergoing resuscitation for sepsis. We did not find evidence to support the hypothesis that degradation of the EG is exacerbated by higher resuscitation fluids volumes in patients with septic shock.
Data availability
The datasets used in the analyses reported in this paper are available from the corresponding author on reasonable request.
References
Rhodes, A. et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock: 2016. Intensive Care Med. 43(3), 304–377 (2017).
Acheampong, A. & Vincent, J. L. A positive fluid balance is an independent prognostic factor in patients with sepsis. Crit. Care 19, 251 (2015).
Sirvent, J. M., Ferri, C., Baro, A., Murcia, C. & Lorencio, C. Fluid balance in sepsis and septic shock as a determining factor of mortality. Am. J. Emerg. Med. 33(2), 186–189 (2015).
Silversides, J. A. et al. Conservative fluid management or deresuscitation for patients with sepsis or acute respiratory distress syndrome following the resuscitation phase of critical illness: a systematic review and meta-analysis. Intensive Care Med. 43(2), 155–170 (2017).
Andrews, B. et al. Effect of an early resuscitation protocol on in-hospital mortality among adults with sepsis and hypotension. JAMA 318(13), 1233–1240 (2017).
Maitland K. KS, Opoka R.O., Engoru C., Olupot-Olupot P., Akech S.O., Nyeko R., Mtove G., Reyburn H., Lang T., Brent B., Evans J.A., Tibenderana J.K., Crawley J., Russell E.C., Levin M., Babiker A.G., Gibb D.M. & On behalf of the FEAST trial group. Mortality after fluid olus in Africal children with severe infection. N. Engl. J. Med. 364(26), 2483–2495 (2011).
Lat, I., Coopersmith, C.M., De Backer, D., Research Committee of the Surviving Sepsis C, Members of the Surviving Sepsis Campaign Research Committee contributing to this article are as f, Co-chair AGA, et al. The surviving sepsis campaign: fluid resuscitation and vasopressor therapy research priorities in adult patients. Crit. Care Med. 49(4), 623–635 (2021).
Keijzers, G. et al. The Australasian Resuscitation In Sepsis Evaluation: Fluids or vasopressors in emergency department sepsis (ARISE FLUIDS), a multi-centre observational study describing current practice in Australia and New Zealand. Emerg. Med. Aust. 32(4), 586–598 (2020).
Chappell, D. et al. Hypervolemia increases release of atrial natriuretic peptide and shedding of the endothelial glycocalyx. Crit. Care 18(5), 538 (2014).
Smart, L. et al. Large-volume crystalloid fluid is associated with increased hyaluronan shedding and inflammation in a canine hemorrhagic shock model. Inflammation 41(4), 1515–1523 (2018).
Fraser, J. R., Laurent, T. C. & Laurent, U. B. Hyaluronan: its nature, distribution, functions and turnover. J. Intern. Med. 242(1), 27–33 (1997).
Uchimido, R., Schmidt, E. P. & Shapiro, N. I. The glycocalyx: a novel diagnostic and therapeutic target in sepsis. Crit. Care 23(1), 16 (2019).
Reitsma, S., Slaaf, D. W., Vink, H., van Zandvoort, M. A. & oude Egbrink, M. G. The endothelial glycocalyx: composition, functions, and visualization. Pflugers Arch. 454(3), 345–359 (2007).
Gopal, S. Syndecans in Inflammation at a Glance. Front Immunol. 11, 227 (2020).
Milford, E. M. & Reade, M. C. Resuscitation fluid choices to preserve the endothelial glycocalyx. Crit. Care 23(1), 77 (2019).
Anand, D., Ray, S., Srivastava, L. M. & Bhargava, S. Evolution of serum hyaluronan and syndecan levels in prognosis of sepsis patients. Clin. Biochem. 49(10–11), 768–776 (2016).
Smart, L. et al. Endothelial glycocalyx biomarkers increase in patients with infection during Emergency Department treatment. J. Crit. Care 42, 304–309 (2017).
Byrne, L. et al. Unintended consequences: fluid resuscitation worsens shock in an ovine model of endotoxemia. Am. J. Respir. Crit. Care Med. 198(8), 1043–1054 (2018).
Ince, C. et al. The endothelium in sepsis. Shock 45(3), 259–270 (2016).
Smart, L. & Hughes, D. The effects of resuscitative fluid therapy on the endothelial surface layer. Front. Vet. Sci. 8, 661660 (2021).
Guerci, P. et al. Glycocalyx degradation is independent of vascular barrier permeability increase in nontraumatic hemorrhagic shock in rats. Anesth. Analg. 129(2), 598–607 (2019).
Ergin, B. et al. Hemodilution causes glycocalyx shedding without affecting vascular endothelial barrier permeability in rats. J. Clin. Transl. Res. 5(5), 243–252 (2020).
Bruegger, D. et al. Release of atrial natriuretic peptide precedes shedding of the endothelial glycocalyx equally in patients undergoing on- and off-pump coronary artery bypass surgery. Basic Res. Cardiol. 106(6), 1111–1121 (2011).
Jedlicka, J., Becker, B. F. & Chappell, D. Endothelial glycocalyx. Crit. Care Clin 36(2), 217–232 (2020).
Hippensteel, J. A. et al. Intravenous fluid resuscitation is associated with septic endothelial glycocalyx degradation. Crit. Care 23(1), 259 (2019).
Saoraya, J., Wongsamita, L., Srisawat, N. & Musikatavorn, K. The effects of a limited infusion rate of fluid in the early resuscitation of sepsis on glycocalyx shedding measured by plasma syndecan-1: a randomized controlled trial. J. Intensive Care 9(1), 1 (2021).
Hahn, R. G., Patel, V. & Dull, R. O. Human glycocalyx shedding: Systematic review and critical appraisal. Acta Anaesthesiol. Scand. 65(5), 590–606 (2021).
Skibsted, S. et al. Biomarkers of endothelial cell activation in early sepsis. Shock 39(5), 427–432 (2013).
Arendts, G. et al. Critical illness in the emergency department: lessons learnt from the first 12 months of enrolments in the Critical Illness and Shock Study. Emerg. Med. Australas. 24(1), 31–36 (2012).
Macdonald, S. P. J. et al. Resistin and NGAL are associated with inflammatory response, endothelial activation and clinical outcomes in sepsis. Inflamm. Res. 66(7), 611–619 (2017).
Hou, P., Filbin, M., Wang, H., Ngo, L., Huang, D. T., Aird, W.C. et al. Endothelial permeability and hemostasis in septic shock: results from the ProCESS Trial. Chest (2017).
Furian, T. et al. Ventricular dysfunction and dilation in severe sepsis and septic shock: relation to endothelial function and mortality. J. Crit. Care 27(3), 319 (2012).
Massey, M. J. & Shapiro, N. I. A guide to human in vivo microcirculatory flow image analysis. Crit Care 20, 35 (2016).
Bruegger, D. et al. Atrial natriuretic peptide induces shedding of endothelial glycocalyx in coronary vascular bed of guinea pig hearts. Am. J. Physiol. Heart Circ. Physiol. 289(5), H1993–H1999 (2005).
Inkinen, N. et al. Association of endothelial and glycocalyx injury biomarkers with fluid administration, development of acute kidney injury, and 90-day mortality: data from the FINNAKI observational study. Ann. Intensive Care 9(1), 103 (2019).
Puskarich, M. A., Cornelius, D. C., Tharp, J., Nandi, U. & Jones, A. E. Plasma syndecan-1 levels identify a cohort of patients with severe sepsis at high risk for intubation after large-volume intravenous fluid resuscitation. J. Crit. Care 36, 125–129 (2016).
Saoraya, J., Wongsamita, L., Srisawat, N. & Musikatavorn, K. Plasma syndecan-1 is associated with fluid requirements and clinical outcomes in emergency department patients with sepsis. Am. J. Emerg. Med. 42, 83–89 (2021).
Montomoli, J., Donati, A. & Ince, C. Acute kidney injury and fluid resuscitation in septic patients: are we protecting the kidney?. Nephron 143(3), 170–173 (2019).
Pouska, J. et al. Impact of intravenous fluid challenge infusion time on macrocirculation and endothelial glycocalyx in surgical and critically Ill patients. Biomed. Res. Int. 2018, 8925345 (2018).
Belavic, M. et al. The effect of various doses of infusion solutions on the endothelial glycocalyx layer in laparoscopic cholecystectomy patients. Minerva. Anestesiol. 84(9), 1032–1043 (2018).
Dogne, S. & Flamion, B. Endothelial glycocalyx impairment in disease: focus on hyaluronan shedding. Am. J. Pathol. 190(4), 768–780 (2020).
Strand, M. E. et al. Innate immune signaling induces expression and shedding of the heparan sulfate proteoglycan syndecan-4 in cardiac fibroblasts and myocytes, affecting inflammation in the pressure-overloaded heart. FEBS J. 280(10), 2228–2247 (2013).
Berg, S., Golster, M. & Lisander, B. Albumin extravasation and tissue washout of hyaluronan after plasma volume expansion with crystalloid or hypooncotic colloid solutions. Acta Anaesthesiol. Scand. 46(2), 166–172 (2002).
Shapiro, N. I. et al. The association of endothelial cell signaling, severity of illness, and organ dysfunction in sepsis. Crit. Care 14(5), R182 (2010).
Scheibner, K. A. et al. Hyaluronan fragments act as an endogenous danger signal by engaging TLR2. J. Immunol. 177(2), 1272–1281 (2006).
Nikaido, T. et al. Serum syndecan-4 as a possible biomarker in patients with acute pneumonia. J. Infect. Dis. 212(9), 1500–1508 (2015).
Nelson, A., Johansson, J., Tyden, J. & Bodelsson, M. Circulating syndecans during critical illness. APMIS 125(5), 468–475 (2017).
Hara, T., Sato, A., Yamamoto, C. & Kaji, T. Syndecan-1 downregulates syndecan-4 expression by suppressing the ERK1/2 and p38 MAPK signaling pathways in cultured vascular endothelial cells. Biochem. Biophys. Rep. 26, 101001 (2021).
Maitland, K. et al. Exploring mechanisms of excess mortality with early fluid resuscitation: insights from the FEAST trial. BMC Med. 11, 68 (2013).
Kuttab, H. I. et al. Evaluation and predictors of fluid resuscitation in patients with severe sepsis and septic shock. Crit. Care Med. 47(11), 1582–1590 (2019).
Kabil, G., Liang, S., Delaney, A., Macdonald, S., Thompson, K., Saavedra, A. et al. Association between intravenous fluid resuscitation and outcome among patients with suspected infection and sepsis: a retrospective cohort study. Emerg. Med. Australas. (2021).
Self, W. H. et al. Liberal versus restrictive intravenous fluid therapy for early septic shock: rationale for a randomized trial. Ann. Emerg. Med. 72(4), 457–466 (2018).
Meyhoff, T. S. et al. Conservative vs liberal fluid therapy in septic shock (CLASSIC) trial-Protocol and statistical analysis plan. Acta Anaesthesiol. Scand. 63(9), 1262–1271 (2019).
Macdonald, S. P. J. et al. Restricted fluid resuscitation in suspected sepsis associated hypotension (REFRESH): a pilot randomised controlled trial. Intensive Care Med. 44(12), 2070–2078 (2018).
Drost, C. C., Rovas, A. & Kumpers, P. Protection and rebuilding of the endothelial glycocalyx in sepsis - Science or fiction?. Matrix Biol. Plus 12, 100091 (2021).
Funding
Funded by a University of Western Australia Research Collaboration Award and the Royal Perth Hospital Research Foundation. SM is a Raine Foundation Clinician Research Fellow.
Author information
Authors and Affiliations
Contributions
S.M., E.B., N.S. and L.S. developed the study hypothesis and methodology. S.M., N.S., D.F. and G.A. oversaw patient recruitment and T.J. undertook data collection. E.B., L.B. and M.H. were responsible for the laboratory work. S.B. undertook the statistical analyses and L.S. assisted with the data interpretatation. S.M. drafted the paper and all authors contributed to revisions for intellectual content and approved the final version for submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Macdonald, S., Bosio, E., Shapiro, N.I. et al. No association between intravenous fluid volume and endothelial glycocalyx shedding in patients undergoing resuscitation for sepsis in the emergency department. Sci Rep 12, 8733 (2022). https://doi.org/10.1038/s41598-022-12752-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-12752-x
- Springer Nature Limited
This article is cited by
-
Association between intravenous fluid administration and endothelial glycocalyx shedding in humans: a systematic review
Intensive Care Medicine Experimental (2024)
-
Fluid bolus increases plasma hyaluronan concentration compared to follow-up strategy without a bolus in oliguric intensive care unit patients
Scientific Reports (2024)
-
Effect of intravenous fluid volume on biomarkers of endothelial glycocalyx shedding and inflammation during initial resuscitation of sepsis
Intensive Care Medicine Experimental (2023)