Abstract
Fasciola hepatica, one of the agents that causes fasciolosis, modulates the host immune system to allow parasite survival in the host. F. hepatica expresses carbohydrate-containing glycoconjugates that are decoded by C-type lectin receptors, such as Dectin-1, mannose receptor, DC-SIGN and MGL, that are mainly present on myeloid antigen presenting cells (APCs) and can mediate immunoregulatory properties on T cells. In particular, Macrophage Gal/GalNAc lectin 2 (MGL2) expands modified Th2 immune responses, while suppressing Th1 polarization, upon recognition of GalNAc-glycosylated parasite components. In this study, by using MGL2-DTR transgenic mice that encode human diphtheria toxin receptor in MGL2+ cells, we demonstrate the role of peritoneal APCs during F. hepatica infection in favoring parasite survival. This process might be mediated by the induction of splenic Tregs in vivo, since the depletion of MGL2+ cells conferred mice with partial resistance to the infection and abrogated the increase of CD4+/CD25+ FoxP3+ Tregs induced by the parasite. Therefore, MGL2+ cells are critical determinants of F. hepatica infection and could constitute immune checkpoints to control parasite infection.
Similar content being viewed by others
Introduction
Fasciola hepatica is a trematode parasite that causes fasciolosis, a zoonotic disease that affects humans1,2. It also infects livestock, causing significant economic losses worldwide1,2. To survive in its mammalian hosts, F. hepatica is capable of modulating the host immune system by inducing a modified type-2 responses characterized by potent immune regulatory processes such as differentiation of regulatory T cells (Tregs), alternative activation of macrophages, involvement of regulatory dendritic cells (DCs), upregulation of IL-10 and TGFβ and down-regulation of Th1 cytokines3,4,5,6,7.
Helminths express carbohydrate-containing glycoconjugates that are extremely important in their life cycles and pathogeny since they can participate in immune escape8. Indeed, glycoconjugates produced by F. hepatica are able to modulate the maturation and function of DCs9,10 and macrophages11,12,13. Furthermore, F. hepatica glycans participate in parasite migration through the intestine in early stages of the infection14,15.
The immunomodulatory role of parasite glycans relies on the ability of lectins to decode their information, such as C-type lectin receptors (CLRs), that are mainly present on myeloid antigen presenting cells (APC)16,17. Previously data have demonstrated that CLRs are key mediators of the immunoregulatory properties induced by F. hepatica. In fact, Dectin-1 on macrophages interacts with F. hepatica excretory-secretory products inducing an alternative activated macrophage phenotype11,12 and likely exerting T cell anergy via selective up-regulation of PD-L2 expression on macrophages in a Dectin-1 dependent way18. However, in-depth studies on the function of these cells are still necessary12. Injection of F. hepatica tegumental antigens induces anergic-like T cells via DCs in a mannose receptor (MR)-dependent manner, although the role of MR during F. hepatica infection has not been determined so far19. In contrast, no implication of the MR was found in the suppression of LPS-induced cytokines by bone marrow derived DCs treated with F. hepatica derived-molecules13. On the other hand, dendritic cell-specific ICAM-3 grabbing non-integrin (DC-SIGN) interacts with F. hepatica glycoconjugates through mannose and fucose residues on regulatory DCs and decreases allogeneic T cell proliferation, via the induction of anergic T cells10.
Macrophage Gal/GalNAc lectin 2 (MGL2, CD301) binds to terminal GalNAc residues, including the Tn antigen (GalNAc-αThr/Ser) and is mainly expressed on immature, tolerogenic or type-2 DCs6,20,21 and alternatively-activated macrophages22. Moreover, MGL activation dampens immune responses, by inducing synthesis of IL-10 by DCs6,23, promoting the differentiation of Tregs24, inducing T cell apoptosis and suppressing T cell activation25. Previous studies from our group demonstrated that human MGL can interact with F. hepatica components through the Tn antigen and modulate the TLR2-induced maturation of human monocyte derived DCs by up-regulating the production of IL-10 and TNFα6. In addition, we have shown that MGL2+ cells in F. hepatica infected mice express a variety of regulatory markers, including IL-10, TNFα and TGFβ, expand modified Th2 immune responses and suppress Th1 polarization6. However, the role of MGL2+ cells during F. hepatica infection in inducing Tregs in vivo has not yet been investigated.
In this study, we demonstrate that MGL2-expressing APCs recruited to the peritoneal cavity during F. hepatica experimental infection in mice are essential for parasite survival as well as for the induction of splenic Tregs in vivo. We used MGL2-DTR transgenic mice that encode human diphtheria toxin receptor (DTR) in MGL2+ cells. Thus, these cells can be depleted with diphtheria toxin (DT) injection24. The depletion of MGL2+ cells conferred mice with partial resistance to the infection and abrogated the increase of CD4+/CD25+ FoxP3+ Tregs induced by the infection. Therefore, MGL2+ cells could constitute immune checkpoints to control F. hepatica infection.
Results
F. hepatica infection in MGL2-DTR transgenic mice is characterized by the recruitment of immunoregulatory F4/80+ cells in the peritoneum and higher levels of splenic Treg
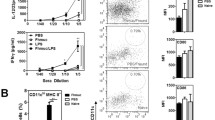
First, we infected MGL2-DTR transgenic mice and analyzed the clinical signs and immunological response induced by F. hepatica infection. After 3 weeks post-infection (wpi), infected animals presented a high clinical score, determined by the general state of the animal (Supplementary Table 1) and alanine transaminase (ALT) activity in serum, a common marker to detect hepatic dysfunction, and liver damage and fibrosis (Fig. 1A). Furthermore, infection was associated with an increase of F4/80+ cells in the peritoneal cavity of infected mice. However, the frequency of these cells was not significantly different from those determined in non-infected mice (Fig. 1B and Supplementary Fig. 1A). In addition, peritoneal F4/80+ cells from infected mice expressed higher CCR2 (Fig. 2A and Supplementary Fig. 1B) and CD11c (Fig. 2B) levels. However, they did not upregulate MerTK expression (Fig. 2C). Additional phenotyping by flow cytometry showed a significantly enhanced expression of Sirpα (Fig. 2D) and MGL2 (Fig. 2E) in infected mice. Furthermore, F4/80+ MGL2+ cells upregulated MHC class II expression with the infection (Fig. 2F). On the other hand, F4/80+ MGL2+ peritoneal APCs upregulated different immunoregulatory molecules, such as PD-L1 (Fig. 3A), but not ICOS-L (Fig. 3B). Last, they also expressed higher levels of immunomodulatory molecules such as IL-10 (Fig. 3C), TNFα (Fig. 3D) and hemoxygenase-1 (HO-1) (Fig. 3E).
F. hepatica infected MGL2-DTR mice present an increase of F4/80+ cells in the peritoneal cavity. (A) Clinical score and ALT in serum after 3 wpi. MGL2-DTR mice were infected with 10 F. hepatica metacercariae (INF). Non infected mice injected with PBS were used as controls (Ctl). (B) Frequency and number of F4/80+ cells in the peritoneal cavity determined by flow cytometry. A representative figure of three independent experiments is shown (± SEM, indicated by error bars). Asterisks indicate statistically significant differences (****p < 0.0001).
Peritoneal F4/80+ cell phenotype in F. hepatica MGL2-DTR mice. Expression of CCR2 (A), CD11c (B), MerTK (C), Sirpα (D), MGL2 (E) and MHC class II (F) is shown in F4/80+ peritoneal cells from F. hepatica infected mice (INF) and non-infected animals (Ctl). Grey lines in histograms represent infected mice while black lines indicated control mice. A representative figure of three independent experiments is shown (± SEM, indicated by error bars). Asterisks indicate statistically significant differences (*p < 0.05, **p < 0.01).
Peritoneal F4/80+ cells in F. hepatica MGL2-DTR mice express immunoregulatory molecules. Expression of PD-L1 (A), ICOS-L (B), IL-10 (C), TNFα (D) and HO-1 (E) is shown in F4/80+ peritoneal cells from F. hepatica infected mice (INF) and non-infected animals (Ctl). Grey lines in histograms represent infected mice while black lines indicated control mice. A representative figure of three independent experiments is shown (± SEM, indicated by error bars). Asterisks indicate statistically significant differences (*p < 0.05, **p < 0.01).
Then, we analyzed the presence of Tregs in spleens of 3 wpi MGL2-DTR mice. As depicted in Fig. 4A, a lower frequency of CD4+ T cells in spleens of infected mice was detected, although their number was higher than in non-infected mice, likely due to prominent splenomegaly in response to the infection (Supplementary Fig. 2). In addition, FoxP3+ CD25+ in CD4+ T cells increased with infection, both in frequency and number, in relation to non-infected mice (Fig. 4B).
F. hepatica infection is associated with an increase in splenic Tregs. Frequency and number of CD4+ cells (A) and FoxP3+ CD25+/CD4+ cells. (B) in spleens were determined by flow cytometry. Infected MGL2-DTR mice were infected with 10 F. hepatica metacercariae (INF). Non infected mice were used as controls (Ctl). A representative figure of three independent experiments is shown (± SEM, indicated by error bars). Asterisks indicate statistically significant differences (*p < 0.05, ***p < 0.001).
MGL2+ cell depletion is associated with resistance to F. hepatica infection
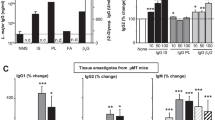
In order to determine the role of MGL2+ F4/80+ cells during F. hepatica infection, we injected MGL2-DTR transgenic mice with DT one day before and after the infection and every 3 days. Mice depleted of MGL2+ cells showed significantly lower clinical signs than control (PBS) mice at 3 wpi (Fig. 5A), which was also related to a significant decrease in serum ALT activity (Fig. 5B), associated with a lower liver damage and fibrosis than the one observed in control (PBS) infected-mice (Fig. 5C). Accordingly, the decrease in clinical signs was associated with a reduction, from 1 day post-infection (dpi), in the frequency and number of MGL2-expressing F4/80+ cells (Figs. 5D,E).
Depletion of F4/80+ MGL2+ cells during F. hepatica infection attenuates the clinical signs induced by the parasite. MGL2-DTR mice were infected with 10 F. hepatica metacercariae and sacrificed at 1 dpi, 1 wpi and 3 wpi. MGL2+ cells were depleted with DT-treatment. Control mice were injected with PBS. Non infected mice were used as controls (NI). Clinical score (A) and ALT (B) in serum of infected and control DT- or PBS-treated mice. (C) Representative images of livers from DT- or PBS-treated 3 wpi mice. (D) Peritoneal F4/80+ MGL2+ cells gating in F. hepatica infected mice treated with DT. (E) Frequency and number of MGL2+ F4/80+ peritoneal cells in infected DT- or PBS-treated mice. (F) Peritoneal F4/80+ cells gating during F. hepatica infection in DT-treated mice. (G) Frequency and number of F4/80+ peritoneal cells in infected DT- or PBS-treated mice at 1 dpi, 1 wpi and 3 wpi. Data from three independent experiments is shown (± SEM, indicated by error bars). Asterisks indicate statistically significant differences (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001).
MGL2+ cell depletion abrogates splenic Treg increase induced by the infection
The depletion of MGL2+ cells in MGL2-DTR infected mice prevented the decrease of CD4+ T cell percentage in the spleen from 1 wpi (Fig. 6A and Supplementary Fig. 3). In contrast, no significant increase in the CD4+ T cell counts was detected in comparison with DT-non treated infected control mice (Fig. 6A). Moreover, the depletion of MGL2+ cells in infected mice abrogated the increase in frequency and number of CD4+/FoxP3+ CD25+ cells (Fig. 6B), suggesting that MGL2+ F4/80+ cells are critical players in the induction of an adaptive regulatory immune response.
Depletion of F4/80+ MGL2+ cells during F. hepatica infection abrogates splenic Treg expansion. Frequency and number of CD4+ cells (A) and FoxP3+ CD25+/CD4+ cells. (B) in spleens were determined by flow cytometry. MGL2-DTR mice were infected with 10 F. hepatica metacercariae and sacrificed at 1 dpi, 1 or 3 wpi. MGL2+ cells were depleted with DT-treatment. Control mice were injected with PBS. Non infected mice were used as controls (NI). (C) Production of IFNγ evaluated by specific ELISA on supernatants of splenocyte culture stimulated with PMA/Ionomicin. A representative figure of three independent experiments is shown (± SEM, indicated by error bars). Asterisks indicate statistically significant differences (*p < 0.05, ***p < 0.001).
Of note, when splenocytes from infected mice depleted of MGL2+ cells were restimulated with molecules derived from the parasite (FhTE), they produced higher levels of IFNγ than those from infected non-depleted mice (Fig. 6C). However, no differences were observed in IL-4 and IL-10 cytokine secretion between infected groups (not shown). We were also unable to detect a difference in IL-4, IFNγ and IL-10 secretion by splenic CD4+ T cells between infected mice with or without MGL2+ cell depletion by flow cytometry (Supplementary Fig. 3).
Peritoneal MGL2+ F4/80+APCs from infected mice are essential for the induction of splenic Tregs
To confirm the role of MGL2+ F4/80+ peritoneal APCs induced by F. hepatica infection in the generation of Tregs in the spleen, we infected MGL2-DTR mice and treated them either with DT to deplete MGL2+ cells or with PBS as control. After 3 dpi, peritoneal cells from both groups of mice were adoptively transferred to the recipient non-infected mice (Fig. 7A,B). The presence of CD4+ CD3+ T cells (Fig. 7C) and Foxp3+ CD24+/CD4+ T cells (Fig. 7D) in the spleen of recipient mice was analyzed by flow cytometry revealing that the group transferred with F4/80+ cells presented an increase in splenic Tregs, while depletion of F4/80+ cells in infected mice did not induce and increase in splenic Tregs.
MGL2+ F4/80+ peritoneal cells induced by F. hepatica infection are necessary for Treg induction. (A) Adoptive transfer from infected MGL2-DTR mice to recipient non-infected mice. (B) F4/80+ peritoneal cell depletion with DT treatment after 3 dpi. Frequency and number of CD4+ CD3+ cells (C) and FoxP3+ CD25+/CD4+ (D) cells in spleens from recipient mice determined by flow cytometry. A representative figure of two independent experiments is shown (± SEM, indicated by error bars). Asterisks indicate statistically significant differences (*p < 0.05).
Discussion
Various studies have independently demonstrated the role of CLRs in recognizing, internalizing and signaling upon the stimulation with helminth glycoconjugates18,26. The uptake and the internalization of parasite molecules are crucial to allow the antigen processing and presentation that may influence the immune response and promote parasite survival in the host20,27,28. In this work, we focused on the role of peritoneal MGL2+ myeloid APC during F. hepatica infection in inducing splenic Tregs in vivo. Macrophages and DCs represent heterogeneous myeloid cell populations specialized in antigen presentation. However, DCs are unique in their capacity to orchestrate the adaptive immune response by activating naïve T cells and inducing their differentiation into different effector T cells depending on the pathogen29,30. Both macrophages and DCs can secrete pro-inflammatory or anti-inflammatory cytokines since they exhibit functional plasticity that enables them to adapt to various local conditions and to restore homeostasis after inflammation29,30,31. Here, we show that MGL2+ F4/80+ cells are recruited to the peritoneal cavity likely through an increase in CCR2, the receptor of the monocyte chemoattractant proteins 1 and 332,33. Indeed, CCR2 is a chemokine receptor associated with the recruitment of inflammatory monocytes in parasite infections34,35 and it mediates the cell migration from the bone marrow and the recruitment of monocytes, myeloid suppressor cells32,33 or monocyte-derived DC-like cells36 that express MHC II together with CD11c and F4/80 into damaged tissue or inflamed tissues.
MGL is a type II transmembrane protein expressed on professional APC. Even though there is only one MGL in humans (hMGL), two orthologues are present in mice that possess different glycan specificity (mMGL1 and mMGL2)37,38. Both mMGL2 and hMGL display similar specificity for terminal GalNAc moieties, including the Tn antigen (αGalNAc-O-Ser/Thr) and LacDiNAc (GalNAcβ1-4GlcNAc)39 and can recognize glycoconjugates from helminth parasites, such as Schistosoma mansoni40, Trichuris suis41 and Taenia crassiceps42. Furthermore, it has been proposed that MGL2+ dermal DCs are specialized in inducing Th2 responses both in allergy and helminth-infection models42.
In order to confirm that MGL2+ cells that express both CD11c and F4/80 may constitute a population of DCs, we evaluated the expression of MerTK, a macrophage specific molecule43. Due to the low expression of MerTK and high levels of Sirpα, PD-L1, IL-10 and TNFα, we suggest that the CD11c+ F4/80+ CCR2+ cells, that also express MGL2 during F. hepatica infection, constitute a population of monocyte-derived DCs with regulatory properties. Moreover, as previously described by our team, and in accordance with our studies performed in MGL2-DTR transgenic mice, regulatory DCs from F. hepatica express high levels of Sirpα and IRF46. Both IRF4 and Sirpα participate in immunoregulation and can promote Treg differentiation44,45,46. In F. hepatica infection in mice, MGL2+ regulatory DCs can suppress Th1 differentiation and induce the production of IL-10 by CD4+ T cell lymphocytes in vitro6. These regulatory functions could be mediated by PD-L1, the ligand of PD-1, expressed on regulatory DCs. This molecule, together with PD-L2, is an immune inhibitor receptor expressed on T cells that limits cell proliferation, induces Treg differentiation and serves to maintain immune homeostasis47,48. Indeed, PD-L2 negatively regulates Th1-mediated immunopathology during F. hepatica infection18. Therefore, PD-L1 on DCs could play a role in controlling the induction of parasite-specific immunity that allows its survival. In addition, a previous work demonstrated that CCR2+ cells recruited in the early stage of infection with T. crssiceps express PDL-1, and suppress T cell proliferation in vitro49. Furthermore, molecules from the tapeworm Hymenolepis diminuta induce a CCR2-dependent recruitment of myeloid monocyte-like cells that express high levels of PD-L1 to the peritoneum. These cells enhance both IL-10 and IL-4 secretion by activating T cells in vitro and have an immunosuppressive role in vivo50.
In a recent report we have shown that F4/80+ myeloid cells in the peritoneum of infected animals express HO-151, the inducible rate-limiting enzyme involved in the catabolism of free heme52 that promotes anti-inflammatory cytokine secretion53,54. In fact, HO-1 expression by DCs induces the production of IL-10 and inhibits T cell proliferation55. In F. hepatica infection it induces production of IL-10 which is necessary for parasite establishment in the host51. Of note, hepatocyte growth factor receptor induces Ras-dependent upregulation of both HO-1 and PD-L1 in cancer56,57. In addition, myeloid cells expressing both HO-1 and PD-L1 in breast tumors suppress T cell activity58. However, further experiments are necessary to explore whether MGL2 signaling is related with HO-1 or PD-L1 expression in peritoneal DC-like cells in F. hepatica infection in mice.
We have previously demonstrated that MGL2+ cells induce the production of IL-10 by CD4+ T cells6. To explore the involvement of these cells in the differentiation of Tregs, we depleted MGL2+ cells from F. hepatica infected mice. We found that they are crucial for infection and differentiation of Tregs, since an increased number of CD4+/FoxP3+CD25+ cells was found in the spleens of infected mice and their depletion abrogated the Treg expansion induced by the infection. Furthermore, adoptive transfer of peritoneal cells from infected mice with or without MGL2+ cell depletion revealed that MGL2+ DCs from infected mice can acquire an immunoregulatory program that licenses them to induce Treg differentiation. Nevertheless, the parasite molecules that trigger this immunoregulatory pathway on peritoneal MGL2+ myeloid cells during F. hepatica infection have not yet been identified. Indeed, although the identification of differentially-expressed genes involved in the glycosylation process of F. hepatica proteins has been performed59 the immunomodulatory role of glycans in certain proteins is still unknown. On the contrary, several studies have identified the glycan motifs from F. hepatica that can interact with CLRs on these cells, such as the Tn antigen that interacts with MGL26,10. Other glycans such as mannosylated glycoconjugates favor anergic T cells or Tregs that silence the immune system of the host through CD209 (or DC-SIGN)10. In addition, MR was described to interact with F. hepatica molecules and to mediate the partial inhibition of TLR-induced maturation of bone marrow-derived DCs13,19. Last, both MR and Dectin-1 immunomodulate Arginase-1 and PD-L2 expression and TGFβ production by macrophages in response to F. hepatica excretory–secretory products12,18. These data suggest that the parasite targets more than one CLR in order to evade immunity. Further experiments are needed to determine whether different factors participate in this process, such as glycans in parasite microvesicles60,61. In addition, other works have reported the anti-inflammatory role of F. hepatica molecules in training the innate immunity of macrophages62. However, the role of glycans in the induction of epigenetic imprinting of cells has not yet been determined.
In conclusion, we demonstrate that MGL2+ DC-like cells are recruited to the peritoneum during experimental F. hepatica infection in mice. They express immunoregulatory molecules that associate with an increase in clinical signs and expansion of Tregs, thus, favoring infection. Altogether, these results suggest that strategies based on MGL2 targeting could be helpful in the control of fasciolosis.
Methods
Ethics statement
Adult worms were collected during the routine work of a local abattoir (Frigorífico Carrasco) in Montevideo (Uruguay). Protocols were approved by the Uruguayan Committee on Animal Research (Comisión Nacional de Experimentación Animal, CNEA, Uruguay).
Mice
MGL2-DTR six- to eight-week-old mice were purchased from Jackson Laboratory (USA). They were used to analyze the role of MGL2+ cells during F. hepatica infection. MGL2+ cells can be depleted with DT injection24. Animals were kept in the animal house (URBE, School of Medicine, UdelaR, Uruguay) with water and food supplied ad libitum. Mouse handling, care and experiments were carried out in compliance with institutional guidelines and regulations from the National Committee on Animal Research (CNEA, http://www.cnea.org.uy/, National Law 18.611, Uruguay) and in accordance with ARRIVE guidelines. Procedures involving animals were approved by the Universidad de la República’s Committee on Animal Research (Comisión Honoraria de Experimentación Animal, CHEA Protocol Number 070153–000,811-19).
Parasite infection, animal treatment and sample obtention
Ten F. hepatica metacercariae (Montevideo, Uruguay) were orally administered per mouse. Viability of metacercariae was analyzed by in vitro excystment (> 70%) as previously evaluated63. Mice were bled at 3 wpi and peritoneal exudate cells (PECs), spleens, and livers were removed after either 1 dpi, 1 or 3 wpi, depending on the experiment. Non-infected animals were used as controls (0 dpi). To evaluate parasite infection while depleting MGL2+ cells, metacercariae were administered on day 0 into MGL2-DTR mice that were previously intraperitoneally (i.p.) injected with diphtheria toxin (DT, 0.5 µg/mouse) (The Native Antigen Company, USA) or PBS on day − 1 and every 2 or 3 days until the end of the experiment. Each experimental group contained at least six mice. PECs and spleens were processed as already described64. Red cells were lysed with ammonium chloride potassium buffer. The alanine aminotransferase (ALT) activity in sera was used to quantify liver damage and was determined with a colorimetric commercial kit (Spinreact, Spain) according to the manufacturer's instructions. The infection severity was assessed with a defined clinical score according to the following parameters: presence or absence of peritoneal hemorrhage, presence of macroscopic liver damage and splenomegaly, and the amount of cell content in the peritoneal cavity51,64,65, where the minimum score was 0 and the maximum was 10 (described in detail in Supplementary Table 1).
Flow cytometry
Cell suspensions from PECs and spleens were washed twice with PBS containing 2% FBS and 0.1% sodium azide (FACS buffer) and stained with specific antibodies for 30 min at 4 °C as previously published51. The following antibodies (Biolegend, USA) were used: anti-Sirp⍺ (P-84), -CD11c (N418), -CCR2 (SA203G11), -PD-L1 (10F9G2) and ICOS-L (HK5.3). Expression of FoxP3, HO-1 and IL-10 was analyzed by intracellular staining by permeabilizing with Cytofix and Perm wash buffers (Biolegend, USA), incubated with anti-IL-10 (JES5-1E3), -FoxP3 (MF14), TNFα (MP6-XT22) and HO-1 (clone ab13248 from Abcam, USA) specific antibodies. Analyses were performed using a BD Accuri C6 Plus cytometer and software (BD-Biosciences, USA).
Proliferation assay and cell culture
Parasite protein extract (FhTE) was prepared from live adult flukes obtained from infected bovines as previously published51. Splenocytes (0.5 × 106/well) from infected mice or uninfected naïve mice (control group) were cultured for 5 days at 37 °C and 5% CO2, in RPMI-1640 with 400 µg/ml glutamine (Capricorn Scientific, Germany) complete medium containing 10% heat-inactivated fetal bovine serum (FBS, Capricorn Scientific, Germany), 50 mM 2-mercaptoethanol, 100 U/ml penicillin, 0.1 mg/ml streptomycin (Merk, Sigma-Aldrich, USA) in presence or absence of FhTE (75 µg/ml) as previously described9. An IFNγ-specific sandwich ELISA assay (Biolegend, USA) was used to quantify IFNγ levels in culture supernatants.
Adoptive transfer of peritoneal cells from infected mice
To evaluate the capacity of MGL2+ cells to induce Tregs, 6 MGL2-DTR mice that were i.p. injected with DT (0.5 µg/mouse) or PBS on days − 1, + 1 and + 3, were infected with 10 F. hepatica metacercariae on day 0. As control groups non-infected MGL2-DTR mice injected either with DT or PBS were used (n = 6). At 4 dpi, PECs from the four groups were collected, red cells were lysed and remaining cells were counted. Depletion of peritoneal MGL2+ cells with DT treatment was verified by flow cytometry. 1 × 106 cells were i.p. injected in recipient non-infected MGL2-DTR mice that did not receive DT treatment. After 10 days, spleens were removed and CD3+ CD4+/FoxP3+ CD25+ cells were analyzed by flow cytometry as described above.
Statistical analysis
The obtained results were expressed as mean ± SEM. Statistical analyses were performed with GraphPad Prism version 6.04 for Windows (GraphPad Software, USA) was used to perform statistical analyses. Results were analyzed using one-way ANOVA followed by Tukey’s test, or two-tailed student's t-test, depending on the experiment. Significant differences shown by asterisks were considered when *p < 0.05, ** p < 0.01, *** p < 0.001, **** p < 0.0001.
Data availability
All data generated or analyzed during this study are included in this published article or available upon request (and its Supplementary Information files).
References
Mas-Coma, S., Valero, M. A. & Bargues, M. D. Fascioliasis. Adv. Exp. Med. Biol. 1154, 71–103. https://doi.org/10.1007/978-3-030-18616-6_4 (2019).
Cwiklinski, K., O’Neill, S. M., Donnelly, S. & Dalton, J. P. A prospective view of animal and human Fasciolosis. Parasite Immunol. 38, 558–568. https://doi.org/10.1111/pim.12343 (2016).
Walsh, K. P., Brady, M. T., Finlay, C. M., Boon, L. & Mills, K. H. Infection with a helminth parasite attenuates autoimmunity through TGF-beta-mediated suppression of Th17 and Th1 responses. J. Immunol. 183, 1577–1586. https://doi.org/10.4049/jimmunol.0803803 (2009).
Donnelly, S. et al. Helminth 2-Cys peroxiredoxin drives Th2 responses through a mechanism involving alternatively activated macrophages. FASEB J. 22, 4022–4032. https://doi.org/10.1096/fj.08-106278 (2008).
O’Neill, S. M. et al. Fasciola hepatica infection downregulates Th1 responses in mice. Parasite Immunol. 22, 147–155. https://doi.org/10.1046/j.1365-3024.2000.00290.x (2000).
Rodriguez, E. et al. Fasciola hepatica Immune Regulates CD11c(+) Cells by Interacting with the Macrophage Gal/GalNAc Lectin. Front. Immunol. 8, 264. https://doi.org/10.3389/fimmu.2017.00264 (2017).
Flynn, R. J. & Mulcahy, G. The roles of IL-10 and TGF-beta in controlling IL-4 and IFN-gamma production during experimental Fasciola hepatica infection. Int. J. Parasitol. 38, 1673–1680. https://doi.org/10.1016/j.ijpara.2008.05.008 (2008).
van Die, I. & Cummings, R. D. Glycan gimmickry by parasitic helminths: A strategy for modulating the host immune response?. Glycobiology 20, 2–12. https://doi.org/10.1093/glycob/cwp140 (2010).
Rodriguez, E. et al. Glycans from Fasciola hepatica Modulate the Host Immune Response and TLR-Induced Maturation of Dendritic Cells. PLoS Negl. Trop. Dis. 9, e0004234. https://doi.org/10.1371/journal.pntd.0004234 (2015).
Rodriguez, E. et al. Fasciola hepatica glycoconjugates immuneregulate dendritic cells through the Dendritic Cell-Specific Intercellular adhesion molecule-3-Grabbing Non-integrin inducing T cell anergy. Sci. Rep. 7, 46748. https://doi.org/10.1038/srep46748 (2017).
Guasconi, L. et al. Dectin-1 on macrophages modulates the immune response to Fasciola hepatica products through the ERK signaling pathway. Immunobiology 223, 834–838. https://doi.org/10.1016/j.imbio.2018.08.004 (2018).
Guasconi, L., Serradell, M. C., Garro, A. P., Iacobelli, L. & Masih, D. T. C-type lectins on macrophages participate in the immunomodulatory response to Fasciola hepatica products. Immunology 133, 386–396. https://doi.org/10.1111/j.1365-2567.2011.03449.x (2011).
Ravida, A. et al. Fasciola hepatica Surface Coat Glycoproteins Contain Mannosylated and Phosphorylated N-glycans and Exhibit Immune Modulatory Properties Independent of the Mannose Receptor. PLoS Negl. Trop. Dis. 10, e0004601. https://doi.org/10.1371/journal.pntd.0004601 (2016).
Garcia-Campos, A. et al. Tegument Glycoproteins and Cathepsins of Newly Excysted Juvenile Fasciola hepatica Carry Mannosidic and Paucimannosidic N-glycans. PLoS Negl. Trop. Dis. 10, e0004688. https://doi.org/10.1371/journal.pntd.0004688 (2016).
Garcia-Campos, A., Baird, A. W. & Mulcahy, G. Migration of Fasciola hepatica newly excysted juveniles is inhibited by high-mannose and oligomannose-type N-glycan-binding lectins. Parasitology 144, 1708–1717. https://doi.org/10.1017/S003118201700124X (2017).
Li, D. & Wu, M. Pattern recognition receptors in health and diseases. Signal Transduct. Target Ther. 6, 291. https://doi.org/10.1038/s41392-021-00687-0 (2021).
McGreal, E. P., Miller, J. L. & Gordon, S. Ligand recognition by antigen-presenting cell C-type lectin receptors. Curr. Opin. Immunol. 17, 18–24. https://doi.org/10.1016/j.coi.2004.12.001 (2005).
Guasconi, L., Chiapello, L. S. & Masih, D. T. Fasciola hepatica excretory-secretory products induce CD4+T cell anergy via selective up-regulation of PD-L2 expression on macrophages in a Dectin-1 dependent way. Immunobiology 220, 934–939. https://doi.org/10.1016/j.imbio.2015.02.001 (2015).
Aldridge, A. & O’Neill, S. M. Fasciola hepatica tegumental antigens induce anergic-like T cells via dendritic cells in a mannose receptor-dependent manner. Eur. J. Immunol. 46, 1180–1192. https://doi.org/10.1002/eji.201545905 (2016).
Higashi, N. et al. The macrophage C-type lectin specific for galactose/N-acetylgalactosamine is an endocytic receptor expressed on monocyte-derived immature dendritic cells. J. Biol. Chem. 277, 20686–20693. https://doi.org/10.1074/jbc.M202104200 (2002).
van Vliet, S. J., van Liempt, E., Geijtenbeek, T. B. & van Kooyk, Y. Differential regulation of C-type lectin expression on tolerogenic dendritic cell subsets. Immunobiology 211, 577–585. https://doi.org/10.1016/j.imbio.2006.05.022 (2006).
Ilarregui, J. M. et al. Macrophage galactose-type lectin (MGL) is induced on M2 microglia and participates in the resolution phase of autoimmune neuroinflammation. J. Neuroinflam. 16, 130. https://doi.org/10.1186/s12974-019-1522-4 (2019).
van Vliet, S. J. et al. MGL signaling augments TLR2-mediated responses for enhanced IL-10 and TNF-alpha secretion. J. Leukoc Biol. 94, 315–323. https://doi.org/10.1189/jlb.1012520 (2013).
da Costa, V. et al. The Tn antigen promotes lung tumor growth by fostering immunosuppression and angiogenesis via interaction with Macrophage Galactose-type lectin 2 (MGL2). Cancer Lett. 518, 72–81. https://doi.org/10.1016/j.canlet.2021.06.012 (2021).
van Vliet, S. J., Gringhuis, S. I., Geijtenbeek, T. B. & van Kooyk, Y. Regulation of effector T cells by antigen-presenting cells via interaction of the C-type lectin MGL with CD45. Nat. Immunol. 7, 1200–1208. https://doi.org/10.1038/ni1390 (2006).
Drickamer, K. & Taylor, M. E. Recent insights into structures and functions of C-type lectins in the immune system. Curr. Opin. Struct. Biol. 34, 26–34. https://doi.org/10.1016/j.sbi.2015.06.003 (2015).
Vazquez-Mendoza, A., Carrero, J. C. & Rodriguez-Sosa, M. Parasitic infections: a role for C-type lectins receptors. Biomed. Res. Int. 2013, 456352. https://doi.org/10.1155/2013/456352 (2013).
Dambuza, I. M. & Brown, G. D. C-type lectins in immunity: recent developments. Curr. Opin. Immunol. 32, 21–27. https://doi.org/10.1016/j.coi.2014.12.002 (2015).
Joardar, N., Mondal, C. & Sinha Babu, S. P. A review on the interactions between dendritic cells, filarial parasite and parasite-derived molecules in regulating the host immune responses. Scand. J. Immunol. 93, e13001. https://doi.org/10.1111/sji.13001 (2021).
Gordon, S. & Pluddemann, A. Tissue macrophages: heterogeneity and functions. BMC Biol. 15, 53. https://doi.org/10.1186/s12915-017-0392-4 (2017).
Jenkins, S. J. & Allen, J. E. The expanding world of tissue-resident macrophages. Eur. J. Immunol. 51, 1882–1896. https://doi.org/10.1002/eji.202048881 (2021).
Kurihara, T., Warr, G., Loy, J. & Bravo, R. Defects in macrophage recruitment and host defense in mice lacking the CCR2 chemokine receptor. J. Exp. Med. 186, 1757–1762. https://doi.org/10.1084/jem.186.10.1757 (1997).
Kuziel, W. A. et al. Severe reduction in leukocyte adhesion and monocyte extravasation in mice deficient in CC chemokine receptor 2. Proc. Natl. Acad. Sci. USA 94, 12053–12058. https://doi.org/10.1073/pnas.94.22.12053 (1997).
Dunay, I. R., Fuchs, A. & Sibley, L. D. Inflammatory monocytes but not neutrophils are necessary to control infection with Toxoplasma gondii in mice. Infect. Immun. 78, 1564–1570. https://doi.org/10.1128/IAI.00472-09 (2010).
Terrazas, C. et al. Ly6C(hi) inflammatory monocytes promote susceptibility to Leishmania donovani infection. Sci. Rep. 7, 14693. https://doi.org/10.1038/s41598-017-14935-3 (2017).
Heyde, S. et al. CD11c-expressing Ly6C+CCR2+ monocytes constitute a reservoir for efficient Leishmania proliferation and cell-to-cell transmission. PLoS Pathog 14, e1007374. https://doi.org/10.1371/journal.ppat.1007374 (2018).
van Vliet, S. J., Saeland, E. & van Kooyk, Y. Sweet preferences of MGL: carbohydrate specificity and function. Trends Immunol. 29, 83–90. https://doi.org/10.1016/j.it.2007.10.010 (2008).
van Vliet, S. J. et al. Carbohydrate profiling reveals a distinctive role for the C-type lectin MGL in the recognition of helminth parasites and tumor antigens by dendritic cells. Int. Immunol. 17, 661–669. https://doi.org/10.1093/intimm/dxh246 (2005).
Denda-Nagai, K. et al. Distribution and function of macrophage galactose-type C-type lectin 2 (MGL2/CD301b): efficient uptake and presentation of glycosylated antigens by dendritic cells. J. Biol. Chem. 285, 19193–19204. https://doi.org/10.1074/jbc.M110.113613 (2010).
van Liempt, E. et al. Schistosoma mansoni soluble egg antigens are internalized by human dendritic cells through multiple C-type lectins and suppress TLR-induced dendritic cell activation. Mol. Immunol. 44, 2605–2615. https://doi.org/10.1016/j.molimm.2006.12.012 (2007).
Klaver, E. J. et al. Trichuris suis-induced modulation of human dendritic cell function is glycan-mediated. Int. J. Parasitol. 43, 191–200. https://doi.org/10.1016/j.ijpara.2012.10.021 (2013).
Terrazas, C. A., Alcantara-Hernandez, M., Bonifaz, L., Terrazas, L. I. & Satoskar, A. R. Helminth-excreted/secreted products are recognized by multiple receptors on DCs to block the TLR response and bias Th2 polarization in a cRAF dependent pathway. FASEB J. 27, 4547–4560. https://doi.org/10.1096/fj.13-228932 (2013).
Gautier, E. L. et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nat. Immunol. 13, 1118–1128. https://doi.org/10.1038/ni.2419 (2012).
Lee, C. G. et al. A distal cis-regulatory element, CNS-9, controls NFAT1 and IRF4-mediated IL-10 gene activation in T helper cells. Mol. Immunol. 46, 613–621. https://doi.org/10.1016/j.molimm.2008.07.037 (2009).
Zheng, Y. et al. Regulatory T-cell suppressor program co-opts transcription factor IRF4 to control T(H)2 responses. Nature 458, 351–356. https://doi.org/10.1038/nature07674 (2009).
Cretney, E. et al. The transcription factors Blimp-1 and IRF4 jointly control the differentiation and function of effector regulatory T cells. Nat. Immunol. 12, 304–311. https://doi.org/10.1038/ni.2006 (2011).
Gerdes, N. & Zirlik, A. Co-stimulatory molecules in and beyond co-stimulation - tipping the balance in atherosclerosis?. Thromb. Haemost. 106, 804–813. https://doi.org/10.1160/TH11-09-0605 (2011).
Daneshmandi, S., Pourfathollah, A. A., Karimi, M. H. & Emadi-Baygi, M. PDL-1/PDL-2 blockade in mice dendritic cells by RNAi techniques to induce antitumor immunity. Immunotherapy 7, 1145–1158. https://doi.org/10.2217/imt.15.80 (2015).
Becerra-Diaz, M. et al. STAT1-dependent recruitment of Ly6C(hi)CCR2(+) inflammatory monocytes and M2 macrophages in a helminth infection. Pathogens 10, 1. https://doi.org/10.3390/pathogens10101287 (2021).
Reyes, J. L. et al. Treatment with cestode parasite antigens results in recruitment of CCR2+ myeloid cells, the adoptive transfer of which Ameliorates Colitis. Infect Immun 84, 3471–3483. https://doi.org/10.1128/IAI.00681-16 (2016).
Costa, M. et al. Heme-Oxygenase-1 Attenuates Oxidative Functions of Antigen Presenting Cells and Promotes Regulatory T Cell Differentiation during Fasciola hepatica Infection. Antioxidants (Basel) 10, 1. https://doi.org/10.3390/antiox10121938 (2021).
Greil, J. et al. Mutating heme oxygenase-1 into a peroxidase causes a defect in bilirubin synthesis associated with microcytic anemia and severe hyperinflammation. Haematologica 101, e436–e439. https://doi.org/10.3324/haematol.2016.147090 (2016).
Tzima, S., Victoratos, P., Kranidioti, K., Alexiou, M. & Kollias, G. Myeloid heme oxygenase-1 regulates innate immunity and autoimmunity by modulating IFN-beta production. J. Exp. Med. 206, 1167–1179. https://doi.org/10.1084/jem.20081582 (2009).
Lee, T. S. & Chau, L. Y. Heme oxygenase-1 mediates the anti-inflammatory effect of interleukin-10 in mice. Nat. Med. 8, 240–246. https://doi.org/10.1038/nm0302-240 (2002).
Chauveau, C. et al. Heme oxygenase-1 expression inhibits dendritic cell maturation and proinflammatory function but conserves IL-10 expression. Blood 106, 1694–1702. https://doi.org/10.1182/blood-2005-02-0494 (2005).
Balan, M. et al. Novel roles of c-Met in the survival of renal cancer cells through the regulation of HO-1 and PD-L1 expression. J. Biol. Chem. 290, 8110–8120. https://doi.org/10.1074/jbc.M114.612689 (2015).
Mukae, Y. et al. Pathological roles of c-Met in bladder cancer: Association with cyclooxygenase-2, heme oxygenase-1, vascular endothelial growth factor-A and programmed death ligand 1. Oncol. Lett. 20, 135–144. https://doi.org/10.3892/ol.2020.11540 (2020).
Muliaditan, T. et al. Repurposing Tin Mesoporphyrin as an Immune Checkpoint Inhibitor Shows Therapeutic Efficacy in Preclinical Models of Cancer. Clin. Cancer Res. 24, 1617–1628. https://doi.org/10.1158/1078-0432.CCR-17-2587 (2018).
McVeigh, P. et al. In silico analyses of protein glycosylating genes in the helminth Fasciola hepatica (liver fluke) predict protein-linked glycan simplicity and reveal temporally-dynamic expression profiles. Sci. Rep. 8, 11700. https://doi.org/10.1038/s41598-018-29673-3 (2018).
Whitehead, B., Boysen, A. T., Mardahl, M. & Nejsum, P. Unique glycan and lipid composition of helminth-derived extracellular vesicles may reveal novel roles in host-parasite interactions. Int. J. Parasitol. 50, 647–654. https://doi.org/10.1016/j.ijpara.2020.03.012 (2020).
Murphy, A. et al. Fasciola hepatica Extracellular Vesicles isolated from excretory-secretory products using a gravity flow method modulate dendritic cell phenotype and activity. PLoS Negl. Trop. Dis. 14, e0008626. https://doi.org/10.1371/journal.pntd.0008626 (2020).
Quinn, S. M. et al. Anti-inflammatory trained immunity mediated by helminth products attenuates the induction of T cell-mediated autoimmune disease. Front. Immunol. 10, 1109. https://doi.org/10.3389/fimmu.2019.01109 (2019).
Gayo, V., Cancela, M. & Acosta, D. Maintenance of life cycle stages of fasciola hepatica in the laboratory. Methods Mol. Biol. 2137, 1–14. https://doi.org/10.1007/978-1-0716-0475-5_1 (2020).
Frigerio, S. et al. Eosinophils control liver damage by modulating immune responses against fasciola hepatica. Front. Immunol. 11, 579801. https://doi.org/10.3389/fimmu.2020.579801 (2020).
Carasi, P. et al. Heme-oxygenase-1 expression contributes to the immunoregulation induced by fasciola hepatica and promotes infection. Front. Immunol. 8, 883. https://doi.org/10.3389/fimmu.2017.00883 (2017).
Acknowledgements
We are particularly grateful to abattoirs ‘Frigorífico Carrasco’ for their help with worm collection. The authors wish to thank to Agencia Nacional de Investigación e Innovación (ANII) for funding (FCE_1_2019_1_1_156295) to TF.
Author information
Authors and Affiliations
Contributions
M.C. performed the experiments, analyzed data and reviewed the manuscript. V.C. assisted with MGL2-DTR mice model to deplete MGL2+ cells and contributed with manuscript revision. S.R-Z, M.L. P.L. and M.F.F. assisted with in vivo animal and organ treatment. M.L. P.L. and M.F.F revised the final manuscript. TF contributed to supervision and design of all experiments shown in this paper, analyzed data and finally prepared and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Costa, M., da Costa, V., Lores, P. et al. Macrophage Gal/GalNAc lectin 2 (MGL2)+ peritoneal antigen presenting cells during Fasciola hepatica infection are essential for regulatory T cell induction. Sci Rep 12, 17661 (2022). https://doi.org/10.1038/s41598-022-21520-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-21520-w
- Springer Nature Limited