Abstract
Interleukin (IL) 1, and its family member, IL-1 receptor antagonist (IL-1ra), are involved in the pathogenesis and inflammation perpetuation of patients with rheumatoid arthritis (RA). Besides, IL-1 has been linked to an increased risk and greater severity of cardiovascular (CV) disease. We aimed to study if IL-1ra is related to the CV manifestations—including lipid pattern and insulin resistance or subclinical atherosclerosis—that accompanies the disease in a large series of patients with RA. Cross-sectional study that encompassed 430 patients with RA. Serum IL-1ra levels were assessed. A multivariable analysis was performed to analyze the relation of IL-1ra to subclinical carotid atherosclerosis, and to traditional CV factors including a complete lipid molecules profile and insulin resistance or beta cell function indices. Body mass index, abdominal circumference, and the presence of obesity were significantly and positively associated with circulating IL-1ra. Similarly, erythrocyte sedimentation rate, and disease activity scores were significantly related to higher IL-1ra serum levels after adjustment for confounders. Neither carotid intima-media thickness nor the presence of carotid plaque were associated with serum levels of IL-1ra. However, after multivariable analysis circulating IL-1ra was independently and positively associated with higher serum levels of total cholesterol, triglycerides, and apolipoproteins B and C-III. Similarly, IL-1ra was related to higher levels of beta-cell function in the univariable analysis, although, in this case, significance was lost after adjustment. Among patients with RA, IL-1ra is associated with both disease activity and several traditional CV risk factors such as obesity and the presence of higher lipid levels. Our findings suggest that IL-1ra can represent a link between the inflammation and the CV disease risk that are present in patients with RA.
Similar content being viewed by others
Introduction
Interleukin (IL) 1 is a highly potent proinflammatory mediator that is important in immune defense and in immune-mediated disease. IL-1 encompasses two distinct cytokines, IL-1α and IL-1β, that signal via the same receptor, IL-1 receptor type 1. A related IL-1 family member, IL-1 receptor antagonist (IL-1ra), serves as an inhibitor that competes with both cytokines for binding to the receptor1.
There is ample evidence on the role of IL-1 in the development, pathogenesis, and inflammation perpetuation of patients with rheumatoid arthritis (RA)2. It is believed that a currently unknown antigenic trigger activates, among others, the production of IL-1 in the joint by macrophages, that eventually further produce or secrete additional cellular messengers such as the proteoglycans and proteases that can lead to the formation of the pannus, which accumulates in the joints. Destructive enzymes can then go on to destroy cartilage and ultimately degrade and erode bone. The role of IL-1 in RA or other inflammatory joint diseases has been supported by the fact that three therapies that target this cytokine (anakinra, canakinumab and rinolacept) have shown efficacy in clinical studies. Similarly, IL-1 cytokines have been associated with the pathogenesis of cardiovascular disease (CVD). In this sense, IL-1 has been found to represent a key pathogenetic mechanism in the formation, progression, and complication of atherosclerosis and the myocardial response to ischemic and nonischemic injury3. Furthermore, as in RA, IL-1 targeted therapies have already been shown to improve CV outcomes in clinical trials in patients with or at risk for myocardial infarction, heart failure, and recurrent pericarditis4,5.
Undoubtedly, rheumatoid arthritis (RA) has been associated with a greater presence of subclinical atheromatosis and a high incidence of CV events6. This accelerated CV disease in RA is believed to be due to the inflammatory activity that accompanies the disease7. However, the exact pathophysiological mechanisms or which specific inflammatory molecules are involved in this increased CV disease of RA are unknown.
In the present study, we have evaluated serum IL-1ra levels in a large series of RA patients who underwent evaluation for subclinical carotid atherosclerosis. In addition, a complete lipid pattern and indices of insulin resistance were measured. Our objective was to study how IL-1ra is related to CV risk factors such as dyslipidemia and insulin resistance, and to the subclinical atherosclerosis that is present in the disease.
Material and methods
Study participants
This was a cross-sectional study that included 430 consecutively recruited patients with RA. All of them were 18 years old or older and fulfilled the 2010 ACR/EULAR classification criteria8. They had been diagnosed by rheumatologists and were periodically followed-up at rheumatology outpatient clinics. For the purpose of inclusion in the present study, the duration of RA disease was required to be ≥ 1 year. Since glucocorticoids are often used in the treatment of RA, patients taking prednisone or an equivalent dose ≤ 10 mg / day were allowed to participate. Patients were excluded if they had a history of myocardial infarction, angina, stroke, a glomerular filtration rate < 60 ml/min/1.73 m2, a history of cancer or any other chronic disease such as hypothyroidism, heart or respiratory diseases, nephrotic syndrome, as well as evidence of active infection.
Data collection and laboratory assessments
Individuals included in the study completed a CV risk factors and medications use questionnaire and underwent a physical examination. Body-mass index –BMI– (the weight in kilograms divided by the square of the height in meters), abdominal circumference, and systolic and diastolic blood pressure were assessed under standardized conditions. Information regarding smoking status, diabetes and hypertension was obtained from the questionnaire. Medical records were reviewed to ascertain specific diagnoses and medications. Disease activity in patients with RA was measured using the Disease Activity Score (DAS28) in 28 joints9, the Clinical Disease Activity Index (CDAI)10 and the Simple Disease Activity Index (SDAI)11. Disease disability was measured through the Health Assessment Questionnaire (HAQ) score12. Cholesterol, triglycerides, and HDL-cholesterol were measured using the enzymatic colorimetric assay. LDL-cholesterol was calculated using the Friedewald formula. A standard technique was used to measure the erythrocyte sedimentation rate (ESR) and high-sensitivity C-reactive protein (CRP). The homeostatic model assessment (HOMA) method was performed to determine IR. Briefly, the HOMA model enabled an estimate of insulin sensitivity (%S) and β-cell function (%B) from fasting plasma insulin, C peptide, and glucose concentrations. In this study we used HOMA2, the updated-computer HOMA model13. Human IL-1ra was measured using an enzyme-linked immunosorbent assay –ELISA– for its quantitative detection (Biovendor, Czech Republic). Both intra and inter-coefficients of variability are < 10% for this assay. Blood samples were taken fasting.
Carotid ultrasound assessment
Carotid ultrasound examination was used to assess carotid intima media thickness (cIMT) in the common carotid artery and to detect focal plaques in the extracranial carotid tree in patients with RA14. A commercially available scanner, the Esaote Mylab 70 (Genoa, Italy), equipped with a 7–12 MHz linear transducer and an automated software-guided radiofrequency technique, Quality Intima Media Thickness in real-time (QIMT, Esaote, Maastricht, Holland), was used for this purpose. As previously reported14, based on the Mannheim consensus, plaque criteria in the accessible extracranial carotid tree (common carotid artery, bulb and internal carotid artery) were defined as follows: a focal protrusion in the lumen measuring at least cIMT > 1.5 mm; a protrusion at least 50% greater than the surrounding cIMT; or arterial lumen encroaching > 0.5 mm15.
Statistical analysis
Demographic and clinical characteristics in patients with RA were described as mean (standard deviation) or percentages for categorical variables. For non-normally distributed continuous variables, data were expressed as median and interquartile range (IQR). Multivariable linear regression analysis, adjusting for confounders, was assessed to analyze the association between disease-related data and IL1-ra serum levels. Confounding variables were selected from demographics and traditional CV risk factors if they had a p value lower than 0.20 in the univariable relationship with IL1-ra. Besides, any variable that because of clinical reasons was considered as confounder was included in the multivariable analysis. Beta coefficients of the linear regression analyses were calculated as non-standardized coefficients. Collinearity in the multivariable regression models was checked through the calculation of the variance inflation factor (a variance inflation factor higher than 10 was considered a violation of collinearity). All the analyses used a 5% two-sided significance level and were performed using Stata software, version 17/SE (StataCorp, College Station, TX, USA). P-values < 0.05 were considered statistically significant.
Ethics approval and consent to participate
The study protocol was approved by the institutional review committees at Hospital Universitario de Canarias and Hospital Universitario Doctor Negrín, and all subjects provided written informed consent (approval no. 2019–452-1). All research was performed in accordance with relevant guidelines/regulations and in accordance with the Declaration of Helsinki.
Results
Demographic and disease-related data
A total of 430 patients with RA were included in this study. Demographic- and disease-related characteristics of the participants are shown in Table 1. The mean age was 55 ± 10 years and 81% of the patients were women. Traditional CV risk factors were common. In this regard, 22% were current smokers, 13% had type 2 diabetes, 32% were considered obese (BMI equal to or greater than 30 kg/m2) and 34% had hypertension. Additionally, 32% of the patients were taking statins at the time of the study. The mean cIMT was 696 ± 131 microns, and 42% of the patients had carotid plaques. Full lipid profile and insulin resistance indices are additionally described in Table 1.
IL1-ra serum levels were 390 (IQR 14-2004) pg/ml. The median duration of the disease in this series of patients with RA was 8 (IQR 4-15) years. Seventy-two percent of patients were positive for rheumatoid factor, and 65% for anti-citrullinated protein antibodies. Disease activity measured by DAS28-ESR was 3.1 ± 1.4. Regarding this, 40% of the patients met the definitions of remission, and 18% and 42% were in the low, moderate/high disease activity categories, respectively. Thirty-six percent of the patients were being treated with prednisone and 87% were taking at least one conventional DMARD in any of its types, methotrexate being the most widely used (73%). Nineteen percent of the patients were receiving anti-tumor necrosis factor therapies. The frequency of use of other treatments and historical disease related data are shown in Table 1.
Association of disease characteristics and cardiovascular risk factors with IL-1ra
The relation of CV risk factors and disease related data to IL-1ra (as dependent variable) is shown in Table 2. BMI (beta coefficient –coef. − 22 [95% confidence interval –CI– 14–30] pg/ml, p < 0.001), abdominal circumference (beta coef. 8 [95%CI 5–11) pg/ml, p < 0.001) and the presence of obesity (beta coef. 169 [95%CI 78–260] pg/ml, p < 0.001) were positively associated with circulating IL-1ra. Age showed a trend for a negative relationship to IL-1ra although in this case statistical significance was not reached. Besides, neither cIMT nor the presence of carotid plaque were associated with serum levels of IL-1ra.
Regarding disease-related data, several significant associations were found, after multivariate analysis, for acute phase reactants and disease activity scores. For example, ESR, but not CRP, and DAS28-ESR, DAS 28-CRP and CDAI were significant related to IL-1ra after adjustment for confounders (Table 2). This was not the case for other disease characteristics such as the presence of rheumatoid factor or anti-citrullinated protein antibodies, or the use of several treatments. Only the use of hydroxychloroquine disclosed a negative relation to IL-1ra although statistical significance was not achieved (beta coef. − 125 [95%CI − 253–3] pg/ml, p = 0.055) (Table 2).
Relationship of IL1-ra with the lipid profile and the indices of insulin resistance
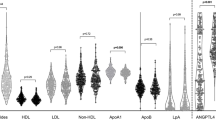
The potential influence of IL-1ra serum levels (as independent variable) on lipid pattern molecules and insulin resistance indices is illustrated in Table 3. After multivariable analysis, IL1-ra was associated with higher serum levels of total cholesterol, triglycerides, non-HDL cholesterol, and apolipoprotein B and C-III. Remarkably, this was found after adjustment for age, diabetes, BMI and waist circumference (Table 3).
The analysis of glucose homeostasis molecules and indices of insulin resistance was only performed in patients with RA who were not diabetic and had a blood glucose lower than 110 mg/dl (Table 3). In this analysis, IL-1ra was associated with higher levels of beta cellfunction (HOMA2-B%) in the univariable analysis (beta coef. 24 [95%CI 138–4], p = 0.019). However, after full multivariable adjustment this relationship was lost (Table 3).
Discussion
Our study is the largest in which IL-1ra serum levels have been analyzed in patients with RA. According to our results, IL-1ra may not only act as an acute phase reactant associated with disease activity, but is also related to some CV comorbidities such as BMI, abdominal circumference and the presence of dyslipidemia. These findings suggest that IL-1 can be involved in both the inflammation present in RA and in the CV comorbidity that accompanies this disease.
We have found in our work a clear relationship between body mass index and abdominal circumference and serum levels of IL-1ra. This is in agreement with the notion that adipose tissue can produce cytokines that have crucial roles in regulating metabolism and immune responses. In obese patients in the general population, it has been described that adipose tissue is responsible for low-grade inflammation through dysregulatory proinflammatory factors16. In obesity, the increased level of adipose tissue-derived IL-1 can result in low-grade inflammation, which is the main culprit of obesity-related complications, such as insulin resistance and liver fibrosis which can facilitate type 2 diabetes and atherosclerosis development17. Probably, and based on our findings, this relationship observed in the general population would be maintained in patients with RA.
A positive and significant relationship between IL-1ra and several molecules related to lipid metabolism such as total cholesterol, triglycerides and lipoproteins B and C-III was found in our study. This is in accordance with the knowledge that IL-1 has been suggested to directly modulate lipid metabolism by suppressing lipoprotein lipase activity18. However, we and others have shown an inverse relationship between inflammation and lipid levels in RA patients19. This does not appear to be the case for IL-1ra where this acute phase reactant is positively associated with higher levels of lipid molecules. However, the fact that most of the patients in our series had low and moderate disease activity and the cross-sectional design of our study does not allow definitive conclusions to be drawn about the influence of IL-1ra on the lipid pattern.
In pancreatic beta cells, IL-1 participates in cytokine-mediated apoptosis and mediates oxidative stress-induced suppression of insulin gene transcription20. Moreover, IL-1β decreases expression of the insulin receptor-1, inhibits glucose transporter translocation to the plasma membrane, and reduces insulin-stimulated glucose uptake and lipogenesis21. This has been supported by the fact that emerging clinical studies have shown that treatment with IL-1 receptor antagonists (anakinra) and IL-1β-specific antibodies (canakinumab) improved glucose metabolism and insulin secretion in patients with type 2 diabetes22. Our result of a relationship between IL-1ra and beta cell function, although only in univariable analysis, is in agreement with what was previously described in a healthy population.
We acknowledge the limitation that in our study we assessed IL1-ra but not IL-1α and IL-1β. However, the assessment of these two cytokines in serum has difficulties due to several factors23. In this sense, circulating IL-1α is usually absent because most of the IL-1α remains in the cytosol of cells in its precursor form, where it can function as an autocrine messenger. Similarly, plasma concentrations of IL-1β are usually below the limit of detection of the available assays (5 pg per milliliter)23. In addition, it is recommended to measure the production of IL-1β in peripheral blood mononuclear cells rather than in serum, since it is generally absent in the latter24. Furthermore, IL-1ra concentrations primarily reflect IL-1 production, so serum levels of the former are proportional to those of the latter25. Taking these considerations together, we believe that measuring IL-1ra in our work was a valid representation of global IL-1 metabolism.
The results shown in this study support the clinical and therapeutic relevance of the biological aspects of IL-1 in CV disease of RA. Three agents are currently available for pharmaceutical inhibition of IL-1: the recombinant IL-1 receptor antagonist (IL-1ra) anakinra; the anti-IL-1β antibody canakinumab; and the IL-1 “trap” fusion molecule rilonacept. In the first large-scale blinded and placebo-controlled, randomized clinical trial that targeted inflammation but not lipids, CANTOS (Canakinumab Anti-Inflammatory Thrombosis Outcome Study) supported the inflammatory hypothesis of atherosclerosis26. Additionally, the CANTOS results highlighted the utility of assessing both LDL-cholesterol and CRP, as targeting either of these biomarkers can reduce recurrent events. We believe that IL-1 inhibition can have a beneficial effect on CV disease and CV events in RA patients.
Conclusion
In conclusion, in patients with RA, serum levels of IL1-ra are significantly associated with the clinical and inflammatory activity of the disease, as well as with different CV comorbidities such as dyslipidemia, beta cell function and obesity. Our findings therefore emphasize the beneficial effect that IL1 inhibition drugs can have both in controlling RA disease activity and the accelerated CV disease that these patients manifest.
Data availability
The data sets used and/or analyzed in the present study are available from the corresponding author upon request.
Abbreviations
- ACPA:
-
Anti-citrullinated peptide/protein antibody
- BMI:
-
Body mass index
- CDAI:
-
Clinical disease activity index
- CI:
-
Confidence of interval
- cIMT:
-
Carotid intima-media thickness
- CRP:
-
C-reactive protein
- CV:
-
Cardiovascular
- DAS28:
-
Disease activity score in 28 joints
- DMARD:
-
Disease-modifying antirheumatic drug
- ELISA:
-
Enzyme-linked immunosorbent assay
- ESR:
-
Erythrocyte sedimentation rate
- HAQ:
-
Health assessment questionnaire
- HDL:
-
High-density lipoprotein
- HOMA:
-
Homeostatic model assessment
- IL-1:
-
Interleukin 1; IL-1ra: Interleukin 1 receptor antagonist
- IQR:
-
Inter quartile range
- LDL:
-
Low-density lipoprotein
- LPL:
-
Lipoprotein lipase
- NSAID:
-
Nonsteroidal anti-inflammatory drug
- RA:
-
Rheumatoid arthritis
- SD:
-
Standard deviation
- SDAI:
-
Simplified disease activity index
- TNF-α:
-
Tumor necrosis factor-α
References
Mantovani, A., Dinarello, C. A., Molgora, M. & Garlanda, C. Interleukin-1 and related cytokines in the regulation of inflammation and immunity. Immunity 50, 778–795 (2019).
Schiff, M. H. Role of interleukin 1 and interleukin 1 receptor antagonist in the mediation of rheumatoid arthritis. Annal Rheum. Dis. 59, i103–i108 (2000).
Abbate, A. et al. Interleukin-1 and the inflammasome as therapeutic targets in cardiovascular disease. Circ. Res. 126(9), 1260–1280 (2020).
Abbate, A. et al. Interleukin-1 blockade inhibits the acute inflammatory response in patients with st-segment–elevation myocardial infarction. J. Am. Heart Assoc. 9(5), e014941 (2020).
Ridker, P. M. et al. Effect of interleukin-1β inhibition with canakinumab on incident lung cancer in patients with atherosclerosis: exploratory results from a randomised, double-blind, placebo-controlled trial. Lancet 390, 1833–1842 (2017).
Corrales, A. et al. Carotid plaques as predictors of cardiovascular events in patients with Rheumatoid Arthritis. Results from a 5-year-prospective follow-up study. Semin. Arthritis Rheum. 50, 1333–1338 (2020).
Ferraz-Amaro, I. et al. Moderate and high disease activity predicts the development of carotid plaque in rheumatoid arthritis patients without classic cardiovascular risk factors: Six years follow-up study. J. Clin. Med. 10(21), 4975 (2021).
Aletaha, D. et al. 2010 rheumatoid arthritis classification criteria: An American college of rheumatology/European league against rheumatism collaborative initiative. Arthritis Rheum. 62, 2569–2581 (2010).
Prevoo, M. L. L. et al. Modified disease activity scores that include twenty-eight-joint counts development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 38, 44–48 (1995).
Smolen, J. S. et al. A simplified disease activity index for rheumatoid arthritis for use in clinical practice. Rheumatology (Oxford) 42, 244–257 (2003).
Aletaha, D. & Smolen, J. The simplified disease activity index (SDAI) and the clinical disease activity index (CDAI): A review of their usefulness and validity in rheumatoid arthritis. Clin. Exp. Rheumatol. 23(5), S100 (2005).
Pincus, T., Swearingen, C. & Wolfe, F. Toward a multidimensional health assessment questionnaire (MDHAQ): Assessment of advanced activities of daily living and psychological status in the patient-friendly health assessment questionnaire format. Arthritis Rheum. 42, 2220–2230 (1999).
Wallace, T. M., Levy, J. C. & Matthews, D. R. Use and abuse of HOMA modeling. Diabet. Care 27, 1487–1495 (2004).
Corrales, A. et al. Carotid ultrasound is useful for the cardiovascular risk stratification of patients with rheumatoid arthritis: Results of a population-based study. Ann. Rheum. Dis. 73, 722–727 (2014).
Touboul, P.-J. et al. Mannheim carotid intima-media thickness consensus (2004–2006). An update on behalf of the advisory board of the 3rd and 4th watching the risk symposium, 13th and 15th European stroke conferences, Mannheim, Germany, 2004, and Brussels, Belgium, 2006. Cerebrovasc. Dis. 23, 75–80 (2007).
Ghanbari, M. et al. Interleukin-1 in obesity-related low-grade inflammation: From molecular mechanisms to therapeutic strategies. Int. Immunopharmacol. 96, 107765 (2021).
Longo, M. et al. Adipose tissue dysfunction as determinant of obesity-associated metabolic complications. Int. J. Mol. Sci. 20, 2358 (2019).
Matsuki, T., Horai, R., Sudo, K. & Iwakura, Y. IL-1 plays an important role in lipid metabolism by regulating insulin levels under physiological conditions. J. Exp. Med. 198, 877–888 (2003).
Tejera-Segura, B. et al. HDL cholesterol efflux capacity in rheumatoid arthritis patients: contributing factors and relationship with subclinical atherosclerosis. Arthritis Res. Ther. 19(1), 1–10 (2017).
Ammendrup, A. et al. The c-Jun amino-terminal kinase pathway is preferentially activated by interleukin-1 and controls apoptosis in differentiating pancreatic β-cells. Diabetes 49, 1468–1476 (2000).
Jager, J., Grémeaux, T., Cormont, M., Le Marchand-Brustel, Y. & Tanti, J. F. Interleukin-1β-induced insulin resistance in adipocytes through down-regulation of insulin receptor substrate-1 expression. Endocrinology 148, 241–251 (2007).
Larsen, C. M. et al. Interleukin-1–receptor antagonist in type 2 diabetes mellitus. N. Engl. J. Med. 356, 1517–1526 (2007).
Epstein, F. H., Dinarello, C. A. & Wolff, S. M. The role of interleukin-1 in disease. N. Engl. J. Med. 328, 106–113 (1993).
Dinarello, C. A. Interleukin-1β. Crit. Care Med. 33(12), S460–S462 (2005).
Arend, W. P. The balance between IL-1 and IL-1Ra in disease. Cytokine Growth Factor Rev. 13, 323–340 (2002).
Ridker, P. M. et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 377, 1119–1131 (2017).
Funding
This work was supported by a grant to I.F-A. and J.C.Q–A from Fundación Canaria Instituto de Investigación Sanitaria de Canarias—FIISC–(PI19/00012).
Author information
Authors and Affiliations
Contributions
I.F.A., M.A.G.G.: Conception, design and interpretation of the data; C.A.S., J.C.Q.A., V.H.H., A.V.G., A.G.D.: Adquisition of the data. All the authors have agreed both to be personally accountable for the author’s own contributions and to ensure that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, are appropriately investigated, resolved, and the resolution documented in the literature. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that there are no conflicts of interest. Nevertheless, Professor MA Gonzalez-Gay and Dr. Iván Ferraz Amaro would like to acknowledge that they received grants/research supports from Abbott, MSD, Jansen and Roche, and received consultation fees from company-sponsored speakers bureaus associated with Abbott, Pfizer, Roche, Sanofi, Sobi, Amgen, Celgene and MSD.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Almeida-Santiago, C., Quevedo-Abeledo, J.C., Hernández-Hernández, V. et al. Interleukin 1 receptor antagonist relation to cardiovascular disease risk in patients with rheumatoid arthritis. Sci Rep 12, 13698 (2022). https://doi.org/10.1038/s41598-022-18128-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-18128-5
- Springer Nature Limited
This article is cited by
-
Prevalence of cardiovascular diseases and traditional cardiovascular risk factors in patients with rheumatoid arthritis: a real-life evidence from BioSTAR nationwide registry
Rheumatology International (2023)
-
Rheumatoid Arthritis Treatment Options and Type 2 Diabetes: Unravelling the Association
BioDrugs (2022)