Abstract
Type 2 diabetes mellitus (T2DM) is a progressive metabolic disorder mainly caused by overweight and obesity that accumulates pro-inflammatory factors in adipose tissue. Studies have confirmed the efficacy of exercise and vitamin D supplementation in preventing, controlling, and treating diabetes. While, reduced physical activity and vitamin D deficiency are related to increased adiposity, blood glucose level, insulin concentration, and insulin resistance. This study purposed to investigate the effect of 8-week aerobic training with vitamin D supplementation on the expression of AMPK, PGC-1α, and UCP-1 genes expression in the visceral adipose tissue of obese rats with T2DM. In this experimental study, fifty male Wistar rats were divided into 5 groups (n = 10): aerobic training and vitamin D supplementation (AT + Vit D), aerobic training (5 days/week for 8 weeks; AT), vitamin D supplementation (Vit D), diabetic control (C) and NC (Non-Diabetic Control). AT + Vit D and AT groups practiced an 8-week aerobic training, 5 days a week. Vit D and AT + Vit D groups receive 5000 IU of vitamin D by injection once a week while AT and C received sesame oil. After blood sampling, visceral fat was taken to measure AMPK, PGC-1α, and UCP1 gene expression. Data were statistically analyzed by One-way ANOVA and paired sample t-test at a significance level of p < 0.05. Based on our results BW, BMI, WC, visceral fat, insulin, glucose, and HOMA-IR were significantly lower in the AT + Vit D, AT, and Vit D groups compared with the C group (p < 0.01). Furthermore, AT + Vit D, AT, and Vit D upregulated AMPK, PGC-1α, and UCP1 gene expression compared to the C. Based on the results compared to AT and Vit D, AT + Vit D significantly upregulated AMPK (p = 0.004; p = 0.001), PGC-1α (p = 0.010; p = 0.001), and UCP1 (p = 0.032; p = 0.001) gene expression, respectively. Also, AT induced more significant upregulations in the AMPK (p = 0.001), PGC-1α (p = 0.001), and UCP1 gene expression (p = 0.001) than Vit D. Vitamin D supplementation enhanced the beneficial effects of aerobic training on BW, BMI, WC, visceral fat, insulin, glucose, and HOMA-IR in diabetic rats. We also observed that separate AT or Vit D upregulated the gene expression of AMPK, PGC-1α, and UCP1 however, combined AT + Vit D upregulated AMPK, PGC-1α, and UCP1 more significantly. These results suggested that combining aerobic training and vitamin D supplementation exerted incremental effects on the gene expressions related to adipose tissue in animal models of diabetes.
Similar content being viewed by others
Introduction
Type 2 diabetes mellitus (T2DM) is a progressive metabolic disease with a growing prevalence. Studies have shown 382 million cases in 2013 globally, which is expected to reach 592 million by 20351. This multi-risk-factored disease is caused by defects in the function and secretion of insulin leading to increased blood glucose and B cell destruction consequently2. Although several lines of action have been considered to manage T2DM, there is an increasing need to find better approaches to control T2DM more effectively3,4. Recently, studies have focused on investigating the biological pathways involved in maintaining energy homeostasis to reduce the adverse effects of insulin resistance and metabolic disorders resulting from excessive food consumption and obesity5. For instance, AMP-Activated Protein Kinase (AMPK) is one of these biological pathways that stimulate FFA oxidation, inhibit FFA synthesis and lipolysis in adipose tissue, and signal the hypothalamus to increase food intake6. In addition, T2DM is associated with the reduced conversion of White Adipose Tissue (WAT) to brown adipose tissue, probably due to decreased expression and activation of specific browning genes such as Uncoupling Protein 1 (UCP1) and Peroxisome Proliferator-Activated Receptor Gamma Coactivator 1-Alpha (PGC-1α)7. The results of studies have shown that aerobic training is a powerful non-pharmacological way to confront obesity-related diseases, especially T2DM8,9, which has been an important lifestyle alteration to manage T2DM in recent years10,11. Cao et al. investigated the role of exercise on AMPK showing both acute and chronic exercises increased the phosphorylation and expression of AMPK and its downstream signaling pathway (e.g. PI3K) in diabetic rats12. Meanwhile, the results of Takekoshi et al. research showed that long-term aerobic training on a treadmill induced an increase in AMPK activity by about 1.30-fold compared with that of sedentary controls in visceral adipose tissue but has no effect on AMPK mRNA levels in subcutaneous adipose tissue13. Kang et al. also showed that long-term swimming training increases the expression of PGC-1α in obese mice14. Handschin and Spiegelman reported that 3-week moderate-intensity endurance training on a treadmill increases the expression of UCP1 mRNA in subcutaneous adipose tissue and visceral adipose tissue of rats leading to increased expression of PGC-1α15. However, some studies observed decreased expression of PGC-1α, and UCP1 while some reported no change in PGC-1α following exercise16,17. On the other hand, various studies have investigated the effect of supplements on patients with T2DM; Among them, studies have associated vitamin D deficiency with changes in insulin and blood glucose, as well as the sensitivity of specific tissues to insulin and consequently the risk of developing T2DM18,19. Also, since vitamin D is adipose-soluble and is stored in large amounts in adipose tissue, the serum level of vitamin D is probably lower in obese individuals20,21. A recent study has shown that vitamin D supplementation increased the effectiveness of aerobic training in reducing visceral fat, insulin, and blood glucose in Rats with metabolic syndrome and concluded that adequate vitamin D level is essential to attain the metabolic effects of physical activity22. Also, Cordeiro et al. reported reduced body weight gain and abdominal adiposity in obese rats following 30 days of vitamin D supplementation in western diet-fed obese rats23. Vitamin D alters insulin sensitivity in two main ways; regulating insulin receptor gene expression, which can be related to insulin resistance, and mediating calcium metabolism, thus increasing glucose transporter bioactivity21.
Accordingly, we hypothesized the possibility of the effect of vitamin D on the secretion and sensitivity of insulin through activating AMPK, UCP-1, and PGC-1α gene expression in visceral adipose tissue.
Although it is well established that separate aerobic training and vitamin D supplementation improve glucose tolerance and diabetes-related factors in skeletal muscle, the long-term effect of combined aerobic training with vitamin D supplementation on the expression of AMPK, PGC-1α, and UCP-1 genes in the visceral adipose tissue remains unknown. Considering the complications of T2DM and the increasing prevalence, this study aimed to investigate the effect of 8-week aerobic training with vitamin D supplementation on AMPK, PGC-1α, and UCP-1 gene expression in the visceral adipose tissue of obese rats with T2DM.
Methods
Ethical approval
All the procedures of the present study were submitted and approved by the Committee of Ethics and Research on the Use of Animals of the Razi University of Kermanshah (IR.RAZI.REC.1401.013) and experiments were performed following the specific Iranian laws on the Bioethics in Experiments with Animals (nº 11.794/2008) and complied with the ARRIVE guidelines for the Care and Use of Laboratory Animals.
Animals
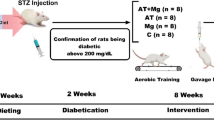
In this experimental study, fifty 4–5-week-old male Wistar rats weighing 180 ± 10 g were selected and divided into two main groups; diabetic (n = 40 rats) and Non-Diabetic Control (NC; n = 10 rats) groups. After inducing diabetes through intraperitoneal streptozotocin (STZ) and nicotinamide with a High-Fat Diet (HFD), diabetic rats were randomly assigned to 4 groups (10 in each group); aerobic training + vitamin D supplementation (AT + Vit D), aerobic training (AT), vitamin D supplementation (Vit D), control (C). AT and AT + Vit D groups practiced an 8-week aerobic training, 5 days a week. Vit D and AT + Vit D groups receive 5000 IU of vitamin D by injection once a week. While, AT and C were injected with sesame oil instead. This placebo-controlled randomized and single-blinded study was registered in the Iranian registry of clinical trials at http://www.irct.ir: number, IRCT20220510054809N1. Also, the Research Ethics Committee of Razi University approved and supervised the study design and conduction (no. IR.RAZI.REC.1401.013), Iran. Animal care, maintenance and sacrifice have been conducted according to the Danish “Animal Welfare Act” (LBK 1343 of 04/12/2007).
Obesity induction method
Two weeks prior to the commencement of the interventional program rats were acclimatized to the new environment. Then, male rats were fed high-fat diet to increase weight in the form of pellets (purchased from Beh-Parvar Company); a mixture of standard mouse food powder (365 mg/kg), sheep fat (310 mg/kg), mixed vitamins and minerals (60 mg/kg), dl-methionine (3 mg/kg), yeast powder (1 mg/kg), and chloride sodium (1 mg/kg). Animals were kept in transparent polycarbonate cages with a length of 30 cm, and width and height of 15 cm. Animals were maintained under a 12-h light/dark cycle, temperature of 25 ± 2 °C, and 45–55% humidity. All animals had free access to special food and water in a 500 ml bottles16,24; the ethical principles of working with laboratory animals were considered in the present study.
Diabetes induction method
After increasing weight (more than 300 g) following 2 weeks, 60 mg/kg BW streptozotocin (dissolved in 0.1 M citrate buffer with pH = 4.5) and then, after 15 min, 110 mg/kg BW of nicotinamide were intraperitoneally injected to induce diabetes in male Wister rats. To ensure the induction of T2DM, blood sugar was measured 2 weeks after the injection with the help of a glucometer and blood samples of the lateral tail vein after immersing in 42 °C water for 40–50 s; Blood sugar above 200 mg/l was considered as an indicator of diabetes25. Animals received no insulin treatment during the study.
Intervention programs
Aerobic training protocol
After the induction of diabetes, the rats were placed in 4 groups of 10 randomly and also through weight equalization. One week before the start of the main aerobic training sessions rats ran on the treadmill for 5 min at a speed of 8–10 m/min, 5 days a week to get familiar with the treadmill. The aerobic training program consisted of 8 weeks of running on a zero-incline treadmill, at 15–25 m/min, for 30–60 min, 5 days a week. In the first week, rats performed 30 min of aerobic training on a treadmill at a speed of 15 m/min. Then, the exercise intensity and duration increased gradually to reach 25 m/min and 60 min in the 8th week. The training intensity was equal to 50–60% of VO2max26. Animals also warmed up and cooled down for 10 min at 5 m/min before and after the aerobic training. To stimulate stopped rats to run, a combination of sound (hitting the wall of the rotating tape) and low voltage electric stimulations were used. In the first week, the low voltage electric stimulus was used along with the sound stimulus. Then, only the sound stimulus was used considering the ethical guidelines of working with laboratory animals16,24 (Fig. 1).
Vitamin D supplementation
In the present study, Vit D and AT + Vit D groups received 5000 international units (IU) of vitamin D per week by single-dose injection. AT and C were injected with sesame oil instead of vitamin D. Vitamin D serum concentration was measured using a vitamin D enzyme-linked immunosorbent assay (ELISA) rat kit (immune diagnostics system Ltd, Boldon, UK; with intraassay coefficient of variation = 1.63%, and sensitivity of method = 1.33 mg/dL)22.
Food intake, bodyweight, and body mass index
All animals were weighed weekly, between 09:00 and 11:30 a.m. using a scale. Body length (nose-to-anus) and Body Mass Index (BMI) were calculated. Food Intake (FI) was measured by subtracting the weight of the uneaten food from the total equal amount of food (20 g/d) given in each cage.
Tissue and blood sampling
In order to eliminate the acute effects of training and uncontrollable variables caused by the training program, 48 h after the last training session, the animals were anesthetized (46, 15) by intraperitoneal injection of a combination of ketamine (70 mg/kg BW) and xylazine (3–5 mg/kg BW) considering the ethical principles. The blood sample was taken from the vena cava; The plasma concentrations of glucose and insulin were measured by the Hitachi Auto Analyzer (type 7170; Hitachi Electronics, Hitachi, Japan). The Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) was calculated using the concentration of glucose and insulin as follows:
Visceral adipose tissue was immediately separated and weighed. After washing with saline, to prevent RNA degradation, it was placed in tubes containing RNA Later, transferred to liquid nitrogen, and then stored in a refrigerator at − 80 °C for further measurements.
RNA extraction/real-time PCR
RNA isolation from 20 mg of adipose tissue was performed using the combined protocol of TRI Reagent® (MRC Inc., US) and miRNeasy methods (Viragene, Iran). First, the frozen tissue was homogenized as recommended by the manufacturer using a gentleMACS™ Octo Dissociator system, M tubes (Viragene, Iran), and the RNA_02 program in 2 ml of TRI Reagent® buffer. After incubating at room temperature for 5 min, the samples were centrifuged at 12,000g at 4 °C for 10 min. The desired layer was piped into a 1.5 ml tube, mixed with 400 μl of chloroform, and centrifuged at 12,000g at 4 °C for 30 min after keeping the sample for 3 min at room temperature. Then the upper layer was mixed with ethanol by inverting the tube several times and loaded on the miRNeasy spin column (Viragene, Iran) performing the manufacturer’s protocol. Finally, RNA was eluted in 30 μl of RNAse-free water. Reverse transcription into cDNA was performed using 1 μg of total RNA with a Prime Script RT reagent kit (Viragene, Iran). Quantitative RT-PCR was performed using TB Green Premix Ex-Taq II (TaKaRa, Dalian, China). Beta-2-Microglobulin (B2M) was used as the reference gene to measure relative gene expression. The results were evaluated by using 2−ΔΔCt comparative method and Light Cycler SW1.1 software. The sequence of the primers is shown in Table 1. The Viragene kit, Iran was used to measure AMPK gene expression with a sensitivity of 0.021; to measure PGC-1α gene expression Viragene mini kit50, Iran was used with a sensitivity of 0.06; And to measure UCP-1 gene expression, Viragene kit, Iran was used with a sensitivity of 0.06.
Statistical analysis
Data were analyzed using Statistical Package for Social Sciences (SPSS, Inc., Chicago, IL, USA) version 26. To check the normality of the distribution in the continuous variables the Shapiro–Wilk test was used. The paired sample t-test was used to do within-group comparisons. Also, one-way ANOVA and Tukey's post-hoc to compare between-groups. The significance level was set at p < 0.05.
Ethical approval
This study was approved by the Ethics Committee of Razi University of Kermanshah (IR.RAZI.REC.1401.013) and was registered in the Iranian Clinical Trial Registration Center (code: IRCT20220510054809N1 on 27/06/2022).
Results
Mean body weight, BMI, and food intake (FI) are shown in Table 2. There was a significant difference between groups in mean BW, BMI, and FI after 8-week of intervention; BW, BMI, and FI reduced significantly in the AT + Vit D, AT, and Vit D groups at the end of the study compared with the beginning (p < 0.01). The highest reduction in BW, BMI, and FI was in the AT + Vit D group, whereas mean BW, BMI, and FI increased significantly in the C and NC groups.
As Table 3 shows, there was a statistically significant difference between groups in visceral fat, insulin, glucose, and HOMA-IR; with the lowest level in AT + Vit D and the highest in the C group. While, the results show a statistically significant difference between groups in serum 25-hydroxyvitamin D; with the highest level in AT + Vit D and the lowest in the C group. Based on the results, the mean visceral fat, insulin, glucose, and HOMA-IR were significantly lower in the AT + Vit D, AT, and Vit D than in the C group. In addition, significant differences were observed in visceral fat, insulin, glucose, HOMA-IR, and serum 25-hydroxyvitamin D between the NC group and other groups (p < 0.05 for all three variables).
The results of AMPK, PGC-1α, and UCP1 gene expression are shown in Figs. 2, 3, 4. There was a significant difference in AMPK, PGC-1α, and UCP1 gene expression between the diabetic and NC groups at the end of the intervention. One-way ANOVA showed a significant difference in Akt, PEPCK, and G6Pase gene expression between diabetic groups. Furthermore, AT + Vit D, AT, and Vit D upregulated AMPK, PGC-1α, and UCP1 compared to the C. Based on the results, AT + Vit D significantly upregulated AMPK (p = 0.004; p = 0.001), PGC-1α (p = 0.010; p = 0.001), and UCP1 (p = 0.032; p = 0.001) compared to AT and Vit D, respectively. Also, AT induced more significant upregulations in the AMPK (p = 0.001), PGC-1α (p = 0.001), and UCP1 gene expression (p = 0.001) than Vit D.
Comparison between mean ± SD of AMPK gene expression between groups. Values were calculated using a one-way analysis of variance followed by post hoc Tukey's test. AT + Vit D Aerobic Training + Vitamin D Supplement, AT Aerobic Training, Vit D Vitamin D Supplement, C Control, NC Non-Diabetic Control. µSignificantly different compared to AT. €Significantly different compared to Vit D. αSignificantly different compared to C. βSignificantly different compared to NC.
Comparison between mean ± SD of PGC-1α gene expression between groups. Values were calculated using a one-way analysis of variance followed by post hoc Tukey's test. AT + Vit D Aerobic Training + Vitamin D Supplement, AT Aerobic Training, Vit D Vitamin D Supplement, C Control, NC Non-Diabetic Control. µSignificantly different compared to AT. €Significantly different compared to Vit D. αSignificantly different compared to C. βSignificantly different compared to NC.
Comparison between mean ± SD of UCP1 gene expression between groups. Values were calculated using a one-way analysis of variance followed by post hoc Tukey's test. AT + Vit D Aerobic Training + Vitamin D Supplement, AT Aerobic Training, Vit D Vitamin D Supplement, C Control, NC Non-Diabetic Control. µSignificantly different compared to AT. €Significantly different compared to Vit D. αSignificantly different compared to C. βSignificantly different compared to NC.
Discussion
In the present study, we initially demonstrated that separate aerobic training and vitamin D improved body weight and serum parameters such as fasting blood glucose, insulin, and HOMA-IR. However, better results were observed when combining aerobic training and vitamin D. Our results were consistent with those of previous studies indicating the same improvements following aerobic training27,28. Also, studies showed that 8 weeks of Vitamin D supplementation reduced body weight, BMI, visceral fat, waist circumference, serum non-HDL cholesterol, and TG29,30. Vitamin D is a hormone that is synthesized in the skin and plays a vital role in maintaining optimal body function, particularly in the regulation of calcium homeostasis and bone health. The level of vitamin D in the body is influenced by various factors, including dietary intake, sun exposure, and physical activity31. The cellular and molecular mechanism of the observed differences in vitamin D values can be traced to the effect of exercise-induced changes on vitamin D metabolism32. During physical activity, there is an increase in the circulating levels of parathyroid hormone (PTH), a hormone that plays a crucial role in the regulation of calcium and phosphate homeostasis33. The increase in PTH levels stimulates the conversion of 25-hydroxyvitamin D [25(OH)D], the primary circulating form of vitamin D, to its active form, 1,25-dihydroxy vitamin D [1,25(OH)2D], in the kidneys32,33.
Moreover, exercise stimulates the increase in vitamin D receptor (VDR) expression in muscle cells as well as the synthesis of vitamin D binding protein (DBP) in the liver, which facilitates the transport and uptake of vitamin D into the muscles34. These changes ultimately result in an increase in the intracellular concentration of 1,25(OH)2D in the muscle fibers, which contributes to the beneficial effects of exercise on muscle health and function27.
However, the observed differences in vitamin D values may have been influenced by exercise-induced changes in vitamin D metabolism, as well as variations in initial levels. Understanding the cellular and molecular mechanisms of these factors can help researchers design more effective interventions to optimize vitamin D status and promote overall health. Therefore, it is crucial to control for these factors in any study examining the relationship between exercise and vitamin D status.
The molecular mechanisms of aerobic training in the improvement of diabetes might include the upregulation of insulin transporters in the cell membrane, reduction of adipokines, inflammatory and oxidative stress responses, and improvement of insulin signal transduction leading to higher insulin sensitivity. Another interesting point made in this study was that 8-week aerobic training increased gene expressions of AMPK in WAT of type 2 diabetes rats. In line with these results, studies have reported an increment in AMPK gene expression following acute and chronic exercises; Li et al. showed a simultaneous increase in AMPK and PGC-1α activity following chronic exercise training35. Park et al. observed a stimulatory role for short-term exercise on AMPK activation in visceral adipose tissue suggesting that aerobic training could stimulate basal AMPK activity in visceral adipose tissue36. Based on the results of the present study, alteration of AMPK mRNA levels in visceral adipose tissue could be involved in ameliorating T2DM following exercise adaptations. AMPK is involved in carbohydrate and fat metabolism, insulin signaling, and inflammation which are reduced in obesity37. Villena et al. reported increased body weight and adipose tissue mass in AMPK knockout mice which supports our results38. Besides the central role of AMPK as an energy sensor, AMPK has also been elucidated to have a paramount function in the browning of WAT and regulating the energy expenditure of brown/beige adipose tissue39. Our result also suggests that chronic exercise along with increased AMPK expression helps increase UCP-1 and PGC-1α gene expression. There are contradictory results in the literature regarding UCP1; Matteis et al. nominate exercise as a new physiological stimulus for brown adipose tissue activity but showed an insignificant increase in UCP-140. While, Brandao et al. investigated physical training and UCP1 expression, in adipose tissue of obese women reporting reduced UCP1 mRNA expression in WAT following 8 weeks of exercise41. It seems that increased intracellular regulatory factors (such as PGC-1α) following aerobic training and vitamin D supplementation might have altered the UPC-1 expression in this study. Furthermore, studies showed that increased secretion of hormones (e.g. thyroid hormones42, norepinephrine43, and Irisin44) during aerobic training might upregulate the UCP-1 gene expression in adipose tissue through different mechanisms. Moreover, a wide spectrum of studies support the increment of PGC-1α genes following aerobic training in skeletal muscle of humans45 and visceral adipose tissue of obese rats16. However, the observed weight loss in this study might be due to the reduced food intake following aerobic training and vitamin D supplementation. According to body weight and food intake results obtained in this study, the most significant reduction was in the group with aerobic training combined with vitamin D. The mechanism by which an increase in the concentration of vitamin D in the body weight and food intake affects body composition is difficult to explain. However, some studies reported no effect from vitamin D on body weight change and energy expenditure and little improvement in cardiovascular risks with cholecalciferol46,47. In agreement with our study, Hoseini et al. reported that vitamin D supplementation combined with aerobic training significantly improves BW and FI in ovariectomized rats22. Also, the decreased appetite hormone, and increased AMPK gene expression, and PGC-1α/UCP-1 pathway following aerobic training might be among the possible mechanisms. In addition, a proposed model related to the relationship between vitamin D deficiency and more food intake is that the decrease in the concentration of calcidiol flow in the hypothalamus induces an increase in the set point of body weight. On the other hand, the appetite increases and the energy consumption decreases through activating the hypothalamic NPY/AgRP neurons and POMC/CART inhibiting.
Furthermore, the results of the present study showed that AMPK, PGC-1α, and UCP1 expression increased after 8 weeks of vitamin D supplementation. Recent evidence has demonstrated that vitamin D is a potential stimulating factor in increasing AMPK activity and gene expression48,49. Gauthier et al. observed low AMPK activation in inflamed adipose tissue of obese human participants50. Chang investigated the role of vitamin D in oxidative stress and mitochondrial changes suggesting that the observed potent inhibitory effect of 1,25(OH)2D on muscle oxidative stress and mitochondrial dynamics might be at least involved in the activation of AMPK49. A recent research also studied the effects of vitamin D supplementation on adipose tissue inflammation and NF-κB/AMPK activation in obese mice fed a high-fat diet indicating a significant increase in AMPK activity by 1.4-fold following 16-week supplementation of vitamin D48. Vitamin D alters the genetic responses by binding to the Vitamin D Nuclear Receptor (VDR) and producing the Vitamin D Response Element (VDRE) which modifies AMPK, PGC-1α and UCP1 gene expression in WAT probably by binding to activating factors (COAC) or repressing factors (CORE)51. Additionally, vitamin D is believed to regulate lipolysis dependent on its localization; stimulating lipogenesis in subcutaneous tissue but inhibiting it in visceral adipose tissue52. Vitamin D supplementation was demonstrated to increase the expression of genes such as PGC-1α and PDK4 and thereby enhance the oxidation of fatty acids53. Vitamin D has long been known as a hormonal regulator of the cAMP signaling pathway54. In turn, cAMP activates cAMP-dependent PKA, the endocrine system, and AMPK activity55 which phosphorylates and activates lipolysis enzymes and leads to cascades of phosphorylation that stimulate the activity of PGC1α, PPARα, and UCP156,57. The results also showed a significant increase in the gene expression of AMPK, PGC-1α, and UCP1 in WAT in AT + Vit D compared to the other three groups (Vit D, AT, and control). However, the main mechanism for combined AT + Vit D is not elucidated, it might be a better approach in inducing AMPK, PGC-1α, and UCP1 expression in WAT of T2DM rats.
Strength and limitations of the study
Our study had several strengths using a randomized, placebo-controlled trial with no dropouts and evaluating the gene expression alterations in animal model of diabetes. However, it is premature to conclude a definitive answer. Also, not evaluating the levels of vitamin D before and after the induction of diabetes or at the beginning of the study and other important transcriptional and post-transcriptional factors were among the limitations of the present study.
Conclusion
Vitamin D supplementation enhanced the beneficial effects of aerobic training on BW, BMI, WC, visceral fat, insulin, glucose, and HOMA-IR in diabetic rats. We also observed that separate AT or Vit D upregulated the gene expression of AMPK, PGC-1α, and UCP1 however, combined AT + Vit D upregulated AMPK, PGC-1α, and UCP1 more significantly. These results suggested that combining aerobic training and vitamin D supplementation exerted incremental effects on the gene expressions related to adipose tissue in animal models of diabetes.
Data availability
The datasets generated and analysed during the current study are not publicly available due to ongoing data analysis but are available from the corresponding author on reasonable request.
Abbreviations
- AT:
-
Aerobic training
- Vit D:
-
Vitamin D
- T2DM:
-
Type 2 diabetes mellitus
- AMPK:
-
AMP-activated protein kinase
- WAT:
-
White adipose tissue
- UCP1:
-
Uncoupling protein 1
- PGC-1α:
-
Peroxisome proliferator-activated receptor gamma coactivator 1-alpha
- VDR:
-
Vitamin D nuclear receptor
- VDRE:
-
Vitamin D response element
- POMC:
-
Pro-opiomelanocortin
- CART:
-
Cocaine- and amphetamine-regulated transcript
References
Batrakoulis, A., Jamurtas, A. Z. & Fatouros, I. G. Exercise and type II diabetes mellitus: A brief guide for exercise professionals. Strength Cond. J. 44, 64–75 (2022).
Nanda, M., Sharma, R., Mubarik, S., Aashima, A. & Zhang, K. Type-2 diabetes mellitus (T2DM): Spatial-temporal patterns of incidence, mortality and attributable risk factors from 1990 to 2019 among 21 world regions. Endocrine 77(3), 444–454 (2022).
Davies, M., Tringham, J., Troughton, J. & Khunti, K. Prevention of type 2 diabetes mellitus. A review of the evidence and its application in a UK setting. Diabetic Med. 21(5), 403–414 (2004).
Mahluji, S. et al. Nutritional management of diabetes mellitus during the pandemic of COVID-19: A comprehensive narrative review. J. Diabetes Metab. Disord. 20(1), 963–972 (2021).
Ruderman, N. & Prentki, M. AMP kinase and malonyl-CoA: Targets for therapy of the metabolic syndrome. Nat. Rev. Drug Discov. 3(4), 340–351 (2004).
Minokoshi, Y. et al. AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 428(6982), 569–574 (2004).
Desjardins, E. M. & Steinberg, G. R. Emerging role of AMPK in brown and beige adipose tissue (BAT): Implications for obesity, insulin resistance, and type 2 diabetes. Curr. Diabetes Rep. 18(10), 1–9 (2018).
Botezelli, J. D. et al. Different exercise protocols improve metabolic syndrome markers, tissue triglycerides content and antioxidant status in rats. Diabetol. Metab. Syndr. 3(1), 1–8 (2011).
Hoseini, R., Damirchi, A. & Babaei, P. The interaction effect of aerobic training and different doses of intramuscular vitamin D on body weight, visceral fat and food intake in female Wistar rats. J. Arak Univ. Med. Sci. 18(7), 24–33 (2015).
Pedersen, B. K. & Saltin, B. Exercise as medicine—Evidence for prescribing exercise as therapy in 26 different chronic diseases. Scand. J. Med. Sci. Sports 25, 1–72 (2015).
Hoffmann, C. et al. Comparison of exercise training effects on mitochondrial substrate oxidation of skeletal muscle and adipose tissue of humans. Diabetologie und Stoffwechsel 14(S 01), 49 (2019).
Cao, S. et al. Effects of exercise on AMPK signaling and downstream components to PI3K in rat with type 2 diabetes. PLoS One 7(12), e51709 (2012).
Takekoshi, K. et al. Long-term exercise stimulates adenosine monophosphate-activated protein kinase activity and subunit expression in rat visceral adipose tissue and liver. Metabolism 55(8), 1122–1128 (2006).
Kang, Y.-S., Kim, J.-C., Kim, J.-S. & Kim, S. H. Effects of swimming exercise on serum irisin and bone FNDC5 in rat models of high-fat diet-induced osteoporosis. J. Sports Sci. Med. 18(4), 596 (2019).
Handschin, C. & Spiegelman, B. M. The role of exercise and PGC1α in inflammation and chronic disease. Nature 454(7203), 463–469 (2008).
Mostafavian, M., Abdi, A., Mehrabani, J. & Barari, A. Effect of eight weeks of aerobic progressive training with capsaicin on changes in PGC-1α and UPC-1 expression in visceral adipose tissue of obese rats with diet. Complement. Med. J. 10(2), 106–117 (2020).
Norheim, F. et al. The effects of acute and chronic exercise on PGC-1α, irisin and browning of subcutaneous adipose tissue in humans. FEBS J. 281(3), 739–749 (2014).
Dimitrov, V., Salehi-Tabar, R., An, B.-S. & White, J. H. Non-classical mechanisms of transcriptional regulation by the vitamin D receptor: Insights into calcium homeostasis, immune system regulation and cancer chemoprevention. J. Steroid Biochem. Mol. Biol. 144, 74–80 (2014).
Heshmat, R. et al. Vitamin D deficiency in Iran: A multi-center study among different urban areas. Iran. J. Public Health 37(1), 72–78 (2008).
Fu, J. et al. Vitamin D levels are associated with metabolic syndrome in adolescents and young adults: The BCAMS study. Clin. Nutr. 38(5), 2161–2167 (2019).
Moaya, M. & Ziaei, S. Effect of tibolone and hormone therapy on body composition and fat distribution in postmenopausal women. J. Arak Univ. Med. Sci. 15(3), 49–57 (2012).
Hoseini, R., Damirchi, A. & Babaei, P. Vitamin D increases PPARγ expression and promotes beneficial effects of physical activity in metabolic syndrome. Nutrition 36, 54–59 (2017).
Cordeiro, M. M. et al. Vitamin D supplementation decreases visceral adiposity and normalizes leptinemia and circulating TNF-α levels in western diet-fed obese rats. Life Sci. 278, 119550 (2021).
Mohammadi, K. & Khajehlandi, A. The effect of a period of swimming training with different temperatures on the expression of β3-AR and ERK2 genes in the visceral adipose tissue of streptozotocin-induced diabetic rats. Feyz J. Kashan Univ. Med. Sci. 25(6), 1303–1312 (2022).
Khalili, A., Nekooeian, A. A. & Khosravi, M. B. Oleuropein improves glucose tolerance and lipid profile in rats with simultaneous renovascular hypertension and type 2 diabetes. J. Asian Nat. Prod. Res. 19(10), 1011–1021 (2017).
Rocha-Rodrigues, S. et al. Effects of physical exercise on myokines expression and brown adipose-like phenotype modulation in rats fed a high-fat diet. Life Sci. 165, 100–108 (2016).
Babaei, P., Shirkouhi, S. G., Hosseini, R. & Soltani Tehrani, B. Vitamin D is associated with metabotropic but not neurotrophic effects of exercise in ovariectomized rats. Diabetol. Metab. Syndr. 9(1), 1–9 (2017).
Nakhaei, H., Mogharnasi, M. & Fanaei, H. Effect of swimming training on levels of asprosin, lipid profile, glucose and insulin resistance in rats with metabolic syndrome. Obes. Med. 15, 100111 (2019).
Botella-Carretero, J. I. et al. Vitamin D deficiency is associated with the metabolic syndrome in morbid obesity. Clin. Nutr. 26(5), 573–580 (2007).
Trowman, R., Dumville, J. C., Hahn, S. & Torgerson, D. J. A systematic review of the effects of calcium supplementation on body weight. Br. J. Nutr. 95(6), 1033–1038 (2006).
Abrams, G. D., Feldman, D. & Safran, M. R. Effects of vitamin D on skeletal muscle and athletic performance. JAAOS J. Am. Acad. Orthop. Surg. 26(8), 278–285 (2018).
Choi, M., Park, H., Cho, S. & Lee, M. Vitamin D3 supplementation modulates inflammatory responses from the muscle damage induced by high-intensity exercise in SD rats. Cytokine 63(1), 27–35 (2013).
Lombardi, G., Ziemann, E., Banfi, G. & Corbetta, S. Physical activity-dependent regulation of parathyroid hormone and calcium-phosphorous metabolism. Int. J. Mol. Sci. 21(15), 5388 (2020).
Girgis, C. M. et al. The vitamin D receptor (VDR) is expressed in skeletal muscle of male mice and modulates 25-hydroxyvitamin D (25OHD) uptake in myofibers. Endocrinology 155(9), 3227–3237 (2014).
Li, L. et al. Mitochondrial biogenesis and PGC-1α deacetylation by chronic treadmill exercise: Differential response in cardiac and skeletal muscle. Basic Res. Cardiol. 106(6), 1221–1234 (2011).
Park, H. et al. Coordinate regulation of malonyl-CoA decarboxylase, sn-glycerol-3-phosphate acyltransferase, and acetyl-CoA carboxylase by AMP-activated protein kinase in rat tissues in response to exercise. J. Biol. Chem. 277(36), 32571–32577 (2002).
Canbolat, E. & Cakıroglu, F. P. The importance of AMPK in obesity and chronic diseases and the relationship of AMPK with nutrition: A literature review. Crit. Rev. Food Sci. Nutr. 63, 449–456 (2023).
Villena, J. A. et al. Induced adiposity and adipocyte hypertrophy in mice lacking the AMP-activated protein kinase-α2 subunit. Diabetes 53(9), 2242–2249 (2004).
Wang, Q. et al. The new role of AMP-activated protein kinase in regulating fat metabolism and energy expenditure in adipose tissue. Biomolecules 11(12), 1757 (2021).
De Matteis, R. et al. Exercise as a new physiological stimulus for brown adipose tissue activity. Nutr. Metab. Cardiovasc. Dis. 23(6), 582–590 (2013).
Brandao, C. F. C. et al. Physical training, UCP1 expression, mitochondrial density, and coupling in adipose tissue from women with obesity. Scand. J. Med. Sci. Sports 29(11), 1699–1706 (2019).
Yau, W. W. & Yen, P. M. Thermogenesis in adipose tissue activated by thyroid hormone. Int. J. Mol. Sci. 21(8), 3020 (2020).
Mattsson, C. L. et al. β1-Adrenergic receptors increase UCP1 in human MADS brown adipocytes and rescue cold-acclimated β3-adrenergic receptor-knockout mice via nonshivering thermogenesis. Am. J. Physiol. Endocrinol. Metab. 301(6), E1108–E1118 (2011).
Li, H. et al. The effect of irisin as a metabolic regulator and its therapeutic potential for obesity. Int. J. Endocrinol. 2021, 6572342 (2021).
Jung, S. & Kim, K. Exercise-induced PGC-1α transcriptional factors in skeletal muscle. Integr. Med. Res. 3(4), 155–160 (2014).
Jorde, R., Sneve, M., Torjesen, P. & Figenschau, Y. No improvement in cardiovascular risk factors in overweight and obese subjects after supplementation with vitamin D3 for 1 year. J. Intern. Med. 267(5), 462–472 (2010).
Sneve, M., Figenschau, Y. & Jorde, R. Supplementation with cholecalciferol does not result in weight reduction in overweight and obese subjects. Eur. J. Endocrinol. 159(6), 675–684 (2008).
Chang, E. Effects of vitamin D supplementation on adipose tissue inflammation and NF-κB/AMPK activation in obese mice fed a high-fat diet. Int. J. Mol. Sci. 23(18), 10915 (2022).
Chang, E. 1, 25-Dihydroxyvitamin D decreases tertiary butyl-hydrogen peroxide-induced oxidative stress and increases AMPK/SIRT1 activation in C2C12 muscle cells. Molecules 24(21), 3903 (2019).
Gauthier, M.-S. et al. Decreased AMP-activated protein kinase activity is associated with increased inflammation in visceral adipose tissue and with whole-body insulin resistance in morbidly obese humans. Biochem. Biophys. Res. Commun. 404(1), 382–387 (2011).
Park, C. Y. & Han, S. N. The role of Vitamin D in adipose tissue biology: Adipocyte differentiation, energy metabolism, and inflammation. J. Lipid Atheroscler. 10(2), 130 (2021).
Szymczak-Pajor, I., Miazek, K., Selmi, A., Balcerczyk, A. & Śliwińska, A. The action of vitamin d in adipose tissue: Is there the link between vitamin D deficiency and adipose tissue-related metabolic disorders?. Int. J. Mol. Sci. 23(2), 956 (2022).
Marcotorchino, J. et al. Vitamin D protects against diet-induced obesity by enhancing fatty acid oxidation. J. Nutr. Biochem. 25(10), 1077–1083 (2014).
Berg, J. P. & Haug, E. Vitamin D: A hormonal regulator of the cAMP signaling pathway. In Biochemistry and Function of Sterols (ed. Vani, M.) 221–229 (CRC Press, 2020).
Manna, P., Achari, A. E. & Jain, S. K. Vitamin D supplementation inhibits oxidative stress and upregulate SIRT1/AMPK/GLUT4 cascade in high glucose-treated 3T3L1 adipocytes and in adipose tissue of high fat diet-fed diabetic mice. Arch. Biochem. Biophys. 615, 22–34 (2017).
Xue, B., Greenberg, A. G., Kraemer, F. B. & Zemel, M. B. Mechanism of intracellular calcium ([Ca2+]i) inhibition of lipolysis in human adipocytes. FASEB J. 15(13), 2527–2529 (2001).
Chang, E. & Kim, Y. Vitamin D decreases adipocyte lipid storage and increases NAD-SIRT1 pathway in 3T3-L1 adipocytes. Nutrition 32(6), 702–708 (2016).
Acknowledgements
The authors would like to thank the participants for their cooperation.
Author information
Authors and Affiliations
Contributions
R.H.: designed the study. K.H.H. and A.G.H.: experimented. R.H.: analyzed the data and wrote the manuscript. K.H.H. and A.G.H.: was involved in the interpretation of data, reviewed and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Khaledi, K., Hoseini, R. & Gharzi, A. Effects of aerobic training and vitamin D supplementation on glycemic indices and adipose tissue gene expression in type 2 diabetic rats. Sci Rep 13, 10218 (2023). https://doi.org/10.1038/s41598-023-37489-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-37489-z
- Springer Nature Limited
This article is cited by
-
Aerobic training with moderate or high doses of vitamin D improve liver enzymes, LXRα and PGC-1α levels in rats with T2DM
Scientific Reports (2024)
-
Impact of Combined Aerobic Training and Magnesium Supplementation on Serum Biomarkers and microRNA-155 and microRNA-21 Expression in Adipose Tissue of Type 2 Diabetic Rats: An Eight-Week Interventional Study
Biological Trace Element Research (2024)
-
Vitamin D3 supplementation could improve the effect of exercise training on type 2 diabetes-induced metabolic disorders via BDNF/irisin axis in elderly women
Sport Sciences for Health (2024)