Abstract
A series of recent studies have indicated that the Caveolin-1 (CAV-1) gene variant may be associated with metabolic and inflammatory markers and anthropometric measures. Furthermore, it has been shown that a plant-based dietary index (PDI) can elicit a positive impact on these metabolic markers. Therefore, we sought to examine whether PDI intakes may affect the relationship between CAV-1 (rs3807992) and metabolic factors, as well as serum inflammatory markers and anthropometric measures, in women with obesity. This current study consisted of 400 women with overweight and obesity, with a mean (SD) age of 36.67 ± 9.10 years. PDI was calculated by a food frequency questionnaire (FFQ). The anthropometric measurements and serum profiles were measured by standard protocols. Genotyping of the CAV-1(rs3807992) was conducted by the PCR–RFLP method. The following genotypic frequencies were found among the participants: GG (47.8%), AG (22.3%), and AA (2.3%). In comparison to GG homozygotes, risk-allele carriers (AA + AG) with higher PDI intake had lower ALT (P: 0.03), hs-CRP (P: 0.008), insulin (P: 0.01) and MCP-1 (P: 0.04). Furthermore, A-allele carriers were characterized by lower serum ALT (P: 0.04), AST (P: 0.02), insulin (P: 0.03), and TGF-β (P: 0.001) when had the higher following a healthful PDI compared to GG homozygote. Besides, risk-allele carriers who consumed higher unhealthful PDI had higher WC (P: 0.04), TC/HDL (P: 0.04), MCP-1 (P: 0.03), and galactin-3 (P: 0.04). Our study revealed that A-allele carriers might be more sensitive to PDI composition compared to GG homozygotes. Following a healthful PDI in A-allele carriers may be associated with improvements in metabolic and inflammatory markers and anthropometric measures.
Similar content being viewed by others
Introduction
Obesity is defined by an excess in the accumulation of lipids in adipose tissue; this accumulation becomes detrimental when it happens in visceral fat1. Indeed, waist circumference (WC) (an indirect measure of visceral fat accumulation) is correlated with the progression of particular metabolic disorders containing cardiovascular diseases (CVDs) and type 2 diabetes (T2DM)2. The prevalence of obesity is universally higher in women3; additionally, women with high body mass index (BMI) are considered at greater risk of T2DM4, atherosclerotic cardiovascular disease5, hypertension6, dyslipidemia7, and endocrine disorders8. Moreover, increased levels of hepatic enzymes, such as alanine aminotransferase (ALT) and aspartate aminotransferase (AST), are common in obesity and their prevalence increases gradually with increasing BMI9. Numerous population studies show that low-grade and chronic inflammation is often augmented in patients with obeisty10. This is characterized by incremented circulating levels of pro-inflammatory cytokines—particularly Interleukin-6 (IL- 6)—and of the monocyte chemoattractant protein (MCP-1), both generated by the adipose tissue11,12,13,14.
Diet is one of the most notable variables that have life-long effects on obesity and metabolic risk factors15. There is abundant evidence to suggest that dietary patterns are related to inflammatory mediators and obesity as an inflammatory-related disease16,17,18,19; indeed, a review article demonstrated that a plant-based diet (PBD) could alleviate certain inflammatory markers, such as IL-620. This kind of dietary pattern has been described as possessing a high intake of vegetables, fruits, grains, and legumes, which could lead to reductions in both inflammation and obesity21. Also, dietary inflammatory index, in which the consumption of food items that can increase body inflammations, such as saturated fatty acid, trans fatty acids, and refined grains, were positively associated with fat mass and concentration of MCP-122. Several dietary intervention studies have shown that healthy plant foods, such as flavonoids, can modulate inflammatory cytokines23,24,25. Therefore, PBD may be associated with an improvement in obesity-related inflammatory profiles and inhibition of chronic diseases risks. Indeed, recent studies have revealed that there might be an interaction between specific genes such as Caveolin-1 and particular dietary patterns26,27.
Caveolin-1 (CAV-1), is the major structural protein of caveolae, which is located on human chromosome 7 (7q31.1) and it includes 3 exons that select intronic single nucleotide polymorphisms (SNPs). CAV-1 works in a scaffolding capacity and has been involved in transmembrane signaling28. CAV-1 is a vital constituent of the lipid raft that controls their activity and cooperates with numerous signaling pathways, including steroid receptors29. Moreover, CAV-1 gene variants have been shown to be correlated with insulin resistance (IR), diabetes mellitus, dyslipidemia, and metabolic syndrome30. Based on previous studies, the adipocyte seems to have higher concentrations of caveolae than any other cell31,32. Additionally, some studies have shown that diets containing foods with antioxidant features such as PBD, can modulate CAV-1 levels and caveolin function for the debarment of chronic diseases33,34,35,36.
Given the rising prevalence of obesity and the lack of studies performed on Caveolin-1 (rs3807992) and plant-based dietary index (PDI), as well as its association with metabolic and inflammatory markers among women, the present study sought to evaluate the interactions between Caveolin-1 (rs3807992) and PDI on metabolic and inflammatory markers in Iranin women with overweight and obesity.
Methods
Study population
This cross-sectional study, including 400 women with obesity, was conducted in the health care center of Tehran, Iran. The subjects were chosen via a random cluster random sampling method, where the inclusion criteria were: women with overweight (BMI ≥ 25 kg/m2) or obesity (BMI ≥ 30 kg/m2), aged above 18 years, before menopause, not being pregnant or lactating and smoker. The women who had any chronic disease (T2DM), CVDs, polycystic ovary syndrome (PCOS), non-alcoholic fatty liver disease, etc.), taking any supplements or medications (Atorvastatin, Cholestyramine, etc.), and weight loss program were excluded. The general characteristics of the participants were collected via a demographic questionnaire. The study protocol was approved by the Ethics Commission of Tehran University of Medical Sciences (IR.TUMS.VCR.REC. 97-03-161-41017)37. Total calorie intake was accepted in the range between 800 and 4200 kcal per day, with intakes outside of this range leading to exclusion from the study38. All of the participants completed a written informed consent form before taking part in the study.
General, anthropometric and physical activity assessments
Height was measured using Seca 216 to the nearest 0.1 cm and weight of subjects was also assessed using Seca scale to the nearest 0.1 kg, with light clothes and unshod, in a standing position. WC was measured at the point of the smallest girth by a trained expert. BMI was calculated as weight/(height)2. In addition, other anthropometric variables were measured using a multi-frequency bioelectrical impedance analyzer InBody 770 scanner (Inbody Co., Seoul, Korea), with participants wearing light clothes without any metal subjects39. In addition, physical activity (PA) was assessed via the validated International Physical Activity Questionnaire40. All participants' blood pressure was monitored using an automatic sphygmomanometer according to established procedures (OMRON, Germany).
Dietary assessments and plant-based dietary intake
A validated 147-items semi-quantitative food frequency questionnaire (FFQ) was used by an expert to assess the dietary intake of the subjects. Grams of foods and beverages were entered into NUTRITIONIST IV software (version 7.0; N-Squared Computing, Salem, OR)41,42. According to the plant-based dietary intake, which was divided to overall plant-based diet index (PDI), healthy PDI (hPDI), and unhealthy PDI (uPDI), the intakes of the subjects were divided to 18 groups, which were: animal (Dairy, animal fat, egg, meat, fish and seafood, and miscellaneous animal-based foods), healthy (whole grains, vegetables, fruits, legumes, nuts, vegetable oils, tea, and coffee), and unhealthy plant-based foods (fruit juices, sugar-sweetened beverages, refined grains, sweets and desserts, and potatoes)43,44. Foods were scored 1–10, after they were transformed to deciles. PDI, hPDI, and uPDI were assessed as scoring 10 and 1 for the highest and lowest deciles of plant food intake, healthy plant foods, and unhealthy plant foods, respectively. Conversely, unhealthy plant foods and animal food items were scored 1 to 10 for the highest and lowest deciles of animal foods and unhealthy plants foods, respectively.
Biochemical measurements
After 10–12 h night fasting, blood samples were gathered at the Nutrition and Biochemistry laboratory of the School of Nutritional Sciences and Dietetics, TUMS. Triacylglycerol kits (Pars Azmoon Inc, Tehran, Iran) were used to measure serum triglycerides (TG) by colorimetric method tests with Glycerol-3-phosphate oxidase Phenol 4-Aminoantipyrine Peroxidase (GPO-PAP). The cholesterol oxidase Phenol 4-Aminoantipyrine Peroxidase (CHOD-PAP) was used to calculate total cholesterol, and the direct method and immunoinhibition were used to measure low-density lipoprotein (LDL) and high-density lipoprotein (HDL). Pars Azmoon (Pars Azmoon Inc., Tehran, Iran) provided all of the kits. ALT, AST, high-sensitivity C-reactive protein (hs-CRP), MCP-1, interleukin 1 beta (IL-1β), and transforming growth factor-beta (TGFβ) were measured via standard protocols. Plasminogen activator inhibitor-1 (PAI-1) (Human PAI-1*96 T ELISA kit Crystal Company) was measured in triplicate. TC was divided into HDL to discern the ratio of TC/HDL, in addition, LDL was divided by HDL to discern the ratio of LDL/HDL. Serum insulin level was also measured by radioimmune assay. More detailed about biochemical measuring are reported in our previous study45.
DNA analysis
Based on a previous study27, the Mini Columns kit (Type G; Genall; Exgene) was used for DNA extraction. The CAV-1 SNP (rs3807992) was genotyped by PCR–RFLP method, using primers, Forward: 3′AGTATTGACCTGATTTGCCATG5′ Reverse: 5′GTCTTCTGGAAAAAGCACATGA-3′. The sequencing process was performed using the ABI PRISM 3730 automated sequencer (Applied Biosystems, Foster City, Calif, USA)27.
Statistical analyses
The Kolmogorov–Smirnov test was used for assessing the normality of the data. The Hardy–Weinberg equilibrium and comparison of categorical variables were assessed with the x2 test. PDI was transformed to tertiles based on the trends. Quantitative (mean ± SD) and qualitative variables (n (%)) were evaluated via one-way analysis of variance (ANOVA) and chi-square among PDI tertiles, respectively. Genotype groups were considered as a dominant inherent model (AA + AG) versus GG homozygous. Analysis of covariance (ANCOVA) was performed for the adjustment model (adjusting for age, physical activity and energy intake and diastolic blood pressure (DBP)). A generalized linear regression model (GLZM) was used to analyze the interactions between CAV-1 polymorphism (rs3807992) and PDI. Data were analyzed using Statistical Package for Social Sciences (SPSS Inc., Chicago, IL, version 25) and a P-value < 0.05 was considered as significant; but for interactions, P < 0.1 was considered significant.
Ethics approval and consent to participate
All methods were performed in accordance with the relevant guidelines and regulations. The protocol of the study was approved by the ethics committee of TUMS (Ethics number: 97-03-161-41017). All participants completed a written informed consent.
Results
Study population characteristics
The participants in this study consisted of 400 women, with a mean (SD) age of 36.67 ± 9.10 years. The majority of individuals (72.4%) were married, had a university education (47.8%), and had a history of obesity (71.2 percent). The following genotypic frequencies were found among the participants: GG (47.8%), AG (22.3%), and AA (2.3%).
The difference in means of biochemical parameters and body composition between CAV-1 rs3807992 genotypes
The comparison of variables including anthropometrics and biochemical parameters according to two genotypes groups (GG and AG + AA) is given in Table 1. The results of this study revealed that DBP (P = 0.007), body fat mass (BFM) (P = 0.01), and fat mass index (FMI) (P = 0.009) were higher in participants carrying the A allele, compared with individuals in the GG genotype. Moreover, A-allele carriers had significantly lower serum HDL (P < 0.001) and LDL (P = 0.01) compared to GG homozygotes; although, the TC/HDL (P = 0.008) ratio was significantly higher. Furthermore, there was no significant association between this polymorphism and other biochemical parameters including TG, inflammatory markers (hs-CRP, MCP-1, TGFβ), and liver enzymes (ALT and AST) in both crude and adjusted models (P > 0.05) (Table 1).
The difference in means of biochemical parameters and body composition between PDI, hPDI and uPDI
The result of comparison of biochemical parameters and body composition between PDI, hPDI and uPDI are shown in Tables 2, 3 and 4. These results displayed that women with higher PDI were older (P = 0.03) and had higher physical activity (P < 0.001) than those with lower PDI. Women with higher healthful PDI had significantly lower LDL/HDL ratios in both crude (P = 0.04) and adjusted models (P = 0.03); although there was no statistically significant difference (P > 0.05) from the low to high unhealthful PDI.
Interactions between PDIs and CAV-1 rs3807992 genotypes on metabolic and inflammatory markers and anthropometric measures
The interaction effects between CAV-1 variants at rs3807992 and PDIs (healthful and unhealthful) on metabolic and inflammatory markers, including TC, HDL, LDL, TC, TC/HDL, LDL/HDL, TG, ALT, AST, hs-CRP, MCP-1, PAI-1, and IL-1β, are shown in Tables 5 and 6.
Interaction between PDI and CAV-1 rs3807992 genotypes on metabolic and inflammatory markers and anthropometric measures
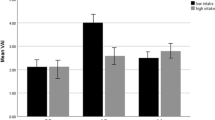
There is a gene-diet interaction for PDI and CAV-1 polymorphism (rs3807992) on ALT (β: − 7.51, 95% CI − 14.45 to − 0.57, P: 0.03), hs-CRP (β: − 3.30, 95% CI − 5.76 to − 0.85, P: 0.008), insulin (β: − 0.14, 95% CI − 0.26 to − 0.02, P: 0.01), and MCP-1 (β: − 54.51, 95% CI − 107.72 to − 1.30, P: 0.04) in both crude and adjusted models (adjusting for age, energy intake, physical activity education, job and marriage status) (Table 5). Thus, the A-allele carriers who consumed higher PDI had lower ALT, insulin, MCP-1, and hs-CRP, compared to GG homozygotes (Fig. 1).
The interaction between CAV-1 SNP rs3807992 and plant-based diet index (PDI) on; (a) ALT, (b) hs-CRP, (c) insulin, (d) MCP-1. *P-value for curd (unadjusted) model. **P-value for the adjusted model by age, physical activity level, energy intake, education, job and marriage status. ALT alanine aminotransferase, hs-CRP high-sensitivity C-reactive protein, MCP-1 monocyte chemoattractant protein-1.
Interaction between healthful PDI and CAV-1 rs3807992 genotypes on metabolic and inflammatory markers and anthropometric measures
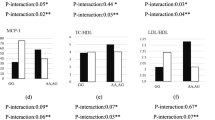
There were significant interactions between healthful PDI and rs3807992 on ALT (β: − 6.52, 95% CI − 13.48 to − 0.43, P: 0.04), AST (β: − 4.36, 95% CI − 8.27 to − 0.44, P: 0.02), insulin (β: − 0.18, 95% CI − 0.28 to − 0.03, P: 0.03), and TGF-b (β: − 56.05, 95% CI − 78.96 to − 3.14, P: 0.001) in both crude and adjusted adjustment model (Table 6). In particular, A-allele carriers were characterized by lower serum ALT, AST, insulin, and TGF-β, when following a healthful PDI, compared to GG homozygote. (Fig. 2).
The interaction between CAV-1 SNP rs3807992 and healthy plant-based diet index (hPDI) on; (a) ALT, (b) AST, (c) TGF-β, (d) insulin. *P-value for curd (unadjusted) model. **P-value for the adjusted model by age, physical activity level, energy intake, education, job and marriage status. AST aspartate aminotransferase, ALT alanine aminotransferase, TGFβ transforming growth factor beta.
Interaction between unhealthful PDI and CAV-1 rs3807992 genotypes on metabolic and inflammatory markers and anthropometric measures
Risk-allele carriers who consumed higher unhealthful PDI had higher WC (β: 0.06, 95% CI 0.001–0.12, P: 0.04), TC/HDL (β: 0.72, 95% CI 0.07–1.53, P: 0.04), MCP-1(β: 0.37, 95% CI 0.02–0.73, P: 0.03), and galactin-3 (β: 6.60, 95% CI 0.26–12.94, P: 0.04). All data are shown in Table 6 and Fig. 3.
The interaction between CAV-1 SNP rs3807992 and Unhealthy plant-based diet index (uPDI) on; (a) WC, (b) TC/HDL, (c) Galactin-3, (d) MCP-1. *P-value for curd (unadjusted) model. **P-value for the adjusted model by age, physical activity level, energy intake, education, job and marriage status. WC waist circumference, MCP-1 monocyte chemoattractant protein-1.
Discussion
To our knowledge, this is the first study to have investigated the interactions between genetic variants of the CAV-1 gene with PDI on metabolic and inflammatory markers among Iranian women with overweight and obesity.
Our findings highlight a gene-diet interaction for PDI and CAV-1 polymorphism (rs3807992) on ALT, hs-CRP, insulin, and MCP-1 in both crude and adjusted models; such that the A- allele carriers who had higher PDI, had lower ALT, insulin, MCP-1, and hs-CRP compared to GG homozygotes. Based on our previous findings46, CAV-1 (rs3807992) might be related to incremented metabolic disease risk factors in women with overweight and obesity. We also previously posited that among minor allele carriers, insulin pathways may responsible for the relationship between CAV-1 rs3807992 and metabolic factors46.
In the present study, DBP, BFM, and FMI were higher in participants carrying the A allele compared to those in the GG genotype. There are some possible functional roles for CAV-1 may be inherited. It has been established by previous studies that CAV-1 has a major regulator function in fat distribution and genetic lipodystrophies in humans47,48. Furthermore, experimental studies have reported an association between CAV-1 mRNA expression in adipose tissues in obese women compared with lean subjects49,50. CAV-1- deficient mice had a smaller lean body phenotype than their wild-type counterparts51,52. Lipodystrophy has been observed in CAV-1 null mice due to a variety of functions attributed to caveolae in adipocytes, including lipid droplet dysfunction, adipocyte differentiation pathway disruption, abnormalities in cholesterol and fatty acid binding, transport, and storage, and an increase in insulin signaling47,53.
Moreover, A-allele carriers had significantly lower serum HDL and LDL compared to GG homozygotes. Additionally, there was no significant relationship between this polymorphism and other biochemical parameters containing TG, inflammatory markers (hs-CRP, MCP-1, PAI-1, and IL-1β), and liver enzymes in both crude and adjusted models. Concordant with our study, Khatibi et al. also found a significant association between CAV-1 genotypes with DBP26, and observed that participants with prevailing alleles had a lower risk of dyslipidemia, and that risk allele carriers, who adhered more to the Mediterranean‑DASH Intervention for Neurodegenerative Delay (MIND) diet, might present with lower dyslipidemia26.
In our study, we also observed a significant interaction between hPDI and rs3807992 on ALT, AST, insulin, and TGF-b in both crude and adjusted models. Indeed, A-allele carriers who had higher hPDI scores, had lower serum ALT, AST, insulin, and TGF-b compared to GG homozygote. Further, risk-allele carriers who had higher uPDI scores, had higher WC, TC/HDL, MCP-1, and galactin-3. No significant interactions were observed between CAV-1 rs3807992 variants and uPDI for other metabolic-related traits. A cross-sectional study in 2021 revealed that, among people following antioxidant-rich diets, ‘A’ allele carriers might be more vulnerable to cardiovascular disorders; the authors also reported no significant relationship between higher adherence to dietary total antioxidant capacity (DTAC) and changes in lipid profile, HOMA-IR, and body composition components27.
It is well accepted that the prevalence of obesity is directly related to dietary patterns54. PBD alleviates body fat due to the decreased caloric intake and incremented energy expenditure because of augmented thermogenesis55. Reduced use of saturated fats, which are usually derived from animal-based foods, might improve insulin sensitivity55; indeed, plant-based foods are also a main source of phytochemicals56. The consumption of phytochemicals, notably polyphenols, which exist in a variety of plant foods (e.g., berries, grapes, onions, apples, cacao, green tea, soy, whole grains, etc.), is related to reduced mortality and chronic disease risk57,58,59,60. The food compound of an uPDI might be associated with higher intakes of unpleasant nutrients and lower intakes of micronutrients and antioxidants, which can negatively affect metabolic syndrome and its factors. A high intake of simple carbohydrates from uPDI could also affect glucose control, lipid metabolism, and weight gain61, whilst decreased dietary fiber might impact glycemic control, insulin sensitivity, and augment inflammation. These effects could be related to reduced inflammation and oxidative stress62,63. Chronic inflammation is often evident in those with poor feeding habits and a sedentary lifestyle, features which are concomitantly associated with obesity progression64,65, in addition to various other pathologies, including asthma and Alzheimer disease, and different diseases related to unbalanced metabolisms such as T2DM, atherosclerosis, and cardiovascular diseases66,67,68,69. Metabolic inflammation consists of a complex mechanism containing crosstalk between different tissues (like adipose tissue and liver) through the whole body. Generally, this low-grade inflammation emerges when cellular stress is distinguished by the immune system70. Another mechanism posited to contribute to the progression of chronic inflammation involves the redundant storage of triglyceride (TG) lipids within adipose tissues. In murine models, exceeding TG storage in white adipose tissue (WAT) induces secretion of pro-inflammatory adipokines, such as IL-1, TNF-α, MCP-1, and IL-6, triggering systemic metabolic inflammation71.
Although the exact mechanism of the interaction between CAV-1 and diet has not yet been fully elucidated, some explanations have been proposed. Several studies have expressed that CAV-1 binds to endothelial nitric oxide synthase (eNOS) and HDL receptors in the caveolae and prevents their activity. Some diets, such as the PBD or anti-inflammatory diets, can dislocate CAV-1 from caveolae to the cytoplasm, leading to a reduction in the CAV-1 level, ameliorating the prohibitory effects on HDL and eNOS receptors72. Decreased nitric oxide (NO) generation, as a result of increased CAV-1 expression, has been thought to occur from prolonged exposure to high glucose, may play a vital role in inflammatory pathways and expansion of inflammation73. Hence, it is not surprising that higher adherence to a uPDI can augment MCP-1 by changing the expression of CAV-1 and other genes.
Diet and plasma-derived nutrients may influence metabolic markers by interacting with caveolae-associated cellular signaling, according to new research36. In this regard, Oberleithner et al. suggest that serum sodium and potassium can control eNOS binding to the caveolae membrane and activity74. As a result, the beneficial benefits of a healthy PDI might be attributed to components like vegetables and fruits, which have a greater impact on potassium and sodium balance.
Beside, the lower liver enzyme in A-allele carriers following an hPDI in the present study can be dependent on the caffeine intake during the hPDI. In this term, we previously found a decreased AST in A-allele carriers who consumed more caffeine in a healthy dietary pattern45.Interestingly, a candidate gene for the favorable impact of caffeine consumption is located near the CAV-1 gene75, Therefor, we suggested a possible interaction between caffeine and CAV-1, which we hope will be confirmed in future study.
In the current study, we observed a relationship between CAV-1 with body composition, which could suggest a CAV-1 lipogenic pathway interaction. The expression of CAV-1 is found in most normal organs but notably assuming that CAV-1 is greatly expressed in adipose tissue76 and the interplay between CAV-1 gene and lipogenic genes has been expressed77. Visceral fat could also be associated with changes in circulating fatty acid composition78. CAV‐1 interacts with two popular receptors that augment oxidative stress: the angiotensin II receptor and mineralocorticoid receptor (MR)79. MR blockade has been shown to reduce NADPH Oxidase 4 (NOX4) expression and lower reactive oxygen species in the kidney and heart80,81.
Several studies have revealed that CAV-1 is associated with oxidative stress, as reactive species could impact the expression, degradation, post-translational changes, and trafficking of Caveolae membranes82. Hence, because of the impact of the CAV-1 gene with oxidative stress, it seems that adherence to hPDI could be capable of lowering the metabolic markers, and, as a result, reducing the likelihood of metabolic diseases in women with obesity carrying a risk the allele of ‘A’. Indeed, the current study provides some support for the consideration of particular dietary guidelines based on genotypes.
Although we provide novel findings of gene-diet interactions, some limitations should be considered in the interpretation of the study. First, the results of this cross-sectional study, although nationally illustrative, cannot indicate a causal relationship. Second, even though we applied a validated FFQ, measurement errors are possible. Third, our study only included women, therefore, results are not generalizable to men. Finally, although we considered potential confounders, residual confounding may still exist. We did not have any data related to family history of cancer; however, the expression of CAV-1 can be effected by this cofounding marker.
Conclusion
In conclusion, the results of the current study suggest that diet, gene variants, and their interaction, should be considered in metabolic diseases risk assessment. PDIs may provide advantages in the prohibition of chronic disease. In future studies, investigating samples from a greater geographic area with wide ranges of ages in both sexes and larger sample sizes might prove more important findings. This work clearly highlights the value of considering genotypes in dietary planning and modification. Therefore, these findings from nutrigenetic studies in future may be combine with a patient's genetic history to give more relevant and customized nutritional recommendations for women with obesity in order to avoid or reduce cardiovascular disease.
Data availability
The data are not publicly available due to containing private information of participants. Data are however available from the authors upon reasonable request and with permission of Khadijeh Mirzaei.
Abbreviations
- AST:
-
Aspartate aminotransferase
- BIA:
-
Bioelectrical impedance analysis
- BMI:
-
Body mass index
- bp:
-
Base pair
- BFM:
-
Body fat mass
- CRP:
-
C-reactive protein
- CVD:
-
Cardiovascular disease
- DBP:
-
Diastolic blood pressure
- DNA:
-
Deoxyribonucleic acid
- DP:
-
Dietary pattern
- eNOS:
-
Endothelial nitric oxide synthase
- FMI:
-
Fat mass index
- FFQ:
-
Food frequency questionnaire
- GLM:
-
General linear model
- GWAS:
-
Genome-wide association studies
- HDL:
-
High-density lipoprotein cholesterol
- IR:
-
Insulin Receptor
- hs-CRP:
-
High sensitivity C-reactive protein
- IPAQ:
-
International physical activity questionnaire
- LDL:
-
Low-density lipoprotein
- MA:
-
Minor allele
- PBD:
-
Plant-based diet
- PDI:
-
Plant-based dietary index
- PCR:
-
Polymerase chain reaction
- PDI:
-
Plant-based dietary index
- RFLP:
-
Restriction fragment length polymorphism
- SNP:
-
Single nucleotide polymorphism
- TC:
-
Total cholesterol
- TG:
-
Triglyceride
- WC:
-
Waist circumference
- WHO:
-
World Health Organization
References
Antonopoulos, A. S. & Tousoulis, D. The molecular mechanisms of obesity paradox. Cardiovasc. Res. 113(9), 1074–1086 (2017).
Tchernof, A. & Després, J. P. Pathophysiology of human visceral obesity: An update. Physiol. Rev. 93(1), 359–404 (2013).
Chooi, Y. C., Ding, C. & Magkos, F. The epidemiology of obesity. Metabolism 92, 6–10 (2019).
Maskarinec, G. et al. Type II diabetes, obesity, and breast cancer risk: The multiethnic cohort. Cancer Epidemiol. Biomarkers Prev. 26(6), 854–861 (2017).
Garcia, M., Mulvagh, S. L., Merz, C. N., Buring, J. E. & Manson, J. E. Cardiovascular disease in women: Clinical perspectives. Circ. Res. 118(8), 1273–1293 (2016).
Seravalle, G. & Grassi, G. Obesity and hypertension. Pharmacol Res. 122, 1–7 (2017).
Palmer, M. K. & Toth, P. P. Trends in lipids, obesity, metabolic syndrome, and diabetes mellitus in the United States: An NHANES analysis (2003–2004 to 2013–2014). Obesity 27(2), 309–314 (2019).
Joham, A. E., Palomba, S. & Hart, R. Polycystic ovary syndrome, obesity, and pregnancy. Semin. Reprod. Med. 34(2), 93–101 (2016).
Marchesini, G., Moscatiello, S., Di Domizio, S. & Forlani, G. Obesity-associated liver disease. J. Clin. Endocrinol. Metab. 93(11), s74–s80 (2008).
Saltiel, A. R. & Olefsky, J. M. Inflammatory mechanisms linking obesity and metabolic disease. J. Clin. Invest. 127(1), 1–4 (2017).
Weiss, R. et al. Obesity and the metabolic syndrome in children and adolescents. N. Engl. J. Med. 350(23), 2362–2374 (2004).
Nieuwdorp, M., Stroes, E. S., Meijers, J. C. & Büller, H. Hypercoagulability in the metabolic syndrome. Curr. Opin. Pharmacol. 5(2), 155–159 (2005).
Dandona, P., Aljada, A. & Bandyopadhyay, A. Inflammation: the link between insulin resistance, obesity and diabetes. Trends Immunol. 25(1), 4–7 (2004).
Dandona, P., Aljada, A., Chaudhuri, A., Mohanty, P. & Garg, R. Metabolic syndrome: A comprehensive perspective based on interactions between obesity, diabetes, and inflammation. Circulation 111(11), 1448–1454 (2005).
Peeters, A. Obesity and the future of food policies that promote healthy diets. Nat. Rev. Endocrinol. 14(7), 430–437 (2018).
Calle, M. C. & Andersen, C. J. Assessment of dietary patterns represents a potential, yet variable, measure of inflammatory status: A review and update. Dis. Mark. 2019, 3102870 (2019).
Galland, L. Diet and inflammation. Nutr. Clin. Pract. 25(6), 634–640 (2010).
Dias, J. A. et al. A high quality diet is associated with reduced systemic inflammation in middle-aged individuals. Atherosclerosis 238(1), 38–44 (2015).
Barbaresko, J., Koch, M., Schulze, M. B. & Nöthlings, U. Dietary pattern analysis and biomarkers of low-grade inflammation: A systematic literature review. Nutr. Rev. 71(8), 511–527 (2013).
Eichelmann, F., Schwingshackl, L., Fedirko, V. & Aleksandrova, K. Effect of plant-based diets on obesity-related inflammatory profiles: A systematic review and meta-analysis of intervention trials. Obes. Rev. 17(11), 1067–1079 (2016).
Hu, F. B. Plant-based foods and prevention of cardiovascular disease: An overview. Am. J. Clin. Nutr. 78(3 Suppl), 544s-s551 (2003).
Ghodoosi, N. et al. Associations of dietary inflammatory index, serum levels of MCP-1 and body composition in Iranian overweight and obese women: A cross-sectional study. BMC. Res. Notes 13(1), 544 (2020).
Chun, O. K., Chung, S. J., Claycombe, K. J. & Song, W. O. Serum C-reactive protein concentrations are inversely associated with dietary flavonoid intake in US adults. J. Nutr. 138(4), 753–760 (2008).
Oyama, J. et al. Green tea catechins improve human forearm vascular function and have potent anti-inflammatory and anti-apoptotic effects in smokers. Intern. Med. 49(23), 2553–2559 (2010).
Zern, T. L. et al. Grape polyphenols exert a cardioprotective effect in pre- and postmenopausal women by lowering plasma lipids and reducing oxidative stress. J. Nutr. 135(8), 1911–1917 (2005).
Khatibi, N. et al. Interactions between caveolin 1 polymorphism and the Mediterranean and Mediterranean-DASH Intervention for Neurodegenerative Delay diet (MIND) diet on metabolic dyslipidemia in overweight and obese adult women: a cross-sectional study. BMC Res. Notes. 14(1), 364 (2021).
Mirzababaei, A. et al. The effect of dietary total antioxidant capacity (DTAC) and Caveolin-1 gene variant interaction on cardiovascular risk factors among overweight and obese women: A cross-sectional investigation. Clin. Nutr. (Edinburgh, Scotland). 40(8), 4893–4903 (2021).
Razani, B., Woodman, S. E. & Lisanti, M. P. Caveolae: From cell biology to animal physiology. Pharmacol. Rev. 54(3), 431–467 (2002).
Brown, D. A. & London, E. Functions of lipid rafts in biological membranes. Annu. Rev. Cell Dev. Biol. 14, 111–136 (1998).
González-Muñoz, E. et al. Caveolin-1 loss of function accelerates glucose transporter 4 and insulin receptor degradation in 3T3-L1 adipocytes. Endocrinology 150(8), 3493–3502 (2009).
Napolitano L. The differentiation of white adipose cells. An electron microscope study. J Cell Biol. 18(3), 663–679 (1963).
Fan, J. Y. et al. Morphological changes of the 3T3-L1 fibroblast plasma membrane upon differentiation to the adipocyte form. J. Cell Sci. 61(1), 219–230 (1983).
Li, W. et al. Caveolin-1 inhibits expression of antioxidant enzymes through direct interaction with nuclear erythroid 2 p45-related factor-2 (Nrf2). J. Biol. Chem. 287(25), 20922–20930 (2012).
Zheng, Y. et al. Epigallocatechin-gallate stimulates NF-E2-related factor and heme oxygenase-1 via caveolin-1 displacement. J. Nutr. Biochem. 23(2), 163–168 (2012).
Guo, Z. et al. Baicalein inhibits prostate cancer cell growth and metastasis via the caveolin-1/AKT/mTOR pathway. Mol. Cell Biochem. 406(1–2), 111–119 (2015).
Majkova, Z., Toborek, M. & Hennig, B. The role of caveolae in endothelial cell dysfunction with a focus on nutrition and environmental toxicants. J. Cell. Mol. Med. 14(10), 2359–2370 (2010).
Abaj, F., & Mirzaei, K. Caveolin-1 genetic polymorphism interacts with PUFA to modulate metabolic syndrome risk. Br. J. Nutr. 2021:1–8.
Banna, J. C., McCrory, M. A., Fialkowski, M. K. & Boushey, C. Examining plausibility of self-reported energy intake data: Considerations for method selection. Front. Nutr. 4, 45 (2017).
Bahrampour, N., Mirzababaei, A., Shiraseb, F., Clark, C. C. T. & Mirzaei, K. The mediatory role of inflammatory markers on the relationship between dietary energy density and body composition among obese and overweight adult women: A cross-sectional study. Int. J. Clin. Pract. 75(10), e14579 (2021).
Hagströmer, M., Oja, P. & Sjöström, M. The International Physical Activity Questionnaire (IPAQ): A study of concurrent and construct validity. Public Health Nutr. 9(6), 755–762 (2006).
Mirmiran, P.H.-E.F., Jessri, M., Mahan, L. K., Shiva, N. & Azizi, F. Does dietary intake by Tehranian adults align with the 2005 dietary guidelines for Americans? Observations from the Tehran lipid and glucose study. J. Heal. Popul. Nutr. 29(1), 1 (2011).
Morgan, K. J., Zabik, M. E. & Stampley, G. L. The role of breakfast in diet adequacy of the US adult population. J. Am. Coll. Nutr. 5(6), 551–63 (1986).
Satija, A. et al. Plant-based dietary patterns and incidence of type 2 diabetes in US men and women: Results from three prospective cohort studies. PLoS Med. 13(6), e1002039 (2016).
Pourreza, S. et al. Association of plant-based diet index with inflammatory markers and sleep quality in overweight and obese female adults: A cross-sectional study. Int. J. Clin. Pract. 75(9), e14429 (2021).
Abaj, F. et al. Interactions between Caveolin-1 (rs3807992) polymorphism and major dietary patterns on cardio-metabolic risk factors among obese and overweight women. BMC Endocr. Disord. 21(1), 138 (2021).
Abaj, F., Saeedy, S. A. G. & Mirzaei, K. Are caveolin-1 minor alleles more likely to be risk alleles in insulin resistance mechanisms in metabolic diseases?. BMC. Res. Notes 14(1), 185 (2021).
Garg, A. & Agarwal, A. K. Caveolin-1: A new locus for human lipodystrophy. J. Clin. Endocrinol. Metab. 93(4), 1183–1185 (2008).
Abaj, F., Saeedy, S. A. G. & Mirzaei, K. Mediation role of body fat distribution (FD) on the relationship between CAV1 rs3807992 polymorphism and metabolic syndrome in overweight and obese women. BMC Med. Genomics 14(1), 1–8 (2021).
Catalán, V. et al. Expression of caveolin-1 in human adipose tissue is upregulated in obesity and obesity-associated type 2 diabetes mellitus and related to inflammation. Clin. Endocrinol. 68(2), 213–219 (2008).
Fernández-Real, J. M. et al. Study of caveolin-1 gene expression in whole adipose tissue and its subfractions and during differentiation of human adipocytes. Nutr. Metab. (Lond). 7, 20 (2010).
Schrauwen, I. et al. A frame-shift mutation in CAV1 is associated with a severe neonatal progeroid and lipodystrophy syndrome. PLoS ONE 10(7), e0131797 (2015).
Viljanen, A. P. M. et al. Effects of weight loss on visceral and abdominal subcutaneous adipose tissue blood-flow and insulin-mediated glucose uptake in healthy obese subjects. Ann. Med. 41(2), 152–160 (2009).
Razani, B. et al. Caveolin-1-deficient mice are lean, resistant to diet-induced obesity, and show hypertriglyceridemia with adipocyte abnormalities. J. Biol. Chem. 277(10), 8635–8647 (2002).
Swinburn, B. A., Caterson, I., Seidell, J. C. & James, W. P. Diet, nutrition and the prevention of excess weight gain and obesity. Public Health Nutr. 7(1a), 123–146 (2004).
Najjar, R. S. & Feresin, R. G. Plant-based diets in the reduction of body fat: Physiological effects and biochemical insights. Nutrients 11(11), 1 (2019).
Dillard, C. J. & German, J. B. Phytochemicals: Nutraceuticals and human health. J. Sci. Food Agric. 80(12), 1744–1756 (2000).
Rienks, J., Barbaresko, J. & Nöthlings, U. Association of polyphenol biomarkers with cardiovascular disease and mortality risk: A systematic review and meta-analysis of observational studies. Nutrients 9(4), 1 (2017).
Nachvak, S. M. et al. Soy, soy isoflavones, and protein intake in relation to mortality from all causes, cancers, and cardiovascular diseases: A systematic review and dose-response meta-analysis of prospective cohort studies. J. Acad. Nutr. Diet. 119(9), 1483–500.e17 (2019).
Kimble, R., Keane, K. M., Lodge, J. K. & Howatson, G. Dietary intake of anthocyanins and risk of cardiovascular disease: A systematic review and meta-analysis of prospective cohort studies. Crit. Rev. Food. Sci. Nutr. 59(18), 3032–3043 (2019).
Knekt, P. et al. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr. 76(3), 560–568 (2002).
Stanhope, K. L. Sugar consumption, metabolic disease and obesity: The state of the controversy. Crit. Rev. Clin. Lab. Sci. 53(1), 52–67 (2016).
Najjar, R. S., Moore, C. E. & Montgomery, B. D. Consumption of a defined, plant-based diet reduces lipoprotein(a), inflammation, and other atherogenic lipoproteins and particles within 4 weeks. Clin. Cardiol. 41(8), 1062–1068 (2018).
Romeu, M. et al. Diet, iron biomarkers and oxidative stress in a representative sample of Mediterranean population. Nutr. J. 12, 102 (2013).
Rodier, F. & Campisi, J. Four faces of cellular senescence. J. Cell Biol. 192(4), 547–556 (2011).
Cevenini, E., Monti, D. & Franceschi, C. Inflamm-ageing. Curr. Opin. Clin. Nutr. Metab. Care. 16(1), 14–20 (2013).
Weissmann, G. It’s complicated: Inflammation from Metchnikoff to Meryl Streep. Faseb J. 24(11), 4129–4132 (2010).
Murdoch, J. R. & Lloyd, C. M. Chronic inflammation and asthma. Mutat Res. 690(1–2), 24–39 (2010).
Chambers, J. C. et al. C-reactive protein, insulin resistance, central obesity, and coronary heart disease risk in Indian Asians from the United Kingdom compared with European whites. Circulation 104(2), 145–150 (2001).
Donath, M. Y. & Shoelson, S. E. Type 2 diabetes as an inflammatory disease. Nat Rev Immunol. 11(2), 98–107 (2011).
Hotamisligil, G. S. Inflammation and metabolic disorders. Nature 444(7121), 860–867 (2006).
Kanda, H. et al. MCP-1 contributes to macrophage infiltration into adipose tissue, insulin resistance, and hepatic steatosis in obesity. J. Clin. Invest. 116(6), 1494–1505 (2006).
Matveev, S., Uittenbogaard, A., van Der Westhuyzen, D. & Smart, E. J. Caveolin-1 negatively regulates SR-BI mediated selective uptake of high-density lipoprotein-derived cholesteryl ester. Eur. J. Biochem. 268(21), 5609–5616 (2001).
Haddad, D., Al Madhoun, A., Nizam, R. & Al-Mulla, F. Role of Caveolin-1 in diabetes and its complications. Oxid. Med. Cell Longev. 2020, 9761539 (2020).
Oberleithner, H., Kusche-Vihrog, K. & Schillers, H. Endothelial cells as vascular salt sensors. Kidney Int. 77(6), 490–494 (2010).
Cornelis, M. C. et al. Genome-wide meta-analysis identifies regions on 7p21 (AHR) and 15q24 (CYP1A2) as determinants of habitual caffeine consumption. PLoS Genet. 7(4), e1002033 (2011).
Carver, L. A. & Schnitzer, J. E. Caveolae: Mining little caves for new cancer targets. Nat. Rev. Cancer 3(8), 571–581 (2003).
Fernández-Real JM, Catalán V, Moreno-Navarrete JM, Gómez-Ambrosi J, Ortega FJ, Rodriguez-Hermosa JI, et al. Study of caveolin-1 gene expression in whole adipose tissue and its subfractions and during differentiation of human adipocytes. Nutr Metab (Lond). 2010;7:20-.
Otis, J. P., Shen, M. C., Quinlivan, V., Anderson, J. L. & Farber, S. A. Intestinal epithelial cell caveolin 1 regulates fatty acid and lipoprotein cholesterol plasma levels. Dis Model Mech. 10(3), 283–295 (2017).
Baudrand, R., Pojoga, L. H., Romero, J. R. & Williams, G. H. Aldosterone’s mechanism of action: Roles of lysine-specific demethylase 1, caveolin and striatin. Curr. Opin. Nephrol. Hypertens. 23(1), 32–37 (2014).
Bayorh, M. A., Rollins-Hairston, A., Adiyiah, J., Lyn, D. & Eatman, D. Eplerenone suppresses aldosterone/ salt-induced expression of NOX-4. J. Renin. Angiotensin. Aldosterone Syst. 12(3), 195–201 (2011).
Stas, S. et al. Mineralocorticoid receptor blockade attenuates chronic overexpression of the renin-angiotensin-aldosterone system stimulation of reduced nicotinamide adenine dinucleotide phosphate oxidase and cardiac remodeling. Endocrinology 148(8), 3773–3780 (2007).
Wang, S. et al. Caveolin-1: An oxidative stress-related target for cancer prevention. Oxid. Med. Cell Longev. 2017, 7454031 (2017).
Acknowledgements
We would like to thank all participants who gave their time to carry out this project.
Funding
This study was supported by a grant from Tehran University of Medical Sciences (97-03-161-41017).
Author information
Authors and Affiliations
Contributions
F.A. and A.M. conceptualization; methodology; investigation; formal analysis; D.H. and N.B. writing original draft. F.A. and A.M. carried out the experiment. C.C. editing. Kh.M.: supervision; validation; project administration. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abaj, F., Mirzababaei, A., Hosseininasab, D. et al. Interactions between Caveolin-1 polymorphism and Plant-based dietary index on metabolic and inflammatory markers among women with obesity. Sci Rep 12, 9088 (2022). https://doi.org/10.1038/s41598-022-12913-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-12913-y
- Springer Nature Limited
This article is cited by
-
Personalized optimal nutrition lifestyle for self obesity management using metaalgorithms
Scientific Reports (2022)