Abstract
Axial spondyloarthritis (axSpA) increases the risk of osteoporosis and vertebral fractures. Bone mineral density (BMD) measured by dual X-ray absorptiometry (DXA) has limitations in axSpA patients. Trabecular bone score (TBS) indirectly assesses bone microarchitecture and can be used to predict fracture risk. However, few studies have investigated the role of TBS in axSpA patients. The objective of this study were to compare TBS between axSpA patients and 1:1 sex- and age-matched healthy volunteers and determine factors associated with low TBS in axSpA patients. A cross-sectional study was conducted in two tertiary-care hospitals. A total of 137 axSpA patients and healthy volunteers were enrolled. Demographics, disease characteristics, and risk factors for osteoporosis were recorded. TBS, BMD at the lumbar spine, hip, and vertebral fractures were assessed by DXA. Low TBS was defined as a TBS value < 1.230. Factors associated with low TBS were examined by logistic regression. Most patients were male (75.9%) and tested positive for HLA-B27 (88.3%). The mean (SD) age was 42.8 (12.0) years. The mean (SD) of TBS in the axSpA patients was lower than those in the healthy volunteers [1.402 (0.107) vs 1.440 (0.086), respectively; p = 0.002]. The mean (SD) of lumbar BMD in the axSpA patients was higher than in healthy volunteers [1.186 (0.212) vs 1.087 (0.124), p < 0.001], whereas the mean (SD) of femoral neck BMD in the axSpA group was lower than that in the healthy volunteers [0.867 (0.136) vs 0.904 (0.155), p = 0.038]. Disease severity as indicated by sacroiliac joint fusion and a high ASDAS score were associated with low TBS with the odds ratios (95% confidence interval) of 11.8 (1.2–115.4) and 5.2 (1.6–16.9), respectively. In conclusion, axSpA patients had a higher prevalence of low TBS than healthy volunteers. Sacroiliac joint fusion and a high ASDAS score were associated with low TBS.
Similar content being viewed by others
Introduction
Axial spondyloarthritis (axSpA) is a chronic inflammatory disease that primarily affects the axial joints (spine and sacroiliac (SI) joints), as well as peripheral joints and entheses. Extraspinal manifestations such as peripheral arthritis, anterior uveitis, psoriasis, colitis, and rare manifestations such as pulmonary fibrosis and amyloidosis are also affected by axSpA1. Although the pathogenesis is unknown, several hypotheses have been proposed, including the arthritogenic peptide, misfolding, molecular mimicry, and cell-surface HLA-B27 homodimer. As a result, a variety of cytokines are produced, which play a role in both the inflammatory process and the formation of new bone. During the inflammatory process, those cytokines, namely interleukin-1 (IL-1), IL-6, IL-17/23, and tumor necrosis factor –α (TNF-α), stimulate the production of receptor activator of nuclear factor kappa-B ligand, which is a potential stimulator of osteoclast differentiation, resulting in bone loss in axSpA patients2,3. Meanwhile, bone morphogenetic protein and Wnt signaling pathways induce osteoblast differentiation to contribute to new bone formation, leading to ankylosis and disability3,4.
Osteoporosis is one of the most common comorbidities in axSpA patients5. Paradoxically, new bone formation occurs resulting in ankylosis of the spine and SI joints. The prevalence of osteoporosis in axSpA patients were reported between 11.7 and 34.4%6,7. Several studies have found that long-standing forms, spinal ankylosis, high inflammatory markers, alcohol consumption, corticosteroid use, and vitamin D deficiency were risk factors for low bone mineral density (BMD) in axSpA patients5,6. Importantly, patients with axSpA are at a higher risk of vertebral fractures (VFs) than the general population8. Furthermore, elevated C-Reactive Protein (CRP), disease duration, and hyperkyphosis have also been linked to an increased risk of VFs5.
The dual-energy X-ray absorptiometry (DXA) is the gold standard tool to assess bone mineral density (BMD). BMD is calculated by measuring bone mineral content divided by the area of interest. Every one standard deviation (SD) different from a mean healthy young adult is reported as T-score. According to the WHO criteria, osteoporosis is defined as a BMD T-score of −2.5 or less measured by DXA at the spine, hip, or forearm9. Furthermore, DXA-BMD at the femoral neck is used in the fracture risk assessment tool FRAX which has been implemented in several guidelines. The 2019 update of the American College of Rheumatology (ACR)/Spondylitis Association of America/Spondyloarthritis Research and Treatment Network Recommendations for the Treatment of Ankylosing Spondylitis and Non-radiographic axSpA suggested that axSpA patients be screened for osteoporosis with DXA10. However, the BMD from DXA may inadvertently increase in axSpA patients due to syndesmophyte formation11.
In addition to BMD measurement, DXA can assess other factors that are helpful in determining the risk of developing fragility fracture risk, such as bone geometry by hip structural analysis and hip axis length12, vertebral fracture assessment (VFA), bone strength by bone strain index (BSI)13, and bone microarchitecture by trabecular bone score (TBS).
The TBS evaluates the subtle variations in pixel grey level within the lumbar DXA image, thereby providing an indirect measurement of bone microarchitecture as trabecular number, trabecular separation, and connectivity density without additional radiation exposure to the patients. A high TBS value is indicative of a strong microarchitecture that exhibits resistance against fractures, whereas a low TBS value implies a fragile microarchitecture that is prone to fractures14. Several studies have reported that TBS was associated with fracture risk in postmenopausal women, particularly VFs. TBS, unlike DXA, is less likely to be affected by syndesmophytes. Therefore, TBS may be valuable in fracture risk assessment in axSpA patients14,15.
The objective of this study was to compare TBS in Thai axSpA patients with age- and sex-matched normal individuals and examine the factors associated with low TBS in axSpA patients.
Methods
Setting and subjects
A cross-sectional study was conducted at Phramongkutklao and Khon Kaen University Hospitals (tertiary care settings) between January 2020 and February 2021. Patients aged 18 years and older who fulfilled the ASAS classification criteria for axSpA were consecutively recruited16. Patients with a history of cancer or a first-time cancer diagnosis (regardless of stage), chronic kidney disease at least stage IV17, hyperthyroidism, hyperparathyroidism, pregnancy, and/or lactation were excluded. Each patient’s healthy volunteer participants were randomly selected from the general Thai population and matched for age and sex with a ratio of 1:1. The Royal Thai Army Medical Department Institutional Board Review and the Khon Kaen Hospital Institutional Review Board in Human Research approved the study with the approved study numbers R178h/62 and HE631141, respectively and confirmed that this research complied with the Declaration of Helsinki. The written informed consent was obtained from all participants and/or their legal guardians prior to entry to the study.
Bone mineral density and trabecular bone score measurements
BMD was measured at the lumbar spine (LS), femoral neck (FN), and total hip (TH) using GE-Lunar iDXA (#210754) and GE-Lunar DPX Duo densitometer (GE Healthcare, Madison, WI, USA). Osteoporosis was defined as a BMD T-score of -2.5 or less in postmenopausal women and men aged ≥ 50 years. Whereas, a Z-score of -2.0 or less was defined as being below the expected range for age (low BMD) in premenopausal patients and men aged < 50 years18. The TBS was obtained from the DXA scan (Medimaps TBS iNsight) at the lumbar spine in both settings. The TBS cut-offs used to categorise TBS groups are obtained from the metanalyses of individual-level data covering 17,809 men and women in 14 prospective population-based cohorts around the world. These groups are categorised as normal TBS, partially degraded (or intermediate TBS), and degraded (or low TBS) when TBS values are above > 1.310, between 1.230 and 1.310, and below < 1.230, respectively19. Fractured vertebrae were excluded from lumbar DXA analysis.
Radiography and vertebral fracture assessment
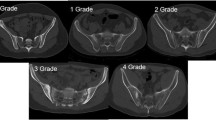
The vertebral fracture assessment (VFA) by DXA and/or the lateral thoracolumbar X-rays were used to determine the presence of VFs. VFs were defined using Genant’s semiquantitative method20 and reviewed by a certified nuclear radiologist. The spinal deformity index (SDI) was calculated by summing the score from 13 vertebrae (T4 to L4). For each vertebra, a visual semiquantitative score of 0, 1, 2, or 3 was assigned for no fracture or mild, moderate, or severe fracture, respectively21. In this study, the SI joints were assessed by radiograph. The modified New York criteria were used to classify sacroiliitis22.
Measurements in axial spondyloarthritis patients
Demographic and clinical data including age, gender, body mass index (BMI), history of alcohol and smoking, duration of disease (defined as the time since axSpA diagnosis), family history of axSpA, current medications [i.e., non-steroidal anti-inflammatory drugs (NSAIDs), proton pump inhibitors (PPIs), sulfasalazine, anti-TNF, anti-IL-17, and anti-osteoporotic agents (anti-OP)] were recorded.
In the present study, patient global assessment (PGA), the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) in Thai version23, the Ankylosing Spondylitis Disease Activity Score [ASDAS-CRP or ASDAS-ESR if C-reactive protein (CRP) was not available], the Bath Ankylosing Spondylitis Functional Index (BASFI) in Thai version23, and the Bath Ankylosing Spondylitis Metrology Index (BASMI) were used to assess disease activity24.
Statistical analysis
Statistical analysis included computing the percentage frequencies for categorical variables and means (standard deviation, SD) or medians (interquartile ranges, IQR) for continuous variables. Comparisons of categorical variables were made using the chi-squared or Fisher’s exact test, as appropriate. Continuous variables were tested for normality using a Shapiro–Wilk test and were compared using Student’s t-test and one-way ANOVA or the Mann–Whitney U test and the Kruskal–Wallis test to compare two and three groups’ means, respectively. Medians with IQR were used for non-normally distributed variables. Univariate and multiple logistic regression analyses were used to determine the relevant risk factors for low TBS. Only risk factors that had univariate associations of P-values < 0.20 were further considered in subsequent multivariate models. The lack of collinearity was confirmed by testing variance inflation factors. P-values < 0.05 were considered statistically significant. All statistical analyses were performed using SPSS software (IBM SPSS Statistics for Windows, Version 23.0. Armonk, NY: IBM Corp.).
Ethics approval and consent to participate
The ethical approval was approved by the Institutional Review Board of the Royal Thai Army Medical Department and the Human Research Ethics Committee of Khon Kaen University. The reference numbers are R178h/62 and HE631141, respectively. This research complied with the Declaration of Helsinki. The written informed consent was obtained from all participants and/or their legal guardians prior to entry to the study.
Results
As shown in Table 1, the study population recruited 137 axSpA patients (115 from Phramongkutklao Hospital and 22 from Khon Kaen University Hospital) and 137 matched healthy volunteers. The majority of participants were male (75.9%), and 34.3% of the women were post-menopausal. The mean (SD) of age and mean duration of disease were 42.8 (12.0) years and 86.8 (96.9) months, respectively. HLA-B27 was found in 88.3%, whereas the family history of axSpA was found in only 13.1%. Patients with axSpA had concomitant anterior uveitis and psoriasis (5.8% and 1.5%, respectively). In axSpA patients, alcohol consumption who used greater than or equal to 3 units/day was found in 19.7%, whereas patients with smoking including ex-smoker were found in 35.8%.
The mean LS-BMD was significantly higher in axSpA patients compared with the healthy volunteers. In contrast, the mean FN-BMD was significantly lower in axSpA patients than in the healthy volunteers. There was no significant difference in TH-BMD between the two groups. There were 13 patients (9.6%) within the axSpA group who met the criteria for osteoporosis or bone mass below the expected range for age. The prevalence of low BMD/osteoporosis at TH was significantly higher in axSpA patients than that in the healthy volunteers, but it was not significantly different between axSpA patients and the healthy volunteers at LS and FN (Table 1). The mean (SD) TBS was significantly lower in patients with axSpA compared to healthy volunteers, with values of 1.402 (0.107) and 1.444 (0.107), respectively (p = 0.002), and exhibiting a statistical power of 94.7%. The computed effect size was 0.391. The prevalence of degraded bone/low TBS in the axSpA patients was also significantly higher than that in the healthy volunteers (7.35% vs 1.46%, p = 0.010). Due to the limited number of patients in the low TBS and osteoporosis group, the clinical significance of these findings might be unclear, thus warranting a cautious interpretation.
As depicted in Table 2, axSpA patients with degraded bone/low TBS group had higher CRP levels, disease activity scores (including PGA, BASDAI, BASFI, BASMI, and ASDAS), and prevalence of grade 4 sacroiliitis and current NSAIDs use than those in normal and high TBS groups. In addition, axSpA patients with degraded bone/low TBS group had a significantly shorter disease duration when compared with other groups. We found that patients with axSpA who had never received anti-TNF or anti-OP medication had lower TBS than patients who had received those medications. (p = 0.037 and p = 0.028, respectively). Furthermore, axSpA patients with degraded bone/low TBS group had significantly lower BMD at LS, FN, and TH than those with intermediate/ normal TBS groups. The radiographic VFs were found in 10 patients (7.5%, 10/134). The prevalence of VFs was significantly higher in patients with degraded bone/low TBS group as compared with patients with intermediate and normal TBS groups (30.0% vs 6.67% vs 5.5%, p = 0.019, respectively).
Factors associated with the trabecular bone score
In the univariate linear regression, ex- or current smoker, alcohol intake ≥ 3 units/day, BMI, current NSAIDs use, all parameters of disease activity, presence of syndesmophyte, and grade 4 sacroiliitis were associated with low TBS. In multivariate linear regression analysis, TBS was negatively associated with female gender, ex- or current smoker, alcohol intake ≥ 3 units/day, ASDAS, and grade 4 sacroiliitis, whereas BMI was positively associated with TBS.
The univariate logistic regression model showed that higher disease severity measurements including PGA, ESR, hsCRP, BASDAI, BASMI, BASFI, ASDAS, grade 4 sacroiliitis, and the presence of syndesmophyte, alcohol intake ≥ 3 units/day, shorter disease duration, and PPI uses were significantly associated with degraded bone/low TBS. The ASDAS and grade 4 sacroiliitis were respectively selected to represent current disease severity and damage in multivariate models. In multivariate logistic regression, ASDAS, and grade 4 sacroiliitis were independently associated with degraded bone/low TBS with the adjusted odds ratios (confidence interval: CI) of 5.228 (1.611–16.963) and 11.820 (1.211–115.412), respectively. Short disease duration was also found to be associated with degraded bone/low TBS; although the association was modest with an adjusted odds ratio (CI) of 0.982 (0.966–0.998), (Table 3).
VFs were found in ten patients in this study (7.5%). The prevalence of VFs was significantly higher in the degraded bone/low TBS group than in the intermediate and normal TBS groups (30.0% vs 6.7% vs 5.5%, p = 0.019, respectively), (Fig. 1). For identifying patients with VFs, the sensitivity (95%CI) and specificity (95%CI) of degraded bone/low TBS were 30% (6.7% to 65.3%) and 94.4% (88.7% to 97.7%), respectively, whereas the sensitivity (95%CI) and specificity (95%CI) of osteoporosis/low bone mass at LS-BMD and FN-BMD were 10.0% (0.25%-44.5%) and 94.4% (88.8%-97.7%) and 16.67% (0.42%-64.12%) and 92.56% (86.35%-96.54%), respectively.
The prevalence of vertebral fractures (two cases were not included in which the radiologist could not interpret vertebral fractures due to artifacts) (VFs) stratified by low bone mineral density (BMD) or osteoporosis (OP) at the lumbar spine (LS), total hip (TH), femoral neck (FN), and trabecular bone score (one patient, whose TBS data could not be analysed due to morbid obesity, was excluded) (TBS) risks in patients with axial spondyloarthritis (axSpA). *p < 0.05, compared with normal TBS group.
Combining the results from TBS and BMD, axSpA patients who had both degraded bone/low TBS and osteoporosis/low bone mass at the femoral neck or total hip were at the highest risk of having VFs as compared to patients with normal TBS and FN-BMD group with the OR (95%CI) of 15.8 (0.88–285.48), p = 0.061 for both femoral and total hip site. In contrast, axSpA patients that had normal BMD and degraded bone/low TBS at the lumbar spine had an increased risk of VFs as compared with those with normal BMD and normal TBS at the same site with the OR (95%CI) of 11.3 (1.58–81.23), p = 0.016), (Fig. 2).
The prevalence of vertebral fractures, stratified by trabecular bone score (TBS) and osteoporosis/low bone mineral density (BMD) according to DXA. Asterisk: statistically significant when compared with patients with ax-SpA with normal BMD and normal TBS. The values displayed in the center of each bar represent the odds ratios for vertebral fractures when compared to individuals with both normal TBS and normal BMD at the lumbar spine, total hip, and femoral neck in patients with axSpA.
Discussion
In this study, patients with axSpA had significantly lower TBS values than sex-age-matched healthy volunteers. The prevalence of degraded bone/low TBS was higher in axSpA patients when compared with healthy volunteers. VFs were more common in patients in the degraded bone/low TBS group than in the intermediate and normal groups. Female gender, smoking status, alcohol intake greater than or equal to 3 units/day, shorter disease duration, elevated hs-CRP, higher disease activities, current NSAIDs use, and not receiving anti-TNF or anti-OP drugs were all significantly associated with lower TBS in patients with axSpA.
The findings from this study were consistent with previous studies25,26,27,28. High disease activity related to inflammation and new bone formation, impaired function, and spinal mobility as assessed by BASFI and BASMI were found to be associated with low TBS in axSpA patients. These findings were supported by an MRI study of the spine, which revealed that local inflammation, known as bone marrow edema, was detected in trabecular bone regions29 as well as the presence of new bone formation on plain films, such as fusion of sacroiliac joints and syndesmophytes, were associated with low TBS in axSpA patients30,31,32.
A meta-analysis of seven longitudinal studies and one randomised controlled trial reported that anti-TNF treatment increased LS-BMD in AS patients by 5.1% and 8.6% after 1 and 2 years, respectively33. Another prospective study in 12 AS patients found that, despite a significant increase in LS-BMD, there was no significant improvement in TBS after 2 years of anti-TNF treatment34. This study found that axSpA patients who did not receive anti-TNF treatment had lower TBS than those who did and that low TBS was associated with high disease activity. These findings emphasized the significance of controlling disease activity with anti-TNF agents or other agents, as they could improve bone quality (TBS) and bone mass (LS-BMD) in axSpA patients.
In this study, alcohol and smoking were associated with degraded bone/low TBS in axSpA patients. Several studies in postmenopausal women found that alcohol consumption and smoking were strongly associated with low BMD33,35; although, these associations had not previously been reported in axSpA patients27,28. Furthermore, those risk factors have been linked to increased radiographic progression in patients with axSpA36,37. As a result, physicians should encourage axSpA patients to discontinue the modified risk factors.
This study found that shorter disease duration was independently associated with degraded bone/low TBS; although, the association was modest. This finding supported the Danish Health Registries’ findings that patients with AS had an increased risk of clinical vertebral and non-vertebral fractures, with the highest risk occurring within the first 2.5 years after diagnosis38. In addition, previous studies demonstrated that a decrease in LS-BMD was associated with active inflammatory lesions at the lumbar spine and sacroiliac joints in early AS patients39,40, and the 2-year follow-up of the DESIR cohort also showed that the prevalence of low BMD at the lumbar spine was 22.4% in patients with early axSpA41. Patients with a briefer disease duration may experience more severe symptoms and disease progression, leading them to seek medical attention at an earlier stage. The appropriate bone health assessment should be performed as part of an initial treatment plan in axSpA patients.
AxSpA patients in the degraded/low TBS groups had a higher prevalence of VFs and a higher risk of developing new VFs when compared to those in the normal or intermediate TBS groups27,28,42,43,44. Degraded bone/low TBS had higher sensitivity than FN-BMD and LS-BMD for detecting VFs in axSpA patients without losing specificity. Although the 2019 ACR recommendations suggested patients with axSpA should be screened for osteoporosis with a DXA scan of the spine and hip10, the results of previous studies together with findings from this study suggested TBS provided additional benefit for vertebral fracture assessment than LS-BMD and FN-BMD alone since vertebral fractures in axSpA patients from this study occurred at a normal BMD.
The clinical application of TBS for fracture risk assessment in patients with axSpA is not well established. TBS could be used in two ways to assess fracture risk in axSpA patients. First, degraded bone/low TBS level could be a tool for fracture risk assessment in AS patients. The cut-off for degraded bone/low TBS used in this study was derived from a meta-analysis of 14 prospective population-based cohorts from countries around the world examining TBS in fracture risk prediction19. However, it is unclear whether this threshold could be used in axSpA patients of any ethnicity since ethnicity affects the fracture-predictive ability of TBS45. Secondly, the value of TBS could be incorporated in FRAX (fracture assessment tool) along with clinical risk factors and FN-BMD. Further research into the clinical use of FRAX and TBS is needed to determine the appropriate adjustment to assess fracture risk in axSpA patients.
There were some limitations in this study. First, this study did not examine some other risk factors for osteoporosis such as vitamin D level, serum calcium, and iPTH. Second, there were limited numbers of patients with low TBS and VFs and only a few patients had other major osteoporotic fractures. Third, as a cross-sectional study, this study data might not represent overall disease activities in patients with axSpA, although the results showed that all parameters of disease activity were consistent with the severity of the disease. Furthermore, only a few patients were menopause or had already received anti-osteoporotic treatment; although, after excluding post-menopausal status or anti-osteoporotic treatment, the results were indifferent.
Conclusion
Patients with axSpA have lower TBS than healthy volunteers. The proportion of axSpA patients with degraded bone/low TBS was higher than the proportion of healthy volunteers. TBS was found to be independently associated with gender, former or current smoker, ≥ 3 units/day alcohol consumption, low BMI, high disease activity including PGA, BASDAI, BASFI, BASMI, and ASDAS, and fusion of sacroiliac joints. Therefore, TBS is a good non-invasive tool for assessing fracture risk in axSpA patients. TBS can be used alone or in conjunction with BMD measurement to identify axSpA patients at high risk for vertebral fractures.
Data availability
The data that support the findings of this study are available at reasonable request from the corresponding author.
Abbreviations
- axSpA:
-
Axial spondyloarthritis
- SI:
-
Sacroiliac
- IL:
-
Interleukin
- TNF-α:
-
Tumor necrosis factor-α
- BMD:
-
Bone mineral density
- VFs:
-
Vertebral fractures
- CRP:
-
C-reactive protein
- DXA:
-
Dual-energy X-ray absorptiometry
- ACR:
-
The American College of Rheumatology
- LS:
-
Lumbar spine
- FN:
-
Femoral neck
- TH:
-
Total hip
- VFA:
-
Vertebral fracture assessment
- BMI:
-
Body mass index
- NSAIDs:
-
Non-steroidal anti-inflammation drugs
- PPIs:
-
Proton pump inhibitors
- PGA:
-
Patient global assessment
- BASDAI:
-
The Bath Ankylosing Spondylitis Disease Activity Index
- ASDAS:
-
The Ankylosing Spondylitis Disease Activity Score
- BASFI:
-
The Bath Ankylosing Spondylitis Functional Index
- BASMI:
-
The Bath Ankylosing Spondylitis Metrology Index
- CI:
-
Confidence interval
References
Deesomchok, U. & Tumrasvin, T. Clinical study of Thai patients with ankylosing spondylitis. Clin. Rheumatol. 4(1), 76–82 (1985).
Amarasekara, D. S., Yu, J. & Rho, J. Bone loss triggered by the cytokine network in inflammatory autoimmune diseases. J. Immunol. Res. 2015, 832127 (2015).
Ronneberger, M. & Schett, G. Pathophysiology of spondyloarthritis. Curr. Rheumatol. Rep. 13(5), 416–420 (2011).
Zhu, W. et al. Ankylosing spondylitis: Etiology, pathogenesis, and treatments. Bone Res. 7(1), 22 (2019).
Moltó, A. & Nikiphorou, E. Comorbidities in spondyloarthritis. Front. Med. (Lausanne). 5, 62 (2018).
Ramírez, J., Nieto-González, J. C., Curbelo Rodríguez, R., Castañeda, S. & Carmona, L. Prevalence and risk factors for osteoporosis and fractures in axial spondyloarthritis: A systematic review and meta-analysis. Semin. Arthritis Rheum. 48(1), 44–52 (2018).
Danve, A. & Raychaudhuri, S. P. Comorbidities in spondyloarthritis. Curr. Treat. Opt. Rheumatol. 3(1), 63–74 (2017).
Weiss, R. J., Wick, M. C., Ackermann, P. W. & Montgomery, S. M. Increased fracture risk in patients with rheumatic disorders and other inflammatory diseases—A case–control study with 53,108 patients with fracture. J. Rheumatol. 37(11), 2247–2250 (2010).
World Health Organization Collaborating Centre for Metabolic Bone Diseases, University of Sheffield, UK. Assessment of Osteoporosis at the Primary Health Care Level [Internet]. https://www.sheffield.ac.uk/FRAX/pdfs/WHO_Technical_Report.pdf (2007).
Ward, M. M. et al. 2019 update of the American College of Rheumatology/Spondylitis Association of America/Spondyloarthritis research and treatment network recommendations for the treatment of ankylosing spondylitis and nonradiographic axial spondyloarthritis. Arthritis Care Res. 71(10), 1285–1299 (2019).
Syed, Z. & Khan, A. Bone densitometry: Applications and limitations. J. Obstet. Gynaecol. Can. 24(6), 476–484 (2002).
Beck, T. J. & Broy, S. B. Measurement of hip geometry-technical background. J. Clin. Densitom. 18(3), 331–337 (2015).
Ulivieri, F. M. & Rinaudo, L. The bone strain index: An innovative dual X-ray absorptiometry bone strength index and its helpfulness in clinical medicine. J. Clin. Med. 11(9), 2284 (2022).
Pothuaud, L. et al. Evaluation of the potential use of trabecular bone score to complement bone mineral density in the diagnosis of osteoporosis: A preliminary spine BMD-matched, case–control study. J. Clin. Densitom. 12(2), 170–176 (2009).
Rabier, B., Heraud, A., Grand-Lenoir, C., Winzenrieth, R. & Hans, D. A multicentre, retrospective case-control study assessing the role of trabecular bone score (TBS) in menopausal Caucasian women with low areal bone mineral density (BMDa): Analysing the odds of vertebral fracture. Bone. 46(1), 176–181 (2010).
Rudwaleit, M. et al. The development of Assessment of Spondyloarthritis International Society Classification criteria for axial spondyloarthritis (part I): Classification of paper patients by expert opinion including uncertainty appraisal. Ann. Rheum. Dis. 68(6), 770–776 (2009).
Levey, A. S. et al. National Kidney Foundation practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Ann. Intern. Med. 139(2), 137–147 (2003).
Shuhart, C.R., Yeap, S.S., Anderson, P.A., Jankowski, L.G., Lewiecki, E.M., Morse, L.R. et al. Executive summary of the 2019 ISCD position development conference on monitoring treatment, DXA cross-calibration and least significant change, spinal cord injury, peri-prosthetic and orthopedic bone health, transgender medicine, and pediatrics. J. Clin. Densitom. 22(4), 453–471 (2019).
McCloskey, E. V. et al. A meta-analysis of trabecular bone score in fracture risk prediction and its relationship to FRAX. J. Bone Miner. Res. 31(5), 940–948 (2016).
Genant, H. K., Wu, C. Y., van Kuijk, C. & Nevitt, M. C. Vertebral fracture assessment using a semiquantitative technique. J. Bone Miner. Res. 8(9), 1137–1148 (1993).
Minne, H. W. et al. A newly developed spine deformity index (SDI) to quantitate vertebral crush fractures in patients with osteoporosis. Bone Miner. 3(4), 335–349 (1988).
van der Linden, S., Valkenburg, H. A. & Cats, A. Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum. 27(4), 361–368 (1984).
Kittiyanpanya, C., Chaiamnuay, S., Asavatanabodee, P. & Narongroeknawin, P. Reliability and validity of the Thai version of bath ankylosing spondylitis indices. J. Med. Assoc. Thailand (Chotmaihet thangphaet) 97(4), 381–385 (2014).
Jones, S. D. et al. A new scoring system for the Bath Ankylosing Spondylitis Metrology Index (BASMI). J. Rheumatol. 22(8), 1609 (1995).
Kang, K. Y., Ju, J. H., Park, S.-H. & Hong, Y. S. Longitudinal association between trabecular bone loss and disease activity in axial spondyloarthritis: A 4-year prospective study. J. Rheumatol. 47(9), 1330–1337 (2020).
Boussoualim, K. et al. Evaluation of bone quality with trabecular bone score in active spondyloarthritis. Jt. Bone Spine 85(6), 727–731 (2018).
Kang, K.Y., Goo, H.Y., Park, S.-H. & Hong, Y.S. Trabecular bone score as an assessment tool to identify the risk of osteoporosis in axial spondyloarthritis: A case–control study. Rheumatology 57(3), 462–469 (2017).
Adel, Y., El Adalany, M.A. & Bassiouni, S.A.R.A.K. Performance of bone mineral density and trabecular bone score in assessment of bone quality in Egyptian male patients with ankylosing spondylitis. Egypt. Rheumatol. Rehabil. 47(1), 35 (2020).
Navarro-Compán, V. et al. In patients with axial spondyloarthritis, inflammation on MRI of the spine is longitudinally related to disease activity only in men: 2 years of the axial spondyloarthritis DESIR cohort. Ann. Rheum. Dis. 77(3), 470–472 (2018).
Jung, J.-Y. et al. Inflammation on spinal magnetic resonance imaging is associated with poor bone quality in patients with ankylosing spondylitis. Mod. Rheumatol. 29(5), 829–835 (2019).
Kang, K. Y. et al. Trabecular bone score value is associated with new bone formation independently of fat metaplasia on spinal magnetic resonance imaging in patients with ankylosing spondylitis. Scand. J. Rheumatol. 49(4), 292–300 (2020).
Kang, K. Y. et al. Severity of sacroiliitis and erythrocyte sedimentation rate are associated with a low trabecular bone score in young male patients with ankylosing spondylitis. J. Rheumatol. 45(3), 349–356 (2018).
Berg, K. M. et al. Association between alcohol consumption and both osteoporotic fracture and bone density. Am. J. Med. 121(5), 406–418 (2008).
Toussirot, E. et al. Paradoxical elevation of serum TRACP5b levels despite increase in lumbar spine bone mineral density during anti-TNFalpha therapy in patients with inflammatory rheumatic disease: A 2-year prospective assessment of bone mass, bone metabolism, and the trabecular bone score. Eur. J. Rheumatol. 4(3), 189–193 (2017).
Ward, K. D. & Klesges, R. C. A meta-analysis of the effects of cigarette smoking on bone mineral density. Calcif. Tissue Int. 68(5), 259–270 (2001).
Min, H. K., Lee, J., Ju, J. H., Park, S.-H. & Kwok, S.-K. Alcohol consumption as a predictor of the progression of spinal structural damage in axial spondyloarthritis: Data from the Catholic Axial Spondyloarthritis COhort (CASCO). Arthritis Res. Ther. 21(1), 187 (2019).
Poddubnyy, D. et al. Baseline radiographic damage, elevated acute-phase reactant levels, and cigarette smoking status predict spinal radiographic progression in early axial spondylarthritis. Arthritis Rheum. 64(5), 1388–1398 (2012).
Prieto-Alhambra, D. et al. Ankylosing spondylitis confers substantially increased risk of clinical spine fractures: A nationwide case–control study. Osteoporos. Int. 26(1), 85–91 (2015).
Gubar, E. et al. AB0689 association between MRI inflammation and bone loss in early axial spondyloarthritis. Ann. Rheum. Dis. 73(Suppl 2), 1032–1033 (2014).
Briot, K. et al. Bone oedema on MRI is highly associated with low bone mineral density in patients with early inflammatory back pain: Results from the DESIR cohort. Ann. Rheum. Dis. 72(12), 1914–1919 (2013).
Briot, K., Etcheto, A., Miceli-Richard, C., Dougados, M. & Roux, C. Bone loss in patients with early inflammatory back pain suggestive of spondyloarthritis: Results from the prospective DESIR cohort. Rheumatology 55(2), 335–342 (2015).
Orenes Vera, A. V. et al. AB0711 usefulness of the trabecular bone score as a predictor of vertebral fracture in patients with axial spondyloarthropathy. Ann. Rheum. Dis. 79(Suppl 1), 1650–1651 (2020).
Richards, C., Hans, D. & Leslie, W. D. Trabecular bone score (TBS) predicts fracture in ankylosing spondylitis: The Manitoba BMD Registry. J. Clin. Densitom. 23(4), 543–548 (2020).
Caparbo, V. F. et al. Assessing bone impairment in ankylosing spondylitis (AS) using the trabecular bone score (TBS) and high-resolution peripheral quantitative computed tomography (HR-pQCT). Bone 122, 8–13 (2019).
Jain, R. K., Narang, D. K., Hans, D. & Vokes, T. J. Ethnic differences in trabecular bone score. J. Clin. Densitom. 20(2), 172–179 (2017).
Acknowledgements
The authors would like to express our deepest appreciation to Phramongkutklao Hospital and College of Medicine and Faculty of Medicine, Khon Kaen University for funding and supporting this study. We appreciated Ms. Supak Ukritchon, who works at the Office of Research and Development, Phramongkutklao College of Medicine, for assisting in data analyses. We also thank Ms. Wajee Matkummee, who works at the Division of Nuclear Medicine, Department of Radiology, Phramongkutklao Hospital, for giving us practical suggestions. Finally, we would like to thank all participants for joining this study.
Funding
This work was supported by Phramongkutklao Hospital and College of Medicine and Faculty of Medicine, Khon Kaen University (funding number IN63324).
Author information
Authors and Affiliations
Contributions
P.S. and S.C. were involved in the conception of the study, performed the analyses, drafted the manuscript, and revised the manuscript. P.S. and A.M. collected data from axSpA patients from Phramongkutklao and Khon Kaen University hospitals, respectively. C.P. provided data from healthy volunteers and revised the manuscript. The other authors assisted in the interpretation of the results from the analyses and drafting of the manuscript. All authors have read and approved the final manuscript and ensured that this is the case. All authors consent for the publication and grant the publisher exclusive license of the full copyright.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Saisirivechakun, P., Mahakkanukrauh, A., Pongchaiyakul, C. et al. Prevalence of low trabecular bone score and its association with disease severity and activity in patients with axial spondyloarthritis. Sci Rep 13, 16258 (2023). https://doi.org/10.1038/s41598-023-43321-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-43321-5
- Springer Nature Limited
This article is cited by
-
Surgical treatment of distal radius fractures: impact on forearm rotation in non-elderly patients
Scientific Reports (2024)