Abstract
We investigated the reference values of liver enzymes based on cardiometabolic risks among children and adolescents using the Korea National Health and Nutrition Examination Survey. A total of 8091 subjects aged 10–18 years were included from the data from 2007–2017. Overall, aspartate aminotransferase (AST), alanine aminotransferase (ALT), and the AST/ALT ratio varied with sex and age. AST levels tended to decrease with age, but ALT levels had a U-shaped curve, which resulted in a gradual increase in the AST/ALT ratio after age 13. The prevalence of MetS was strongly associated with elevated AST or ALT and a decreased AST/ALT ratio. The prevalence ratios of the development of MetS were also elevated in groups with high levels of AST and ALT and a low AST/ALT ratio. Particularly in the combined ALT and AST/ALT analyses, borderline-high levels also showed a high prevalence ratio of MetS. Liver enzymes were also involved in the increase in the adjusted mean values for each risk factor for MetS. Here, we provided updated reference values for liver enzymes based on the analysis between population-based data and cardiometabolic risk factors; AST, ALT and the AST/ALT ratio might be useful in the early diagnosis and treatment of MetS.
Similar content being viewed by others
Introduction
The rates of childhood obesity and metabolic syndrome are increasing worldwide1. Metabolic syndrome (MetS) is a pathophysiological state that has been used to associate overweight and obesity and their consequences such as cardiovascular diseases and diabetes mellitus. Since the National Cholesterol Education Program (NCEP) defined MetS in the adult population in 2001, subsequent studies have been conducted using modified criteria for MetS for children and adolescents and have reported an increasing prevalence of MetS. Although many different criteria resulted in difficulty estimating the exact prevalence in pediatric populations, a recent systematic review reported that the estimated prevalence of pediatric MetS ranged from 0.3–26.4% according to different geographic locations and populations2. In Korean studies, the prevalence of MetS has been reported to range from 5.7 to 10.9% according to the modified NCEP-ATP III criteria3,4. In particular, it its generally accepted that overweight and obese children have a higher risk of developing MetS than the normal population5. Indeed, a meta-study analysis showed that the prevalence of overweight and obese populations was high, from 24.09 to 56.32%6.
Hepatic involvement of MetS is commonly observed as hepatic steatosis in nonalcoholic fatty liver disease (NAFLD); pathological fat accumulation associated with chronic inflammation in the liver results in detrimental consequences, including impairment of glucose and lipid metabolism and an increase in cardiovascular events combined with oxidative stress, endothelial dysfunction and hypercoagulability7. Therefore, the recent current consensus recommends screening for NAFLD in children with accompanying MetS8. However, there is wide variability in the upper limit of normal for AST or ALT across different ages and between the sexes. The distinction of liver disease usually depends on the cutoff values that define an abnormal test, which is also critical for sensitivity and specificity9; this is the reason it is necessary to analyze population based data to establish reference intervals.
Several lines of studies have reported the upper limits of normal for liver enzymes in children and adolescents. In the United States, a population-based study suggested that the upper limits of normal foe ALT in metabolically normal individuals without liver disease were 26 mg/dl for boys and 22 mg/dl for girls10. Furthermore, in a Canadian study, the cutoff value for ALT was suggested to be 30 mg/dl in children between 1 and 12 years of age and 24 mg/dl in those between 13 and 19 years11. In other regions, including Europe12,13, the United Kingdom14, Mexico15, Sweden16, Iran17, Taiwan18 and China19, the reference ranges of liver enzymes in children were evaluated in a similar manner. However, in Korean children and adolescents, the similar studies were not population-based, and the association with MetS was not analyzed20 or adult data21.
In this study, we aimed to establish sex- and age-stratified reference intervals based on a healthy nonhospitalized pediatric population using data from KNHANES 2007–2017. We analyzed liver enzyme levels and other biochemical, physical, and social data from KNHAES to assess the association of AST, ALT and the AST/ALT ratio with MetS. Related components of MetS were also analyzed based on the normal, borderline-high and high levels of liver enzymes. Through the results of our population-based group study, we expect to add to our understanding of the roles of liver enzymes and a related ratio in MetS and provide a basis for novel liver enzyme reference ranges for children and adolescents in assessing the risk for MetS.
Results
Clinical characteristics of the study participants
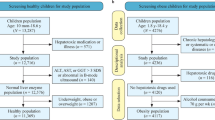
Among the KNHANES from 2007 to 2017, a total of 8091 children and adolescents were selected for this study (4307 male and 3784 female) (Fig. 1). The clinical characteristics of the participants are summarized in Table 1. The mean ages of the subjects were 14.30 ± 2.51 and 14.36 ± 2.51 for males and females, respectively. There were significant sex differences in the height SDS, the WC SDS, BP, AST, ALT and the AST/ALT ratio, glucose, T-C, TG, HDL-c, LDL-c, alcohol consumption, smoking, physical activity and rural residence.
Distribution of liver function according to age and sex
The age- and sex-specific distributions AST, ALT and the AST/ALT ratio are shown in Fig. 2. The percentile values for each age and sex, corresponding to LMS variables, are summarized in Table 2. Values of AST and ALT varied considerably according to sex and age; the percentile curve of AST tended to decrease as both boys and girls aged, but the ALT curves were slightly U-shaped in both sexes. Overall variations in AST and ALT according to age were higher in the upper percentile groups (≥ 75th percentile) than in the lower percentile groups (3rd—50th percentiles). Trends of the AST/ALT ratio decreased according to age and skewed to the left in both boys and girls; however, the levels were significantly higher in girls than in boys. Moreover, the overall AST/ALT ratio was relatively constant and decreased slightly over time.
The distribution of sex- and age-specific percentiles for levels of AST, ALT, and the AST/ALT ratio in children aged 10–18 years (n = 8091). Each figure represents the distribution of percentile by age of (A) AST percentile of boys, (B) AST percentile of girls, (C) ALT percentile of boys, (D) ALT percentile of girls, (E) AST/ALT ratio percentile of boys and (F) AST/ALT ratio percentile of girls.
Comparison of adjusted mean values of cardiometabolic risk factors according to AST, ALT, and the AST/ALT ratio
We compared the mean values of cardiometabolic risk factors, namely, WC, systolic/diastolic BP, glucose, T-C, TG, HDL-c and LDL-c stratified into normal (< 75th percentile), borderline-high (75th ≥ and < 95th percentile) and high (≥ 95th percentile) groups for AST and ALT and normal (> 25th percentile), borderline-high (> 5th and ≤ 25th percentile) and high (≤ 5th percentile) groups for the AST/ALT ratio. The upper limits of normal for boys and girls were estimated to be AST values of 23 and 20 IU/L, ALT values of 19 and 14 IU/L, and the AST/ALT ratios of 0.65 and 0.90, respectively.
The influences of AST, ALT and the AST/ALT ratio on cardiometabolic risk factors are summarized in Table 3. After adjustment for potential confounders, a comparison revealed that the adjusted mean values of T-C, TG, HDL-c and LDL-c were significantly higher in the borderline-high AST group than in the normal group. ALT levels were associated not only with lipid profiles but also with WC and BP. In the analysis of the AST/ALT ratio on cardiometabolic risk factors, the borderline-high group had a higher WC SDS, systolic BP, T-C, TG, HDL-c and LDL-c than the normal group. Moreover, the high AST/ALT ratio group had a higher WC SDS, diastolic BP, glucose, T-C, TG, HDL-c and LDL-c than the normal group, in which diastolic BP, glucose, T-C, TG and LDL-c were significantly higher than those in the borderline-high group.
Prevalence and adjusted prevalence ratios for MetS according to liver function and weight increase
The prevalence of MetS was significantly increased in the overweight and obese groups as the AST level increased but not in the normal weight group. In particular, the difference was significant between the borderline-high and high groups rather than between the normal group and the borderline-high group. Although there was a sex difference regarding the prevalence ratio for some detailed components, there was no significant difference in the contribution to the overall risk increase between boys and girls. An increase in the prevalence of MetS was similarly observed in the overweight and obesity groups as ALT levels increased (Fig. 3A). The number of components of MetS, especially more than two components, also increased as AST increased in the overweight and obesity groups (Fig. 4A). In contrast to the AST analysis results, the prevalence of MetS increased even in the borderline-high group compared to the normal group as the ALT level increased (Fig. 3B). In the analysis of the number of components of MetS, the presence of more than one component increased as ALT increased in the normal, overweight and obese groups (Fig. 4B). The analysis of the AST/ALT ratio also demonstrated that the prevalence of MetS increased as the AST/ALT level decreased in the overweight and obesity groups; in contrast to the ALT values, the differences among the normal, borderline-high, and high groups were more clearly observed in the AST/ALT values (Fig. 3C). Moreover, analysis of the number of components of MetS also clearly demonstrated that more than one component increased as the AST/ALT ratio increased not only in the overweight and obesity group but also in the normal group (Fig. 4C).
The differences in the prevalence of MetS for groups according to levels of AST, and ALT, and the AST/ALT ratio in children aged 10–18 years based on obesity (n = 8091). Subjects were classified as normal (< 85th percentile), overweight (≥ 85 and < 95th percentile), and obese (≥ 95th percentile) according to BMI. (A) The prevalence of MetS was presented according to AST, (B) ALT, and (C) the AST/ALT ratio.
The differences in the number of MetS components for groups according to the levels of AST, and ALT, and the AST/ALT ratio in children aged 10–18 years based on the obesity (n = 8091). Subjects were subdivided into normal (< 85th percentile), overweight (≥ 85 and < 95th percentile), and obese (≥ 95th percentile) groups according to BMI. (A) The number of components of MetS was presented according to AST, (B) ALT, and (C) the AST/ALT ratio.
The adjusted prevalence ratios for MetS and its components increased as AST and ALT increased and the AST/ALT ratio deceased, which is summarized in Table 4. Adjusted prevalence ratios for elevated glucose, elevated TG and MetS were increased for subjects with a high level of AST. Moreover, adjusted prevalence ratios for all analyzed metabolic components were considerably increased in subjects with a high level of ALT. A decreased AST/ALT ratio was most closely associated with an increased risk of MetS. In addition, the combination of high ALT and a low AST/ALT was also associated with an increased risk of elevated WC, elevated BP, elevated glucose, elevated TG, reduced HDL-c and MetS, which is summarized in Table 5.
Discussion
We found that clear differences in AST, ALT, and AST/ALT ratio curves between age- and sex- different subgroups; the AST levels tended to decrease with age, but the ALT curves were U-shaped, which resulted in trends of the AST/ALT ratio to reduce with age and skewed to the left. In the cardiometabolic risk assessment based on our novel references, components of MetS significantly increased in high levels of AST, ALT, and AST/ALT ratio; some of the components were increased even in borderline high levels of liver enzymes in the analysis of ANCOVA with correction of age, sex, BMI, etc. Similarly, the odds ratio of metabolic risk factors also increased in the high AST, ALT, and the low AST/ALT ratio; some components of MetS were significantly increased even in borderline high levels.
In our findings, AST and ALT were higher in boys than in girls and differed over time; AST tended to decrease with age but ALT levels were U-shaped and increased with age. These tendencies were consistent with previous reports in Korean subjects20 and those of other ethnicities; for instance, an obvious increase in ALT levels was observed in males after the age of 11 years19, and a sex difference with a continuous decrease in concentration from ages 2 to 14 years was observed for AST11,19,22,23. Percentile distributions of the AST/ALT ratio showed sex differences across all observed ages in our data; the ratio was significantly higher in girls than in boys. Despite conflicts with the data of the HELENA study12, our results were similar to those of a recent Chinese study24, which seems to be due to ethnic or geographic differences.
Regarding MetS, an increase in the prevalence of MetS according to AST, ALT, and the AST/ALT ratio was more clearly observed as BMI increased (Figs. 3 and 4). This result suggested that although a strong correlation exists between high levels of AST, ALT, and the AST/ALT ratio and most cardiometabolic risk factors exist, the interpretation of liver markers in children and adolescents could be adapted differentially depending on the differential BMI values. Previously, AST or AST/ALT ratios were known to indicate progression to diabetes25. Similarly, in our findings, high serum AST, ALT, and the AST/ALT ratio were all associated with a high prevalence ratio of elevated glucose (Table 4). These results strongly supported that a decreased AST/ALT ratio might effectively predict increased cardiometabolic risk, especially insulin resistance when high ALT levels exist. Of interest, our results showed that the associations of AST, ALT, and the AST/ALT ratio with cardiometabolic risk factors were significant both in boys and girls, which partially conflicts with a previous European study12. The differential distribution of overweight or obese children in the study population might affect the analysis. In addition, differential body adiposity might also influence screening tests of liver markers. Therefore, detailed comparative studies on this point will be needed in future studies.
Since the importance of childhood MetS on adulthood transition has been raised, several studies have suggested various criteria for the evaluation of MetS; however, there are still controversies regarding the establishment of uniform diagnostic criteria2. Among them, the modified NCEP-ATP III criteria26,27,28 adapted in our study and the International Diabetes Federation (IDF) criteria29 are the two most widely used definitions. The characteristic differences in the IDF criteria on MetS were the presence of obesity as a mandatory condition for the diagnosis and the cutoff value > 150 mg/dL of high TG compared to our diagnostic definition30. If we followed the IDF criteria, the prevalence of MetS according to liver enzyme levels in normal or overweight subjects would have been excluded in the metabolic syndromes. However, the prevalence of metabolic syndrome and its risk factors according to liver enzymes were also significantly higher even in the overweight group as in the obesity group in our results (Figs. 3 and 4). Although it is not yet clear what the exact contribution of metabolic risk factors is to the development of CVD and T2DM in adulthood, these findings might support that nonobese and overweight pediatric subjects with metabolic risk factors are also potentially at risk for adult metabolic syndromes. Therefore, detailed long-term observational and cohort studies on this point will be needed in future studies.
Based on the percentile distribution, we suggested that the upper limits of normal of AST, ALT and the AST/ALT ratio were 23 IU/L and 20 IU/L (< 75th percentile), 19 IU/L and 14 IU/L (< 75th percentile), and 1.1 and 1.35 (< 25 percentile) in boys and girls, respectively. Similar studies with non-overweight adolescents proposed sex-specific thresholds for ALT levels < 25 IU/L in males and < 22 IU/L in females to detect pediatric chronic liver disease10. However, Labayen et al. suggested upper limits of normal for ALT of 24–25 (75th percentile) and 22–24 IU/L (75th percentile) and thresholds of the AST/ALT ratio associated with high cardiometabolic risk of 1.0–0.74 and 0.86–0.87 (ranging from 13-35th percentile) for boys and girls, respectively12. In those studies, the estimated upper limits of normal of ALT in adolescents were higher than those in our report. This might be due to differences in ethnicities between the studies; other possibilities are differences in the proportion of subjects with obesity and severity of central adiposity levels. Although direct comparison of the AST/ALT ratio between our data and previous reports is difficult, it seems that stricter levels of the AST and ALT should be applied for Korean adolescents, especially for the overweight or obese subgroup for the precise estimation of cardiometabolic prognosis.
The main limitation of this study is the cross-sectional nature of the analysis, which cannot identify the temporal association of MetS with AST, ALT, and the AST/ALT ratio. A large population-based, longitudinal cohort study could address this limitation in the future by serial measurements of liver enzymes and follow-up for the occurrence of cardiometabolic events. The other limitation is that our data were from subjects of one ethnicity in a single country. Thus, comparisons and meta-analyses with other ethnic groups will be needed for the broad application of pediatric reference intervals. Despite information about a family history of premature coronary heart disease, we could not exclude familial hyperlipidemia during subject selection due to limitations on laboratory tests. Combined familial hyperlipidemia frequently accompanies NAFLD in approximately 49–76% of cases31, which implies that the possible effects of these comorbidities were not completely excluded in our data. Other liver markers such as gamma-GT have also been suggested to be strong predictors of cardiovascular disease and T2DM in adults32, and metabolic risks in adolescents12; however, we did not analyze other possible markers in our current study. The definition of physical activity used in this study was not consistent with the current recommendations of The American Academy of Pediatrics (AAP); a daily moderate to vigorous intensity of exercise for more than 60 min33. This is due to the difference in the KHANES protocol and needs improvement in future investigations.
In conclusion, we newly established reference values for AST, ALT, and the AST/ALT ratio based on the risk assessment of MetS components. High levels of AST and ALT and a low AST/ALT ratio were closely associated with the prevalence of MetS and its components. In particular, overweight and obese children and adolescents have a considerably higher prevalence of MetS and its components when liver enzymes exceed the upper limits of normal than do normal subjects. Both ALT and the AST/ALT ratio were effective in screening for metabolic risk in both sexes in a Korean population. Therefore, the age- and sex-specific reference values provided in this study may contribute to the early diagnosis and treatment of MetS.
Materials and methods
This study was based on data from the KNHANES. The KNHANES is a cross-sectional, nationally representative survey that is conducted by the Korean Centers for Disease Control and Prevention (KCDC) annually. The survey consists of a health questionnaire, health examination, and nutritional assessment. Every participant in the KNHANES gave informed consent at the time of data collection and the survey protocols were approved by the KCDC Institutional Review Board. KNHANES was performed in accordance with relevant guidelines and regulations of the National Health Promotion Act in Korea. The database is available to the public at the KNHANES website (http://knhanes.cdc.go.kr) and details of the KNHANES have been described previously34. This study was also approved by the Institutional Review Board of Hallym University Chuncheon Sacred Heart Hospital (IRB No. CHUNCHEON 2021–10-004). All methods were performed in accordance with the relevant guidelines and regulations.
Subjects
The subjects in this study were from the KNHANES from 2007 to 2017. The initial subjects comprised 10,033 children and adolescents among a total of 89,630 subjects. Subjects who had missing anthropometric data (n = 733) and those missing blood laboratory results or had any type of hepatitis were excluded (n = 1193). Subjects with abnormal triglyceride (TG) levels (≥ 400 mg/dL) (n = 16) were excluded because low-density lipoprotein cholesterol (LDL-c) levels were calculated with Friedewald’s Equation35.
Measurements
Anthropometric data and blood pressure (BP) were measured by trained experts according to standardized protocols. Details of the anthropometric measurements have been described previously26. Standard deviation scores (SDSs) were used for height, weight, BMI, and WC which were calculated with the LMS methods using the 2017 Korean reference36. Blood samples were collected after the participants fasted for at least 8 h. Samples were immediately centrifuged, transported to a central laboratory (NeoDin Medical Institute, Seoul, Korea) and analyzed within 24 h. Serum levels of total cholesterol (T-C), high-density lipoprotein cholesterol (HDL-c), TG, and glucose were measured enzymatically using a Hitachi 7600 automatic analyzer (Hitachi, Tokyo, Japan). LDL-c was calculated with Friedewald’s equation35.
Collection of lifestyle parameters and socioeconomic status
Lifestyle-related parameters and socioeconomic information were collected by questionnaires. Smoking was defined as having smoked more than five packs of cigarettes throughout one’s lifetime. Alcohol consumption was defined as at least two alcoholic beverages/month during the previous year. Physical activity was defined as: (1) intense physical activity for 30 min at least three days/week, (2) moderate physical activity for 30 min at least five days/week, or (3) walking for 30 min at least five days/week. Based on physical activity, subjects were classified as exercising or non-exercising. For socioeconomic information, household income was categorized as being within the lowest quartile or not. The residence area was divided into urban and rural areas.
Definitions of MetS and its components
Obesity and overweight were defined as ≥ 95th and ≥ 85th but < 95th percentile, respectively, as shown in a previous study36. The definition of MetS followed the modified criteria of the National Cholesterol Education Program Adult Treatment Panel III (NCEP-ATP III), as previously described in our published paper26; subjects who met 3 of the following 5 criteria were defined as having MetS: (1) increased WC based on the Korean pediatric population; ≥ 90th percentile for age and sex according to the 2017 Korean growth chart36; (2) elevated BP, namely, SBP or DBP ≥ 90th percentile for age- and sex-matched reference data from the Korean pediatric population36 or treatment with antihypertensive medication; (3) fasting blood glucose ≥ 100 mg/dL or treatment for type 2 diabetes mellitus (T2DM); (4) elevated TG (≥ 110 mg/dL); and (5) low HDL-c (< 40 mg/dL). T2DM was diagnosed satisfying one or more of the following criteria: (1) subjects who self-reported their disease using a questionnaire, (2) current medication or insulin use to manage T2DM, or (3) subjects with a fasting glucose level ≥ 126 mg/dL during the survey period.
Statistical analysis
The basic characteristics consisted of continuous variables and categorical variables; each variable is presented as the mean ± standard deviation (SD) and frequencies or percentages (%), respectively. Student’s t-test was used to compare the means of the demographic and biochemical characteristics. The chi-square (χ2) test was used to compare clinical categorical variables between boys and girls.
We obtained percentile curves of AST, ALT and the AST/ALT ratio as a function of age as a continuous variable, stratified by sex using the LMS model to fit smoothed L (skew), M (median), and S (coefficient of variation) curves using the General Additive Model for Location Scale and Shape (GAMLSS) package version 4.2.6. of the R statistical package. The Box–Cox Cole and Green, gamma or inverse Gaussian distributions were fitted to the observed distribution of AST, ALT and the AST/ALT ratio. Percentile curves were generated for the 3rd, 5th, 10th, 15th, 25th, 50th, 75th, 85th, 90th, 95th and 97th percentiles. The adjusted mean values of cardiometabolic risk factors were compared between three groups (normal < 75th; borderline high, ≥ 75th and < 95th; and high, ≥ 95th percentile) using analysis of covariance (ANCOVA) followed by Bonferroni’s post-hoc test after adjusting for sex, age, BMI SDS, alcohol consumption, smoking, physical activity, residence, household income, and diagnosis of hypertension, T2DM, and dyslipidemia. We estimated the adjusted prevalence ratios and 95% confidence intervals (CIs) for MetS among the normal, borderline high and high groups by multiple logistic regression analysis. The estimates were adjusted for sex, age, BMI SDS, alcohol consumption, smoking, physical activity, residence, household income, diagnosis of hypertension, T2DM, and dyslipidemia. P < 0.05 was considered indicative of statistical significance. All statistical analyses in this study were performed using R statistical package version 3.5.1 (The R Foundation for Statistical Computing, Vienna, Austria). The prevalence ratios were calculation using the prLogistic package. The prLogistic package was implemented under the FLOSS (Free/Libre Open Source Software) paradigm in the R system for statistical computing (R Development Core Team 2021) and it was available from the Comprehensive R Archive Network (CRAN) at https://cran.r-project.org/package=prLogistic and the experimental updates at GitHub repository https://github.com/Raydonal/prLogistic.
Data availability
The data that support the findings of this study are available in [the KNHANES website] at [https://knhanes.kdca.go.kr]. The data that support the findings of this study are available from the corresponding author, [Shim YS], upon reasonable request.
Abbreviations
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- BP:
-
Blood pressure
- BMI:
-
Body mass index
- CI:
-
Confidence interval
- HDL-c:
-
High-density lipoprotein cholesterol
- KCDC:
-
Korean Centers for Disease Control and Prevention
- KNHANES:
-
The Korea National Health and Nutrition Examination Survey
- LDL-c:
-
Low-density lipoprotein cholesterol
- MetS:
-
Metabolic syndrome
- NAFLD:
-
Nonalcoholic fatty liver disease
- NCEP-ATP III:
-
National Cholesterol Education Program Adult Treatment Panel III
- NCEP:
-
National Cholesterol Education Program
- SDS:
-
Standard deviation scores
- T2DM:
-
Type 2 diabetes mellitus
- TG:
-
Triglyceride
- T-C:
-
Total cholesterol
- WC:
-
Waist circumference
References
Di Cesare, M. et al. The epidemiological burden of obesity in childhood: a worldwide epidemic requiring urgent action. BMC Med. 17, 212. https://doi.org/10.1186/s12916-019-1449-8 (2019).
Reisinger, C., Nkeh-Chungag, B. N., Fredriksen, P. M. & Goswami, N. The prevalence of pediatric metabolic syndrome-a critical look on the discrepancies between definitions and its clinical importance. Int. J. Obes. (Lond) 45, 12–24. https://doi.org/10.1038/s41366-020-00713-1 (2021).
Kim, S. & So, W. Y. Prevalence and correlates of metabolic syndrome and its components in elderly Korean adults. Exp. Gerontol. 84, 107–112. https://doi.org/10.1016/j.exger.2015.09.012 (2016).
Chung, J. Y. et al. Prevalence of metabolic syndrome in children and adolescents - the recent trends in South Korea. J. Pediatr. Endocrinol. Metab. 26, 105–110. https://doi.org/10.1515/jpem-2012-0294 (2013).
Gepstein, V. & Weiss, R. Obesity as the main risk factor for metabolic syndrome in children. Front. Endocrinol. 10, 1. https://doi.org/10.3389/fendo.2019.00568 (2019).
Bitew, Z. W. et al. Metabolic syndrome among children and adolescents in low and middle income countries: a systematic review and meta-analysis. Diabetol. Metab. Syndr. 12, 1. https://doi.org/10.1186/s13098-020-00601-8 (2020).
Bhatia, L. S., Curzen, N. P., Calder, P. C. & Byrne, C. D. Non-alcoholic fatty liver disease: a new and important cardiovascular risk factor?. Eur. Heart. J. 33, 1190–1200. https://doi.org/10.1093/eurheartj/ehr453 (2012).
Vos, M. B. et al. NASPGHAN Clinical Practice Guideline for the Diagnosis and Treatment of Nonalcoholic Fatty Liver Disease in Children: Recommendations from the Expert Committee on NAFLD (ECON) and the North American Society of Pediatric Gastroenterology, Hepatology and Nutrition (NASPGHAN). J. Pediatr. Gastroenterol. Nutr. 64, 319–334. https://doi.org/10.1097/MPG.0000000000001482 (2017).
Ruhl, C. E. & Everhart, J. E. Trunk fat is associated with increased serum levels of alanine aminotransferase in the United States. Gastroenterology 138, 1346–1356. https://doi.org/10.1053/j.gastro.2009.12.053 (2010).
Schwimmer, J. B. et al. SAFETY study: alanine aminotransferase cutoff values are set too high for reliable detection of pediatric chronic liver disease. Gastroenterology 138, 1357–1364. https://doi.org/10.1053/j.gastro.2009.12.052 (2010).
Colantonio, D. A. et al. Closing the gaps in pediatric laboratory reference intervals: a CALIPER database of 40 biochemical markers in a healthy and multiethnic population of children. Clin. Chem. 58, 854–868. https://doi.org/10.1373/clinchem.2011.177741 (2012).
Labayen, I. et al. Liver enzymes and clustering cardiometabolic risk factors in European adolescents: the HELENA study. Pediatr. Obes. 10, 361–370. https://doi.org/10.1111/ijpo.273 (2015).
Bibiloni, M. M. et al. Relation between liver transaminases and dyslipidaemia among 2–10 y.o. Northern Mexican Children. PLoS ONE 11, 1–12 (2016).
Sekkarie, A. et al. ALT Trends through Childhood and Adolescence Associated with Hepatic Steatosis at 24 Years: A Population-Based UK Cohort Study. Children (Basel) 7, 1. https://doi.org/10.3390/children7090117 (2020).
Elizondo-Montemayor, L. et al. Association of ALT and the metabolic syndrome among Mexican children. Obes. Res. Clin. Pract. 8, e79-87. https://doi.org/10.1016/j.orcp.2012.08.191 (2014).
Ridefelt, P. Pediatric reference intervals - the Swedish experience. Clin. Biochem. 47, 740–741. https://doi.org/10.1016/j.clinbiochem.2014.05.038 (2014).
Kelishadi, R. et al. Association of alanine aminotransferase concentration with cardiometabolic risk factors in children and adolescents: the CASPIAN-V cross-sectional study. Sao Paulo Med. J. 136, 511–519. https://doi.org/10.1590/1516-3180.2018.0161161118 (2018).
Lai, D. S. et al. Pediatric reference intervals for several biochemical analytes in school children in Central Taiwan. J. Formos Med. Assoc. 108, 957–963. https://doi.org/10.1016/S0929-6646(10)60009-5 (2009).
Li, X. et al. Establishment of age- and gender-specific pediatric reference intervals for liver function tests in healthy Han children. World J. Pediatr. 14, 151–159. https://doi.org/10.1007/s12519-018-0126-x (2018).
Cho, S. M., Lee, S. G., Kim, H. S. & Kim, J. H. Establishing pediatric reference intervals for 13 biochemical analytes derived from normal subjects in a pediatric endocrinology clinic in Korea. Clin. Biochem. 47, 268–271. https://doi.org/10.1016/j.clinbiochem.2014.09.010 (2014).
Kim, H. C., Choi, K. S., Jang, Y. H., Shin, H. W. & Kim, D. J. Normal serum aminotransferase levels and the metabolic syndrome: Korean National Health and Nutrition Examination Surveys. Yonsei Med. J. 47, 542–550. https://doi.org/10.3349/ymj.2006.47.4.542 (2006).
Loh, T. P. & Metz, M. P. Trends and physiology of common serum biochemistries in children aged 0–18 years. Pathology 47, 452–461. https://doi.org/10.1097/PAT.0000000000000274 (2015).
Hilsted, L., Rustad, P., Aksglaede, L., Sorensen, K. & Juul, A. Recommended Nordic paediatric reference intervals for 21 common biochemical properties. Scand. J. Clin. Lab. Invest. 73, 1–9. https://doi.org/10.3109/00365513.2012.721519 (2013).
Lin, S. et al. The Relationship Between Aspartate Aminotransferase To Alanine Aminotransferase Ratio And Metabolic Syndrome In Adolescents In Northeast China. Diabetes Metab. Syndr. Obes. 12, 2387–2394. https://doi.org/10.2147/DMSO.S217127 (2019).
Botros, M. & Sikaris, K. A. The de ritis ratio: the test of time. Clin. Biochem. Rev. 34, 117–130 (2013).
Lee, H. J. et al. Distribution of waist-to-height ratio and cardiometabolic risk in children and adolescents: A population-based study. Sci Rep 11, 9524. https://doi.org/10.1038/s41598-021-88951-9 (2021).
Cook, S., Weitzman, M., Auinger, P., Nguyen, M. & Dietz, W. H. Prevalence of a metabolic syndrome phenotype in adolescents: findings from the third National Health and Nutrition Examination Survey, 1988–1994. Arch. Pediatr. Adolesc. Med. 157, 821–827. https://doi.org/10.1001/archpedi.157.8.821 (2003).
de Ferranti, S. D. et al. Prevalence of the metabolic syndrome in American adolescents: findings from the Third National Health and Nutrition Examination Survey. Circulation 110, 2494–2497. https://doi.org/10.1161/01.CIR.0000145117.40114.C7 (2004).
Zimmet, P. et al. The metabolic syndrome in children and adolescents. Lancet 369, 2059–2061. https://doi.org/10.1016/S0140-6736(07)60958-1 (2007).
Magge, S. N. et al. The metabolic syndrome in children and adolescents: Shifting the focus to cardiometabolic risk factor clustering. Pediatrics 140, 1. https://doi.org/10.1542/peds.2017-1603 (2017).
Riggio, S. et al. Assessment of liver stiffness in subjects affected by familial combined hyperlipidaemia with hepatic steatosis. Eur. J. Clin. Invest. 40, 722–728. https://doi.org/10.1111/j.1365-2362.2010.02323.x (2010).
Sakamoto, A. et al. Comparison of the impact of changes in waist circumference and body mass index in relation to changes in serum gamma-glutamyltransferase levels. J. Atheroscler. Thromb. 20, 142–151. https://doi.org/10.5551/jat.14415 (2013).
Expert Panel on Integrated Guidelines for Cardiovascular, H., Risk Reduction in, C., Adolescents, National Heart, L. & Blood, I. Expert panel on integrated guidelines for cardiovascular health and risk reduction in children and adolescents: summary report. Pediatrics 128(5), S213–256. https://doi.org/10.1542/peds.2009-2107C (2011).
Kweon, S. et al. Data resource profile: The Korea National Health and Nutrition Examination Survey (KNHANES). Int. J. Epidemiol. 43, 69–77. https://doi.org/10.1093/ije/dyt228 (2014).
Friedewald, W. T., Levy, R. I. & Fredrickson, D. S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 18, 499–502 (1972).
Kim, J. H. et al. The 2017 Korean National Growth Charts for children and adolescents: development, improvement, and prospects. Kor. J. Pediatr. 61, 135–149. https://doi.org/10.3345/kjp.2018.61.5.135 (2018).
Author information
Authors and Affiliations
Contributions
S.Y.-J. designed the study, drafted the manuscript, and analyzed the publicly available data set. L.H.S. and H.J.S. reviewed and revised the manuscript and provided important intellectual content, including conceptualization of the study design. S.Y.S. supervised all aspects of manuscript preparation and assisted with the study formulation, data analysis, manuscript writing, and interpretation of the findings. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Seo, YJ., Shim, Y.S., Lee, H.S. et al. Updated reference ranges for aminotransferase levels of Korean children and young adolescents based on the risk factors for metabolic syndrome. Sci Rep 12, 15739 (2022). https://doi.org/10.1038/s41598-022-20104-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-20104-y
- Springer Nature Limited