Abstract
Learning and negative outcome expectations can increase pain sensitivity, a phenomenon known as nocebo hyperalgesia. Here, we examined how a targeted pharmacological manipulation of learning would impact nocebo responses and their brain correlates. Participants received either a placebo (n = 27) or a single 80 mg dose of d-cycloserine (a partial NMDA receptor agonist; n = 23) and underwent fMRI. Behavioral conditioning and negative suggestions were used to induce nocebo responses. Participants underwent pre-conditioning outside the scanner. During scanning, we first delivered baseline pain stimulations, followed by nocebo acquisition and extinction phases. During acquisition, high intensity thermal pain was paired with supposed activation of sham electrical stimuli (nocebo trials), whereas moderate pain was administered with inactive electrical stimulation (control trials). Nocebo hyperalgesia was induced in both groups (p < 0.001). Nocebo magnitudes and brain activations did not show significant differences between d-cycloserine and placebo. In acquisition and extinction, there were significantly increased activations bilaterally in the amygdala, ACC, and insula, during nocebo compared to control trials. Nocebo acquisition trials also showed increased vlPFC activation. Increased opercular activation differentiated nocebo-augmented pain aggravation from baseline pain. These results support the involvement of integrative cognitive-emotional processes in nocebo hyperalgesia.
Similar content being viewed by others
Introduction
Pain can arise as a debilitating symptom that is malleable and highly susceptible to an individual’s internal and external environment1,2. Outcome expectations are shown to play a role in shaping pain responses to a given event or treatment3,4,5. While positive outcome expectations can produce beneficial effects from inert treatments (placebo effects), negative outcome expectations can blunt the effect of active interventions and even increase pain sensitivity in response to inert treatments, a phenomenon termed nocebo hyperalgesia6,7,8,9.
An important process proposed to be involved in nocebo effects is associative learning10,11,12,13. Classical conditioning is used in experimental nocebo models to form expectations through associative learning11. In nocebo conditioning, negative associations form by pairing an inert nocebo stimulus (a sham treatment) to surreptitiously increased pain stimulations. After repeated trials, the nocebo stimulus evokes increases in perceived pain. Negative suggestions are commonly used to enhance conditioning9,10,14. Concurrently, conditioned nocebo effects have been shown to effectively reduce using extinction paradigms in which learned associations are discontinued14,15,16. Such paradigms can be adapted for use in MRI settings, using thermal pain stimulations paired with sham electrodes to serve as the nocebo manipulation in conditioning paradigms.
One of the major neural components mediating associative learning processes are the N-methyl-d-aspartate (NMDA) receptors17,18 whose agonism has been found to augment learning19,20,21,22. Enhanced NMDA receptor activity promotes local neuroplasticity, which in turn is believed to enhance the acquisition and consolidation of learned material in both animals23,24 and humans25,26. Studies that used pharmacological agents such as d-cycloserine (DCS) to enhance NMDA-dependent learning support an implication of NMDA receptors in associative learning27,28. DCS is a compound that impacts NMDA-mediated neuroplasticity differently in different doses. In lower doses (in most studies varying between 50 and 250 mg) it acts as a partial agonist at the glycine modulatory site of NMDA receptors29. To our knowledge, no studies have examined the role of NMDA-mediated learning in nocebo effects.
Findings on extinction-learning and exposure therapy indicate that DCS may be a promising agent for augmenting NMDA-dependent learning27,30,31. DCS has also been shown to enhance performance on declarative learning32 and generalization of conditioned effects to novel contexts33. Studies show that DCS enhances the extinction of phobias and other symptoms resulting from aversive learning29,34. Learning studies have shown that DCS can facilitate procedural learning35, and extinction or memory consolidation24,27. This evidence suggests that by agonizing NMDA receptors, DCS enhances specific learning processes, and can be used to manipulate and investigate how particular learning mechanisms may be involved in pain effects. Previous studies on experimentally induced nocebo and placebo effects indicate an involvement of brain areas that integrate prior experiences and memory into the processing of pain, such as the insula and amygdala36,37,38,39,40. Yet, neuroimaging findings in the field of nocebo and learning are still limited and somewhat inconsistent36. By utilizing fMRI while pharmacologically agonizing NMDA-mediated learning during nocebo induction, precise neural processes involved in learned pain can be examined.
In the present study we aim to investigate for the first time the role of NMDA-receptor dependent learning in the acquisition and extinction of nocebo effects. Detailed hypotheses and planned analyses were listed in the study pre-registration. Briefly, as compared to placebo administration, we hypothesize that DCS will augment the acquisition of nocebo hyperalgesia and will induce nocebo effects that are more resistant to extinction. We further hypothesize that differential brain activation will be detected between the DCS and placebo groups during nocebo acquisition, evocation, and extinction, in a number of a priori regions of interest such as the prefrontal cortex, anterior cingulate cortex, amygdala, and hippocampus, that were implicated in previous nocebo studies36. We also hypothesize that neural activation will differ between the experience of nocebo-augmented pain and the experience of pain stimulations of the same high intensity.
Materials and methods
Experimental design
This randomized controlled trial utilizes a placebo-controlled, double-blind design with respect to the pharmacological administration. A double-blind randomization list was created by the Leiden University Medical Center (LUMC) pharmacy. Participants were randomly allocated into blocks of one of two pharmacological groups: DCS or placebo. The random allocation sequence was thus carried out by a party independent from the study and the study investigators enrolled participants and assigned them to the double-blinded pharmacological group. All participants underwent nocebo pre-conditioning outside the scanner and acquisition/extinction procedures in the MR scanner, by use of conditioning and negative verbal suggestions. The entire study consisted of two parts in the same testing day. The screening part lasted approximately 1 h and took place at the department of Social and Behavioral Sciences, Leiden University, the Netherlands. The fMRI part lasted approximately 3 h, of which approximately 1 h took place in the 3 Tesla MRI scanner of the Leiden Institute of Brain and Cognition (LIBC) scanning facilities at the LUMC. This study was pre-registered on ClinicalTrials.gov (NCT04762836; trial registration date 21/02/2021), approved by the Medical Ethics Committee Leiden, The Hague, Delft (P19.003; trial approval date 29/06/2020), and all experiments were performed in accordance with relevant guidelines and regulations for research on human subjects.
Participants
The required sample size for the primary analysis was calculated based on a previous imaging study that, similar to our primary study objective, investigated the effects of DCS in a learning task32. The analysis was conducted in G*power 3.141 for a mixed model analysis of variance (ANOVA). In the experiment by Onur et al.32 an ANOVA revealed a main effect of the pharmacological agent (DCS vs. placebo) [F(1,27) = 5.454; P = 0.027] on performance in a declarative learning task. We derived partial η2 from the F statistic and degrees of freedom42,43. With an effect size of η2 = 0.17, alpha error probability set at α = 0.05, and desired power set at 0.9, the sample size indicated 22 participants per pharmacological group. Given the potential for dropout and artefacts in imaging data, we recruited 25 participants per group in this study.
Inclusion criteria were: age between 18 and 35 years, a good command of the English language, and (corrected to) normal vision and hearing. Exclusion criteria were any history of chronic pain, serious medical or psychiatric conditions, experiencing pain on the day of the study or use of analgesic medication in the 24 h prior to testing, use of psychotropic drugs in the month prior to testing, and being pregnant or breastfeeding. A physician performed a brief health screening based on our exclusion criteria and to assess vital signs. Participants also needed to be eligible to undergo MRI and were screened for standard MRI-compatibility exclusion criteria. Participants were recruited via the recruitment website Sona (Sona Systems, Tallinn, Estonia) and were only able to sign-up for participation if they had not previously participated in similar studies. All participants signed written informed consent and were reimbursed with a 90-euro payment.
Thermal pain stimulation
Thermal pain stimuli were delivered to participants’ right volar forearm and pain intensities were rated on a numeric rating scale (NRS) ranging from 0 (no pain) to 10 (worst pain imaginable on the arm). In the screening part, pain stimuli were delivered via a Thermal Sensory Analyzer with a 3 × 3 cm thermode probe (TSA-II; Medoc Advanced Medical Systems, Ramat Yishai, Israel). In the MRI part, pain was delivered with an MR-compatible ATS 3 × 3 thermode attached to a Pathway device (Medoc Advanced Medical Systems, Ramat Yishai, Israel).
Sensory thresholds
We followed a sensory-thresholds method that follows published standardized and protocolled procedures44. To test warmth and pain threshold levels, heat stimuli were applied from a baseline of 32 °C on the forearm and participants were asked to indicate the first moment that they perceived warmth and the first moment that they perceived pain. After a practice trial for each, the average of 3 warmth and 3 pain detection values were calculated as thresholds for warmth and pain, respectively.
Pain calibration and administered stimuli
Throughout the experiment, each stimulus was initiated from a 32 °C baseline, increased to a target temperature with ramp up and return rates of 8 °C per second, and presented at peak temperature for 5 s. The maximum temperature that could be reached was 50 °C. The inter-stimulus interval consisted of a pain rating screen with a 6 s duration followed by a fixation cross with a mean duration of 5 s, jittered around a normal distribution of + /− 2 s. Pain calibrations were conducted to select the temperatures that would induce moderate and high pain during nocebo conditioning. The calibrations were individually tailored, based on participants’ NRS ratings of maximum 30 pain stimuli of varying intensities. We used the median temperatures that participants consistently rated as NRS 6–9 (high pain) for nocebo trials in the pre-conditioning and acquisition phases. We used median temperatures consistently rated as approximately NRS 3–5 (moderate pain) for all control trials as well as evocation/extinction nocebo trials.
After calibrations, a nocebo pre-conditioning took place and included 7 nocebo and 7 control trials, to increase the time of learning and ensure nocebo effects would be induced. At the start of the MRI session a baseline phase took place during which 5 high and 5 moderate pain stimuli were administered. The acquisition and extinction phases each included 14 nocebo and 14 control trials. All trials were administered in pseudorandom order, so that no more than three trials of the same type were administered in a row. To reduce habituation or sensitization to heat-pain, we moved the thermode higher on the arm between functional scans; the thermode was moved to a more proximal site on the same arm after baseline and at one third and two thirds of the acquisition/extinction procedure).
Nocebo manipulation
A commercial Transcutaneous Electrical Nerve Stimulation (TENS) device (Beurer EM 80) was used to deliver (sham) electrical stimuli, which served as the nocebo manipulation in the procedure. Negative verbal suggestions were used to create expectations regarding the pain-enhancing effects of administering electrical stimuli in combination with thermal pain. Two electrodes were placed in a diagonal line on the base of the thumb and the inner elbow. Participants underwent a short mock calibration procedure during which they felt a light electrical pulse through the electrodes (ConMed MR-compatible Cleartrace ECG electrodes). This pulse was delivered in order to increase the believability of the nocebo manipulation. The device was not actually present during conditioning in the MRI scanner, but messages displayed on a computer monitor via E-Prime 3.0 (Psychology Software Tools, Pittsburgh, PA, USA) indicated the sham activation of the electrical stimulation during nocebo trials. Negative suggestions indicated to all participants that when the messages “on” (nocebo stimulus in either purple or yellow font, counterbalanced) and “off” (control stimulus in grey font) were displayed, their pain would be respectively aggravated (nocebo trials) or not altered (control trials). In the pre-conditioning and acquisition phases, the activation of sham electrical stimulation was paired to covertly increased pain stimulation during nocebo trials.
Pharmacological manipulation
A single dose of DCS was administered at 80 mg for all participants in the DCS group. The LUMC pharmacy prepared DCS (powder form) into capsules, as well as placebo capsules of identical appearance containing the inert agent microcrystalline cellulose. Because plasma concentrations were expected to peak between 1 and 3 h after DCS administration45,46, participants ingested the capsule 2 h before entering the MRI scanner to undergo the main learning paradigm.
Measures
Pain
Throughout the experiment, participants were provided with a 6-s window to rate their pain on a sliding scale representing the pain NRS, following each pain stimulation. A message, presented on the computer monitor 2 s after the pain stimulus returned to skin temperature, prompted the pain rating to be given by use of a keyboard in the screening part and button boxes in the MRI session.
Learning
To assess learning rates before and after the administration of DCS, participants completed the Wechsler Memory Scale–Fourth Edition (WMS–IV) subtest Verbal Paired Associates47. The test was performed twice, once before the administration of DCS and once at the end of the scanning session.
MRI
Data were collected at the Leiden Institute for Brain and Cognition imaging facilities at the Leiden University Medical Center, using a Phillips Achieva 3 Tesla scanner with a maximum gradient strength of 40 mT/m, bore diameter of 60 cm, and field-of-view of 45 cm (head-feet direction), and a 32-channel head coil. A structural MRI was made with a T1 weighted gradient echo sequence. Functional scans were taken utilizing T2* weighted gradient echo planar images (TR = 2200 ms, voxel size = 2.75 × 2.5 × 2.75 mm, TE = 30 ms, flip angle = 80°, matrix = 80 × 80, field of view = 220 × 220, slice thickness = 2.75 mm, slice gap = 0.28 mm, 40 slices per volume, sensitivity encoding factor = 2) with a 32-channel SENSE head coil. Functional scans began with two automatically discarded dummy volumes to allow for magnetic field stabilization. Heart rate (finger pulse oximeter) and respiration (respiratory belt transducer) were measured to correct for physiological artifacts during scanning.
Questionnaires
A questionnaire containing demographic and health questions was used to screen participants for inclusion. An MRI-compatibility questionnaire was also used, to ensure participants were eligible to enter the scanner. The Structured Clinical Interview for the Diagnostic and Statistical manual for Mental disorders (SCID-5-RV48 was used to screen participants for psychiatric disorders. The following four questionnaires were used to measure psychological characteristics: a short version of the State-Trait Anxiety Inventory, State version (STAI-S–s)49,50, the State-Trait Anxiety Inventory, Trait version (STAI-T)50, the Pain Catastrophizing Scale (PCS)51 which assesses catastrophizing thoughts or worrying relating to pain52, and the Body Vigilance Scale (BVS)53 which measures vigilance about bodily sensations. Total scores were used for all questionnaires.
Participants also completed an exit questionnaire at the end of the experiment, containing manipulation checks and questions about their participation and side effects. The questions were: “did you believe the information you received in this study”, “how much did you worry about what the experimenter thought of you or changing your responses to please them”, “were you focused on the pain stimulations during the study”, and “did you notice the association between the electrical stimuli and pain aggravation”. Questions were rated on a 0–10 NRS from “not at all” to “very much”. All questionnaires were displayed on a computer monitor via web-based survey software (Qualtrics, Provo, Utah, USA).
Study procedures
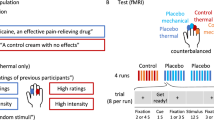
During the screening part (see Fig. 1), participants signed an informed consent form and completed the health, psychiatric, and MRI screening for inclusion in the study. Sensory and pain threshold levels were then tested, and pain stimuli were calibrated for each participant. The electrodes were then attached to the hand and arm and the short mock calibration of the sham electrical stimulation took place. Preconditioning was then completed. During the MRI part, participants first completed the WMS–IV and then received the oral pharmacological administration. During a 2-h waiting time for DCS to take effect, participants had a small, standardized meal, completed the psychological questionnaires, and prepared to enter the MRI scanner. Then, participants entered the scanner, completed a structural scan, and were then exposed to the baseline pain stimulations. Participants then underwent the nocebo acquisition and extinction procedures. After the end of the experiment, participants completed the second WMS–IV, were asked to answer the exit questions, and then were debriefed and reimbursed for their participation.
Timeline of the experimental procedure of this study. After screening for inclusion and pain calibrations, participants underwent a pre-conditioning phase. After the first learning task, D-cycloserine (DCS) or placebo was administered, and the psychological questionnaires were completed. In the fMRI part, participants completed structural scans and thereafter a first functional scan while receiving baseline moderate and high pain stimuli. Thereafter, three scans covered the acquisition and extinction of nocebo hyperalgesia. Finally, the second learning task, exit questionnaire, and a debriefing were completed.
Statistical analysis
Data screening and behavioral measures
Analyses of demographic, psychological, and behavioral measures were performed for descriptive purposes and as manipulation checks. Behavioral data were analyzed and visualized by use of R programming software (version 4.1.2; R Core Team, 2019), including the MASS54, stargazer55, and ggplot256 packages.
The magnitude of reported nocebo hyperalgesia was measured within-subjects and was defined as the difference in pain ratings for the first nocebo trial compared to the first control trial of the extinction phase (i.e., evocation). The first evocation trials were selected to check whether significant nocebo hyperalgesia was induced, as previous studies indicate the effect to be clearest in those trials14,57. We also compared the average of pain ratings of the first 10 trials of extinction (i.e., evocation) for a significant nocebo response, as these were used for brain imaging analysis where more trials are required. Repeated measures ANOVAs were conducted with trial type as within-subjects factor with two levels (nocebo trial, control trial), as a separate model, to test whether significant nocebo hyperalgesia was induced.
The first and last pairs of trials of the extinction phase were used to calculate the magnitude of extinction of nocebo responses. The reduction of nocebo responses was measured as the change in magnitude of nocebo responses (nocebo minus control trial difference score) between the start and the end of the extinction phase. A repeated-measures ANOVA was performed with time of measurement (pre to post) as within-subjects factor with two levels (nocebo magnitude before extinction, nocebo magnitude after extinction).
For the purpose of a thorough exploration of the data, we additionally performed the main behavioural, pharmacological, and fMRI analyses on a group of consistent nocebo responders which was drawn based on a 3SD method adapted from previous studies58. To test nocebo responses, the effects of DCS, and brain activations among only those participants who were had clear and consistent nocebo responses, we drew a group including those participants that reported nocebo evocation trials within 3 SD from their mean nocebo ratings during conditioning. We identified 28 participants as consistent nocebo responders, of which 13 received placebo and 15 received DCS. The detailed results of these additional exploratory analyses are reported in Supplementary material.
Pearson correlation analyses were performed between all questionnaire data (psychological questionnaires, exit questions) and the magnitude of nocebo responses (nocebo minus control trial difference score), to establish whether these factors impacted nocebo responding. We also conducted post-hoc exploratory mediation analyses, to explore potential between-groups mediating effects of the questionnaire scores on nocebo magnitudes.
For all behavioral analyses the threshold for significance was set at p < 0.05 and partial eta-squared (ηp2) was computed as a measure of effect size, with ηp2 of 0.01 considered small, 0.06 considered medium, and 0.14 considered a large effect size59,60. To conduct analysis of variance (ANOVA) and correlations, potential outliers and the assumptions of normality and homogeneity were checked.
Pharmacological manipulation
To test the first hypothesis, that DCS would lead to larger nocebo responses than a placebo, we examined whether nocebo hyperalgesia differed between the DCS and Control groups. A 2 × 2 mixed model ANOVA was performed, with group as the between-subjects factor and trial type in evocation as within-subjects factor (first extinction nocebo trial, first extinction control trial). We also examined the reduction of nocebo magnitudes after extinction. To compare extinction between the pharmacological groups, a 2 × 2 mixed model ANOVA was performed with group as the between-subjects factor and time of measurement (pre to post) as within-subjects factor with two levels (nocebo-control trial difference before extinction, nocebo-control trial difference after extinction).
fMRI analyses
In preprocessing the data, anatomical scans were skull stripped with the Brain Extraction Toolbox (BET;61. Subsequent preprocessing was conducted in SPM12 (Wellcome Department of Cognitive Neurology, London; www.fil.ion.ucl.ac.uk/spm) running on MATLAB 2021A (MathWorks, Natick MA, USA; https://www.mathworks.com/products/matlab.htm. Functional scans were realigned to correct for motion artifacts and low frequency drift, and six motion regressors (x, y, z, pitch, roll, yaw) were obtained to account for temporally correlated head motion artefacts in the design matrix. Low frequency drift was corrected with a 128 s high pass filter. Structural scans were coregistered to the mean functional image calculated during realignment. Functional images were rigid-body coregistered to the structural scan, which was then normalized to Montreal Neurological Institute (MNI) space with 2 × 2 × 2 mm voxels. The resulting deformation field was then applied to the functional images. A Gaussian spatial smoothing kernel of 6 mm full width at half maximum was applied to the functional images. Data were visually inspected for successful co-registration. The four functional scans were preprocessed separately, then concatenated to one set of functional images per participant. Heart rate and respiratory data were preprocessed with the PhysIO toolbox62.
For statistical analysis, functional images were modeled on a design matrix consisting of columns for 1, Baseline moderate pain trials; 2, Baseline high pain trials; 3, Acquisition control trials; 4, Acquisition nocebo trials; 5, Evocation control trials; 6, Evocation nocebo trials; 7, Extinction control trials; 8, Extinction nocebo trials (columns 1–8 of the design matrix, each modeled with the onset and duration of the approximately 7000 ms pain stimulus); 9, Pain rating periods; 10, Control and nocebo anticipatory cues; 11, RETROICOR regressors for heart rate, respiration, and heart rate-respiration interaction63; 12, respiratory volume per time; 13, heart rate variability (HRV; 11–13 estimated with the PhysIO toolbox); and 14, six motion regressors for the rigid body transformation. All task regressors (1–10) were convolved with the hemodynamic response function. Data were additionally high-pass filtered with a cut-off of 128 s and corrected for temporal autocorrelation with a first-order autoregressive model.
First level analyses pertaining to our hypotheses contrasted acquisition control trials with acquisition nocebo trials, evocation control trials with evocation nocebo trials, and baseline high pain trials with evocation nocebo trials. The evocation phase consisted of the first 10 extinction trials (evocation) and exploratory analyses of the extinction phase included the remaining 18 extinction trials (labeled as exploratory because hypotheses for extinction of nocebo responses were not explicitly stated in the pre-registration of this study). Second level analyses compared these contrasts between and across pharmacological groups. Masks pertaining to a priori regions of interest (ROI) including the ventrolateral prefrontal cortex (vlPFC), dorsolateral prefrontal cortex (dlPFC), amygdala, anterior cingulate cortex (ACC), operculum, and insula were drawn from the Harvard–Oxford Atlas (HOA;64 in FSLeyes65. Masked ROI analyses were conducted separately per ROI. Statistical significance for all contrasts was corrected with a familywise error rate (FWE) correction to a p value of pFWE < 0.05. This was further adjusted with a Bonferroni correction per hypothesis to pFWE < 0.01 to correct for five ROI analyses per hypothesis, with a minimum cluster size of 10 voxels (2 mm MNI space). Imaging data visualizations were carried out using FSL66.
Results
Participants and pain reports
Fifty-three participants were enrolled in this study and 2 were excluded upon screening for inclusion, based on their medical history (Fig. 2). The data of 1 participant that completed the study were excluded due to technical errors in the experiment. A total of 50 participants (39 women) were included in the final analyses. The mean age of participants was 23 years (SD = 3.3; Table 1). Table 1 also displays the mean warmth and pain detection thresholds, mean temperatures used to induce moderate and high pain, and reported pain differences during baseline as well as nocebo acquisition and extinction. Five participants that received DCS and two that received placebo self-reported noticing mild dizziness (n = 2 in the DCS group), or sleepiness/tiredness (n = 2 in the DCS group, n = 1 in the placebo group).
On average, participants reported that they believed the information they received during the study (M = 7.3, SD = 2.3), they were not concerned about what the researcher thought of them or changing their responses out of compliance (M = 0.6, SD = 1.1), they were focused on the heat stimuli (M = 8.2, SD = 1.2), and they noticed the increased pain association with the nocebo electrical stimuli (Mean = 9.2, SD = 1.2). We ran Pearson’s correlations between the magnitude of nocebo hyperalgesia and manipulation check exit questions and none of the responses to exit questions where significantly correlated with the magnitude of nocebo responses (all p > 0.05).
Behavioral results
The regression assumptions of linearity and homogeneity were met, and behavioral data were normally distributed. No outliers were present, determined by Mahalanobis distance. Correlation analyses of psychological questionnaire scores (Table 1) did not yield significant associations with nocebo magnitude or any other pain measures (all p > 0.05). Results on the between-groups mediating effect of questionnaire scores on nocebo magnitudes also yielded non-significant results (for all paths p ≥ 0.05). Baseline and post-experimental learning rates are shown in Table 1 and indicate that WMS-IV learning rates remained stable from before to after DCS administration.
The nocebo manipulation successfully induced nocebo responses as measured during the first trials of extinction (Fig. 3a). Across both groups, there was a significant difference between pain reports for the first nocebo and first control trial of the extinction phase (F (1,49) = 73.03, p < 0.001, ηp2 = 0.19) indicating the presence of nocebo hyperalgesia. We also found a significant nocebo effect in the first ten extinction trials which were used as an evocation phase for fMRI analysis (F (1,49) = 59.73, p < 0.001, ηp2 = 0.08; Fig. 3b). Finally, there was significant extinction of nocebo responses, with the magnitude of nocebo responses being significantly lower in the last pair of (nocebo/control) extinction trials, as compared to the first pair of extinction trials (F (1,49) = 13.17, p = 0.001, ηp2 = 0.08). When testing nocebo extinction in the last 10 (nocebo/control) extinction trials compared to the first 10 extinction trials, the extinction effect was not significant (F (1,49) = 0.53, p = 0.46, ηp2 = 0.004).
Behavioral results represented as group means and standard deviations. Dots in the figure represent individual data points. There was a significant nocebo effect in the first pair (A) and first 10 trials of extinction (B). Nocebo responses were not affected by D-cycloserine (DCS) compared to placebo (C). Learning rates were measured on the WMS-IV before and after DCS administration (D). There was only a slight non-significant reduction in learning rates of all participants, irrespective of drug group.
Pharmacological manipulation
A mixed ANOVA indicated that there was no significant interaction between drug group and the magnitude of nocebo responses based on trial type (F (1,48) = 0.002, p = 0.97, ηp2 < 0.001; Fig. 3c). This was aligned with no increases in learning rate from pre to post drug administration in the DCS as compared to the placebo group (Figs. 3d, 4). We did not find an effect of DCS on the magnitude of extinction either, as there was no significant interaction between drug group and the reduction of nocebo responses at the end of extinction (F (1,48) = 0.11, p = 0.73, ηp2 = 0.001).
Timeseries of all trials. Mean pain ratings are shown in pre-conditioning (Pre) baseline (Basel), nocebo induction (Indu), and extinction (Ext) including the first two extinction trials (Evocation) where behavioral nocebo effects were measured. D-cycloserine (DCS) administration did not significantly affect the magnitude of nocebo responses. The slight group difference in ratings observed indicates a minor average difference in individually calibrated pain intensities that was already present prior to DCS administration and had no significant effects on outcomes (all p > 0.05).
fMRI results
Increased activity in evocation nocebo trials compared to baseline high pain was found in the right operculum (Table 2, Fig. 5a). During the acquisition phase, we detected an increased BOLD response during nocebo trials compared to control trials in bilateral ACC, bilateral amygdala, bilateral insula, and bilateral vlPFC (all clusters from a priori analyses are presented in Table 2, Fig. 5b, e–i).
Results of the fMRI analysis. Differences in signal intensity of BOLD activations between baseline pain and nocebo-augmented increased pain responses were found in the operculum (A). Contrasting nocebo and control trials resulted in differential BOLD signal intensity during nocebo acquisition (B), evocation (first 10 extinction trials; C), and extinction (last 18 extinction trials; D). For nocebo acquisition, the significant activations in the vlPFC, insula, and amygdala in sagittal plane (E), the insula and amygdala in axial plane (F), the ACC in sagittal (G) and axial planes (H), and the amygdala in axial plane (I) are also shown. Location coordinates for sagittal (X), coronal (Y), and axial (Z) slices are shown above each fMRI image. Brain images are in neurological display convention.
No clusters reached the threshold for significance in the evocation (first 10 extinction trials) control and nocebo contrast initially, but notably, in exploratory analyses of the evocation phase with no minimum cluster size, we detected an increased BOLD response during nocebo trials in the left insula (all clusters from exploratory findings in Table 3, Fig. 5c), albeit this did not reach significance. Exploratory analysis of the remaining 18 extinction trials detected increased BOLD signal during nocebo trials in bilateral amygdala and insula, as well as a small, below-threshold cluster of the ACC (Table 3, Fig. 5d). Parameter estimates were computed with MarsBaR67 and are plotted for all clusters (Fig. 6).
Parameter estimates are plotted for all clusters and contrasts. Raw parameter estimates were extracted using MarsBar in SPM, with no scaling applied. Parameter estimates represent the mean beta value of the entire ROI, as derived from the Harvard Oxford cortical and subcortical structures atlases. The first two, highlighted bars represent the contrast between baseline high pain stimulations and evocation phase nocebo trials when pain was augmented by the nocebo manipulation. The remaining bars represent the contrast between nocebo and control trials for each of the experimental phases.
No differences between pharmacological groups were detected in any a priori ROIs, or during exploratory whole brain analyses, for the hypothesized contrasts between acquisition control and nocebo trials, evocation control and nocebo trials, or baseline high pain to evocation nocebo trials (Table 4). We further explored whether DCS would lead to differences in brain activations, as compared to placebo, only among participants that were labeled as consistent nocebo responders. We found no significant between-groups differences in any a priori ROIs (see Supplementary material).
Discussion
This study investigated the role of DCS on the acquisition and extinction of nocebo hyperalgesia in an fMRI study. Significant nocebo effects were induced but DCS did not influence nocebo magnitudes or brain activations, suggesting that the pharmacological manipulation did not influence learning in this nocebo paradigm. fMRI results indicated that in acquisition and extinction phases, there were significantly increased signal intensity of BOLD activations bilaterally in the amygdala, ACC, and insula, during nocebo compared to control trials. Nocebo acquisition trials also showed increased vlPFC activation. Increased opercular activation further differentiated nocebo-augmented pain aggravation from baseline high pain. These results are in line with previous nocebo studies and support the involvement of cognitive processes in nocebo hyperalgesia.
The learning paradigm induced significant nocebo responses across both groups, as was anticipated, and these effects were stronger among a group of consistent nocebo responders. The pharmacological manipulation in this study did not affect learning of verbal pairs or nocebo associations. DCS generally tended towards larger effect sizes, but still non-significant, for the group of consistent nocebo responders. Although DCS is known to impact neuroplasticity29 previous findings are mixed. Many studies show effects of DCS on phobia and symptoms that are known to result from aversive learning29,34. Yet, other studies have shown differential effects of DCS, for example facilitating procedural but not declarative learning35, and extinction or memory consolidation, but not necessarily acquisition of learned responses24,27. These differential findings could theoretically be related to the dosage used, with doses in the relevant studies mentioned above varying from 50 to 250 mg and fixed, rather than measured based on body weight. We choose a moderate dose of 80 mg. Generally, there does not seem to be an apparent dose-related efficacy of DCS, with one review of the literature reporting that neither the dose nor the time of administration had an effect on the learning outcomes68.
Interestingly, DCS augmentation effects have mainly been studied in phobic stimuli and for fear memory29. These results suggest that DCS is effective in modulating limbic NMDA circuits engaged in paradigms with a heavy fear load69. We did find increased amygdala activation for nocebo trials over control trials during acquisition and extinction of the nocebo effect irrespective of pharmacological group, and there seems to be some involvement of fear in nocebo40,70,71. Speculatively, the type of pain-learning task employed in our nocebo experiment may potentially not primarily rely on the same fear-learning circuits that DCS has been found to affect in previous studies. DCS not affecting nocebo responding may point to a potential differentiation between the specific mechanisms involved in pain-learning versus fear-learning. In other words, we speculate that learning a negative nocebo association may not involve the NMDA-mediated learning that DCS may be able to augment in more fear-specific contexts.
The amygdala has been consistently implicated in fear-learning32,72, but amygdala involvement may not be an essential feature or necessary prerequisite for nocebo induction. Other brain areas are shown to underlie nocebo hyperalgesia in the absence of an amygdala involvement73,74,75. Interestingly, the amygdala seems to be involved when experimental contexts or suggestions are especially negative or frightening, such as in visceral pain studies70 or studies of a higher threat-load that include extensive conditioning and negative suggestions40. In line with this, our study with pre-conditioning and negative suggestions showed increased activation of the amygdala on nocebo compared to control trials. Involvement of the amygdala in the more negative experimental contexts could suggest that fear may be a secondary modulatory factor in nocebo hyperalgesia71,76. Pain-related learning seems to take place on two conceptual levels. On one hand, cortical-level associative learning mechanisms may be at the core of acquiring learned pain effects. On the other hand, fear-related learning, that may take place in subcortical loops involving the amygdala, mediates pain worsening, and may be a secondary modulatory factor in pain chronification77.
Distinct learning mechanisms mediated via the vlPFC may also have engaged during nocebo acquisition in our study. The vlPFC is linked to learning, belief formation, and stimulus–response associations78,79,80,81,82. Neural circuits involving the vlPFC are thought to communicate through oscillations in gamma-band (60–160 Hz) frequency channels83. This relates to previous studies implicating gamma-band oscillations as a marker of learning in nocebo acquisition16,84,85. The vlPFC, as the present study also suggests, may be implicated in sensory stimuli whose properties are processed bottom-up83. This corresponds to participants engaging in this type of bottom-up processing of nocebo versus control stimuli, only in the acquisition phase of our experiment, before top-down processing based on learned information of nocebo associations begins taking place.
The insula and more broadly the operculum are also thought to be central cognitive features of sensory perception86,87,88. Opercular involvement is consistently found in nocebo hyperalgesia and marks mechanisms of sensory discrimination and cognitive pain modulation37. We also found differences in insular and opercular activations between nocebo and control stimuli. It is perhaps unsurprising that sensory modulation is involved in the acquisition of nocebo hyperalgesia. Crucially, however, we found a persistence of insular activations even when all heat administrations were equal in intensity, during extinction, albeit this was only a small cluster of activations. This may indicate that the brain continues engaging in cognitive pain discrimination during nocebo responding, when nocebo stimuli were generally perceived as more painful while all heat intensities were actually identical. Indeed, this is in line with findings of the present study indicating that nocebo responses were not completely attenuated at the end of the extinction phase. Indeed, when we tested nocebo extinction across a broader range of trials (in the last to the first 10 trials), the extinction effect was not significant, potentially reflecting that any attenuation of learned effects that does occur takes place quickly in the beginning of the extinction phase.
We also found further differences in opercular activations between evoked nocebo responses and baseline pain. Before learning took place, we administered participants with baseline high pain stimuli. Our results show increased activation of the operculum during nocebo-augmented high pain in the evocation phase, as compared to the baseline high pain stimuli. The operculum was significantly less engaged in experiencing high pain before learning took place, while increased cognitive sensory processing seems to take place when pain sensitivity is increased under nocebo hyperalgesic conditions. The consistent involvement of subregions of the operculum, ACC, and PFC in nocebo responding may underscore a primary role of cognitive and sensory integration and modulation in nocebo hyperalgesia.
A limitation of this study was in analyzing the small number of initial nocebo evocation trials, before prolonged extinction, which underpowered the evocation results. Given that extinction begins soon after the pairing of the cues and varying stimulus intensities is discontinued, only the first few extinction trials can be considered to distinctly represent nocebo evocation. One solution that would allow future studies to examine brain activity during evocation, before extinction occurs, is to reinforce nocebo associations throughout extinction. Some nocebo studies employed such continued reinforcement paradigms and achieved persistent nocebo responding that may have been less contaminated by extinction effects37,89. Additionally, while we conducted our power analysis for this study based on the behavioral-pharmacological primary outcome, a sample of 25 participants per group is considered a minimum required sample for fMRI analyses, and this may have led to some of the imaging results for smaller brain clusters being underpowered90.
This study also had other limitations that future research should address. It is important to note that the generalizability of our results may be limited, due to the recruitment of a healthy, young participant sample. Results of this study may not represent pain processing correlates in patients or individuals who have experienced severe or chronic pain, as their neurophysiology may differ from that of healthy people91. It is imperative for future research to replicate our findings both in patient populations and in more realistic clinical contexts.
Albeit the pharmacological manipulation using a partial NMDA receptor agonist did not affect nocebo responses, this study provided important support for the integration of cognitive-emotional and sensory processes in nocebo hyperalgesia. The acquisition of nocebo hyperalgesia was primarily characterized by increased activation in brain regions that cognitively integrate and modulate pain inputs. We showed that cognitive-emotional processing of pain signals in the operculum and ACC may integrate prior negative experiences. Understanding the intricate relationship of learning and sensory modulation in the formation of negative pain associations is highly relevant for the effective management of pain.
Data availability
The primary data files containing extracted raw data are submitted to the journal as supporting materials. The full set of materials, protocols, raw behavioral and neuroimaging files, and scripts for preprocessing and analyses will be made available via a complete publication package on the online data repository DANS (https://dans.knaw.nl/en/data-stations/life-health-and-medical-sciences/), within the first month after this study has been accepted for publication, according to Leiden University policy. The corresponding author will make available all data and materials to the journal’s Editorial Board Members and Referees immediately upon request.
References
Yosipovitch, G., Greaves, M. W. & Schmelz, M. Itch. The Lancet 361(9358), 690–694. https://doi.org/10.1016/S0140-6736(03)12570-6 (2003).
Colloca, L. & Miller, F. G. The nocebo effect and its relevance for clinical practice. Psychosom. Med. 73(7), 598–603. https://doi.org/10.1097/PSY.0b013e3182294a50 (2011).
Albu, S. & Meagher, M. W. Expectation of nocebo hyperalgesia affects EEG alpha-activity. Int. J. Psychophysiol. 109, 147–152. https://doi.org/10.1016/j.ijpsycho.2016.08.009 (2016).
Manaï, M., van Middendorp, H., Veldhuijzen, D. S., Huizinga, T. W. J. & Evers, A. W. M. How to prevent, minimize, or extinguish nocebo effects in pain. PAIN Reports 4(3), e699–e699. https://doi.org/10.1097/PR9.0000000000000699 (2019).
Peci, S. & Peci, F. Nocebo phenomenon. Neuropsychol. Trends https://doi.org/10.7358/neur-2016-020-peci (2016).
Amanzio, M. Expert review of clinical pharmacology Nocebo effects and psychotropic drug action nocebo effects and psychotropic drug action. Expert Rev. Clin. Pharmacol. 8(2), 159–161. https://doi.org/10.1586/17512433.2015.992877doi.org/10.1586/17512433.2015.992877 (2015).
Benedetti, F., Lanotte, M., Lopiano, L. & Colloca, L. When words are painful: Unraveling the mechanisms of the nocebo effect. Neuroscience 147(2), 260–271. https://doi.org/10.1016/j.neuroscience.2007.02.020 (2007).
Blasini, M., Corsi, N., Klinger, R. & Colloca, L. Nocebo and pain: An overview of the psychoneurobiological mechanisms. PAIN Rep. https://doi.org/10.1097/PR9.0000000000000585 (2017).
Thomaidou, M. A. et al. Learning mechanisms in nocebo hyperalgesia: The role of conditioning and extinction processes. Pain https://doi.org/10.1097/j.pain.0000000000001861 (2020).
Blythe, J. S., Peerdeman, K. J., Veldhuijzen, D. S., van Laarhoven, A. I. M. & Evers, A. W. M. Placebo and nocebo effects on itch. Itch 4(3), e27. https://doi.org/10.1097/itx.0000000000000027 (2019).
Bräscher, A.-K., Witthöft, M. & Becker, S. The underestimated significance of conditioning in placebo hypoalgesia and Nocebo hyperalgesia. Pain Res. Manage. 2018, 1–8. https://doi.org/10.1155/2018/6841985 (2018).
Icenhour, A. et al. Learning by experience? Visceral pain-related neural and behavioral responses in a classical conditioning paradigm. Neurogastroenterol. Motil. 29, e13026. https://doi.org/10.1111/nmo.13026 (2017).
van de Sand, M. F., Menz, M. M., Sprenger, C. & Büchel, C. Nocebo-induced modulation of cerebral itch processing—an fMRI study. Neuroimage 166, 209–218. https://doi.org/10.1016/J.NEUROIMAGE.2017.10.056 (2018).
Colagiuri, B., Quinn, V. F. & Colloca, L. Nocebo hyperalgesia, partial reinforcement, and extinction. J. Pain 16(10), 995–1004. https://doi.org/10.1016/J.JPAIN.2015.06.012 (2015).
Colagiuri, B. & Quinn, V. F. Autonomic arousal as a mechanism of the persistence of nocebo hyperalgesia. J. Pain 19(5), 476–486. https://doi.org/10.1016/j.jpain.2017.12.006 (2018).
Thomaidou, M. A. et al. Temporal structure of brain oscillations predicts learned nocebo responses to pain. Sci. Rep. 11(1), 9807. https://doi.org/10.1038/s41598-021-89368-0 (2021).
Castellano, C., Cestari, V. & Ciamei, A. NMDA receptors and learning and memory processes. Curr. Drug Targets 2(3), 273–283 (2001).
Rezvani, A. H. Involvement of the NMDA System in Learning and Memory (CRC Press/Taylor & Francis, 2006).
Glanzman, D. L. Associative learning: Hebbian flies. Curr. Biol. 15(11), R416–R419. https://doi.org/10.1016/J.CUB.2005.05.028 (2005).
Karel, P. et al. d-Cycloserine enhanced extinction of cocaine-induced conditioned place preference is attenuated in serotonin transporter knockout rats. Addict. Biol. 23(1), 120–129. https://doi.org/10.1111/adb.12483 (2018).
Wu, I.-T., Tang, T.-H., Ko, M.-C., Chiu, C.-Y. & Lu, K.-T. Amygdaloid zif268 participated in the D-cycloserine facilitation effect on the extinction of conditioned fear. Psychopharmacology 232(20), 3809–3819. https://doi.org/10.1007/s00213-015-4042-7 (2015).
Henneberger, C., Papouin, T., Oliet, S. H. R. & Rusakov, D. A. Long-term potentiation depends on release of d-serine from astrocytes. Nature 463(7278), 232–236. https://doi.org/10.1038/nature08673 (2010).
Horio, M., Mori, H. & Hashimoto, K. Is D-cycloserine a prodrug for D-serine in the brain?. Biol. Psychiat. 73(12), e33–e34. https://doi.org/10.1016/j.biopsych.2012.07.013 (2013).
Ledgerwood, L., Richardson, R. & Cranney, J. d-Cycloserine facilitates extinction of learned fear: Effects on reacquisition and generalized extinction. Biol. Psychiat. 57(8), 841–847. https://doi.org/10.1016/j.biopsych.2005.01.023 (2005).
Levin, R. et al. Behavioral and cognitive effects of the N-methyl-d-aspartate receptor co-agonist d-serine in healthy humans: Initial findings. J. Psychiatr. Res. 61, 188–195. https://doi.org/10.1016/j.jpsychires.2014.12.007 (2015).
Guastella, A. J., Dadds, M. R., Lovibond, P. F., Mitchell, P. & Richardson, R. A randomized controlled trial of the effect of d-cycloserine on exposure therapy for spider fear. J. Psychiatr. Res. 41(6), 466–471. https://doi.org/10.1016/J.JPSYCHIRES.2006.05.006 (2007).
Goff, D. C. D-Cycloserine: An evolving role in learning and neuroplasticity in schizophrenia. Schizophr. Bull. 38(5), 936–941. https://doi.org/10.1093/schbul/sbs012 (2012).
Inslicht, S. et al. S14 randomized controlled trial of hydrocortisone and D-cycloserine on fear extinction in PTSD. Biol. Psychiatry 83(9), 352. https://doi.org/10.1016/J.BIOPSYCH.2018.02.905 (2018).
Norberg, M. M., Krystal, J. H. & Tolin, D. F. A meta-analysis of D-cycloserine and the facilitation of fear extinction and exposure therapy. Biol. Psychiat. 63(12), 1118–1126. https://doi.org/10.1016/J.BIOPSYCH.2008.01.012 (2008).
Davis, M., Ressler, K., Rothbaum, B. O. & Richardson, R. Effects of D-cycloserine on extinction: Translation from preclinical to clinical work. Biol. Psychiat. 60(4), 369–375. https://doi.org/10.1016/J.BIOPSYCH.2006.03.084 (2006).
Ebrahimi, C. et al. Combining D-cycloserine with appetitive extinction learning modulates amygdala activity during recall. Neurobiol. Learn. Memory 142, 209–217. https://doi.org/10.1016/j.nlm.2017.05.008 (2017).
Onur, O. A. et al. The N-Methyl-D-aspartate receptor co-agonist D-cycloserine facilitates declarative learning and hippocampal activity in humans. Biol. Psychiat. 67(12), 1205–1211. https://doi.org/10.1016/J.BIOPSYCH.2010.01.022 (2010).
Brom, M. et al. d-Cycloserine reduces context specificity of sexual extinction learning. Neurobiol. Learn. Mem. 125, 202–210. https://doi.org/10.1016/J.NLM.2015.09.010 (2015).
Nave, A. M., Tolin, D. F. & Stevens, M. C. Exposure therapy, D-cycloserine, and functional magnetic resonance imaging in patients with snake phobia: a randomized pilot study. J. Clin. Psychiatry 73(9), 1179–1186. https://doi.org/10.4088/JCP.11m07564 (2012).
Kuriyama, K., Honma, M., Koyama, S. & Kim, Y. d-cycloserine facilitates procedural learning but not declarative learning in healthy humans: A randomized controlled trial of the effect of d-cycloserine and valproic acid on overnight properties in the performance of non-emotional memory tasks. Neurobiol. Learn. Mem. 95(4), 505–509. https://doi.org/10.1016/J.NLM.2011.02.017 (2011).
Thomaidou, M. A., Peerdeman, K. J., Koppeschaar, M. I., Evers, A. W. M. & Veldhuijzen, D. S. How negative experience influences the brain: A comprehensive review of the neurobiological underpinnings of nocebo hyperalgesia. Front. Neurosci. 15, 2021. https://doi.org/10.3389/fnins.2021.652552 (2021).
Freeman, S. et al. Distinct neural representations of placebo and nocebo effects. Neuroimage 112, 197–207. https://doi.org/10.1016/j.neuroimage.2015.03.015 (2015).
Egorova, N. et al. Neuromodulation of conditioned placebo/nocebo in heat pain: Anodal vs cathodal transcranial direct current stimulation to the right dorsolateral prefrontal cortex. Pain 156(7), 1342–1347. https://doi.org/10.1097/j.pain.0000000000000163 (2015).
Jensen, K. et al. A neural mechanism for nonconscious activation of conditioned placebo and nocebo responses. Cereb. Cortex 25(10), 3903–3910. https://doi.org/10.1093/cercor/bhu275 (2015).
Tinnermann, A., Geuter, S., Sprenger, C., Finsterbusch, J. & Büchel, C. Interactions between brain and spinal cord mediate value effects in nocebo hyperalgesia. Science 358, 105–108 (2017).
Faul, F., Erdfelder, E. & Lang, A. G. G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 39(2), 175–191. https://doi.org/10.3758/BF03193146 (2007).
Kennedy, J. J. The eta coefficient in complex anova designs. Cohen 30, 885 (1970).
Cohen, J. Eta-squared and partial eta-squared in fixed factor anova designs. Educ. Psychol. Meas. 33, 107–112 (1973).
Rolke, R. et al. Quantitative sensory testing: A comprehensive protocol for clinical trials. Eur. J. Pain 10(1), 77–88. https://doi.org/10.1016/J.EJPAIN.2005.02.003 (2006).
D’Souza, D. C. et al. IV glycine and oral d-cycloserine effects on plasma and CSF amino acids in healthy humans. Biol. Psychiat. 47(5), 450–462. https://doi.org/10.1016/S0006-3223(99)00133-X (2000).
Meftah, A., Hasegawa, H. & Kantrowitz, J. T. D-serine: A cross species review of safety. Front. Psych. 12, 1346. https://doi.org/10.3389/fpsyt.2021.726365 (2021).
Wechsler, D. Wechsler Adult Intelligence Scale–Fourth Edition (WAIS–IV) Elsevier, 2014).
First, M., Williams, J. B. W., Karg, R. S. & Spitzer, R. L. The Structured Clinical Interview for the DSM-5, Research Version (SCID-RV). Arlington, VA: American Psychiatric Association [Online]. https://www.columbiapsychiatry.org/research/research-labs/diagnostic-and-assessment-lab/structured-clinical-interview-dsm-disorders-14 (2015).
Marteau, T. M. & Bekker, H. The development of a six-item short-form of the state scale of the Spielberger State-Trait Anxiety Inventory (STAI). Br. J. Clin. Psychol. 31(3), 301–306. https://doi.org/10.1111/j.2044-8260.1992.tb00997.x (1992).
Spielberger, C., Gorsuch, R., Lushene, P., Vagg, P. & Jacobs, A. Manual for the state-trait anxiety inventory STAI (Form Y) (‘Self-Evaluation Questionnaire’). In Man State-Trait Anxiety Invent STAI 4–6 (Consulting Psychologists Press Inc., 1983).
Sullivan, M., Bishop, S. & Pivik, J. The pain catastrophizing scale: Development and validation. Psychol. Assess. 7(4), 524–532 (1995).
Crombez, G. et al. Let’s talk about pain catastrophizing measures: an item content analysis. PeerJ 8, e8643. https://doi.org/10.7717/peerj.8643 (2020).
Schmid, N. B., Lerew, D. R. & Trakowski, J. H. Body vigilance in panic disorder: Evaluating attention to bodily perturbations. J. Consult. Clin. Psychol. 65(2), 214–220. https://doi.org/10.1037/0022-006X.65.2.214 (1997).
Venables, W. N. & Ripley, B. D. Modern Applied Statistics with S—MASS, 4th ed. (Springer, 2002). https://www.stats.ox.ac.uk/pub/MASS4/.
Hlavac, M. stargazer: Well-formatted regression and summary statistics tables. R package version 5.2.2. https://CRAN.R-project.org/package=stargazer (2018).
Wickham, H. ggplot2: Elegant Graphics for Data Analysis (Springer, 2016). https://ggplot2.tidyverse.org
Bartels, D. J. P. et al. Role of conditioning and verbal suggestion in placebo and Nocebo effects on itch. PLoS ONE 9(3), e91727. https://doi.org/10.1371/journal.pone.0091727 (2014).
Youssef, A. M., Macefield, V. G. & Henderson, L. A. Pain inhibits pain; human brainstem mechanisms. Neuroimage 124, 54–62. https://doi.org/10.1016/j.neuroimage.2015.08.060 (2016).
Richardson, J. T. E. Eta squared and partial eta squared as measures of effect size in educational research. Educ. Res. Rev. 6(2), 135–147. https://doi.org/10.1016/j.edurev.2010.12.001 (2011).
Cohen, J. A power primer. Psychol. Bull. 112(1), 155–159. https://doi.org/10.1037/0033-2909.112.1.155 (1992).
Smith, S. M. Fast robust automated brain extraction. Hum. Brain Mapp. 17(3), 143–155. https://doi.org/10.1002/hbm.10062 (2002).
Kasper, L. et al. The PhysIO toolbox for modeling physiological noise in fMRI data. J. Neurosci. Methods 276, 56–72. https://doi.org/10.1016/j.jneumeth.2016.10.019 (2017).
Glover, G. H., Li, T.-Q. & Ress, D. Image-based method for retrospective correction of physiological motion effects in fMRI: RETROICOR. Magn. Reson. Med. 44(1), 162–167. https://doi.org/10.1002/1522-2594(200007)44:1%3c162::AID-MRM23%3e3.0.CO;2-E (2000).
Desikan, R. S. et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31(3), 968–980. https://doi.org/10.1016/j.neuroimage.2006.01.021 (2006).
McCarthy, P. FSLeyes V.1.3.0. Zenodo https://doi.org/10.5281/zenodo.3937147 (2021).
Jenkinson, M., Beckmann, C. F., Behrens, T. E. J., Woolrich, M. W. & Smith, S. M. FSL. Neuroimage 62(2), 782–790. https://doi.org/10.1016/J.NEUROIMAGE.2011.09.015 (2012).
Brett, M., Anton, J.-L., Valabregue, R. & Poline, J.-B. Region of interest analysis using an SPM toolbox. Neuroimage 16, 5. https://doi.org/10.1016/S1053-8119(02)90013-3 (2002).
Mataix-Cols, D. et al. D-cycloserine augmentation of exposure-based cognitive behavior therapy for anxiety, obsessive-compulsive, and posttraumatic stress disorders: A systematic review and meta-analysis of individual participant data. JAMA Psychiat. 74(5), 501–510. https://doi.org/10.1001/jamapsychiatry.2016.3955 (2017).
Millecamps, M. et al. d-Cycloserine reduces neuropathic pain behavior through limbic NMDA-mediated circuitry. Pain 132(1–2), 108–123. https://doi.org/10.1016/j.pain.2007.03.003 (2007).
Schmid, J. et al. Neural underpinnings of nocebo hyperalgesia in visceral pain: A fMRI study in healthy volunteers. Neuroimage 120, 114–122. https://doi.org/10.1016/j.neuroimage.2015.06.060 (2015).
Thomaidou, M. A., Veldhuijzen, D. S., Meulders, A. & Evers, A. W. M. An experimental investigation into the mediating role of pain-related fear in boosting nocebo hyperalgesia. Pain 162(1), 287–299. https://doi.org/10.1097/j.pain.0000000000002017 (2021).
Etkin, A. & Wager, T. D. Functional neuroimaging of anxiety: a meta-analysis of emotional processing in PTSD, social anxiety disorder, and specific phobia. Am. J. Psychiatry 164(10), 1476–1488. https://doi.org/10.1176/appi.ajp.2007.07030504 (2007).
Ellerbrock, I., Wiehler, A., Arndt, M. & May, A. Nocebo context modulates long-term habituation to heat pain and influences functional connectivity of the operculum. Pain 156(11), 2222–2233. https://doi.org/10.1097/j.pain.0000000000000297 (2015).
Keltner, J. R. Isolating the modulatory effect of expectation on pain transmission: A functional magnetic resonance imaging study. J. Neurosci. https://doi.org/10.1523/JNEUROSCI.4463-05.2006 (2006).
Rodriguez-Raecke, R. et al. Insular cortex activity is associated with effects of negative expectation on nociceptive long-term habituation. J. Neurosci. 30(34), 11363–11368. https://doi.org/10.1523/JNEUROSCI.2197-10.2010 (2010).
Egorova, N., Benedetti, F., Gollub, R. L. & Kong, J. Between placebo and nocebo: response to control treatment is mediated by amygdala activity and connectivity. Eur. J. Pain 24(3), 580–592. https://doi.org/10.1002/ejp.1510 (2020).
Timmers, I. et al. Amygdala functional connectivity mediates the association between catastrophizing and threat-safety learning in youth with chronic pain. Pain 162, 12. https://doi.org/10.1097/j.pain.0000000000002410 (2021).
Donohue, S. E., Wendelken, C., Crone, E. A. & Bunge, S. A. Retrieving rules for behavior from long-term memory. Neuroimage 26(4), 1140–1149. https://doi.org/10.1016/j.neuroimage.2005.03.019 (2005).
Sakagami, M. et al. A Code for behavioral inhibition on the basis of color, but not motion, in ventrolateral prefrontal cortex of macaque monkey. J. Neurosci. 21(13), 4801–4808. https://doi.org/10.1523/JNEUROSCI.21-13-04801.2001 (2001).
Salmi, J., Nyberg, L. & Laine, M. Working memory training mostly engages general-purpose large-scale networks for learning. Neurosci. Biobehav. Rev. 93, 108–122. https://doi.org/10.1016/j.neubiorev.2018.03.019 (2018).
Toni, I., Ramnani, N., Josephs, O., Ashburner, J. & Passingham, R. E. Learning arbitrary visuomotor associations: temporal dynamic of brain activity. Neuroimage 14(5), 1048–1057. https://doi.org/10.1006/nimg.2001.0894 (2001).
Versteeg, M., Hafkemeijer, A., de Beaufort, A. J. & Steendijk, P. An Understanding of (Mis)understanders: Exploring the underlying mechanisms of concept learning using functional magnetic resonance imaging. Mind Brain Educ. 15(1), 129–138. https://doi.org/10.1111/mbe.12273 (2021).
Zeithamova, D. et al. Brain Mechanisms of Concept Learning. J. Neurosci. 39(42), 8259–8266. https://doi.org/10.1523/JNEUROSCI.1166-19.2019 (2019).
Miltner, W. H. R., Braun, C., Arnold, M., Witte, H. & Taub, E. Coherence of gamma-band EEG activity as a basis for associative learning. Nature 397(6718), 434–436. https://doi.org/10.1038/17126 (1999).
Tu, Y. et al. A neural mechanism of direct and observational conditioning for placebo and nocebo responses. Neuroimage https://doi.org/10.1016/j.neuroimage.2018.10.020 (2019).
Craig, A. D. Interoception: The sense of the physiological condition of the body. Curr. Opin. Neurobiol. 13(4), 500–505 (2003).
Veldhuijzen, D. S., Greenspan, J. D., Kim, J. H. & Lenz, F. A. Altered pain and thermal sensation in subjects with isolated parietal and insular cortical lesions. Eur. J. Pain 14(5), 535.e1-535.e11. https://doi.org/10.1016/j.ejpain.2009.10.002 (2010).
Wiech, K. et al. Anterior insula integrates information about salience into perceptual decisions about pain. J. Neurosci. 30(48), 16324–16331. https://doi.org/10.1523/JNEUROSCI.2087-10.2010 (2010).
Kong, J. et al. A functional magnetic resonance imaging study on the neural mechanisms of hyperalgesic Nocebo effect. J. Neurosci. 28(49), 13354–13362. https://doi.org/10.1523/JNEUROSCI.2944-08.2008 (2008).
Cremers, H., Wager, T. & Yarkoni, T. The relation between statistical power and inference in fMRI. PLoS ONE 12, e0184923. https://doi.org/10.1371/journal.pone.0184923 (2017).
Pinheiro, E. S. S. et al. Electroencephalographic patterns in chronic pain: A systematic review of the literature. PLoS ONE 11(2), 49085. https://doi.org/10.1371/journal.pone.0149085 (2016).
Acknowledgements
This work was supported by a Vici grant from the Netherlands Organization for Scientific Research (grant number 45316004) granted to A. Evers. The authors would like to thank Prof. Erik Vermetten for serving as the independent expert on this study, as well as Elio Sjak-Shie and Evert Dekker for their valuable technical support in this project. We also thank Dr Donald Goff, for his valuable advice on the pharmacological manipulation for this study.
Author information
Authors and Affiliations
Contributions
M.T.: conceptualion and design; acquisition, analysis, and interpretation of data; investigation; methodology; visualization; manuscript drafting and writing revisions. J.B.: conceptualion and design; acquisition, analysis, and interpretation of data; investigation; methodology; writing revisions. D.V.: conceptualion and design; resources; interpretation of data; supervision; methodology; writing revisions. K.P.: conceptualion and design; writing revisions. JvL.: acquisition of data; writing revisions. E.G.: conceptualion and design; interpretation of data; writing revisions. H.C.: analysis and interpretation of data; writing revisions. A.E.: conceptualion and design; supervision; funding acquisition; writing revisions. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Thomaidou, M.A., Blythe, J.S., Veldhuijzen, D.S. et al. A randomized pharmacological fMRI trial investigating d-cycloserine and brain plasticity mechanisms in learned pain responses. Sci Rep 12, 19080 (2022). https://doi.org/10.1038/s41598-022-23769-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-23769-7
- Springer Nature Limited