Abstract
We investigated the characteristics of neovascular age-related macular degeneration (AMD), which rarely recurs after initial remission. This study retrospectively analyzed 392 neovascular AMD patients treated with anti-vascular endothelial growth factor (VEGF). All patients received three monthly loading doses of anti-VEGF injections, followed by a pro re nata (as needed) regimen for 24 months. The baseline characteristics associated with the odds of having no recurrence within 24 months were evaluated using multivariate modeling. After the initial three loading injections over 24 months, 58 (14.8%) eyes showed no exudative recurrence and did not require additional anti-VEGF injections. These patients without exudative recurrence had significantly better best-corrected visual acuity (P = 0.003) and lower central subfoveal thickness (P = 0.035) at 24 months than those with exudative recurrence. Additionally, the incidence of macular atrophy was significantly lower in the former than in the latter (8.6% vs. 21.9%; P = 0.020). Multivariate analysis revealed that younger age (odds ratio [OR], 0.901; P = 0.033), smaller lesion size (OR, 0.589; P = 0.016), and absence of fibrovascular pigment epithelial detachment (PED) (OR, 1.349; P = 0.028) were associated with higher odds of no recurrence during follow-up. Approximately 15% of the neovascular AMD patients showed no exudative recurrence after initial remission during the 24-month follow-up. The infrequent recurrence after initial remission correlated with younger age, smaller lesion size, and absence of fibrovascular PED.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Age-related macular degeneration (AMD) is the leading cause of blindness among elderly people in developed countries1. Neovascular AMD is an advanced form of AMD characterized by the development of macular neovascularization (MNV)2. Since vascular endothelial growth factor (VEGF) is a key factor promoting neovascularization3, intravitreal anti-VEGF injections have been the main treatment option for neovascular AMD for over a decade4.
Anti-VEGF therapy comprises several treatment regimens for the management of neovascular AMD. Monthly fixed injection regimens are mainly used in clinical trials5. In real-world practice, the pro re nata (PRN; as-needed injection) and treat-and-extend (TAE) regimens are commonly used to reduce the treatment burden on patients. Patients receiving the TAE regimen are treated for extended intervals in case of dry macula, and the treatment interval is shortened when exudation is detected. The maximal interval between injections in the TAE protocol is generally 12 weeks6,7. Although TAE involves fewer injections, it is reportedly as effective as a monthly fixed injection regimen administered for 24 months. Therefore, the TAE regimen is currently the primary treatment strategy for patients with neovascular AMD8. When treating neovascular AMD with intravitreal anti-VEGF injections, the requirement for additional treatment varies among patients regardless of the treatment protocols9,10,11.
The Comparison of Age-related Macular Degeneration Treatment Trials (CATT) evaluated a subset of neovascular AMD patients with prolonged remission without additional anti-VEGF treatment. The study reported that 14.8% (n = 96) of the patients did not require additional treatment after the end of the 2-year treatment up to the 5-year follow-up completion. Additionally, among these 96 patients, 43 were treated with the PRN protocol9. For such neovascular AMD patients who maintain long-term remission without the need for additional injection, the TAE or fixed regimen could be an excessive or increased treatment burden.
To date, the characteristics of neovascular AMD cases without recurrence in the long term after initial loading injections have not been reported sufficiently. The prediction of infrequent recurrence may help provide a tailored approach to administer anti-VEGF treatment in patients with neovascular AMD. This study aimed to analyze the characteristics of a subset of neovascular AMD that does not recur after initial remission.
Materials and methods
The AMD database of Kim’s Eye Hospital was reviewed through a computerized search. Patients who were diagnosed with neovascular AMD and had received intravitreal anti-VEGF therapy between January 2016 and March 2019 were identified retrospectively. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the Institutional Review Board of Kim’s Eye Hospital for the review and analysis of patient data. The requirement for informed consent from the patients was waived by the Institutional Review Board.
Study population
The inclusion criteria for this study were as follows: (1) age > 50 years; (2) active MNV confirmed by multimodal imaging including spectral-domain optical coherence tomography (SD-OCT), fluorescein angiography (FA), and indocyanine green angiography (ICGA) at the first visit; (3) treatment-naïve MNV treated with anti-VEGF with ranibizumab (0.5 mg/0.05 mL) or aflibercept (2 mg/0.05 mL) using the PRN regimen; and (4) 24-month follow-up completed after the initial diagnosis.
The exclusion criteria were as follows: (1) treatment with a fixed regimen or proactive treatment, such as TAE; (2) foveal scars or fibrosis at the initial evaluation; and (3) concomitant retinal disorders that could affect visual outcomes, including high myopia, diabetic retinopathy, or retinal vein occlusion. When a patient had bilateral neovascular AMD, only the eye with the earlier diagnosis was included in the analysis.
All patients with neovascular AMD had received consecutive monthly loading anti-VEGF injections. After the initial loading injections, the patients were followed up for 2 years at intervals of 4–8 weeks. Based on the SD-OCT findings, additional PRN regimen anti-VEGF injections were administered when exudative recurrence was detected. Standardized examinations, including best-corrected visual acuity (BCVA), intraocular pressure, fundus examination, and SD-OCT (comprising 31 horizontal lines in 6 mm × 6 mm area) were performed at every visit for all the patients.
Image analysis
Two retinal specialists (W.Y. and J.K.) independently analyzed all SD-OCT scans of the enrolled patients to detect exudative recurrence during the 24-month maintenance phase. Subretinal fluid (SRF) was defined by hyporeflective spaces between the photoreceptor layer and retinal pigment epithelium (RPE) on the SD-OCT images, while intraretinal fluid (IRF) was defined by hyporeflective spaces within the neurosensory retina12. Other macular changes, including subretinal hyperreflective material (SHRM)13 and retinal hemorrhage, were also evaluated.
Initial remission after anti-VEGF treatment was defined as complete resolution of the macular fluid, including SRF and IRF, after the three loading injections. Persistent pigment epithelial detachment (PED) was not considered a criterion indicating initial remission. Exudative recurrence was defined as the occurrence of SRF, IRF, retinal hemorrhage, or SHRM on SD-OCT after initial remission.
Various baseline characteristics of the neovascular AMD patients were analyzed. Using SD-OCT and FA/ICGA images, the lesions were classified as type 1 MNV (sub-RPE choroidal neovascularization [CNV]), type 2 MNV (subretinal CNV), or type 3 MNV (retinal angiomatous proliferation [RAP])2. Type 1 MNV cases with typical polypoidal lesions on ICGA were further classified as polypoidal choroidal vasculopathy (PCV; aneurysmal type 1 MNV)2. Additionally, the lesions were localized, and the lesion size was manually measured on the FA/ICGA image. Central foveal thickness was measured as the distance between the internal limiting membrane and surface of the RPE at the foveal center, while subfoveal choroidal thickness was measured as the distance from the outer portion of the hyperreflective line of the RPE to the hyporeflective line of the sclerochoroidal interface at the foveal center. All measurements were estimated using built-in software of the HRA-2 machine (Heidelberg Eye Explorer software, version 6.0.9.0; Heidelberg Engineering).
The PED definition used in this study was in line with that of our previous investigations and other clinical trials14,15,16, i.e., RPE elevation > 400 µm in width and > 75 µm in height or RPE elevation > 200 µm in vertical height. Furthermore, based on the SD-OCT findings, PED was classified as fibrovascular, when there was moderately reflective space adherent under the surface of the PED, or serous, when the PED was optically clear15,17. When the PED subtypes could not be determined on SD-OCT, the dominant component of the PED was evaluated using the corresponding FA/ICGA images.
Visual and anatomical outcomes
The change in BCVA (converted from Snellen visual acuity to the logarithm of the minimal angle of resolution for statistical analysis) from baseline to 3, 6, 12, 18, and 24 months post-treatment was determined as the visual outcome. The proportion of patients who gained or lost more than three lines of BCVA when compared with the baseline was evaluated. Additionally, those with BCVA > 20/40 or < 20/200 after the treatment were evaluated.
The change in the central foveal thickness and incidence of macular atrophy (MA) during the study period were assessed as the anatomical outcomes. Based on the method reported in our previous publications, the development of MA was evaluated at 24 months from baseline18,19. In case of a hypopigmented area > 250 µm within the macular vascular arcades, the lesion was determined as MA after confirming the following: (1) visibility of the underlying choroidal vasculature; (2) SD-OCT findings of increased signal transmission in the choroid in the absence of RPE; or (3) reduced autofluorescence signal on autofluorescence images.
Statistical analysis
A chi-square test was performed for comparing the categorical variables between the groups and student’s t-test for comparing the continuous variables. Stepwise multivariate logistic regression analysis was performed to identify the relationship between neovascular AMD without exudative recurrence during the maintenance phase and the patients’ baseline clinical characteristics. SPSS software, version 18.0 (SPSS Inc., Chicago, IL, USA), was used for all statistical analyses; P-values < 0.05 were considered statistically significant.
Results
In total, 608 eyes with neovascular AMD that had been followed up for 24 months were initially identified from the database. Among these, 216 eyes were excluded due to treatment with the TAE regimen (n = 178), foveal scar or atrophy at baseline (n = 32), and other concurrent macular diseases including diabetic retinopathy or retinal vein occlusion (n = 6).
The mean age of the study group was 69.8 ± 8.4 years, and the average number of anti-VEGF injections administered in all the patients during the 24-month study period was 9.3 ± 4.8 (range, 3–17). All the patients were South Korean. Table 1 presents detailed clinical data of the enrolled patients.
Neovascular AMD without exudative recurrence during the maintenance phase
Among the 392 eyes included, 329 (83.9%) showed initial remission after the three loading injections. Moreover, 58 eyes (14.8%) showed no exudative recurrence after the initial loading injection during the 24-month follow-up and did not need additional injections. Exudative recurrence was observed in 334 eyes (85.2%) more than once during the 24-month maintenance phase.
Several significant differences in the baseline characteristics were observed between the groups with (n = 334) and without (n = 58) exudative recurrence. Patients without recurrence were significantly younger in age than those with recurrence (63.6 ± 8.8 years vs. 70.0 ± 9.1 years; P = 0.013) (Table 1). The mean lesion size of the eyes without exudative recurrence was significantly smaller than that of the eyes with exudative recurrence (1.08 ± 0.81 mm2 vs. 2.25 ± 1.88 mm2; P = 0.008) (Table 1). Fibrovascular PED was less frequent in eyes without exudative recurrence than in those with exudative recurrence (55.2% vs. 70.4%; P = 0.022) (Table 1). The mean number of injections administered was significantly lower in the group without recurrence than in the group with exudative recurrence, as the former did not receive additional injections after the three loading injections during the 24-month period (3.0 ± 0.0 vs. 10.2 ± 5.3; P < 0.001) (Table 1).
There were no differences in sex, mean baseline BCVA, mean central subfoveal thickness, mean subfoveal choroidal thickness, lesion location, presence of choroidal vascular hyperpermeability, baseline fluid features, or MNV subtype between the two groups (Table 1).
Visual outcomes
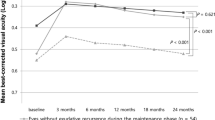
The time course of the BCVA changes was compared between the eyes with and without exudative recurrence during the 24-month follow-up (Fig. 1). The visual gain after the three loading injections tended to be maintained in the eyes without exudative recurrence for 24 months, whereas that of the eyes with exudative recurrence showed a gradual decline, and eventually, the BCVA showed no significant difference compared to that at baseline (Fig. 1). After the 24-month follow-up, the BCVA of the eyes without recurrence was significantly better than that of the eyes with recurrence during the maintenance phase (0.35 ± 0.30 [Snellen equivalent 20/44] vs. 0.47 ± 0.29 [20/59]; P = 0.003) (Fig. 1).
Mean best-corrected visual acuity (BCVA) of the groups with and without exudative recurrence during the 24-month follow-up. The improved visual acuity after three loading injections was maintained in the eyes without exudative recurrence during the 24 months, whereas that of the eyes with exudative recurrence showed a gradual decline (*P < 0.05, compared with baseline BCVA). At 24 months, the eyes without exudative recurrence showed significantly improved BCVA than those having exudative recurrence more than once (P = 0.003).
No significant difference was found in the proportion of improved visual acuity (gain of three or more lines in BCVA) between the eyes with and without exudative recurrence at 24 months. On the contrary, the proportion of eyes showing worsening of more than three lines of visual acuity in the group without exudative recurrence was significantly lower than that in the group with exudative recurrence (5.2% [3/58 eyes] vs. 15.3% [51/334 eyes]; P = 0.039) (Table 2). The proportion of eyes with 20/40 or better vision at 24 months tended to be higher in the group without recurrence than in the group with recurrence, without a significant difference between them (44.8% vs. 33.2%; P = 0.083) (Table 2).
Anatomical outcomes
The mean central foveal thickness significantly decreased during the 24-month follow-up in both groups (Fig. 2). After 24 months of treatment, the mean central foveal thickness of the eyes without exudative recurrence was significantly lesser than that of the eyes with exudative recurrence during the maintenance phase (268 ± 133 µm vs. 297 ± 186 µm; P = 0.035) (Fig. 2). The incidence of MA development was significantly lower in the eyes without exudative recurrence than in the eyes with exudative recurrence (8.6% [5/58 eyes] vs. 21.9% [73/334 eyes]; P = 0.020) (Table 2).
Central foveal thickness of the groups with and without exudative recurrence during the 24-month follow-up. The central foveal thickness of both the groups was maintained after the initial three loading injections during the 24 months. The central foveal thickness of the eyes without exudative recurrence was significantly lesser than that of the eyes with recurrence after 24 months (P = 0.035).
Odds for no exudative recurrence during the maintenance phase
Several significant baseline characteristics associated with the absence of exudative recurrence were identified using the multivariate logistic regression analysis (Table 3). Younger age at baseline (odds ratio [OR], 0.901; 95% confidence interval [CI], 0.831–0.979; P = 0.033) was associated with increased odds of no recurrence during the maintenance phase. Similarly, smaller lesion size (OR, 0.589; 95% CI, 0.383–0.901; P = 0.016) and absence of fibrovascular PED (OR, 1.349; 95% CI, 1.226–2.884; P = 0.028) were significantly associated with an increased likelihood of the absence of recurrence. However, serous PED was not significantly associated with absence of recurrence in the maintenance phase (P = 0.652). Other factors, including sex, baseline BCVA, baseline subfoveal choroidal thickness, presence of choroidal vascular hyperpermeability, baseline fluid features, PED height, MNV subtype, and type of anti-VEGF, showed no correlation with the absence of recurrence (Table 3).
Discussion
In this study, only patients treated with the PRN regimen were included because a proactive treatment such as the TAE regimen could decrease the recurrence rate of neovascular AMD20. Hence, our study identified and analyzed the characteristics of a stable subset of neovascular AMD cases that did not show recurrence after initial remission during the long-term follow-up.
Approximately 15% of the patients with neovascular AMD did not require additional anti-VEGF injections after the initial three loading injections during the 24-month follow-up. Despite administering only three anti-VEGF injections in 2 years, patients without exudative recurrence demonstrated better visual and anatomical outcomes and a lower incidence of MA than those with exudative recurrence.
Similar to our results, several previous studies reported stable neovascular AMD cases that did not show recurrence after the initial loading injections. In the SUSTAIN study, which applied the PRN regimen in 513 patients, approximately 20% of the patients did not require an injection after the first three treatments during the 12-month follow-up21. In another retrospective study of 139 eyes, 25.2% of the patients presenting dry macula after the loading treatment did not need additional injections during the 24-month follow-up22. In the EVEREST study, 14.1% of PCV patients in the ranibizumab monotherapy group did not require additional anti-VEGF injections after the three loading injections during 12 months23.
In neovascular AMD patients who do not experience recurrence after the initial loading injection, the TAE or fixed regimen might involve unnecessary anti-VEGF injections and increased treatment burden. Furthermore, although debatable, there are concerns that an increased number of anti-VEGF injections may lead to MA progression9. Therefore, the prediction that stable neovascular AMD shows infrequent recurrence could assist the clinicians in managing patients in an individualized manner. In this study, several baseline characteristics relevant to stable neovascular AMD without recurrence during the 24-month follow-up were identified.
Infrequent exudative recurrence correlated with younger age and smaller lesion size. A previous study reported that younger age was associated with infrequent recurrence of neovascular AMD22. Additionally, the CNV size correlates with visual prognosis. There is evidence regarding an association between smaller CNV lesions and better visual acuity outcomes in several phase 3 trials24,25. Furthermore, smaller CNV areas were associated with a decreased risk of exudative recurrence, particularly IRF development, during anti-VEGF treatment26. In addition to these lines of evidence regarding the favorable prognosis of smaller CNV lesions, our results suggest that during anti-VEGF treatment, consideration of lesion size could be a predictor for recurrence and can assist in deciding the treatment regimen after the loading injections.
The absence of fibrovascular PED was identified as another predictive factor for the low recurrence risk during the long-term follow-up. On the other hand, the presence of serous PED was not significantly associated with recurrence. It has been reported that PED is associated with an increased treatment frequency27. Particularly, PED at baseline was more likely to cause recurrence and activity when shifting from a fixed dosing regimen to a flexible dosing regimen12. More recently, it was reported that a higher PED height increases the risk of IRF development during anti-VEGF treatment26. Hence, neovascular AMD with fibrovascular PED should be monitored cautiously. Moreover, the PRN regimen might be insufficient to preserve the visual gain obtained after the first remission.
The frequency of recurrence during anti-VEGF treatment may be affected by the subtype of neovascular AMD, because the response to anti-VEGF varies slightly among the neovascular AMD subtypes28. Previously, several studies reported that the subtype of neovascular AMD is associated with the frequency of exudative recurrence. Kuroda et al. reported that PCV tended to recur frequently after the loading injection22. Compared to the other types of MNV, RAP (type 3 MNV) could reportedly achieve a long remission period of 6 or 12 months without recurrence29,30. However, in our study, the subtype of neovascular AMD did not demonstrate any association with the absence of recurrence after initial remission. This could be attributed to the difference between the remission periods of each study; our study evaluated a longer remission period of 24 months compared to that of previous studies29,30. Since type 3 MNV is almost reactivated within 7–12 months after the loading injection28, it did not show a difference in the recurrence frequency as compared to the other MNV types during the 24-month follow-up.
Our study has several limitations, including its retrospective design. A large proportion of participants treated with the TAE regimen were excluded, which possibly introduced a bias. Future studies should evaluate the association between the frequency of exudative recurrence and TAE regimen. Furthermore, studies should be performed to determine the optimal strategy to manage patients with neovascular AMD, who are anticipated to experience infrequent recurrence after initial remission. Additionally, the stability of neovascular AMD, which does not recur, should be evaluated over a longer follow-up period.
In conclusion, 14.8% of the eyes with neovascular AMD in our study showed no exudative recurrence during the 24-month follow-up and did not require additional anti-VEGF injections after the initial three loading injections. Younger age, smaller lesion size, and absence of fibrovascular PED correlated with infrequent recurrence after initial remission. Identifying the predictive factors at baseline might be clinically relevant, as this information allows clinicians to categorize patients better and provide individualized anti-VEGF treatments.
Data availability
The datasets used and analyzed in this study will be available from the corresponding author on reasonable request.
References
Bressler, N. M. et al. Potential public health impact of age-related eye disease study results: AREDS report no. 11. Arch. Ophthalmol. 121, 1621–1624. https://doi.org/10.1001/archopht.121.11.1621 (2003).
Spaide, R. F. et al. Consensus nomenclature for reporting neovascular age-related macular degeneration data: Consensus on neovascular age-related macular degeneration nomenclature study group. Ophthalmology 127, 616–636. https://doi.org/10.1016/j.ophtha.2019.11.004 (2020).
Otani, A. et al. Vascular endothelial growth factor family and receptor expression in human choroidal neovascular membranes. Microvasc. Res. 64, 162–169. https://doi.org/10.1006/mvre.2002.2407 (2002).
Fu, D. J. et al. Insights from survival analyses during 12 years of anti-vascular endothelial growth factor therapy for neovascular age-related macular degeneration. JAMA Ophthalmol. 139, 57–67. https://doi.org/10.1001/jamaophthalmol.2020.5044 (2021).
Group, C. R. et al. Ranibizumab and bevacizumab for neovascular age-related macular degeneration. N. Engl. J. Med. 364, 1897-1908, https://doi.org/10.1056/NEJMoa1102673 (2011).
Wykoff, C. C. et al. Prospective trial of treat-and-extend versus monthly dosing for neovascular age-related macular degeneration: TREX-AMD 1-year results. Ophthalmology 122, 2514–2522. https://doi.org/10.1016/j.ophtha.2015.08.009 (2015).
Gupta, O. P. et al. A treat and extend regimen using ranibizumab for neovascular age-related macular degeneration clinical and economic impact. Ophthalmology 117, 2134–2140. https://doi.org/10.1016/j.ophtha.2010.02.032 (2010).
Aslanis, S., Amren, U., Lindberg, C. & Epstein, D. Recurrent neovascular age-related macular degeneration after discontinuation of vascular endothelial growth factor inhibitors managed in a treat-and-extend regimen. Ophthalmol. Retina 6, 15–20. https://doi.org/10.1016/j.oret.2021.03.010 (2022).
Comparison of Age-related Macular Degeneration Treatments Trials Research, G. et al. Five-year outcomes with anti-vascular endothelial growth factor treatment of neovascular age-related macular degeneration: The comparison of age-related macular degeneration treatments trials. Ophthalmology 123, 1751-1761, https://doi.org/10.1016/j.ophtha.2016.03.045 (2016).
Madhusudhana, K. C. et al. UK neovascular age-related macular degeneration database. Report 6: time to retreatment after a pause in therapy. Outcomes from 92 976 intravitreal ranibizumab injections. Br. J. Ophthalmol. 100, 1617–1622. https://doi.org/10.1136/bjophthalmol-2015-308077 (2016).
Rayess, N., Houston, S. K., Gupta, O. P., Ho, A. C. & Regillo, C. D. Treatment outcomes after 3 years in neovascular age-related macular degeneration using a treat-and-extend regimen. Am. J. Ophthalmol. 159, 3–8. https://doi.org/10.1016/j.ajo.2014.09.011 (2015).
Schmidt-Erfurth, U. & Waldstein, S. M. A paradigm shift in imaging biomarkers in neovascular age-related macular degeneration. Prog. Retin. Eye Res. 50, 1–24. https://doi.org/10.1016/j.preteyeres.2015.07.007 (2016).
Dansingani, K. K. et al. Subretinal hyperreflective material imaged with optical coherence tomography angiography. Am. J. Ophthalmol. 169, 235–248. https://doi.org/10.1016/j.ajo.2016.06.031 (2016).
Cho, H. J. et al. Development of intraretinal fluid in neovascular age-related macular degeneration during anti-vascular endothelial growth factor treatment. Am. J. Ophthalmol. https://doi.org/10.1016/j.ajo.2021.07.026 (2021).
Cho, H. J. et al. Response of pigment epithelial detachment to anti-vascular endothelial growth factor treatment in age-related macular degeneration. Am. J. Ophthalmol. 166, 112–119. https://doi.org/10.1016/j.ajo.2016.03.039 (2016).
Waldstein, S. M. et al. Morphology and visual acuity in aflibercept and ranibizumab therapy for neovascular age-related macular degeneration in the view trials. Ophthalmology 123, 1521–1529. https://doi.org/10.1016/j.ophtha.2016.03.037 (2016).
Skondra, D., Papakostas, T. & Vavvas, D. G. Enhanced depth imaging optical coherence tomography in age-related macular degeneration. Semin. Ophthalmol. 27, 209–212. https://doi.org/10.3109/08820538.2012.708807 (2012).
Cho, H. J. et al. Risk factors for geographic atrophy after intravitreal ranibizumab injections for retinal angiomatous proliferation. Am. J. Ophthalmol. 159, 285–292. https://doi.org/10.1016/j.ajo.2014.10.035 (2015).
Cho, H. J. et al. Progression of macular atrophy in patients undergoing anti-vascular endothelial growth factor therapy for neovascular age-related macular degeneration. Acta Ophthalmol. 99, e540–e546. https://doi.org/10.1111/aos.14631 (2021).
Hwang, R. Y., Santos, D. & Oliver, S. C. N. Rates of exudative recurrence for eyes with inactivated wet age-related macular degeneration on 12-week interval dosing with bevacizumab therapy. Retina 40, 679–685. https://doi.org/10.1097/IAE.0000000000002446 (2020).
Holmes, J. P. et al. Use of booster inoculations to sustain the clinical effect of an adjuvant breast cancer vaccine: from US Military Cancer Institute Clinical Trials Group Study I-01 and I-02. Cancer 117, 463–471. https://doi.org/10.1002/cncr.25586 (2011).
Kuroda, Y. et al. Factors associated with recurrence of age-related macular degeneration after anti-vascular endothelial growth factor treatment: A retrospective cohort study. Ophthalmology 122, 2303–2310. https://doi.org/10.1016/j.ophtha.2015.06.053 (2015).
Koh, A. et al. Efficacy and safety of ranibizumab with or without verteporfin photodynamic therapy for polypoidal choroidal vasculopathy: A randomized clinical trial. JAMA Ophthalmol. 135, 1206–1213. https://doi.org/10.1001/jamaophthalmol.2017.4030 (2017).
Kaiser, P. K. et al. Ranibizumab for predominantly classic neovascular age-related macular degeneration: subgroup analysis of first-year ANCHOR results. Am. J. Ophthalmol. 144, 850–857. https://doi.org/10.1016/j.ajo.2007.08.012 (2007).
Ying, G. S. et al. Baseline predictors for one-year visual outcomes with ranibizumab or bevacizumab for neovascular age-related macular degeneration. Ophthalmology 120, 122–129. https://doi.org/10.1016/j.ophtha.2012.07.042 (2013).
Cho, H. J. et al. Development of Intraretinal fluid in neovascular age-related macular degeneration during anti-vascular endothelial growth factor treatment. Am. J. Ophthalmol. 234, 6–14. https://doi.org/10.1016/j.ajo.2021.07.026 (2022).
Ashraf, M., Souka, A. & Adelman, R. A. Age-related macular degeneration: using morphological predictors to modify current treatment protocols. Acta Ophthalmol. 96, 120–133. https://doi.org/10.1111/aos.13565 (2018).
Kim, J. H., Chang, Y. S., Kim, J. W., Kim, C. G. & Lee, D. W. Long-term incidence and timing of reactivation in patients with type 3 neovascularization after initial treatment. Graefes Arch. Clin. Exp. Ophthalmol. 257, 1183–1189. https://doi.org/10.1007/s00417-019-04306-1 (2019).
Muftuoglu, I. K. et al. Long-term remission of neovascular age-related macular degeneration with as-needed anti-vascular endothelial growth factor therapy. Retina 38, 516–522. https://doi.org/10.1097/IAE.0000000000001572 (2018).
Lin, T. et al. Factors associated with extended remission in neovascular age-related macular degeneration on pro re nata treatment protocol. Br. J. Ophthalmol. 104, 58–63. https://doi.org/10.1136/bjophthalmol-2018-313447 (2020).
Acknowledgements
This study was supported by Kim’s Eye Hospital Research Center.
Funding
The authors have no proprietary or commercial interest in any materials discussed in this article.
Author information
Authors and Affiliations
Contributions
Design and conduction of the study H.J.C.; Data collection Y.J.J., W.Y., J.Y., J.K., and H.J.C.; Analysis and interpretation of data H.J.C. and W.Y.; Writing of the article H.J.C.; Critical revision and final approval of article J.W.K., and H.J.C.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cho, H.J., Jeon, Y.J., Yoon, W. et al. Neovascular age-related macular degeneration without exudative recurrence over 24 months after initial remission. Sci Rep 12, 15662 (2022). https://doi.org/10.1038/s41598-022-19400-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-19400-4
- Springer Nature Limited
This article is cited by
-
Factors associated with the development of exudation in treatment-naive eyes with nonexudative macular neovascularization
Graefe's Archive for Clinical and Experimental Ophthalmology (2024)