Abstract
Diabetic macular edema (DME), a complication of diabetes mellitus, is a leading cause of adult-onset blindness worldwide. Recently, intravitreal anti-VEGF injection has been used as a first-line treatment. This study analyzed the association between the genetic profile of patients with DME and their response to treatment. Intravitreal anti-VEGF injections were administered monthly for three months to Korean patients diagnosed with DME, who were classified into two groups depending on whether they responded to anti-VEGF therapy or showed recurrence within six months. Peripheral blood samples were used for genetic analyses. Genome-wide association analysis results sowed that the genes DIRC3 on chromosome 2 (rs16857280, p = 1.2 × 10–6), SLCO3A1 on chromosome 15 (rs12899055, p = 2.5 × 10–6), and RAB2A on chromosome 8 (rs2272620, p = 4.6 × 10–6) were associated with treatment response to intravitreal anti-VEGF injection. SLC35F1, TMEM132D, KIAA0368, HPCAL1, IGF2BP3, SPN2S, COL23A1, and CREB5 were also related to treatment response (p < 5.0 × 10–5). Using the KEGG pathway analysis, RAB2A and CREB5 were found to be associated with AMPK signaling related to VEGF (p = 0.018). The identified genetic biomarkers can elucidate the factors affecting patient response to intravitreal anti-VEGF injection and help select appropriate therapeutic strategy.
Similar content being viewed by others
Introduction
Diabetic retinopathy (DR) is one of the leading causes of vision loss worldwide1. Diabetic macular edema (DME) is the most common cause of vision loss in patients with type 2 diabetes2. DME can occur at any stage of DR. There is neuronal loss, local inflammation, and pericyte loss, leading to microaneurysms that can leak fluid into the retina3,4. When fluid leaks into the macular area, subretinal and intraretinal fluid accumulate in the inner and outer reticular layers, leading to the swelling or thickening of the macula and development of DME5,6.
Based on the Early Treatment Diabetic Retinopathy Study (ETDRS), macular laser photocoagulation has been used at the point of leakage to treat non-center-involved DME and has been the gold standard for managing DME for many years. Refractory DME has been treated by removing tractional components via vitrectomy, increasing the oxygen level of the vitreous, and increasing the clearance rates of inflammatory cytokines and vascular endothelial growth factor (VEGF). Recently, VEGF has been considered to play a key role in DME, and intravitreal anti-VEGF injections have been used as the primary treatment. This treatment has also been proven to induce the regression of new vessels and improve the stage of DR7. Despite the widespread use of various anti-VEGF agents, it has been reported that about 30% of the patients do not respond to them. This is because complex mechanisms, such as inflammation, affect the development of DME8,9.
Current treatments are not always effective in patients with DME. Disease progression does not correlate with the patient’s lifestyle, and the response to anti-VEGF treatment also may vary10. The pathophysiology may differ depending on genetic variations. The complex mechanisms involved in the disease progression need to be analyzed further. Genome-wide association studies (GWAS) are an efficient method for identifying genetic variants associated with complex diseases, including DR. Several GWAS for DR-related phenotypes have been reported recently10. GWAS have also been performed using cohorts of patients with DME. Many of these studies claim to have found genetic associations; however, there are few studies related to the therapeutic effect.
The purpose of this study was to analyze the genetic association in patients with DME according to their response to the primary treatment, intravitreal anti-VEGF injection. For the evaluation, Korean patients with underlying diabetes mellitus were classified into two groups; a good responder group in which the macula was maintained without recurrence or edema, and a poor responder group.
Method
This prospective study included patients with defined ophthalmological status who received intravitreal injections to treat non-proliferative diabetic retinopathy (NPDR) with DME from a single physician (H Cho) at the Hanyang University Guri Hospital between May 2016 and August 2017. This study was approved by the Institutional Review Board of Hanyang University Guri Hospital (IRB FILE No. 2016–01-007) and adhered to the tenets of the Declaration of Helsinki. All participants provided written informed consent to participate in the study.
Study subjects
The inclusion criteria were treatment-naïve patients aged 18 years or older, presence of diabetes mellitus type 2, treatment with oral anti-hyperglycemic agents or insulin, and presence of center-involved DME with central subfield thickness (CST) measuring over 300 μm. The medical histories of all patients were reviewed, and those diagnosed with hypertension, dyslipidemia, or nephropathy and being treated or adequately controlled through on-going treatments were identified. Patients with one or more of the following criteria were excluded from the study: history of ocular surgery within the past 6 months, such as cataract extraction, active proliferative diabetic retinopathy (PDR), history of vitreous hemorrhage or preretinal hemorrhage presumably linked to PDR; previous treatment for DR, such as panretinal photocoagulation, intravitreal injection, or vitrectomy; or any history of coexisting pathology that could cause macular edema, such as retinal vascular disease, retinal dystrophies, and degeneration, ocular inflammatory disease, epiretinal membrane or vitreo-macular traction, uncontrolled hypertension (systolic blood pressure > 160 mmHg or diastolic blood pressure > 100 mmHg), uncontrolled diabetes mellitus (HbA1c > 10.0%), end-stage renal disease, heart failure, anemia, autoimmune disease, or infection.
All participants underwent standard ophthalmologic examination, including best-corrected visual acuity, slit lamp biomicroscopy, optical coherence tomography (swept source OCT, Topcon DRI OCT-1 Atlantis; Topcon, Inc., Tokyo, Japan), and wide-field fluorescein angiography (FA; Optos, Dunfermline, Scotland). The scanning protocol consisted of fast macular thickness maps as well as a 3D OCT 12.0 × 9.0 mm scan.
Macular thickness was measured using the built-in OCT software. DME was defined as the thickening of the retina (CST ≥ 300 μm, Fig. 1a)11. Bevacizumab (Avastin, Genentech) was administered intravitreally at the baseline visit, and at 30 ± 3 days and 60 ± 3 days from the baseline visit. Treatment response was defined as CST < 300 μm after intravitreal injection (Fig. 1b). The patients with DME were classified as good or poor responders by an experienced ophthalmologist (H Cho) after the last intravitreal anti-VEGF injection. The good responder group was defined as those who achieved CST < 300 μm for up to 6 months after the last injection. Refractory DME was defined as patients who did not achieve CST < 300 μm even after treatment. The poor responder group included patients with refractory or recurrent DME, defined as CST ≥ 300 μm within 6 months12,13,14.
Genome-wide SNP genotyping and quality controls
Prior to the first intravitreal injection, peripheral blood samples were obtained with informed consent. The Illumina OmniExpressExome-8 BeadChip was used to conduct genome-wide genotyping analysis, according to the Infinium HD protocol (Illumina, CA, USA). 200 ng of genomic DNA from each sample was whole-genome amplified, fragmented, precipitated, and resuspended in the appropriate hybridization buffer. After hybridization of the denatured samples onto the prepared BeadChips, the BeadChips were extended using a single labeled base. Then they were stained and imaged on the Illumina Bead Array Reader to obtain normalized bead intensity data for each sample. The data were loaded into the Illumina GenomeStudio software (v2011.1), which converted the fluorescence intensities into single nucleotide polymorphism (SNP) genotypes. For 960,912 markers on the chip, SNPs with a call rate lower than 95%, a minor allele frequency < 1% in the population, or significant deviation from Hardy–Weinberg equilibrium (HWE) in the whole sample (p < 0.0001) were excluded. A total number of 537,159 SNPs were included. Principal component (PC) analysis was performed to confirm that there were no samples deviating from homogeneity as a whole. The first four PCs were also included as covariates to adjust for the population stratification.
Genotype distributions were compared between the good and poor responder groups with logistic regression control using the SVS software (Golden Helix SNP and Variation Suite™ (Version 8.8.3), Bozeman, MT, USA).
Bioinformatics analysis
Pathway-based enrichment analysis was performed using the Database for Annotation, Visualization, and Integrated Discovery (DAVID; https://david.ncifcrf.gov) to annotate the biological pathways and molecular functions of the identified genes15. To analyze the genes and biochemical links, the Kyoto Encyclopedia of Genes and Genomes (KEGG; https://www.kegg.jp) was used16.
Result
A total of seventy-one treatment-naïve DME patients diagnosed between May 2016 and August 2017 underwent intravitreal injections. No patient was lost during follow-up. Six patients were excluded because they underwent surgery for vitreous hemorrhage, and fourteen patients were excluded due to other ophthalmic diseases identified during the follow-up period. A total of fifty-one patients were treated with intravitreal anti-VEGF injection, and twenty-five of them were classified as good responders, who did not show recurrence for more than 6 months after the last injection. Twenty-six patients were classified as poor responders. Genomic DNA extracted from whole blood in the good responder and poor responder groups were genotyped.
The demographics and clinical characteristics, including duration of DM and baseline HbA1c level, were not significantly different between the good and poor responder groups (p > 0.05, Table 1). Additionally, baseline CST and best-corrected visual acuity (BCVA) were not significantly different between the two groups.
Manhattan plots and high-ranked SNPs
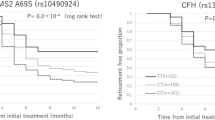
The Manhattan plots show the GWAS results of treatment response to intravitreal anti-VEGF injection in patients with DME (Fig. 2). In the comparison between the good and poor responder groups, no SNPs reached the conventional level of genome-wide significance (p < 5.0 × 10–8), but 11 SNPs were associated with the response to intravitreal anti-VEGF injection (p < 5.0 × 10–5). The highest ranked SNP, which was located in DIRC3 gene on chromosome 2, was associated with treatment response (rs16857280, p = 1.21 × 10–6, Fig. 3a). The minor allele frequency was higher in the good responder group (OR 18.23). On chromosome 15, SLCO3A1 gene was related to treatment response to intravitreal anti-VEGF injection, and its minor allele was also more frequently found in the good responder group (rs12899055, p = 2.5 × 10–6, OR 23.05, Fig. 3b). Another genotyped SNP, rs2772620, located in the RAB2A gene on chromosome 8, was associated with treatment response. The minor allele in this SNP rs2272620 was more frequently found in the poor responder group (p = 4.6 × 10–6, OR 0.08, Fig. 3c). In addition, SLC35F1, TMEM132D, KIAA0368, HPCAL1, IGF2BP3, SPN2S, COL23A1, and CREB5 genes were related to treatment response (p < 5.0 × 10–5, Table 2).
Pathway enrichment analysis
GWAS identified 11 SNPs that showed a suggestive association with response to intravitreal anti-VEGF injection (p < 5.0 × 10–5). Pathway-based enrichment analysis was performed for the genes of these SNPs using the KEGG database. The RAB2A and CREB5 gene were associated with the AMP-activated protein kinase (AMPK) signaling pathway (Fig. 4, p = 0.018). In this signaling pathway, RAB2A was involved in glucose metabolism. CREB5 was found to be related to mitochondrial biogenesis and apoptosis in the AMPK signaling pathway.
Proposed scheme for AMPK signaling pathway. VEGF: vascular endothelial growth factor; AMPK: AMP-activated protein kinase; RAB2A: Ras-related protein 2A; CREB: CAMP responsive element binding protein 5; mTOR: mammalian target of rapamycin; eNOS: endothelial isoform of nitric-oxide synthase; NFκB: nuclear factor kappa-light-chain-enhancer of activated B cells.
Discussion
To our knowledge, this is the first GWAS reported in relation to the treatment response of DME. In this study, we analyzed the relationship between genetic profiles and treatment outcomes of intravitreal anti-VEGF injection of DME in Korean patients. Eleven SNPs were associated with the treatment response. From the enriched pathway analysis based on KEGG, these genes were found to be related to the AMPK signaling pathway.
There are studies reporting the expression of genes associated with treatment response in DME, but these did not utilize GWAS. Shazly et al. reported a significant correlation between the genetic characteristics and response to intravitreal anti-VEGF (bevacizumab) in patients with DME. The distribution and allele frequency of the VEGFA -634C>G polymorphism (rs2010963) were significantly higher in good responders than in poor responders (p < 0.001)17. Tetikoglu et al. studied the association between VEGFA gene polymorphisms and response to intravitreal anti-VEGF (ranibizumab) injection in patients with DME, but no significant association was found18. In a pilot study that performed GWAS to differentiate between bevacizumab responders and non-responders, the majority of expressed genes were in transcriptional regulation or receptor activation pathways19. A recent study in a Middle Eastern population investigated the relationship between serum hyperglycemia-related long noncoding ribonucleic acid (lncRNA) levels and response to anti-VEGF (aflibercept) injection, but no significant association was found20,21. To date, only one recent report has conducted a GWAS to identify genes associated with the response to anti-VEGF treatment in DME, which has been performed in the Australian population22. In this report, the treatment response was determined based on the change in CST 12 months after the initiation of anti-VEGF injections, and several significant loci were identified. However, to date, no such GWAS has been performed in the Asian population.
The highest-ranked SNP rs16857280 is located in DIRC3, which was associated with treatment response. Diseases associated with DIRC3 include renal cell carcinoma, breast cancer, and skin cancer23. In a recent study, DIRC3 was found to act as a tumor suppressor, blocking the growth of human melanoma, and patients with high levels of DIRC3 showed significantly improved survival rates compared to those with low level 24. It has been shown that DIRC3 can be used to identify novel targets for skin cancer treatment. In this study, SLCO3A1 (rs12899055) was related to treatment response, and its minor allele was more frequently found in the good responder group (OR 23.05). In another GWAS, SLCO3A1 (rs207959) mediated inflammatory processes through the nuclear factor kappa-light-chain-enhancer of activated B cells (NFκB) in Crohn's disease25. These genes have not yet been identified as being related to DR or DME. Because these genes were shown to be related to treatment response in this study, the effect and associated mechanism of action on anti-VEGF treatment should also be taken into consideration.
rs2272620 is located in the RAB2A gene. RAB2A is known to be involved in glucose metabolism via the AMPK signaling pathway. AMPK translocates GLUT4 from intracellular storage vesicles to the plasma membrane, increasing glucose uptake into cells. In this process, translocation and fusion of GLUT4 vesicles are regulated by the Rab family, which is activated by AMPK26. RAB2A is also associated with cellular damage and apoptosis in Müller cells27. Müller cells are the primary glial cells in the retinal tissue that perform a wide range of functions, including maintaining the blood-retinal barrier. Hyperglycemia causes Müller cell dysfunction and loss, associated with RAB2A28. As RAB2A was associated with anti-VEGF treatment response, it may be the AMPK signaling pathway or Müller cell dysfunction in the VEGF-related pathways.
AMPK modulates cellular metabolism in response to stress, such as malnutrition, hypoxia, or exercise29. Pathway-based enrichment analysis using the KEGG database revealed that the AMPK signaling pathway was associated with the RAB2A and CREB5 genes (p < 0.05). Activation of AMPK in endothelial cells by VEGF represents a pro-angiogenic pathway30. Intracellular metabolic states are involved in the regulation of angiogenesis, which may involve AMPK. In addition to the glucose metabolism pathways in which RAB2A is involved, AMPK has a protective role in retinopathy31. AMPK promotes angiogenesis by increasing the production of VEGF and the endothelial isoform of nitric-oxide synthase (eNOS), which dilates retinal arterioles to improve circulation32. AMPK may attenuate angiogenesis by inhibiting mammalian target of rapamycin (mTOR) signaling as tumor-promoting and pro-metastatic factors33. Although the mechanisms are not clearly understood, they protect retinal neurons, glial cells, and retinal pigment epithelial cells by inhibiting oxidative stress and inflammation in the AMPK signaling pathway34. AMPK signaling inhibits the inflammatory response induced by NF-κB. Another study reported that SLCO3A1, which was identified in this study, was also associated with NFκB in regulating inflammation35. In a previous study that performed miRNA profiling and cytokine assays of the aqueous humor of DME patients, the aqueous concentrations of not only VEGF, but also inflammatory cytokines such as interleukin-6 and 8, were significantly higher in the DME group compared to the control group36. Inflammation or other factors may play an important role in the pathogenesis of DME and can be potential molecular targets for anti-VEGF injection and future DME treatment.
There are some limitations to this study that may have influenced the conclusions. First, the data were obtained from only one ethnic group (Korean) and may not be applicable to other ethnic groups. To generalize these results, data on genetic characteristics from different ethnic groups should be obtained and analyzed based on clinical characteristics. Second, due to the small sample size, no SNPs reached genome-wide significance (5e−8). Instead, we identified several loci with p values suggestive of association (5e−5). Third, there was also a lack of comparison with normal controls or patients with other disease or treatment response according to the drug. Further analysis may be helpful for initiating intravitreal injection therapy in patients with naive DME. In addition, analysis of the treatment response according to the subtypes of DME (according to morphology) or medication history could have provided meaningful information; however, this was not feasible due to the small sample size (of each subtype). Future studies with larger sample sizes and analyses based on subtypes are needed.
This study is the first to report a GWAS on the treatment response of Korean patients with DME to intravitreal anti-VEGF injection (the primary treatment). This study may allow ophthalmologists to personalize treatment options based on the patient’s genetic profile, improve drug selection, and reduce trial and error. In addition, it will help researchers understand the pathways related to the pathogenesis and treatment of DME.
Data availability
The datasets generated and analysed during the current study are available in ClinVar, https://www.ncbi.nlm.nih.gov/clinvar/submitters/508569.
References
Klein, B. E. Overview of epidemiologic studies of diabetic retinopathy. Ophthalmic Epidemiol. 14, 179–183 (2007).
Romero-Aroca, P. Managing diabetic macular edema: The leading cause of diabetes blindness. World J. Diabetes 2, 98–104 (2011).
Klein, R., Klein, B. E., Moss, S. E. & Cruickshanks, K. J. Relationship of hyperglycemia to the long-term incidence and progression of diabetic retinopathy. Arch. Intern. Med. 154, 2169–2178 (1994).
Caldwell, R. B. et al. Vascular endothelial growth factor and diabetic retinopathy: Pathophysiological mechanisms and treatment perspectives. Diabetes Metab. Res. Rev. 19, 442–455 (2003).
Murakami, T. et al. Foveal cystoid spaces are associated with enlarged foveal avascular zone and microaneurysms in diabetic macular edema. Ophthalmology 118, 359–367 (2011).
Zhang, X., Zeng, H., Bao, S., Wang, N. & Gillies, M. C. Diabetic macular edema: New concepts in patho-physiology and treatment. Cell Biosci. 4, 27 (2014).
Kim, E. J. et al. Treatment of diabetic macular edema. Curr. Diab. Rep. 19, 68 (2019).
Diabetic Retinopathy Clinical Research, N. et al. Aflibercept, bevacizumab, or ranibizumab for diabetic macular edema. N. Engl. J. Med. 372, 1193–1203 (2015).
Nguyen, Q. D. et al. Ranibizumab for diabetic macular edema: Results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology 119, 789–801 (2012).
Broadgate, S., Kiire, C., Halford, S. & Chong, V. Diabetic macular oedema: Under-represented in the genetic analysis of diabetic retinopathy. Acta Ophthalmol. 96 Suppl A111, 1–51 (2018).
Photocoagulation for diabetic macular edema. Early Treatment Diabetic Retinopathy Study report number 1. Early Treatment Diabetic Retinopathy Study research group. Arch. Ophthalmol. 103, 1796–1806 (1985).
Choi, M. Y., Jee, D. & Kwon, J. W. Characteristics of diabetic macular edema patients refractory to anti-VEGF treatments and a dexamethasone implant. PLoS ONE 14, e0222364 (2019).
Ciulla, T. A., Hussain, R. M., Ciulla, L. M., Sink, B. & Harris, A. Ranibizumab for diabetic macular edema refractory to multiple prior treatments. Retina 36, 1292–1297 (2016).
Bressler, N. M. et al. Persistent macular thickening following intravitreous aflibercept, bevacizumab, or ranibizumab for central-involved diabetic macular edema with vision impairment: A secondary analysis of a randomized clinical trial. JAMA Ophthalmol. 136, 257–269 (2018).
Dennis, G. et al. DAVID: Database for annotation, visualization, and integrated discovery. Genome Biol. 4, P3 (2003).
Ogata, H. et al. KEGG: Kyoto Encyclopedia of genes and genomes. Nucleic Acids Res. 27, 29–34 (1999).
El-Shazly, S. F., El-Bradey, M. H. & Tameesh, M. K. Vascular endothelial growth factor gene polymorphism prevalence in patients with diabetic macular oedema and its correlation with anti-vascular endothelial growth factor treatment outcomes. Clin. Exp. Ophthalmol. 42, 369–378 (2014).
Tetikoglu, M., Yuksel, Z., Aktas, S., Sagdik, H. M. & Ozcura, F. VEGF-A gene polymorphisms and responses to intravitreal ranibizumab treatment in patients with diabetic macular edema. Int. Ophthalmol. 38, 2381–2388 (2018).
Dabir, S. S. et al. Differential systemic gene expression profile in patients with diabetic macular edema: Responders versus nonresponders to standard treatment. Indian J. Ophthalmol. 62, 66–73 (2014).
Toraih, E. A., Abdelghany, A. A., Abd El Fadeal, N. M., Al Ageeli, E. & Fawzy, M. S. Deciphering the role of circulating lncRNAs: RNCR2, NEAT2, CDKN2B-AS1, and PVT1 and the possible prediction of anti-VEGF treatment outcomes in diabetic retinopathy patients. Graefes Arch. Clin. Exp. Ophthalmol. 257, 1897–1913 (2019).
Gurung, R. L., FitzGerald, L. M., McComish, B. J., Verma, N. & Burdon, K. P. Identifying genetic risk factors for diabetic macular edema and the response to treatment. J. Diabetes Res. 2020, 5016916 (2020).
Gurung, R. L. et al. Identifying genetic biomarkers predicting response to anti-vascular endothelial growth factor injections in diabetic macular edema. Int. J. Mol. Sci. 23, 4042 (2022).
Bodmer, D., Schepens, M., Eleveld, M. J., Schoenmakers, E. F. & Geurts van Kessel, A. Disruption of a novel gene, DIRC3, and expression of DIRC3-HSPBAP1 fusion transcripts in a case of familial renal cell cancer and t(2;3)(q35;q21). Genes Chromosomes Cancer 38, 107–116 (2003).
Coe, E. A. et al. The MITF-SOX10 regulated long non-coding RNA DIRC3 is a melanoma tumour suppressor. PLoS Genet. 15, e1008501 (2019).
Wei, S. C. et al. SLCO3A1, A novel crohn’s disease-associated gene, regulates nf-kappaB activity and associates with intestinal perforation. PLoS ONE 9, e100515 (2014).
Krishan, S., Richardson, D. R. & Sahni, S. Adenosine monophosphate-activated kinase and its key role in catabolism: Structure, regulation, biological activity, and pharmacological activation. Mol. Pharmacol. 87, 363–377 (2015).
Jayaguru, P. & Mohr, S. Nuclear GAPDH: Changing the fate of Muller cells in diabetes. J. Ocul. Biol. Dis. Infor. 4, 34–41 (2011).
Kusner, L. L., Sarthy, V. P. & Mohr, S. Nuclear translocation of glyceraldehyde-3-phosphate dehydrogenase: A role in high glucose-induced apoptosis in retinal Muller cells. Invest. Ophthalmol. Vis. Sci. 45, 1553–1561 (2004).
Dasgupta, B. & Chhipa, R. R. Evolving lessons on the complex role of AMPK in normal physiology and cancer. Trends Pharmacol. Sci. 37, 192–206 (2016).
Spengler, K. et al. Protein kinase A negatively regulates VEGF-induced AMPK activation by phosphorylating CaMKK2 at serine 495. Biochem. J. 477, 3453–3469 (2020).
Li, Y., Sun, R., Zou, J., Ying, Y. & Luo, Z. Dual roles of the AMP-activated protein kinase pathway in angiogenesis. Cells 8, 752 (2019).
Levine, Y. C., Li, G. K. & Michel, T. Agonist-modulated regulation of AMP-activated protein kinase (AMPK) in endothelial cells. Evidence for an AMPK -> Rac1 -> Akt -> endothelial nitric-oxide synthase pathway. J. Biol. Chem. 282, 20351–20364 (2007).
Li, L. et al. Vandetanib (ZD6474) induces antiangiogenesis through mTOR-HIF-1 alpha-VEGF signaling axis in breast cancer cells. Onco Targets Ther. 11, 8543–8553 (2018).
Jeon, S. M. Regulation and function of AMPK in physiology and diseases. Exp. Mol. Med. 48, e245 (2016).
Salminen, A., Hyttinen, J. M. & Kaarniranta, K. AMP-activated protein kinase inhibits NF-kappaB signaling and inflammation: Impact on healthspan and lifespan. J. Mol. Med. (Berl.) 89, 667–676 (2011).
Cho, H. et al. Micro-RNAs in the aqueous humour of patients with diabetic macular oedema. Clin. Exp. Ophthalmol. 48, 624–635 (2020).
Acknowledgements
This research was supported by the National Research Foundation of Korea (NRF) grants funded by the Korea government (NRF—2020R1A2C1010229 to HC, NRF- 2020R1F1A1048475 to YUS and NRF- 2021R1I1A1A01059690 to EHH), and by a grant of the Korean Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HI19C075300) to EHH.
Author information
Authors and Affiliations
Contributions
E.H.H. and H.Y. wrote the main manuscript text and contributed equally. H.S.Y. and J.E.P. analyzed and interpreted the data. Y.U.S. and S.Y.B. did critical revision of the manuscript for important intellectual content. H.C. and S.Y.B. supervised the study. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hong, E.H., Yeom, H., Yu, H.S. et al. Genome-wide association study of the response of patients with diabetic macular edema to intravitreal Anti-VEGF injection. Sci Rep 12, 22527 (2022). https://doi.org/10.1038/s41598-022-26048-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-26048-7
- Springer Nature Limited