Abstract
This was a prospective cohort study of eighteen patients with large and debilitating vascular malformations with one or more major systemic complications. In all patients, we discovered activating alterations in either TEK or PIK3CA. Based on these findings, targeted treatment using the PI3K inhibitor alpelisib was started with regular check-ups, therapy duration varied from 6 to 31 months. In all patients, marked improvement in quality of life was observed. We observed radiological improvement in fourteen patients (two of them being on combination with either propranolol or sirolimus), stable disease in 2 patients. For 2 patients, an MRI scan was not available as they were shortly on treatment, however, a clinically visible response in size reduction or structure regression, together with pain relief was observed. In patients with elevated D-dimer levels before alpelisib administration, a major improvement was noted, suggesting its biomarker role. We observed overall very good tolerance of the treatment, documenting a single patient with grade 3 hyperglycemia. Patients with size reduction were offered local therapies wherever possible. Our report presents a promising approach for the treatment of VMs harboring different targetable TEK and PIK3CA gene mutations with a low toxicity profile and high efficacy.
Similar content being viewed by others
Introduction
Vascular anomalies (VA) are a large and diverse group of diseases characterized by abnormal growth and development of blood or lymphatic vessels. They are associated with diverse symptomatology and often disabling conditions and remain both diagnostic and therapeutic challenges for medical professionals all over the world1,2. According to the ISSVA 2018 classification (Classification of Vascular Anomalies ©2018 International Society for the Study of Vascular Anomalies Available at “issva.org/classification” Accessed 01-JUN-2023), they comprise two major categories, vascular tumors and vascular malformations, which can be further divided into several subgroups. Vascular malformations (VMs) can be categorized into simple, combined, vascular malformations of major named vessels and vascular malformations associated with other anomalies.
The overall incidence of congenital VMs in the general population is 1.5%, and approximately two-thirds of cases are of venous predominance3. They can present as localized or diffuse lesions, and the symptomatology depends on the localization, extension, and involved anatomical structures. The appearance and symptoms are not static and can often progress during growth spurts and puberty1,2. Because of this varied symptomatology, multidisciplinary cooperation is of vital importance. For extensive lesions with vital organ and body part involvement, surgical procedures are very complicated because of their size and/or location. In these patients, other therapeutic options should be considered.
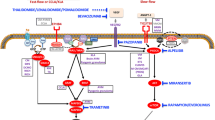
Mutations in genes that are involved in two significant intracellular signaling pathways, RAS/MAPK and PI3K/AKT, have been implicated in the pathophysiology of the majority of vascular malformations4,5,6. This opens the possibility that drugs/inhibitors currently being used in cancer patients may be used to treat patients with VMs7.
In this prospective observation report, we focused on the effect of the PI3K inhibitor alpelisib, which was recently approved by the FDA and EMA for the treatment of PIK3CA-mutated, hormone receptor-negative advanced breast cancer, showing very promising results8. Several authors have already demonstrated its effect in patients with PIK3CA-related overgrowth syndromes or PIK3CA-altered lymphatic malformations, and as a result of the EPIK-P1 clinical study9, it has recently been approved by the FDA for adult and pediatric patients with severe manifestations of the PIK3CA-related overgrowth spectrum10,11,12,13. In addition to patients with activating PIK3CA mutations, alpelisib treatment might also be beneficial for patients with activating TEK alterations, as the PI3K/AKT pathway is considered to be a central part of signaling through the TIE2 receptor encoded by this gene14. This was recently demonstrated in work published by Remy et al., in which the authors described the efficacy and pharmacokinetics of alpelisib in 3 patients with VMs harboring TEK mutation15.
Results
In each patient, activating mutations in either TEK or PIK3CA were found. In 12/18 patients, TEK exon 17 mutation was found, with TEK p.L914F being the most prevalent (9/12). In one of these patients, we found a TEK mutation of germline origin (p.Y897C). One patient harbored a truncating mutation in exon 23 of the TEK gene. Five patients harbored PIK3CA hotspot mutations, all of which have already been observed in patients with VM. In one patient, in addition to the somatic TEK mutation found in VA biopsy, a germline PTPN11 mutation causing Noonan syndrome was detected. The identified variants and the testing methods for each patient are listed in Table 1.
The total duration of alpelisib treatment varied from 6 to 31 months as of May 2023.
In all patients, marked improvement in the quality of life (QoL) was observed. In 12 patients, radiological improvement was documented; in 2 patients, the anomaly size remained stable; in 2 patients, radiological improvement was documented after combining alpelisib with either sirolimus (No. 6) or propranolol (No. 9). For 2 patients (Nos. 16 and 17), an MRI scan was not available due to short period of alpelisib administration. However, rapid clinical response with subjective improvement in QoL was documented. Effect of the treatment for every single patient is summarized in Table 2, percentage of VM size reduction in pre- and posttherapy MRI scans was radiologically measured either as volumatic, or planar change. For this purpose, we excluded two patients on combined treatment and two patients without MRI at time of analysis.
When comparing patients with TEK (9 patients) and PIK3CA (5 patients) mutation, better radiological response was observed in PIK3CA mutation group. For TEK mutation group the average volume reduction was 21% and median value was 19%. For PIK3CA mutation group the average volume reduction was 53% and median value was 45%. Moreover, both patients with stable disease harbored TEK mutation.
In patients with elevated D-dimer levels before alpelisib administration, a major improvement was noted. The D-dimer levels continually decreased, as shown in Fig. 1.
Notable individual cases are further described in more detail below.
We observed overall very good tolerance of the treatment. According to the CTCAE v5.016, we noted grade 3 toxicity of hyperglycemia requiring insulin treatment in one patient and grade 2 toxicity in 4 patients. Those specifically included mucositis in two patients, hyperglycemia requiring oral hypoglycemic medication and recurrent abdominal pain in one. The rest of the reported side effects were only grade 1, most commonly temporary abdominal discomfort, headaches, nausea, hyperglycemia, or liver enzyme elevation. Apart from the patient with hyperglycemia on insulin treatment, none of the patients required admission for treatment of adverse events, and all were managed in an outpatient setting.
Index cases presentation
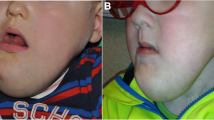
One of the first patients (No. 2) who was administered alpelisib treatment was an 18-year-old woman with severe and debilitating venous malformation of the right lower limb, pelvis, and genitalia. Prior to the start of alpelisib in November 2020, she underwent several surgical procedures over the years with only temporary effects. After 5 months of alpelisib treatment, we observed a rapid size reduction (see Fig. 2) and significant improvements in both QoL measurements and coagulation markers.
Patient No. 8 came to our clinic with a VeM located in her right foot, which resulted in severe pain while walking. Within weeks after the introduction of alpelisib treatment, she was able to walk and soon even run without any limitations. The effect of the treatment on the VeM size reduction and structure regression is well documented on MRI of the right foot prior to and 3 months after alpelisib treatment (see Fig. 3).
The patient with Noonan syndrome (No. 3) and a somatic TEK mutation had a rapid but only partial effect of the single-agent alpelisib treatment; therefore, a MEK inhibitor was added to target PTPN11 germline mutation, which may have contributed to the pathogenesis and thus to enhance the effect of alpelisib. However, very soon after the start of this double-agent therapy, the patient suffered from severe headaches and nausea, leading to the temporary discontinuation of both drugs. Within 2 weeks of discontinuation, we observed a rapid increase in the size of the malformation and D-dimer levels. Therefore, alpelisib treatment was restarted as monotherapy at a 50% dose reduction with good patient compliance and both clinical and laboratory efficacy in decreasing D-dimer levels.
The oldest patient was a 36-year-old woman who underwent a series of 25 surgeries and multiple nontargeted treatments for her VeM of a left lower limb starting in childhood before genetic testing discovered a pathogenic mutation of the TEK gene. After starting alpelisib treatment, we observed rapid clinical effects in QoL and size reduction of the lesions, which was later confirmed with MRI imaging.
Patient no. 12, with VeM of the left lower limb with muscular involvement, started the treatment due to potentially disabling surgery. Six months after the alpelisib administration, we achieved size reduction allowing safe total resection of the lesion without collateral damage to the healthy tissue. Six months after the procedure, the patient remains asymptomatic without any treatment (See Fig. 4).
Discussion
The use of targeted therapeutics for the management of VMs represents a new off-label therapeutic strategy, which could be used as an addition to classic local treatment approaches following the paradigm of precision medicine. Drugs that are currently being used in this setting include mTOR inhibitors17, PI3K inhibitors11, AKT inhibitors18, MEK inhibitors19, and VEGF inhibitors7; however, some of them show only limited efficacy, possibly because they are administered without prior knowledge of the driving gene mutation.
In this observational study, activating mutations in the PIK3CA and TEK genes were discovered in the lesions of 18 patients. Molecular analyses were performed using various sequencing methods ranging from Sanger sequencing to next-generation sequencing of targeted gene panels or WES. These different methods were selected based on current availability and the clinical picture suggesting a particular mutation in a given patient. The best diagnostic algorithm for the future, reflecting both economic and medical needs, remains to be established and is the subject of a future project.
Obtaining a representative piece of the lesion for subsequent analyses can become the first major obstacle due to the high risk of severe bleeding. A multidisciplinary team of specialists is of vital importance in facing such challenges. In patients with a difficult surgical approach, liquid biopsy could potentially help to discover driving mutations and may serve as a potential diagnostic marker; however, further investigation is needed20.
The starting dose of alpelisib was selected based on BSA-modified dosing schemes for the treatment of patients with breast cancer for whom a single dose of 300 mg is recommended (calculated 173 mg/m2 BSA). Our study used a 20% dose reduction (138.6 mg/m2/day) with high efficacy. Thus, for patients with insufficient response, the dose can be increased. In addition, for one of our patients who suffered an adverse event, restarting the drug with a 50% reduction was sufficient to maintain efficacy with a response measurable clinically and in D-dimer levels. In patient with hyperglycemia grade 3 toxicity we observed regrowth of the lesion after alpelisib cessation, restarting drug in 25% of the original dose led to suboptimal clinical and D-dimer response, however, at 37% of the original dose there was a very good both clinical and laboratory effect with only low dose of insulin therapy. Management of alpelisib induced diabetes mellitus was recently described in case report by Pia Peris et al.21.
To evaluate the therapeutic effect, patients underwent regular check-ups consisting of current history taking, clinical investigation, and laboratory testing. With the more recent patients, we also performed MRI scans before and 6 months after the start of the treatment. A measurable response was observed in 14 out of 18 patients on MRI, and a visual clinical response was observed in all patients, proving that alpelisib is highly effective. In all patients, we observed a striking effect on clinical symptomatology within the first few months of the treatment. Similar efficacy was reported by other authors describing their experience in a cohort of 19 patients and two case reports of patients with PIK3CA-related overgrowth syndrome (PROS)11,12,13. In our study, we also report the efficacy of alpelisib on VMs in patients without germline mutations but with somatic mutations, and moreover, this report describes alpelisib efficacy in the largest cohort of patients with somatic mutations in the TEK gene so far, thus following up on work of Remy et al.15.
When compared the radiological response in patients with TEK and PIK3CA mutations, both the average volume reduction and median value were higher in patients harboring PIK3CA mutation, however larger group would be needed for stronger results. The response in QoL parameters and coagulation markers were similar for both groups.
In patients who originally suffered from chronic coagulopathy, alpelisib treatment resulted in a decrease and even normalization of D-dimer levels, which seems to be a very useful marker of treatment success. Furthermore, we observed pain relief with quality-of-life improvement as the most important therapeutic goal. We have not observed any signs of cumulative toxicity comparing the first and second 6 months of drug exposure thus far.
After treatment discontinuation in two patients, we observed both clinical and paraclinical deterioration with a rapid rise of D-dimer levels, suggesting the need for prolonged treatment exposure. However, the optimal treatment duration and minimal effective dose remain unknown, as does the long-term toxicity profile in young patients. Different dosing and scheduling schemes have yet to be tested. Another open question is identifying the so-called “best achievable response” and reintroducing different local therapeutic options. Subjects who achieve size reduction after alpelisib treatment can be recommended for local treatments with a much lower risk of mutilation or complications such as bleeding or anatomical structure destruction. In our cohort, one patient (No. 12) experienced a 87% volume reduction of initially unresectable lesions on alpelisib, and the residual lesions were resected without bleeding or functional deficits of the thigh.
This prospective observational study contributes informations about a new treatment option that is well tolerated and shows objective responses with highly appreciated quality of life improvement.
Methods
All patients or their legal guardians signed informed consent with molecular genetic testing and off-label treatment using alpelisib. A total of 18 patients with large, debilitating or opioid requiring VMs with one or more major systemic complications (e.g., chronic systemic consumption coagulopathy, major organ involvement or multiple bone and joint involvement, scoliosis) underwent molecular genetic analysis using either whole-exome sequencing (WES), a targeted next-generation sequencing (NGS) panel, or direct sequencing of TEK exon 17 by Sanger’s method. The analyses were performed on lesion biopsy specimens. DNA was extracted from FFPE tissue using QIAmp FFPE Tissue Kit (Qiagen, Germany) and treated with NEBNext FFPE DNA Repair Mix (New England Biolabs, MA, USA). For WES, both lesion and matched normal genomic DNA were used. Libraries were prepared using TruSeq DNA Exome (Illumina, CA, USA) according to the manufacturer’s instructions and sequenced on the NextSeq500 platform using NextSeq 500/550 Mid Output Kit v2.5 (150 Cycles) (Illumina). Libraries for targeted NGS were prepared using QIAseq Targeted DNA Panel—Human Actionable Solid Tumor (Qiagen) according to the manufacturer’s instructions and sequenced on the NextSeq500 platform using NextSeq 500/550 Mid Output Kit v2.5 (300 Cycles) (Illumina). For direct sequencing of exon 17 of the TEK gene, custom-made PCR primers (IDT, NJ, USA) were used to amplify the target sequence (F: 5′-CCTGGGTGGTGTTGCTAGAT-3′, R: 5′-AGAGGGAACTCCACAGGAAAG-3′). Sequencing analysis of a purified and labeled PCR product was performed on the ABI 3130xl device (ThermoFisher Scientific, MA, USA). Patients with confirmed mutations in PIK3CA and TEK were recruited for alpelisib treatment and efficacy analysis.
All patients included in our study were Caucasian. Prior to the start of alpelisib treatment, a thorough patient history, clinical examination, baseline imaging, and laboratory tests, including D-dimer levels, were obtained. Initial clinical symptoms with basic demographic data, class of VM according to ISSVA (International society for the study of vascular anomalies) and discovered mutation types are summarized in Table 1.
Based on molecular findings and clinical symptoms, all 18 patients began orally administered alpelisib treatment. The dosage used for our patients was derived from the breast cancer guidelines and individually adjusted to body surface area (BSA) and varied from 50 to 300 mg per dose. The average starting dose was 138.6 mg/m2/day. As our center was not part of the EPIK-P1 study, the dosing of alpelisib in our patients differs from the dosing schedule in EPIK-P1, which led to the FDA approval of alpelisib for PIK3CA-related Overgrowth Spectrum patients in April 202222.
Regular check-ups were performed to evaluate the efficacy of the treatment. Each patient was examined in an outpatient clinic with laboratory and clinical examinations every 4 weeks and imaging, mostly MRI scan, every 3 months ± 2 weeks.
Quality of life (QoL) was assessed with a revised patient control outcome questionnaire which included an assessment of both pain and functional impairment resulting in a four-step semiquantitative scale: worse, stable, improved, and marked improvement.
Adverse events were classified according to CTCAE v5.0.
Discontinuation of the alpelisib treatment was allowed at the patient´s request due to adverse events and at the discretion of the treating physician.
All methods were carried out in accordance with relevant guidelines and regulations.
Conclusion
Novel PI3K inhibitors such as alpelisib represent a promising therapeutic option for patients with confirmed TEK or PIK3CA gene alterations when local treatment methods have only partial or temporary effect or when the lesion is inoperable because of its location. This prospective observational study contributes information about this new treatment option, which is well tolerated documenting only 1 grade 3 toxicity, objective responses, and highly appreciated quality of life improvements.
Several questions still need to be answered, as this is an off-label, agnostic, and experimental treatment. Although we did not observe any side effect requiring permanent treatment discontinuation, long-term toxicity remains to be established. The optimal treatment duration, dosing, and scheduling are also a matter of further investigation, as well as the optimal position of this systemic treatment in the complex management of vascular malformations.
Data availability
The data that support the findings of this study are available from the corresponding author, [P. Mudry], upon reasonable request. Sequencing data are available from: https://www.ebi.ac.uk/ena/browser/view/PRJEB53413, accession number PRJEB53413.
Abbreviations
- BSA:
-
Body surface area
- CTCAE v5.0:
-
Common terminology criteria for adverse events version 5.0
- NGS:
-
Next-generation sequencing
- PIK3CA:
-
Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha
- PROS:
-
PIK3CA-related overgrowth syndrome
- QoL:
-
Quality of life
- TEK:
-
Tyrosine kinase, endothelial
- VA:
-
Vascular anomalies
- VM(s):
-
Vascular malformation(s)
- VeM:
-
Venous malformation
- WES:
-
Whole-exome sequencing
References
Wassef, M. et al. Vascular anomalies classification: Recommendations from the international society for the study of vascular anomalies. Pediatrics 136, e203-214 (2015).
Steiner, J. E. & Drolet, B. A. Classification of vascular anomalies: An update. Semin. Interv. Radiol. 34, 225–232 (2017).
Eifert, S., Villavicencio, J. L., Kao, T. C., Taute, B. M. & Rich, N. M. Prevalence of deep venous anomalies in congenital vascular malformations of venous predominance. J. Vasc. Surg. 31, 462–471 (2000).
Queisser, A., Boon, L. M. & Vikkula, M. Etiology and genetics of congenital vascular lesions. Otolaryngol. Clin. N. Am. 51, 41–53 (2018).
Vikkula, M. et al. Vascular dysmorphogenesis caused by an activating mutation in the receptor tyrosine kinase TIE2. Cell 87, 1181–1190 (1996).
Limaye, N. et al. Somatic mutations in angiopoietin receptor gene TEK cause solitary and multiple sporadic venous malformations. Nat. Genet. 41, 118–124 (2009).
Van Damme, A., Seront, E., Dekeuleneer, V., Boon, L. M. & Vikkula, M. New and emerging targeted therapies for vascular malformations. Am. J. Clin. Dermatol. 21, 657–668 (2020).
André, F. et al. Alpelisib for PIK3CA-mutated, hormone receptor-positive advanced breast cancer. N. Engl. J. Med. 380, 1929–1940 (2019).
Canaud, G. et al. LBA23 EPIK-P1: Retrospective chart review study of patients (pts) with PIK3CA-related Overgrowth Spectrum (PROS) who have received alpelisib (ALP) as part of a compassionate use programme. Ann. Oncol. 32, S1297 (2021).
Delestre, F. et al. Alpelisib administration reduced lymphatic malformations in a mouse model and in patients. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.abg0809 (2021).
López Gutiérrez, J. C. et al. Alpelisib treatment for genital vascular malformation in a patient with congenital lipomatous overgrowth, vascular malformations, epidermal nevi, and spinal/skeletal anomalies and/or scoliosis (CLOVES) syndrome. J. Pediatr. Adolesc. Gynecol. 32, 648–650 (2019).
Pagliazzi, A. et al. PIK3CA-related overgrowth spectrum from diagnosis to targeted therapy: a case of CLOVES syndrome treated with alpelisib. Front. Pediatr. 9, 732836 (2021).
Venot, Q. et al. Targeted therapy in patients with PIK3CA-related overgrowth syndrome. Nature 558, 540–546 (2018).
Du, Z., Zheng, J., Zhang, Z. & Wang, Y. Review of the endothelial pathogenic mechanism of TIE2-related venous malformation. J. Vasc. Surg. Venous Lymphat. Disord. 5, 740–748 (2017).
Remy, A. et al. Repurposing alpelisib, an anti-cancer drug, for the treatment of severe TIE2-mutated venous malformations: Preliminary pharmacokinetics and pharmacodynamic data. Pediatr. Blood Cancer 69, e29897 (2022).
Common Terminology Criteria for Adverse Events (CTCAE) | Common Terminology Criteria for Adverse Events (CTCAE) | Protocol Development | CTEP. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm (2017).
Triana, P. et al. Sirolimus in the treatment of vascular anomalies. Eur. J. Pediatr. Surg. Off. J. Austrian Assoc. Pediatr. Surg. Al Z. Kinderchir. 27, 86–90 (2017).
Kobialka, P. et al. Low dose AKT inhibitor miransertib cures PI3K-related vascular malformations in preclinical models of human disease. bioRxiv https://doi.org/10.1101/2021.07.16.452617 (2021).
Lekwuttikarn, R., Lim, Y. H., Admani, S., Choate, K. A. & Teng, J. M. C. Genotype-guided successful treatment of an arteriovenous malformation in a child. JAMA Dermatol. 155, 256–257 (2019).
Palmieri, M. et al. A pilot study of next generation sequencing–liquid biopsy on cell-free DNA as a novel non-invasive diagnostic tool for Klippel-Trenaunay syndrome. Vascular 29, 85–91 (2021).
Pla Peris, B., Arranz Martin, A., Ballesteros García, A., Sebastián-Valles, F. & Marazuela Azpiroz, M. Alpelisib-Induced diabetes mellitus: Case Report, pharmacodynamics and management considerations. Front. Endocrinol. 13, 10 (2022).
FDA approves Novartis Vijoice® (alpelisib) as first and only treatment for select patients with PIK3CA-Related Overgrowth Spectrum (PROS). Novartis https://www.novartis.com/news/media-releases/fda-approves-novartis-vijoice-alpelisib-first-and-only-treatment-select-patients-pik3ca-related-overgrowth-spectrum-pros (2022).
Acknowledgements
We acknowledge the Core Facility Bioinformatics of CEITEC Masaryk University and the Core Facility Genomics supported by the NCMG research infrastructure (LM2018132 funded by MEYS CR) for their support in obtaining the scientific data presented in this paper. The Ethics Committee for Multicenter Clinical Trials of the University Hospital Brno approved this case series report (ref. number 01-291221/EK). Written informed consent was obtained from the parents or guardians of the children who served as subjects of the investigation, and when appropriate, assent was obtained from the subjects themselves.
Funding
Supported by the Ministry of Health, Czech Republic, Grant Nr. NV19-03-00562 and NU20-03-00240. All rights reserved. Supported by the Ministry of Health, Czech Republic—conceptual development of research organization (FNBr, 65269705). All rights reserved. Supported by Masaryk University, Faculty of Medicine, Czech Republic (MUNI/A/1427/2021). Supported by the project National Institute for Cancer Research (Programme EXCELES, ID Project No. LX22NPO5102)—Funded by the European Union—Next Generation EU. Supported by the European Regional Development Fund - Project ENOCH (No. CZ.02.1.01/0.0/0.0/16_019/0000868). The authors of this publication are members of the European Reference Network on Paediatric Cancer (ERN PaedCan). All rights reserved.
Author information
Authors and Affiliations
Contributions
Conceptualization: M.S., P.P., P.M., J.S.; Methodology: P.P., O.S., H.P.; Investigation: R.F., B.P., J.S.k., A.S., J.B., L.J., O.K., M.M., L.P.; Data curation: R.F., B.P., J.S.k., A.S., J.B., L.J., O.K., M.M., L.P.; Resources: M.S., P.P., H.P., P.J., P.M.; Writing—original draft: M.S.; Writing–Review and Editing: P.P., P.M., J.S.; Supervision: O.S., P.J., P.M., J.S. All authors approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sterba, M., Pokorna, P., Faberova, R. et al. Targeted treatment of severe vascular malformations harboring PIK3CA and TEK mutations with alpelisib is highly effective with limited toxicity. Sci Rep 13, 10499 (2023). https://doi.org/10.1038/s41598-023-37468-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-37468-4
- Springer Nature Limited