Abstract
Identify risk factors associated with mortality in HIV patients admitted to an ICU in the city of Bogotá. Retrospective cohort study of patients treated in an ICU during the years 2017–2019. The analysis included descriptive statistics, association tests, and a logistic regression model. A predictive model of mortality at the time of admission to the ICU was developed. 110 HIV patients were identified. Association was found between a Charlson index ≥ 6 and mortality (OR = 2.3, 95% CI 1.0–5.1) and an increase in mortality in the first 21 days of ICU stay (OR = 2.2, 95% CI 1.0–4.9). In the logistic regression analysis, the absence of highly active antiretroviral therapy (HAART) upon admission to the ICU (OR = 2.5 95% CI 1.0–6.1) and the first 21 days of ICU stay (OR = 2.3 95% CI 1.0–5.4) were associated with an increase in mortality. The predictive mortality model established that mortality was higher in patients admitted to the ICU without having previously received HAART than in those who did receive therapy at the time of admission to the ICU. In patients with HIV admitted to the ICU, the absence of HAART will negatively impact mortality during their hospital stay.
Similar content being viewed by others
Introduction
Starting in the 1980s with the identification of HIV as the cause of opportunistic infections at the lung level in apparently healthy patients1, infectious and noninfectious complications began to be seen at the metabolic and cardiovascular levels2,3, and respiratory compromise was the first manifestation of the disease and the most frequent cause of admission to the ICU4,5. After the introduction of prophylaxis against Pneumocystis jirovecii and the start of highly active antiretroviral therapy, the morbidity and mortality associated with HIV infection has decreased, allowing a progressive increase in the life expectancy of these patients4.
In the era after the introduction of antiretroviral therapy, the epidemiological profile and prognosis of HIV patients admitted to the intensive care unit changed, and severe sepsis emerged as the leading cause of mortality in the intensive care unit6. Mortality has been related to other factors, such as the stage of the disease and associated noninfectious comorbidities, such as cardiovascular disease, chronic kidney disease, and liver disease, due to direct injury or infection by hepatotropic viruses5,7,8,9,10. Among the predictors evaluated in the ICU, ventilatory support, a high APACHE score at admission (greater than 24), and the use of vasopressors are associated with mortality5,11,12,13. In contrast, early admission to the ICU is associated with a decrease in mortality14.
Although ICU mortality in patients with HIV has decreased, the management of these patients continues to be a challenge for the intensivist due to the systemic affection observed at the time of admission, the presence of opportunistic infections, the associated comorbidities, the social context and the antiretroviral drug interactions.
In Colombia, there are no studies of risk factors and their impact on the mortality of patients with human immunodeficiency virus infection that require management in the intensive care unit. The objective of this study was to evaluate the factors associated with mortality in patients with HIV and/or AIDS hospitalized in the intensive care unit.
Methodology
A retrospective cohort study was carried out at Santa Clara Hospital in the city of Bogotá, which included patients older than 18 years with a diagnosis of human immunodeficiency virus (HIV) infection who were cared for in the intensive care unit (ICU) during the years 2017–2019, excluding pregnant women. The medical records of these patients were reviewed, and data variables presented by these patients at the time of admission to the ICU were extracted and entered for analysis in a matrix in Microsoft Excel previously designed by the researchers.
The outcome variable was defined as death from HIV in the ICU as a dichotomous variable (yes/no). The other variables evaluated included those related to demographic characteristics, to the diagnosis and treatment of HIV and those infections associated with the disease such as tuberculosis, the use of prophylaxis for opportunistic germs, the clinical stage of the disease, the presence of comorbidities, the type of diagnosis of admission to the ICU, the need for advanced supports, complications of ICU management, the presence and number of opportunistic infections, laboratory values and variables associated with the length of stay in the ICU. The cutoff points used to evaluate prognostic markers for HIV-1 infection were similar to those used in previous publications15,16.
Statistic analysis
An analysis was carried out from descriptive and inferential statistics, in which the categorical variables are reported as proportions, the continuous variables as the mean ± SD or as median and Interquartile range (IQR) according to the type of distribution. The association between categorical variables was evaluated with the χ2 statistic (p < 0.05) and Fisher's exact test, and odds ratios were evaluated with 95% confidence intervals (95% CIs). To establish differences between the quantitative variables, the t test and the Mann–Whitney U test were used when appropriate, and the differences were considered statistically significant with a value of p < 0.05. Variables with a value of p < 0.1 and others considered clinically relevant were included in a logistic regression model to determine the factors associated with mortality.
By means of the formula: P (Y = 1) = 1/1 + exp (− α − β1x1 − β2x2 − β3x3), using the beta coefficients of the statistically significant variables, a predictive model of mortality was developed according to the presence or absence of the characteristics in the patients upon admission to the ICU.
All statistical analyses were performed using SPSS Statistical Software, version 25 (IBM, 2017). The study was approved by the ethics committee of Santa Clara Hospital.
Ethics approval and consent to participate
The study was approved by the Research and Innovation Committee of the Integrated Subnetwork of Health Services Center East, State Social Entity (S.S.E.) (CIeI. 12/20) and complies with the principles of the Declaration of Helsinki. In accordance with the national regulations on retrospective studies and considering that no intervention would be carried out in humans, it was agreed with the committee of the Research and Innovation of the Integrated Subnetwork of Health Services Center East S.S.E., belonging to the health secretary of the city of Bogotá, that this study did not require informed consent from the patients.
Results
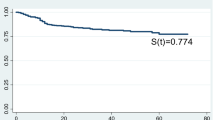
A total of 110 patients with a diagnosis of HIV admitted to the ICU were included of which 64 (58.2%) died. The median age was 39 years (IQR 30–50). Patients older than 60 years represented 11.9% of the total. The male population represented 83.6% of the total. The majority of patients (93.6%) were initially admitted to the emergency department before admission to the ICU. A total of 65.4% of the patients had a previous HIV diagnosis upon admission to the ICU. In those patients without a previous diagnosis of HIV, the diagnosis was established in the emergency room in 18.2% of the cases and in the ICU in 12.7% of the cases. Regarding the severity of the disease, 85.5% had a clinical stage of AIDS, 88.9% had a CD4 lymphocyte count < 200 mm3, and 91.7% had a viral load ≥ 50 mm3. The use of high-intensity antiretroviral therapy before admission to the hospital was documented in 31.8% of the patients; this therapy was started in the ICU in 12.7% of the cases. Regarding comorbidities, a higher frequency of neoplasia associated with AIDS (12.8%), heart disease (6.4%) and COPD (5.5%) was observed. Forty-one percent of the patients had psychoactive substance dependence, and 44.5% of the patients had a Charlson index ≥ 6 upon admission.
Opportunistic infections were documented in 64 patients; among them, 50% had infection by one germ, 34.4% had infection by two germs and 15.6% by three germs. The main opportunistic infections identified were candidiasis (54.6%), tuberculosis (45.3%), cytomegalovirus and cryptococcosis (15%). Seventy percent of the patients were receiving prophylaxis for opportunistic infections, and 41.8% were receiving treatment for tuberculosis at the time of admission (Table 1).
The main diagnoses for admission to the ICU were septic shock (62.8%) and central nervous system infection (41%). Serum lactate values were documented in 94 patients, in whom the median was 1.8 (IQR 1.0–2.5); among these, 45.3% had abnormal values. The serum albumin value on admission was obtained in 57 patients, and the average value was 2.2 (SD ± 0.72), with abnormal values in 8.7% of the cases.
Regarding ICU management, 70% of the patients required vasopressor support, 70.9% ventilatory support, and 61% pharmacological coma. The median time of stay of the patients in the ICU was 21 days (IQR 9–48), and 50.9% of the patients had a time of stay in the ICU equal to or less than 21 days.
The main complication during the ICU stay was acute kidney injury, which was observed in 56.4% of the cases. A total of 58.2% of the patients died during their stay in the ICU (Table 1).
In the bivariate analysis, when comparing the characteristics of the patients who died with those who did not, it was possible to show that the odds ratio (OR) for mortality was 2.3 times higher in those patients with a Charlson index ≥ 6 (95% CI 1.0–5.1). It was also evidenced that the mortality odds ratio was 2.2 times higher in the first 21 days of stay in the ICU (95% CI 1.0–4.9). A protective association was found in those patients with P. jirovecii infection; however, this corresponds to a type 1 error due to a low number of patients with this characteristic (Table 1).
After including in the logistic regression the variables considered statistically significant and those considered relevant in the literature, the model that presented the best fit to explain the outcome was established, finding that the odds ratio of mortality during the first 21 days of ICU stay were 2.3-fold higher (95% CI 1.0–5.4) and that the absence of highly active antiretroviral therapy at ICU admission was associated with a 2.5-fold increase in the odds ratio of mortality (95% CI 1.0–6.1) (Table 2).
Finally, a predictive model of mortality at the time of admission to the ICU was established, including those variables that maintained a statistically significant association with the outcome in the logistic regression model. Highly active antiretroviral therapy was found to be the main determinant of mortality regardless of the length of stay of patients in the ICU, achieving a decrease in mortality from 54.3 to 31.8% in those patients with an ICU stay equal to or less than 21 days and from 73.5 to 55.2% in those patients with a stay longer than 21 days (Table 3).
This decrease in mortality observed in those patients with HAART on admission to the ICU remained stable during the first 21 days of stay in the ICU and in the following days.
Discussion
In our country, we found a high prevalence and mortality (77.2%) in patients with a diagnosis of HIV treated in highly complex hospitals17. Information regarding the clinical characteristics and outcomes in HIV patients admitted to the intensive care unit in our setting is scarce. A previous study carried out at the Hospital Santa Clara in Bogotá showed a mortality close to 50% in this population18, and the cumulative incidence of mortality in this study was 58.2%, considerably high with respect to mortality in developed countries such as the Netherlands (28.2%)19, Spain (25.1%)20, Canada (20%)21, and the United States (19%)22; however, the mortality reported in this study is comparable to that reported by Kwisera et al. in Uganda11.
In the era prior to HAART, ventilatory support had a close relationship with mortality in terms of pulmonary involvement by opportunistic agents, as reported by Morrison23; however, in this study, no relationship with mortality was evidenced. This result reflects the evolution of antiretroviral therapy and advances in the management of critical patients24.
The main complication observed was acute kidney injury, followed by liver failure, unlike that reported in other studies, where the main complication was respiratory failure25.
The proportion of elderly patients in this study was 11.9%, and a cutoff point of 60 years was considered to be associated with mortality in the multivariate analysis. This finding is consistent with the increase in mortality in HIV patients with advancing age26 and in the presence of comorbidities not related to this pathology27. This relationship is more evident with aging28,29.
Regardless of age, comorbidities have an impact on prognosis that can be evaluated with the Charlson index, which, depending on its value, has been associated with increased mortality in critically ill patients30,31. In this study, this association was present with Charlson index values ≥ 6 (OR 2.3, CI 1.0–5.1), a result similar to that reported by Vidal et al.20, but higher than that found in other studies where mortality increases from values of the Charlson index ≥ 332.
After the onset of the HIV pandemic, one of the interventions that changed the natural history of the disease was the introduction of antiretroviral therapy33, significantly modifying mortality. However, the initiation of antiretroviral therapy in critically ill patients is controversial due to the possibility of adverse effects related to the presence of uncontrolled opportunistic infections, the possibility of drug interactions and changes in the pharmacodynamics of critically ill patients due to antiretroviral drugs34. In this study, a predictive model was carried out that showed a clear impact on mortality in critically ill patients who received antiretroviral therapy upon admission to the intensive care unit compared to those who did not receive it (31.8% vs. 54.3%). This benefit prevailed during the entire length of stay in the intensive care unit35.
The median length of stay of patients with HIV infection in the ICU in the era prior to the period before HAART therapy was initiated and after its introduction as standard treatment in these patients is just under 10 days22,36. In this study, the median length of stay was 21 days, with 49.1% of patients exceeding this ICU length of stay, which contrasts with other studies in which 11% of patients had a longer ICU stay37. This could be due to the presence of subacute opportunistic infections at the central nervous system level, given the number of patients who had a documented central nervous system infection at the time of admission (41%), which could contribute to the progression of central respiratory failure with the need for mechanical ventilation.
A decrease in mortality was noted from day 21 of ICU stay, which contrasts with the results of the study by Moitra et al.38, which demonstrated a decreased possibility of discharge home at the time of hospital discharge and an increase in mortality from day seven of ICU stay.
The increase in mortality seen in this study during the first three weeks of ICU stay could be explained by comorbidities and HIV-related patient characteristics. A high percentage of patients with AIDS (85.4%) and with CD4 lymphocyte counts < 200/mm were detected (88.9%), suggesting that this population is at high risk for AIDS-associated diseases.
It is important to emphasize that the high percentage of prophylaxis for opportunistic germs (70.6%) could explain the low isolation of P. jirovecii. In the case of tuberculosis as an opportunistic infection, its presence did not increase mortality when compared with other infections, which would reflect a protective effect of the treatment for this disease that patients had been receiving before admission to the ICU.
Patients with CD4 lymphocyte counts < 200 mm3 and viral loads ≥ 50 mm3 had higher mortality, although most of the patients received treatment for opportunistic infections. Unlike from what was expected, mortality was similar to that of those who did not receive prophylaxis for opportunistic germs, which suggests additional causes of mortality to the presence of this type of infection, such as those associated with sepsis of bacterial origin and their respective complications6,39.
This study is not without its limitations. First, it is a retrospective cohort, monocentric study, which limits the adequate collection of information and may suggest associations that could change with increasing sample size. The information was limited in data on variables that have shown an impact on mortality in previous studies, such as serum albumin values, viral load and CD4 lymphocyte count. Information on the impact of sepsis and its treatment strategies on mortality is also lacking because no information is available to compare data obtained in the days after admission with those obtained at the time of ICU admission.
A protective association was found in those patients with opportunistic infection by P. jirovecii, which is not plausible and is considered a result of the limited sample size; therefore, the results should be analyzed with caution. In the same way, because the analyzed data came from a single hospital institution, it is recommended that in future investigations, multicenter studies be carried out with the calculation of random and representative samples of the population.
In terms of strengths, this study represents a subpopulation of HIV patients who present to the ICU in poor general health due to severity of infection, low CD4 lymphocyte count, and lack of or low adherence to antiretroviral treatment, which is in contrast to what has been observed in the general population of HIV patients. This warrants a more comprehensive approach to finding and treating diseases that may or may not be associated with advanced stages of HIV infection.
Conclusions
This study, which assessed risk factors for mortality in HIV patients admitted to the ICU, found that a Charlson index ≥ 6 and the first 3 weeks of ICU stay were associated with mortality; in addition, advanced age contributed to this fact. In contrast, the use of highly active antiretroviral therapy in HIV patients before admission to the ICU is associated with a decrease in mortality that is seen throughout the duration of hospitalization.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article.
Abbreviations
- AIDS:
-
Acquired immunodeficiency syndrome
- ARDS:
-
Acute respiratory distress syndrome
- APACHE:
-
Acute Physiology and Chronic Health Evaluation
- CI:
-
Confidence interval
- COPD:
-
Chronic Obstrucive Pulmonary Disease
- HAART:
-
Highly active antiretroviral therapy
- HIV:
-
Human immunodeficiency virus
- ICU:
-
Intensive care unit
- IQR:
-
Interquartile range
- IRIS:
-
Immune reconstitution inflammatory syndrome
- SD:
-
Standard deviation
- SE:
-
Standard error
- OR:
-
Odds ratio
References
Delgado, R. Virological characteristics of HIV. Enferm. Infect. Microbiol. Clin. 29, 58–65 (2011).
Hadigan, C. & Kattakuzhy, S. Diabetes mellitus type 2 and abnormal glucose metabolism in the setting of human immunodeficiency virus. Endocrinol. Metab. Clin. N. Am. 43, 685–696 (2014).
Warriner, A. H., Burkholder, G. A. & Overton, E. T. HIV-related metabolic comorbidities in the current ART Era. Infect. Dis. Clin. N. Am. 28, 457–476 (2014).
Sarkar, P. & Rasheed, H. F. Clinical review: Respiratory failure in HIV-infected patients—A changing picture. Crit. Care 17, 66 (2013).
Barbier, F. et al. Etiologies and outcome of acute respiratory failure in HIV-infected patients. Intensive Care Med. 35, 1678–1686 (2009).
Japiassú, A. M. et al. Sepsis is a major determinant of outcome in critically ill HIV/AIDS patients. Crit. Care 14, 66 (2010).
Vachiat, A., McCutcheon, K., Tsabedze, N., Zachariah, D. & Manga, P. HIV and Ischemic heart disease. J. Am. Coll. Cardiol. 69, 73–82 (2017).
Kaspar, M. B. & Sterling, R. K. Mechanisms of liver disease in patients infected with HIV. BMJ Open Gastroenterol. 4, 66 (2017).
Randall, D. W. et al. Acute kidney injury among HIV-infected patients admitted to the intensive care unit. Int. J. STD AIDS 26, 915–921 (2015).
Kenji Yassue, A. et al. AIDS in intensive care unit patients: Epidemiology and outcomes. J. AIDS Clin. Res. 8, 66 (2017).
Kwizera, A. et al. Clinical characteristics and short-term outcomes of HIV patients admitted to an African Intensive Care Unit. Crit. Care Res. Pract. 2016, 66 (2016).
Nichas, G. & Wachter, R. M. Outcomes of intensive care for patients with human immunodeficiency virus infection. Arch. Intern. Med. 160, 541–547 (2000).
Xiao, J. et al. Etiology and outcomes for patients infected with HIV in intensive care units in a tertiary care hospital in China. J. Med. Virol. 87, 366–374 (2015).
Chiang, H. H. et al. Admissions to intensive care unit of HIV-infected patients in the era of highly active antiretroviral therapy: Etiology and prognostic factors. Crit. Care 15, 66 (2011).
Holmes, C. B. et al. CD4 decline and incidence of opportunistic infections in Cape Town, South Africa: Implications for prophylaxis and treatment. J. Acquir. Immune Defic. Syndr. 42, 464–469 (2006).
BHIVA. BHIVA Guidelines for the Treatment of HIV-1-Positive Adults with Antiretroviral Therapy 2015 (2016 Interim Update). Accessed 14/12/2016. 2015, 1–151 (2016).
MontúfarAndrade, F. et al. Epidemiología de la infección por el virus de inmunodeficiencia humana en pacientes hospitalizados en una institución de alta complejidad y enseñanza universitaria en Medellín, Colombia. Infectio 20, 9–16 (2016).
Vergara Erika, A., Pulido-Ramírez, J. A., L. S. Médica Colombiana, A. (2017). Trabajos de Investigación X Congreso Latinoamericano de Medicina Interna XXVI Congreso ACMI-ACP. Acta Médica Colomb. 42, 112 (2017).
Huson, M. A. et al. Characteristics and outcome of patients with AIDS in Dutch ICUs between 1997 and 2014∗. Crit. Care Med. 44, 291–299 (2016).
Vidal-Cortés, P. et al. Epidemiology and outcome of HIV-infected patients admitted to the ICU in the current highly active antiretroviral therapy era. Med. Intensiva 44, 283–293 (2020).
Turvey, S. L., Bagshaw, S. M., Eurich, D. T. & Sligl, W. I. Epidemiology and outcomes in critically ill patients with human immunodeficiency virus infection in the era of combination antiretroviral therapy. Can. J. Infect. Dis. Med. Microbiol. 66, 2017 (2017).
Akgün, K. M. et al. Medical ICU admission diagnoses and outcomes in human immunodeficiency virus-infected and virus-uninfected veterans in the combination antiretroviral era. Crit. Care Med. 41, 1458–1467 (2013).
Morris, A. et al. Intensive care of human immunodeficiency virus-infected patients during the era of highly active antiretroviral therapy. Am. J. Respir. Crit. Care Med. 166, 262–267 (2002).
Nirappil, F. J. et al. Characteristics and outcomes of HIV-1-infected patients with acute respiratory distress syndrome. J. Crit. Care 30, 60–64 (2015).
Afessa, B. & Green, B. Clinical course prognostic factors and outcome prediction for HIV patients in the ICU: The PIP (Pulmonary complications, ICU support, and prognostic factors in hospitalized patients with HIV) study. Chest 118, 138–145 (2000).
Legarth, R. A. et al. Long-term mortality in HIV-infected individuals 50 years or older: A nationwide, population-based cohort study. J. Acquir. Immune Defic. Syndr. 71, 213–218 (2016).
Skiest, D. J., Rubinstien, E., Carley, N., Gioiella, L. & Lyons, R. The importance of comorbidity in HIV-infected patients over 55: A retrospective case-control study. Am. J. Med. 101, 605–611 (1996).
Bernard, C. et al. Aging with HIV: What effect on mortality and loss to follow-up in the course of antiretroviral therapy? The IeDEA West Africa cohort collaboration. HIV/AIDS Res. Palliat. Care 10, 239–252 (2018).
Hentzien, M. et al. HIV-related excess mortality and age-related comorbidities in patients with HIV aged ≥60: A relative survival analysis in the French Dat’AIDS cohort. BMJ Open 9, e024841 (2019).
Stavem, K., Hoel, H., Skjaker, S. A. & Haagensen, R. Charlson comorbidity index derived from chart review or administrative data: agreement and prediction of mortality in intensive care patients. Clin. Epidemiol. 9, 311–320 (2017).
Song, S. E. et al. The prognostic value of the Charlson’s comorbidity index in patients with prolonged acute mechanical ventilation: A single center experience. Tuberc. Respir. Dis. 79, 289–294 (2016).
Yıldız, A., Yiğt, A. & Benli, A. R. The prognostic role of Charlson comorbidity index for critically ill elderly patients. Eur. Res. J. https://doi.org/10.18621/eurj.451391 (2019).
Morris, A., Masur, H. & Huang, L. Current issues in critical care of the human immunodeficiency virus-infected patient. Crit. Care Med. 34, 42–49 (2006).
Garland, J. M., Levinson, A. & Wing, E. Care of critically ill patients with human immunodeficiency virus. Ann. Am. Thorac. Soc. 17, 659–669 (2020).
Andrade, H. B. et al. Highly active antiretroviral therapy for critically ill HIV patients: A systematic review and meta-analysis. PLoS ONE 12, e0186968 (2017).
Van Lelyveld, S. F. L. et al. Short- and long-term outcome of HIV-infected patients admitted to the intensive care unit. Eur. J. Clin. Microbiol. Infect. Dis. 30, 1085–1093 (2011).
Arabi, Y., Venkatesh, S., Haddad, S., Al Shimemeri, A. & Al Malik, S. A prospective study of prolonged stay in the intensive care unit: Predictors and impact on resource utilization. Int. J. Qual. Heal. Care 14, 403–410 (2002).
Moitra, V. K., Guerra, C., Linde-Zwirble, W. T. & Wunsch, H. Relationship between ICU length of stay and long-term mortality for elderly ICU survivors. Crit. Care Med. 44, 655–662 (2016).
Greenberg, J. A., Lennox, J. L. & Martin, G. S. Outcomes for critically ill patients with HIV and severe sepsis in the era of highly active antiretroviral therapy. J. Crit. Care 27, 51–57 (2012).
Acknowledgements
We thank the master's in epidemiology at El Bosque university for their help in analyzing the information. We also thank the Santa Clara hospital and its patients for their participation, and Doctor María Angelica Palencia and the Research and Innovation Committee of the Integrated Subnetwork of Health Services Center East S.S.E. in the city of Bogotá, Colombia, for facilitating the completion of this work.
Author information
Authors and Affiliations
Contributions
Conceptualization, writing—review & editing, G.O.R.; Methodology, formal analysis, writing—original draft, C.F.L.; Conceptualization, investigation, resources, J.A.M.; Formal analysis, visualization, J.E.B. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ruiz, G.O., Herrera, C.F.L., Bohórquez, J.A.M. et al. Mortality in patients with acquired human immunodeficiency virus infection hospitalized in an intensive care unit during the period 2017–2019. Sci Rep 12, 15644 (2022). https://doi.org/10.1038/s41598-022-19904-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-19904-z
- Springer Nature Limited