Abstract
This study aimed to investigate the preventive effect of teriparatide (TPD) administration on medication-related osteonecrosis of the jaw (MRONJ) before tooth extraction due to periodontal lesions in bilaterally ovariectomized female rats treated with zoledronic acid. Thirty skeletally mature Sprague–Dawley rats were randomly divided into three groups: control (CONT, n = 10), zoledronic acid (ZA, n = 10), and zoledronic acid and teriparatide (ZA-TPD, n = 10). The rats were sacrificed 8 weeks after tooth extraction. Micro-computed tomography analysis of the tibia showed that bone mineral density was highest in the CONT, followed by that in the ZA and ZA-TPD groups (CONT/ZA, p = 0.009; CONT/ZA-TPD, p < 0.001; ZA/ZA-TPD, p < 0.001). In the trabecular bone analysis of the extraction site, significant differences in specific bone surface (CONT/ZA, p = 0.010; CONT/ZA-TPD, p = 0.007; ZA/ZA-TPD, p = 0.002) and trabecular thickness (CONT/ZA-TPD, p = 0.002; ZA/ZA-TPD, p = 0.002) were observed. Histological analyses of the extraction sites revealed characteristic MRONJ lesions in the ZA group. Osteonecrosis, inflammatory cells, and sequestrum were less frequently observed in the ZA-TPD group than in the ZA group. In conclusion, TPD administration before tooth extraction helped reduce the occurrence of MRONJ in rats treated with zoledronic acid, confirming its preventative effects.
Similar content being viewed by others
Introduction
Bisphosphonate (BP) is an anti-resorptive agent for the prevention of bone density loss in patients with osteoporosis. Bone tissue helps maintain homeostasis by continuously forming and breaking down bone using osteoblasts and osteoclasts, respectively. BP prevents bone loss by inducing osteoclast apoptosis1. Thus, it is recommended as a first-line treatment for postmenopausal osteoporosis, effective in preventing fractures with long-term use2,3. Zoledronic acid (ZA), used for intravenous therapy in high-risk of fracture groups, and significantly reduces the risk of hip and vertebral fractures4. However, despite the various benefits of BP, MRONJ is known as a potential side effect of anti-resoprtive agents5.
MRONJ is an expanded concept of bisphosphonate-related osteonecrosis of the jaw and diagnosed when certain criteria are present6. Notably, MRONJ is a rare multifactorial disease that involves both inflammation and infection6. Various animal studies have been conducted to understand the pathophysiology of MRONJ7,8. However, most of these studies did not properly reflect the clinical environment by conducting experiments in healthy rats without systemic disease or extracting healthy teeth after BP administration7,8.
Bone necrosis associated with BP occurs only in the jawbone due to its predisposition to bacterial infections9,10. It is known that the main factor triggering MRONJ is invasive treatment such as tooth extraction, but it is argued that bacterial infection, not invasive treatment itself, is the main risk factor 11. Consequently, the importance of preventive treatment, such as removing inflammation before dental treatment, has been emphasized12. Most EXTs are performed to treat inflammatory lesions such as periodontitis6. Osteonecrosis of the jaw (ONJ) has been shown to be aggravated by bacterial infections; however, its incidence decreased with oral hygiene improvement13. This suggests that bacterial infections play an important role in the development of MRONJ.
Treating underlying dental conditions before the use of BP is the key in preventing MRONJ. However, if a patient is already taking BP and requires invasive dental treatment, a withdrawal period may be considered in consultation with their physician. However, according to a recent American Association of Oral and Maxillofacial Surgeons (AAOMS) position paper, drug holidays are controversial6. Although various prevention and treatment methods have been proposed for MRONJ, no gold standard has been established6. Since osteoclast activation is suppressed by BP in MRONJ, TPD treatment, which can stimulate osteoclast activity, is effective in preventing MRONJ14,15. Intermittent TPD can increase new bone formation, bone mass, bone density, and bone quality through anabolic effects16.
Although bacterial infection is a factor in the development of MRONJ, no animal model accurately reflected the clinical environment. Therefore, research is needed to develop an animal model that accurately reflects the clinical environment prior to the development of a standard treatment for MRONJ prevention. In this study, it was expected that TPD would reduce the risk of MRONJ by promoting alveolar bone healing. The null hypothesis of this study was that TPD has no preventive effect on the development of MRONJ. To confirm this hypothesis, the MRONJ rat model in which ZA was administered before tooth extraction was used.
Results
Animals
Evaluations related to animals is described in the Supplementary Appendix.
Proximal tibia: micro-CT
The ZA-TPD group showed the greatest increase in trabecular bone microstructure, followed by the ZA and CONT groups, respectively (Fig. 1A).
Evaluation of the systemic effect of zoledronic acid and teriparatide in the proximal tibia. Teriparatide increases the bone density of the proximal tibia and improves bone quality. (A) Three-dimensional cross-sectional images using micro-CT (lateral view, top view, bottom view). Trabecular bone was the least amount in the CONT group and most amount in the ZA-TPD group. (B) Histological images of epiphyseal cartilage (40, 100, 200, 400 magnification). Primary spongiosa showed a large difference among the groups and gradually thickened in the following order: CONT, ZA, and ZA-TPD. (C) Three-dimensional analysis of trabecular bone using micro-CT. Black double arrow indicates primary spongiosa zone. Asterisk (*) means statistically significant (P < 0.017). CT computed tomography, CONT control, ZA zoledronic acid, ZA-TPD zoledronic acid and teriparatide.
Quantitative analysis showed that BV/TV, BS/BV, BS/TV, Tb.N, and BMD significantly increased in the ZA-TPD group compared with the other groups (p < 0.001) and in the ZA group compared to the CONT group (p = 0.009). Conversely, Tb.Sp significantly decreased in the ZA-TPD group compared with the other groups (p < 0.001) and in the ZA group compared with the CONT group (p = 0.002). Additionally, Tb.Th was highest in the CONT group and decreased in the ZA and ZA-TPD groups, respectively; however, a statistically significant difference was only observed between the CONT and ZA groups (p = 0.006) (Fig. 1C).
Proximal tibia: histology
Differences in the thickness of the growth plates of the proximal tibia were not observed among the groups. However, the proliferative zone of the CONT group could be more clearly observed compared with that of the other groups (Fig. 1B). In contrast, the thickness of the primary spongiosa showed a large difference among the groups, with gradual thickening observed in the following order: CONT, ZA, and ZA-TPD. Additionally, the primary spongiosa was barely observed in the CONT group but remarkably thickened in the ZA-TPD group (Fig. 1B).
EXT socket: micro-CT
In the two-dimensional cross-sectional radiographic image of the EXT socket (Fig. 2A), the EXT socket healed well in the CONT group. In contrast, bone healing was incomplete at the EXT site in the ZA and ZA-TPD groups, and sequestrum was observed in the ZA group. The density of trabecular and cortical bone was higher in the ZA-TPD group than in other groups.
Comparison of changes in extraction site following systemic administration of zoledronic acid and teriparatide. TPD induced healing of the extraction socket. (A) Two-dimensional coronal plane images using micro-CT. TPD induced healing of the extraction socket as much as in the CONT group. (B) Three-dimensional analysis using micro-CT. (C) Bone around extraction sockets on H-E staining (100, 200 magnification). In the ZA group, sequestrum, empty lacunae, and inflammatory cells were observed in all samples, and in the TPD group, extraction socket healing was observed. (D) Epithelium images around extraction socket on H-E staining (40, 100 magnification). (E) Histomorphometric analysis with H-E staining. Black arrowhead indicates inflammatory cell. Black arrow indicates empty lacunae. White arrow indicates reversal line. White arrowhead indicates opened wound. Asterisk (*) means statistically significant (P < 0.017). CT computed tomography, H–E hematoxylin and eosin, CONT control, ZA zoledronic acid, ZA-TPD zoledronic acid and teriparatide.
Three-dimensional microarchitecture analysis of the trabecular bone showed that BV/TV, BS/TV, Tb.Th, and Tb.N were in the least amount in the CONT group and increased the most in the ZA-TPD group (Fig. 2B). Among these, only Tb.Th significantly increased in the ZA-TPD group compared with the other groups (p = 0.002). In contrast, BS/BV and Tb.Sp decreased the most in the ZA-TPD group, followed by the ZA and ZA-TPD groups. Only BS/BV showed statistically significant differences between the groups (CONT/ZA, p = 0.010; CONT/ZA-TPD, p < 0.007; ZA/TPD, p < 0.002).
EXT socket: histology
The EXT socket of the CONT group showed normal healing with no abnormal findings such as bone exposure (Fig. 2C). In the ZA group, bone healing at the extraction site was incomplete, and exposed bones were observed in three specimens (Fig. 2D). Additionally, sequestrum, empty lacunae, and inflammatory cells were observed in all specimens (Fig. 2C). In the ZA-TPD group, necrotic bone was observed in only one specimen, and exposed bone was observed in three specimens. Moreover, inflammatory and sequestered cells and reversal lines were clearly observed in most samples (Fig. 2C).
EXT socket: histomorphometry analysis
Histomorphometric analysis results obtained from the EXT sites of all specimens indicated that the living bone of the ZA group was significantly less than that of the CONT (p = 0.002) and ZA-TPD (p = 0.006) groups, whereas the number of necrotic bones was significantly higher (CONT/ZA, p = 0.002; ZA/ZA-TPD, p = 0.006) (Fig. 2E). Osteocyte density was significantly lower in the ZA group than in the CONT (p = 0.002) and ZA-TPD (p = 0.002) groups, whereas empty lacunae density (CONT/ZA, p < 0.001; ZA/ZA-TPD, p < 0.001) and empty lacunae ratio (CONT/ZA, p < 0.001; ZA/ZA-TPD, p < 0.001) were significantly higher in the ZA group.
qPCR analysis
The total load of oral microbiota collected from the silk was uniform in all groups (Fig. 3A) and have a highly diverse composition (Fig. 3B, C). Proteobacteria and Firmicutes accounted for most of the microorganisms in all groups (Fig. 3B). The composition of Proteobacteria and Bacteroidetes decreased in the following order: CONT, ZA, and ZA-TPD groups. Meanwhile, Firmicutes and Actinobacteria gradually increased in the following order: CONT, ZA, and ZA-TPD groups (Fig. 3B). γ-proteobacteria and bacilli were dominant in all groups (Fig. 3C). However, there were no statistically significant differences between the bacterial population in all groups overall.
Discussion
The null hypothesis of this study was that TPD has no preventive effect on the development of MRONJ. To confirm this hypothesis, the MRONJ rat model in which ZA was administered before tooth extraction was used.
The most likely cause of MRONJ is a mechanism in which dysfunction of osteoclasts and osteoblasts, along with bacterial infection, leads to a decrease in bone remodeling6. In this study, the occurrence of MRONJ increased when EXT was performed after ZA administration in rats with periodontitis; however, short-term administration of TPD before EXT decreased the incidence of MRONJ. Furthermore, systemic bone quality and BMD improved when TPD was administered.
Rodgers et al.17 reported that convenience, relevance, and suitability should be considered when selecting a model suitable for osteoporosis. Rats are widely used as an osteoporosis model because of the ease in handling them and their calcium metabolism similar to humans18. However, since rats often do not have menopause, OVX is used as a means to induce menopause19. They are also known to mature sexually and skeletally at 2.5 and 10 months, respectively20,21. Rodents are highly reproducible when inducing periodontitis and are easier to handle than other animals22,23,24. The periodontitis animal model is classified into a ligation-induced model and pathogenic bacterial infusion model. The ligation-induced model is commonly used because it can reproduce an environment similar to that in human periodontitis25. In the case of a model injected with pathogenic bacteria, periodontitis is induced by directly injecting lipopolysaccharide subgingivally26,27. The bacterial injection model has the disadvantage of low reproducibility of periodontitis and being time-consuming compared to the ligation model. Therefore, we performed OVX at 12 weeks of age, which is the first maturation period, and reproduced MRONJ using a ligation-induced periodontitis model in an osteoporosis-induced rat model28.
Although controversies regarding the treatment method for MRONJ remain, intermittent parathyroid hormone treatment is attracting attention as an adjuvant therapy in addition to surgical procedures29,30. TPD is a recombinant protein of parathyroid hormone, an agent that acts directly on bone tissue and is an 84-amino-acid chain. TPD consists of 34 amino-terminals and has high sequence conservation in rats and humans; hence, the experiment was conducted in rats31. Moreover, because mice and humans show similar calcium metabolism18, they are most often used as animal models for bone metabolic disease studies16,32. In this study, radiological and histological evaluations were performed on the proximal tibia to evaluate the systemic effect of the drug. Results showed that ZA and TPD significantly increased bone quality (Fig. 1–2). TPD (20–200 μg/kg) is used in various ways for fracture treatment, MRONJ management, and bone transplantation16,33,34. Moreover, it was most effective clinically and histologically for MRONJ treatment when administered at 20 μg/kg34. The systemic effect of TPD was confirmed in the proximal tibia as bone quality improved both radiographically and histologically (Fig. 1). Histological observations showed that TPD administration activated the development of the primary spongiosa zone below the growth plate, suggesting that the mineralization process in the cartilage is active and may further promote ossification activity. Furthermore, the administration of TPD 2 weeks before EXT is effective in preventing ONJ 35. Similar to the results of these study, when only ZA was administered at the EXT site, necrotic bone increased in an MRONJ-like state; however, when TPD was administered before EXT, the amount of necrotic bone was at the same level as that in the CONT group (Fig. 2).
The proximal tibia was used to observe the systemic effects of ZA and TPD using micro-CT analysis (Fig. 1A). When only ZA was administered, BV/TV, Tb.N, and BMD increased. When TPD was additionally administered, BV/TV, Tb.N, and BMD increased significantly (Fig. 1C). Both BS/BV and BS/TV increased in the experimental group; however, Tb.Sp significantly decreased (Fig. 1C). These results indicate that the systemic administration of ZA and TPD improved osteoporotic status in osteoporosis-induced rats, which is consistent with the results of previous studies34,35. The appropriate dose and usage of ZA and TPD was then determined. However, in the mandible, BS/BV was highest in the CONT group and lowest in the ZA-TPD group (Fig. 2B). Conversely, Tb.Th was highest in the ZA-TPD group, and a statistically significant difference was found only when compared with the ZA-TPD group (Fig. 2B). This means that the bone quality of the ZA-TPD group is the best. In previous animal experiments, there was a large difference in the analytical method of each experiment, such as the nonperformance of micro-CT analysis in the EXT socket34,35 or different ROI locations within the EXT socket36,37. This may be due to the difficulty of setting ROIs, as various anatomical structures are located around the experimental site, and that histological findings are more meaningful than radiological findings for the evaluation of MRONJ. These problems are may have affected the micro-CT results of the mandible in our study.
Histological evaluation of MRONJ has been conducted in many studies and may be considered as a basic step in understanding the mechanism of MRONJ. In general, histologically evaluating the state of bone healing, epithelial integrity, inflammation, necrotic bone, osteoblasts, osteoclasts, and blood vessels are used as standard indicators38. In our study, inflammation, necrotic bone, sequestrum, and exposed bone, which are the most important features for MRONJ development, were evaluated. In some studies, inflammatory cells were divided into four stages and classified39,40. In our study, only the presence/absence of inflammation was evaluated without classification. Inflammation was observed in 100% of the ZA group, 80% of the ZA-TPD group, and 40% of the CONT group (Fig. 2C). In our experiment, root rest was confirmed in two cases for CONT, one case for ZA, and two cases for TPD. The observed inflammation of 40% in CONT is thought to be influenced by root rest. Necrotic bone is the most representative feature of MRONJ; however, the diagnostic criteria for necrotic bone are very diverse. In our study, it was defined as a devital bone area with 10 or more adjacent empty lacunae41. The amount of necrotic bone was significantly higher in the ZA group, and the ZA-TPD group has an amount similar with that in the CONT group (Fig. 2E). Sequestrum was observed in all specimens in the ZA group, but only in seven specimens in the ZA-TPD group. Meanwhile, bone exposure was confirmed in two and three specimens in the ZA and ZA-TPD groups, respectively, and no difference was observed between the two groups. Based on these histological findings, it is speculated that during the process of inducing MRONJ by ZA, TPD induces osteoblast activity and promotes bone modeling to prevent MRONJ occurrence.
Studies have been conducted on various factors known to increase the risk of MRONJ, such as the type of drug used, method of administration, duration of administration, and dental diseases7,8. Clinically, more severe periodontitis or periapical diseases are treated with EXT, which increases the risk of MRONJ. MRONJ is a disease caused by various factors, including anti-resorptive medication, inflammation, and infection6. Therefore, in this study, MRONJ was induced by extraction after inducing periodontitis through tooth ligation. Most animal models of periodontitis are made by ligating silk into the tooth42. In some studies, animal models of periodontitis have been made by injecting lipopolysaccharide7 or bacterial pathogens42 into the gingival fissure.
According to Masa et al.43, the total amount of bacterial DNA collected from periodontal lesions of IL-17 KO mice, which plays an important role in host defense against bacterial infection, was significantly increased compared to that in wild-type mice. We created an animal model in which periodontitis was induced using silk22, and oral bacteria that accumulated around the silk caused inflammation, leading to alveolar bone destruction. To confirm that periodontitis was induced, oral microbiota from the collected silk was analyzed. Equal amounts of microbiota were detected in all groups (Fig. 3A), and there was no statistical difference in the number and distribution of Bacteroides and Firmicutes associated with periodontitis. Moreover, Proteobacteria and Actinobacteria, which are often observed in healthy environments without inflammatory findings, also showed no statistical differences in the quantity and distribution rate detected. A previous study reported a significant increase in the distribution of γ-proteobacteria in KO mice43. In our study, in addition to Bacteroidia, Clostridia, Bacilli, and Sphingobacteriia, which are associated with periodontitis, we also found oral bacterial, such as α-proteobacteria, β-proteobacteria, γ-proteobacteria, Actinobacteria_c, and coriobacteriia, which are mainly observed in healthy environments. These populations did not differ between groups, suggesting that periodontitis was well-induced in all groups, and that MRONJ was difficult to develop with only periodontitis.
Conclusion
On the other hand, a limitation of our study is that we did not set up a control group in which periodontitis was not induced. Additional studies are needed to evaluate the contribution of the oral microbiota to the occurrence and prevention of MRONJ in a periodontitis-free environment and determine the effective dose and duration of TPD to confirm its preventive effect on MRONJ. In conclusion, TPD administration before EXT is effective in preventing MRONJ.
Methods
Animals
Animal selection, management, surgical protocol followed the protocols approved by the Institutional Animal Care and Use Committee of Yonsei Medical Center (IACUC No. 2018-0210) and conformed to the ARRIVE 2.0 guidelines. More details are described in the Appendix. All the methods in this study were performed in accordance with relevant guidelines and regulation.
Experimental design
The rats were randomly divided into three groups: control (CONT, n = 10), zoledronic acid (ZA, n = 10), and zoledronic acid and teriparatide (ZA-TPD, n = 10). Sample size was determined with an effect size of 0.9 and power of 0.7. A total sample size of 26 was calculated. Finally, a total of 30 samples was employed, 10 per group, considering a dropout rate of 15%. The effect size was calculated by referring to data of a previous study34. Bilateral ovariectomy (OVX) was performed to induce osteoporosis.
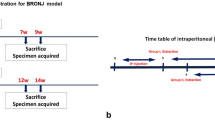
Body weight was measured weekly before OVX until the end of the experiment. The rats were subjected to bilateral OVX to induce osteoporosis. Eight weeks later, periodontal lesions (mandibular 2nd molar) were induced via silk ligation.
Four weeks after the ligation, the ZA and ZA-TPD groups were administered with ZA (Zometa; Novartis, Basel, Switzerland) through weekly intravenous injection for 4 weeks (40 μg/kg × day). The ZA-TPD group was then administered with TPD (Forsteo; Eli Lilly, Houten, The Netherlands) via subcutaneous injection every day for 4 weeks (20 μg/kg × day). During the same period, the CONT and ZA groups (sham-ZA and sham-TPD) received injections of the same volume and method of saline. Ten weeks after the ligation, the ligated teeth were extracted. The rats were sacrificed 8 weeks after EXT (Supplementary Fig. S1). More details are described in the Supplementary Appendix.
Micro-computed tomography(micro-CT)
Micro-CT (skyscan1173; SkyScan, Kontich, Belgium) was performed on the tibia and mandible (7.1, 12.07 µm), which were also analyzed using CTAn software (CTAn; SkyScan, Kontich, Belgium) to evaluate the systemic effects of the drug. The region of interest (ROI) was decided at 0.7 to 3.5 mm below the growth plate to analyze the trabecular bone microstructure of the tibia. The grayscale was set to 30–255 (Supplementary Fig. S2A). The ROIs for the evaluation of the trabecular bone morphometry were set in a cylindrical shape with Ø 0.6 mm and 0.5 mm length, and the grayscale was set to 50–255 (Supplementary Fig. S2B).
The bone volume fraction (BV/TV, %), specific bone surface (BS/BV, mm2/mm3), bone surface density (BS/TV, mm2/mm3), trabecular thickness (Tb.Th, mm), trabecular number (Tb.N, 1/mm), trabecular separation (Tb.Sp, mm), and bone mineral density (BMD, g/cm2) were used to evaluate the trabecular bone microarchitecture.
Histology
The specimens were demineralized with 10% ethylene-diamine-tetraacetic acid for 4 weeks. After paraffin embedding, the blocks were stained with hematoxylin and eosin. The stained tissue was observed under an optical microscope (OLYMPUS BX43; Olympus Corporation, Tokyo, Japan). Living bone (%, the bone area with morphologically normal osteocytes), necrotic bone (%, the devital bone area in which there were ≥ 10 pyknotic osteocyte lacunae), osteocyte density (#/mm2, number of osteocytes), empty lacunae density (#/mm2, number of empty osteocyte lacunae), and empty lacunae ratio (number of empty lacunae per total number of osteocyte lacunae) were quantified in the mandible specimens using 200× magnified images from above the EXT site.
Quantitative polymerase chain reaction (qPCR) analysis: oral microbiota
Silk ligation was maintained for 10 weeks. Bacterial DNA was extracted using a kit (FastDNA Spin Kit; MP Biomedicals, Solon, OH, USA). The primer sequences used were: F341 ‘CCTACGGGNGGCWGCAG’ and R805 ‘GACTACHVGGGTATCTAATCC’.
Statistical analysis
Statistical analyses were performed using IBM SPSS Statistics for Windows (Version 25; IBM, Armonk, NY, USA). After the Kruskal–Wallis test, the three groups were compared using the post-hoc Mann–Whitney U test. Statistical significance was set at p < 0.01744.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Weinstein, R. S., Roberson, P. K. & Manolagas, S. C. Giant osteoclast formation and long-term oral bisphosphonate therapy. N. Engl. J. Med. 360, 53–62. https://doi.org/10.1056/NEJMoa0802633 (2009).
Eriksen, E. F., Diez-Perez, A. & Boonen, S. Update on long-term treatment with bisphosphonates for postmenopausal osteoporosis: A systematic review. Bone 58, 126–135. https://doi.org/10.1016/j.bone.2013.09.023 (2014).
Compston, J. et al. Diagnosis and management of osteoporosis in postmenopausal women and older men in the UK: National Osteoporosis Guideline Group (NOGG) update 2013. Maturitas 75, 392–396. https://doi.org/10.1016/j.maturitas.2013.05.013 (2013).
Serrano, A. J., Begona, L., Anitua, E., Cobos, R. & Orive, G. Systematic review and meta-analysis of the efficacy and safety of alendronate and zoledronate for the treatment of postmenopausal osteoporosis. Gynecol. Endocrinol. 29, 1005–1014. https://doi.org/10.3109/09513590.2013.813468 (2013).
Nicolatou-Galitis, O. et al. Medication-related osteonecrosis of the jaw: Definition and best practice for prevention, diagnosis, and treatment. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 127, 117–135. https://doi.org/10.1016/j.oooo.2018.09.008 (2019).
Ruggiero, S. L. et al. American Association of Oral and Maxillofacial Surgeons’ position paper on medication-related osteonecrosis of the jaws-2022 update. J. Oral Maxillofac. Surg. 80, 920–943. https://doi.org/10.1016/j.joms.2022.02.008 (2022).
Tanaka, J. et al. An osteonecrosis model induced by oral bisphosphonate in ovariectomised rats. Oral Dis. 21, 969–976. https://doi.org/10.1111/odi.12368 (2015).
Soares, M. Q. S. et al. Zoledronic acid induces site-specific structural changes and decreases vascular area in the alveolar bone. J. Oral Maxillofac. Surg. 76, 1893–1901. https://doi.org/10.1016/j.joms.2018.03.007 (2018).
Boff, R. C., Salum, F. G., Figueiredo, M. A. & Cherubini, K. Important aspects regarding the role of microorganisms in bisphosphonate-related osteonecrosis of the jaws. Arch. Oral Biol. 59, 790–799. https://doi.org/10.1016/j.archoralbio.2014.05.002 (2014).
Ewald, F. et al. Retrospective analysis of bacterial colonization of necrotic bone and antibiotic resistance in 98 patients with medication-related osteonecrosis of the jaw (MRONJ). Clin. Oral Investig. 25, 2801–2809. https://doi.org/10.1007/s00784-020-03595-9 (2021).
Aguirre, J. I. et al. Oncologic doses of zoledronic acid induce osteonecrosis of the jaw-like lesions in rice rats (Oryzomys palustris) with periodontitis. J. Bone Miner. Res. 27, 2130–2143. https://doi.org/10.1002/jbmr.1669 (2012).
Kim, T. et al. Removal of pre-existing periodontal inflammatory condition before tooth extraction ameliorates medication-related osteonecrosis of the jaw-like lesion in mice. Am. J. Pathol. 188, 2318–2327. https://doi.org/10.1016/j.ajpath.2018.06.019 (2018).
Ripamonti, C. I. et al. Decreased occurrence of osteonecrosis of the jaw after implementation of dental preventive measures in solid tumour patients with bone metastases treated with bisphosphonates. The experience of the National Cancer Institute of Milan. Ann. Oncol. 20, 137–145. https://doi.org/10.1093/annonc/mdn526 (2009).
Lombard, T., Neirinckx, V., Rogister, B., Gilon, Y. & Wislet, S. Medication-related osteonecrosis of the jaw: New insights into molecular mechanisms and cellular therapeutic approaches. Stem Cells Int. 2016, 8768162. https://doi.org/10.1155/2016/8768162 (2016).
Cheung, A. & Seeman, E. Teriparatide therapy for alendronate-associated osteonecrosis of the jaw. N. Engl. J. Med. 363, 2473–2474. https://doi.org/10.1056/NEJMc1002684 (2010).
Park, K. M. et al. Synergistic effect of hyperbaric oxygen therapy with PTH [1-34] on calvarial bone graft in irradiated rat. Oral Dis. 25, 822–830. https://doi.org/10.1111/odi.13037 (2019).
Rodgers, J. B., Monier-Faugere, M. C. & Malluche, H. Animal models for the study of bone loss after cessation of ovarian function. Bone 14, 369–377. https://doi.org/10.1016/8756-3282(93)90166-8 (1993).
Jung, J. et al. Effect and timing of parathyroid hormone analog administration for preventing medication-related osteonecrosis of the jaws in a murine model. J. Craniomaxillofac. Surg. 49, 719–725. https://doi.org/10.1016/j.jcms.2021.02.023 (2021).
Turner, A. S. Animal models of osteoporosis—Necessity and limitations. Eur. Cell Mater. 1, 66–81. https://doi.org/10.22203/ecm.v001a08 (2001).
Jee, W. S. & Yao, W. Overview: Animal models of osteopenia and osteoporosis. J. Musculoskelet. Neuronal Interact. 1, 193–207 (2001).
Liu, X. L., Li, C. L., Lu, W. W., Cai, W. X. & Zheng, L. W. Skeletal site-specific response to ovariectomy in a rat model: Change in bone density and microarchitecture. Clin. Oral Implants Res. 26, 392–398. https://doi.org/10.1111/clr.12360 (2015).
Abe, T. & Hajishengallis, G. Optimization of the ligature-induced periodontitis model in mice. J. Immunol. Methods 394, 49–54. https://doi.org/10.1016/j.jim.2013.05.002 (2013).
Li, C. H. & Amar, S. Morphometric, histomorphometric, and microcomputed tomographic analysis of periodontal inflammatory lesions in a murine model. J. Periodontol. 78, 1120–1128. https://doi.org/10.1902/jop.2007.060320 (2007).
Graves, D. T., Fine, D., Teng, Y. T., Van Dyke, T. E. & Hajishengallis, G. The use of rodent models to investigate host-bacteria interactions related to periodontal diseases. J. Clin. Periodontol. 35, 89–105. https://doi.org/10.1111/j.1600-051X.2007.01172.x (2008).
Gyorfi, A., Fazekas, A., Suba, Z., Ender, F. & Rosivall, L. Neurogenic component in ligature-induced periodontitis in the rat. J. Clin. Periodontol. 21, 601–605. https://doi.org/10.1111/j.1600-051x.1994.tb00750.x (1994).
Ishida, N. et al. Periodontitis induced by bacterial infection exacerbates features of Alzheimer’s disease in transgenic mice. NPJ Aging Mech. Dis. 3, 15. https://doi.org/10.1038/s41514-017-0015-x (2017).
Liu, R., Desta, T., Raptis, M., Darveau, R. P. & Graves, D. T. P. gingivalis and E. coli lipopolysaccharides exhibit different systemic but similar local induction of inflammatory markers. J. Periodontol. 79, 1241–1247. https://doi.org/10.1902/jop.2008.070575 (2008).
Yousefzadeh, N., Kashfi, K., Jeddi, S. & Ghasemi, A. Ovariectomized rat model of osteoporosis: A practical guide. EXCLI J. 19, 89–107. https://doi.org/10.17179/excli2019-1990 (2020).
Doh, R. M. et al. Teriparatide therapy for bisphosphonate-related osteonecrosis of the jaw associated with dental implants. Implant Dent. 24, 222–226. https://doi.org/10.1097/ID.0000000000000232 (2015).
Jung, J. et al. Short-term teriparatide and recombinant human bone morphogenetic protein-2 for regenerative approach to medication-related osteonecrosis of the jaw: A preliminary study. J. Bone Miner. Res. 32, 2445–2452. https://doi.org/10.1002/jbmr.3237 (2017).
Martin, T. J. Parathyroid hormone-related protein, its regulation of cartilage and bone development, and role in treating bone diseases. Physiol. Rev. 96, 831–871. https://doi.org/10.1152/physrev.00031.2015 (2016).
Dam, C., Jung, U. W., Park, K. M., Huh, J. & Park, W. Effect of teriparatide on early sinus graft healing in the ovariectomized rabbit. Clin. Oral Implants Res. 31, 264–273. https://doi.org/10.1111/clr.13565 (2020).
Hoffmann, D. B. et al. Comparison of parathyroid hormone and strontium ranelate in combination with whole-body vibration in a rat model of osteoporosis. J. Bone Miner. Metab. 35, 31–39. https://doi.org/10.1007/s00774-016-0736-0 (2017).
Zandi, M., Dehghan, A., Zandipoor, N., Amini, P. & Doulati, S. Effect of different doses and durations of teriparatide therapy on resolution of medication-related osteonecrosis of the jaw: A randomized, controlled preclinical study in rats. J. Craniomaxillofac. Surg. 46, 466–472. https://doi.org/10.1016/j.jcms.2017.12.027 (2018).
Zandi, M., Dehghan, A., Mohammadi-Mofrad, A., Amini, P. & Vahdatinia, F. Short-term perioperative teriparatide therapy for the prevention of medication-related osteonecrosis of the jaw: A randomized, controlled preclinical study in rats. J. Craniomaxillofac. Surg. 45, 275–280. https://doi.org/10.1016/j.jcms.2016.12.010 (2017).
Yu, W. & Su, J. The effects of different doses of teriparatide on bisphosphonate-related osteonecrosis of the jaw in mice. Oral Dis. 26, 609–620. https://doi.org/10.1111/odi.13275 (2020).
Kim, J. Y., Jang, H. W., Kim, J. I. & Cha, I. H. Effects of pre-extraction intermittent PTH administration on extraction socket healing in bisphosphonate administered ovariectomized rats. Sci. Rep. 11, 54. https://doi.org/10.1038/s41598-020-79787-w (2021).
Yan, R. et al. Establishment and assessment of rodent models of medication-related osteonecrosis of the jaw (MRONJ). Int. J. Oral Sci. 14, 41. https://doi.org/10.1038/s41368-022-00182-4 (2022).
Rodriguez-Lozano, F. J. et al. Allogeneic bone marrow mesenchymal stem cell transplantation in tooth extractions sites ameliorates the incidence of osteonecrotic jaw-like lesions in zoledronic acid-treated rats. J. Clin. Med. https://doi.org/10.3390/jcm9061649 (2020).
Yadegari, A., Aminzadeh, A., Seyyedkhamesi, S. & Aminian, M. The effect of melatonin on prevention of bisphosphonate-related osteonecrosis of the jaw: An animal study in rats. J. Korean Assoc. Oral Maxillofac. Surg. 46, 266–274. https://doi.org/10.5125/jkaoms.2020.46.4.266 (2020).
Hayano, H. et al. Distinct immunopathology in the early stages between different antiresorptives-related osteonecrosis of the jaw-like lesions in mice. Bone 135, 115308. https://doi.org/10.1016/j.bone.2020.115308 (2020).
Li, C. L., Seneviratne, C. J., Huo, L., Lu, W. W. & Zheng, L. W. Impact of Actinomyces naeslundii on bisphosphonate-related osteonecrosis of the jaws in ovariectomized rats with periodontitis. J. Craniomaxillofac. Surg. 43, 1662–1669. https://doi.org/10.1016/j.jcms.2015.07.001 (2015).
Tsukasaki, M. et al. Host defense against oral microbiota by bone-damaging T cells. Nat. Commun. 9, 701. https://doi.org/10.1038/s41467-018-03147-6 (2018).
Bonferroni, C. Teoria statistica delle classi e calcolo delle probabilita. Pubblic. R Istit. Super. Sci. Econ. Commer. Firenze 8, 3–62 (1936).
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (Grant nos. 2017R1D1A1B03033657 and 2021R1A6A3A01087956).
Author information
Authors and Affiliations
Contributions
K.M.P., contributed to conception, design, data acquisition, analysis and interpretation, drafted and critically revised the manuscript, Funder; N.L., contributed to conception, design, data analysis, and interpretation, drafted and critically revised the manuscript; H.S.K., contributed to data analysis, critically revised the manuscript; J.K., contributed to data interpretation, critically revised the manuscript; W.P., contributed to conception, design and data analysis, critically revised the manuscript, supervised the manuscript, Funder. All authors gave their final approval and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Park, KM., Lee, N., Kim, J. et al. Preventive effect of teriparatide on medication-related osteonecrosis of the jaw in rats. Sci Rep 13, 15518 (2023). https://doi.org/10.1038/s41598-023-42607-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-42607-y
- Springer Nature Limited