Abstract
Therapeutic exercise exerts positive effects by mitigating or reducing the motor or cognitive changes that people with Down syndrome undergo throughout their life. There are no updated systematic reviews that integrate the evidence available in a way that facilitates decision-making for physical rehabilitation teams. This study therefore aimed to consolidate the information available and compare the effects of different types of physical exercise on the motor function of adults with DS. We conducted a systematic review and meta-analysis of randomized clinical trials and quasi-experimental studies. The literature search was performed between January 2023 and February 2023 using the PubMed, SCIELO, Epistemonikos, and Lilacs databases. Studies were selected according to pre-determined inclusion and exclusion criteria. The risk-of-bias assessment was performed using the risk-of-bias rating tool for randomized clinical trial (RoB) and the risk of bias of non-randomized comparative studies was assessed using the risk of bias in non-randomized studies of interventions (ROBINS-I) tool. Risk-of-bias assessment and meta-analyses were performed using the RevMan software package. Sixteen studies met the eligibility criteria for the qualitative synthesis and 4 were included in the meta-analyses. Combined exercise significantly increased muscle strength both in the upper limbs (SMD = 0.74 [95% CI 0.25–1.22]) and lower limbs (SMD = 0.56[95% CI 0.08–1.04]). Aerobic exercise improved spatiotemporal gait parameters. Aerobic exercise showed significant improvements in dynamic balance while combined exercise significantly increased dynamic and static balance. The certainty of the evidence was low to moderate for all outcomes. There was low and moderate certainty of evidence for the outcomes proposed in this review. However, therapeutic exercise could be effective in improving muscle strength and gait functionality.
Similar content being viewed by others
Introduction
Down syndrome (DS) is a medical condition caused by a genetic abnormality where chromosome 21 is either partially or completely duplicated1. It is the most common and prevalent genetic neurological disorder associated with intellectual disability and motor disorders characterized by hypotonia, ligamentous laxity, and limited muscle strength2, in addition to other cardiorespiratory, gastrointestinal, and immunological comorbidities3.
The motor changes in balance, strength, resistance, and mobility2 caused by DS have a direct effect on motor function, defined as the ability or capacity to learn, maintain, coordinate, and assume voluntary control of postures and movement patterns4. Therefore, DS affects quality of life and the ability to perform activities of daily living (ADL), thus increasing the dependency on other individuals and the adoption of a sedentary lifestyle in many cases5.
This is the reason why DS individuals require therapeutic interventions, especially rehabilitation, to improve their motor skills. Therapeutic exercise is included within these interventions, which the World Health Organization defines as “a variation of physical activity, aimed at reaching a pre-established goal, which is generally the improvement or maintenance of physical fitness or of the health condition”6. Exercise is characterized as being planned and repetitive which means that it is performed regularly6 and is divided into different types: aerobic exercise, where large muscle groups are exercised improving cardiovascular capacity; strength exercises; flexibility or stretching exercises; and neuromuscular exercises, including proprioception7,8,9,10, balance, and agility exercises11.
Scientific evidence of the effects of exercise in DS individuals is extensive in this regard. Multiple interventions have been identified in the literature that evaluate the effectiveness of exercise in water1,2, progressive resistance exercise12,13, continuous aerobic exercise5,14,15,16, specific modalities such as Nordic walking17, and combined exercise which is simultaneous aerobic and resistance training18,19,20 on different motor function outcomes, such as aerobic and functional capacity2,5, dynamic balance, muscular strength, endurance1,12,14,18,19,20, and gait17.
However, the many types of therapeutic exercises available in the literature as well as the many motor function outcomes in which the effectiveness of these types of exercise is measured impede rehabilitation teams’ decision-making when attempting to identify the type of exercise that, according to its prescription, is the most effective in improving these motor function outcomes.
Despite having a substantial amount of evidence on the effect of exercise in adult DS individuals, there is no accumulation of this evidence that accounts for the effect of different types of therapeutic exercise, mode of application, and in general, the prescription parameters of effective interventions. The integration of available evidence will facilitate decision-making for physical rehabilitation teams. This study therefore aimed to consolidate the information available and compare the effects of different types of physical exercise on the motor function of adult DS individuals.
Methods
This review was conducted in accordance with Cochrane Handbook of Systematic Reviews of Interventions21 and the recommendations of the methodology proposed in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines22. This analysis was prospectively registered on Open Science Framework (OSF) and it is available in https://doi.org/10.17605/OSF.IO/MRKN2. Ethical and internal review board approval was not required because no human or animal subjects were involved.
Study selection criteria
Participants
DS individuals aged 18 years or older. Studies that included the population in this review as well as populations with other characteristics, and studies whose results for DS individuals were not presented separately, were excluded.
Interventions
Application of any type of therapeutic exercise—either strength or resistance, aerobic, or neuromuscular exercise—with specific prescription parameters such as intensity, duration, and frequency.
Comparison
Control group with no exercise intervention, or another type of exercise intervention with another prescription parameters.
Outcome measures
Studies that did not include at least one of the outcomes proposed for this review were excluded. The outcomes prioritized in this review were as follows:
-
Primary: Strength, defined as the ability of a muscle group to develop contraction against resistance23; balance or equilibrium, understood as the ability to maintain the body’s stability on each side of the axis24; and gait variables, defined as bipedal walking used to move from one place to another with minimal effort and energy consumption25.
-
Secondary: Coordination, defined as the ability to execute and control movements24; posture, defined as the alignment of body segments during movement or a sustained situation25; and functional tasks, such as climbing stairs.
Designs
Experimental studies such as randomized or quasi-experimental clinical trials were included.
Search methods for identification of studies
The literature search was carried out in the PubMed, Epistemonikos, SCIELO and Lilacs databases. The following algorithms were used to search for articles in English:
DATABASE | ALGORITHM |
|---|---|
PubMed | ((((((((((((((Down syndrome) OR Trisomy 21)) AND Adult)) AND (((((((Exercise) OR Physiotherapy) OR Physical) OR Training) OR Neuromuscular exercise))) AND (((((((((Motor function) OR Functionality) OR Balance) OR Posture) OR Coordination) OR Gait) OR Strength)) NOT Physiology)) |
Epistemonikos | (((((((((basic physical abilities) OR (Speed)) OR (Resistance)) OR (Strength)) OR (Skill)) OR (Flexibility)) OR (Coordination)) OR (Balance)) OR (Agility)) AND ((((((((((therapeutic exercise) OR (physiotherapy)) OR (physical therapy)) OR (exercise)) OR (Endurance Training)) OR (Motion Therapy)) OR (Muscle Stretching Exercises)) OR (Plyometric Exercise)) OR (Resistance Training)) AND (((((Syndrome, Down) OR (Mongolism)) OR (Trisomy G)) OR (Down's Syndrome)) OR (Trisomy 21))) |
The search was conducted between January 2023 to February 2023 and no filters by language or publication date were applied.
Other sources
Additionally, other sources of evidence were consulted to allow for the identification and analysis of published or unpublished literature (gray literature) that had not been detected through the systematic search. These other sources included manual searches in the reference list of the systematic reviews found through the search in the databases. Another search was performed through the L·OVE platform (Living Overview of Evidence) in the Down Syndrome section26 which let us review and compare the studies in the Epistemonikos database where an evidence matrix was built to, automatically, list the systematic reviews that share at least one study included as well as all the studies included in each of these reviews27.
Study selection
This was performed by two reviewers independently applying the selection criteria (MMM, YVC). Duplicate studies were initially merged into a bibliographic reference manager, followed by screening through the review of titles and abstracts to identify studies that included the population of interest for the present review, therapeutic exercise intervention, and at least one of the motor function outcomes. Subsequently, the full texts of the selected studies were retrieved, and after a comprehensive reading, studies were excluded based on their design, the population included, or because they did not include at least one of the motor function outcomes. A third reviewer (ERG) intervened to define whether or not studies for which there was no agreement should be included in this review.
Data extraction and management
This was conducted by two reviewers independently (MMM, YVC) in an Excel file. The following items were extracted: year of publication and authors; title; characteristics of participants such as age, sex, and number of participants per group; characteristics of the interventions applied, such as the type and mode of exercise, with their prescription of intensity, duration, and frequency; outcomes evaluated with their respective measuring instruments; and the results obtained by variable and group.
Risk-of-bias assessment
This was performed using the risk-of-bias (RoB) tool for randomized clinical trials21 based on seven domains, namely, sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessors, incomplete outcome data, selective outcome reporting, and “other aspects.” Each of these domains was assigned a rating of “low risk,” “high risk,” or “unclear risk.” The risk of bias of non-randomized comparative studies was assessed using the risk of bias in non-randomized studies of interventions (ROBINS-I) tool5.
The response options for each domain-level judgement are: (1) Low risk of bias; (2) Moderate risk of bias; (3) Serious risk of bias; (4) Critical risk of bias; and (5) No information on which to base a judgement about risk of bias for this domain. Finally, the option response for an overall risk of bias judgement using ROBINS-I are: (1) Low risk of bias (the study is comparable to a well-performed randomized trial); (2) Moderate risk of bias (the study provides sound evidence for a non-randomized study but cannot be considered comparable to a well-performed randomized trial); (3) Serious risk of bias (the study has some important problems); (4) Critical risk of bias (the study is too problematic to provide any useful evidence and should not be included in any synthesis); and (5) No information on which to base a judgement about risk of bias5. Risk of bias assessment figures were developed in RevMan 5.428.
Synthesis of data
The selected body of evidence was assessed according to the prioritized outcomes. Each outcome described the population’s features; parameters of the interventions, including the exercise mode applied, frequency, intensity, and duration of the interventions applied in the said studies; and the quantitative results achieved with their level of significance. The data were synthesized on a Microsoft Excel base, extracting data from the population’s features, randomization methods, outcome measures, duration of follow-up, and assessment methods from each study. The meta-analysis considered direct comparisons between the experimental group that performed the interventions (aerobic exercise and resistance exercise) and a control group that performed educational activities, recreational activities, or continuity with ADL or exercise interventions with different parameters or the comparison between different types of exercise.
Averages and standard deviations of the data available from the selected studies were extracted from the prioritized outcomes included in the studies. When the studies reported standard errors of the mean, the standard deviations were obtained by multiplying standard errors of the mean by the square root of the sample size. Standardized mean differences (SMD) and 95% confidence intervals (95% CI) were calculated to combine the results of the studies using different measures for the same concept or of studies presenting variability in its features.
Heterogeneity between trials was assessed using the chi-squared test, with a p value of < 0.05 considered statistically significant after due consideration of the value of I2 Heterogeneity was reported as low (I2 = 0%–50%) or high (I2 > 50%)29,30. The results were combined using the random effects model and the 95% CI was calculated. All data analysis were performed using the RevMan 5 software28.
Assessment of the certainty of evidence
This was performed using the Grading of Recommendations, Assessment, Development and Evaluations (GRADE) system21 for each outcome. This system specifies four levels of quality evidence: “High,” “Moderate,” “Low,” and “Very Low.” The level is determined by considering the risk of bias in the study, inconsistency, direction of evidence, precision of an effect estimate, and other considerations that include publication bias, whether or not the effect is large, the existence of confounding factors, and the dose–response gradient. These variables, except for “other considerations,” were evaluated on a three-level scale: “not serious,” “serious,” and “very serious.” For “other considerations,” the publication bias scale was classified as “not detected” or “strong suspicion”; large effect was graded as “no,” “large,” or “very large”; confounders were graded as “no,” “will reduce the demonstrated effect,” or “suggests a spurious effect”; and the dose–response gradient was classified as “yes” or “no.”.
Results
Search and selection of studies
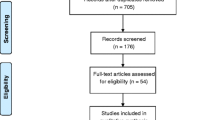
The electronic search yielded 898 studies, and 190 studies were obtained from other sources to yield a total of 1088. Of these, 188 studies were excluded owing to duplication and 798 were excluded after the review by titles and abstracts. In total, 102 studies were assessed in full text, of which 86 were excluded because they did not meet the eligibility criteria, mainly the study design, and because they did not include at least one of the outcomes prioritized in this review. Thus, only 16 studies met the eligibility criteria for the qualitative synthesis and 4 were included in the meta-analyses. This information is presented in a flowchart following the PRISMA model (Fig. 1).
General characteristics of the studies
Table 1 shows that the studies selected for this review included a total of 402 participants aged 18–65 years. Of the 16 studies, 15 included the population aged 21–50 years1,2,5,12,13,17,19,20,31,32,33,34,35,36,37, with only 414,32,34,36 including the population aged 60 years and older, and included the population aged 18–2012,13,33,35,36,37 years. The study with the smallest sample was the one by Post et al.35, with 11 participants, while that with the largest sample was the study by Rimmer et al.20, with 52 participants.
Risk-of-bias assessment
All included studies had some risk of bias. Three studies presented an unclear risk because there was randomization, but the method was not described17,20,32. Related to blinding only one study20 had high risk bias because participants were notified of the group they belonged to with no mention of personnel blinding. Six studies1,17,20,32,33,36 had unclear bias in blinding of outcome assess. Just one study20 had unclear risk of bias of incomplete outcome data and only two studies14,17 obtained a high risk of bias in selective outcome reporting. In other aspects assess, one study12 presented a high risk of bias due to possible confounding bias. Figures 2 and 3 summarize the information on bias risk domains.
The methodological quality of the seven selected quasi-experimental studies2,13,19,31,34,35,37 was moderate to serious Table 2.
Therapeutic exercise interventions used for each outcome
Muscular strength
This outcome was the most frequently reported within the studies included. The interventions addressed aerobic exercise in water1,2, continuous and interval aerobic exercise5, aerobic exercise14,32,33, progressive resistance exercise12,13, resistance exercise35,37, and combined exercise19,20,33,34. The duration of the interventions was between 62 and 25 weeks14 with frequencies ranging from two12,13,32 to three times a week1,2,5,14,19,20,33,34,37. The measuring instruments are shown in Table 1.
Results were found in favor of interventions for abdominal strength1, increasing the number of abdominal push-ups performed. However, these results were not significant (SMD = 0.39[95% CI − 0.38–1.17]). A trend was found in favor of the experimental groups in the lower limb test (Appendix 1), where the number of times to get up and sit down on a chair in 30 s was increased1,5. However, there were no significant results after these interventions (SMD = 0.15 [95% CI −0.37–0.68]).
Finally, significant results were found in the strength of both the upper (SMD = 0.74 [95% CI 0.25–1.22]) and lower limbs (SMD = 0.56 [95% CI 0.08–1.04]), regarding the interventions of combined exercise20 and progressive resistance exercise12 against the muscular strength of the upper and lower limbs (Appendix 1), with increases in the 1-repetition maximum (RM) leg press and 12-RM chest press protocols.
Quasi-experimental studies using resistance13,35,37, or aerobic exercise1,2,34 also showed statistically significant differences in increasing muscle strength (Table 1).
Balance
Interventions used to improve balance included aerobic exercise in aquatic environment1,2 and in the terrestrial environment14,32,33, continuous and interval aerobic exercise5, and combined exercise34,36. These interventions lasted between 62 and 25 weeks14, with a frequency of two32 to three times per week. The measuring instruments are shown in Table 1.
Both static and dynamic balances were assessed. For static balance, Boer et al.1 observed improvements in time maintaining balance on one leg and in the number of consecutive steps on a balance beam; however, these improvements were not significant. Regarding dynamic balance, the improvements were not significant either36. Carmeli et al.14 with aerobic exercise reported significant improvements in dynamic balance between groups p < 0.05 and finally, Cai and Baek33 with a combined exercise plan showed significant increase of dynamic and static balance.
A quasi-experimental studies using aerobic exercise34 also showed statistically significant differences in dynamic balance (Table 1).
Gait
Two studies evaluated the effectiveness of their gait interventions13,17. The interventions applied were aerobic exercise through Nordic walking17 and progressive resistance exercise13. The measuring instruments are shown in Table 1. The interventions lasted 10 weeks with a frequency of two13 to three times per week16. Cowley et al.13 found no significant differences in gait speed after their intervention. Conversely, Skiba et al.16 found an improvement in the space–time parameters, angular changes of the limbs, and mean values of the angular deviations at the joints.
Functional tasks
The interventions applied included aerobic exercise in an aquatic1,2 and terrestrial32 environment, continuous and interval aerobic exercise5, progressive resistance exercise12,13, and combined exercise36. The durations ranged from 62 to 14 weeks32, with frequencies ranging from two times12,13,32 to three times per week1,2,5,36. The measuring instruments are shown in Table 1.
It was found that the interventions applied for this outcome1,5 had not significant results (SMD = 0.47 [95% CI = − 0.07,1.01]) in the 6-min walk performance (Appendix 1). Similarly, the time to perform the test after the interventions was reduced in the 8-foot up and go test1,5. However, these results were not significant (SMD = − 0.48 [95% CI − 1.04–0.07]) (Appendix 1).
Perrot et al.32 in a randomized clinical trial showed significant improvements in functional task using combined exercise via Nintendo Wii (Table 1).
Just one quasi-experimental study using resistance exercise13 showed significant improvements in go up and down 10 steps test, or aerobic exercise also showed statistically significant differences in increasing muscle strength (Table 1).
Assessment of the certainty of the evidence
It was found that the certainty of evidence was low for all studies because some had a high risk of bias in the random sequence generation and an unclear risk in blinding. In other studies, the confidence intervals were wide and exceeded the no effect line and some had unclear risk of bias in 4 out of 7 criteria (Table 3).
Discussion
This systematic review assessed the effect of different types of physical exercise on the motor function of adult DS individuals. Among the types of exercise identified in the literature reviewed to improve muscle strength, balance, and gait are aerobic exercise in different modes, such as aquatic, judo, bicycle, and walking. Modes with mechanotherapy equipment were included within the muscle strength exercise, and endless treadmills and elliptical bike were mainly included within the cardiovascular resistance exercises.
Physical exercise is important throughout life because it improves the health38. Physical exercise also has a therapeutic objective for DS individuals as they may require exercise-based clinical interventions throughout their life to improve physical abilities, such as muscle strength, flexibility, and balance39. These diminished physical abilities in DS individuals hinder the performance of ADL and impact their quality of life40.
Three studies included muscle strength as an outcome. Aguiar et al.31 found no differences between the initial and final measurements after a judo training program, while Boer1 and Boer and de Beer2 observed an increase in muscle strength with aquatic aerobic exercise, as did Boer and Moss5 with a bike or treadmill workout. However, the impact of aerobic exercise on increasing muscle strength remains controversial41. The American College of Sports Medicine recommends combining the intensity, volume, and frequency of training to improve muscle strength in young, middle-aged, and older populations and to optimize muscle hypertrophy and strength gains38,42. Vigorous training intensity and/or high training frequency, however, may be difficult to include in a training program for DS individuals owing to their characteristic comorbidities as well as in older adults.
Studies in other populations have reported improvements in muscle hypertrophy, and thus, in muscle strength among younger and older adults after engaging in a single type and mode of physical exercise that includes walking43,44 and riding a bicycle45,46, which is consistent with the findings of this review.
Aerobic exercise and the exercise combination were used to improve balance in adults with DS in aquatic mode2, with treadmill14, basketball33 and bicycle5, and differences were reported only for the intervention in aquatic mode and with exercise combination. Earlier studies have shown the mechanisms underlying this type of exercise that can improve balance, considering that the aquatic environment can stimulate an increase in the strategies and postural adjustments necessary for the execution of different movements47. This improves the quality of motor function by improving muscle strength and balance48.
The ability to walk and do ADL depends on being able to control dynamic and static balance, thus requiring the control of the trajectory of center of pressure49. Improvements in balance after resistance exercise have been attributed mostly by enhanced muscle strength, neural function and force control, as muscle torque is required to maintain balance50, thus, exercise training without a specific balance component may be effective in improving balance control due to a direct influence on muscle mass and strength. It would be important to consider the specificity principle of training, improvements in balance would be amplified by adding balance training in both static and dynamic conditions using neuromuscular exercise in combination with aerobic or resistance exercise (neuromuscular exercise)51.
There were no differences in functional fitness between aerobic exercise in the aquatic or terrestrial modes. Studies including resistance exercise did not assess this outcome. Functional fitness is a construct in which all the physical abilities, which were also included as outcomes in this review, including muscle strength, balance, and posture, and other outcomes, which were not prioritized in this review, such as flexibility and mobility, were included. Physical exercise should enhance all of these abilities for an impact on functional fitness49. On the other hand, the benefits of strength may not transfer effectively to concomitant improvements in functional outcomes such as balance, functional tasks, or activities of daily living52. The contradictory evidence may be due to differences in training intensity, frequency, type of resistance training, equipment (such as free weights or resistance machines), and types of measurements used to evaluate balance, strength and ADL.
Among the limitations of this study, we can mention the methodological limitations of the studies included. All the studies included met at least one of the criteria for unclear risk of bias and 9 out of 12 studies met at least one criterion for high risk of bias. This is consistent with the certainty of evidence for each of the outcomes proposed in this study, as it was low for all the outcomes included. This is partly attributable to the methodological limitations already stated, but also to the imprecision of the studies, possibly due to the small sample sizes included in the primary studies53.
On the other hand. The meta-analyses report very low statistical heterogeneity or in some cases 0 according to the I2, although there is clearly clinical heterogeneity in the included primary studies. These meta-analyses were performed with a small number of primary studies, which may explain the results obtained in the assessment of heterogeneity. It should be taken into account that within the statistical test used to assess heterogeneity is χ2 (Chi-square) and the inconsistency index or I2. Both tests are influenced by the number of studies included in the meta-analyses, therefore, it is possible that the limited number of studies influence the results of the test and these are interpreted as homogeneous when in fact the opposite may occur. It is necessary that the reader and decision maker keep this information in mind when analyzing the findings of systematic reviews with meta-analyses54.
The strengths of this systematic review are having analyzed the effectiveness and prescription parameters of the different types of therapeutic exercise that can be used in people with DS. As a limitation, no sensitivity analysis was performed nor was publication bias explored due to the small sample size of the studies and the low number of studies included in the meta-analysis.
Future research can study the effect of different types of exercise on clinical rehabilitation goals among adult DS individuals. Studies with robust research designs and sample sizes consistent with the effect measure are required to evaluate the effects of exercise on the physical abilities of DS individuals.
Conclusions
There is low certainty of the evidence for the outcomes proposed in this review. Therapeutic exercise, however, could be effective in improving muscle strength and gait functionality. Standardized instruments that measure the outcomes in motor function and research of better methodological quality that assess the effectiveness of the exercise prescription parameters are required. This would facilitate the evaluation of the effectiveness of the intervention as well as decision-making in the practice regarding the type of exercise that would be indicated for each patient according to his or her therapeutic needs.
Data availability
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
Abbreviations
- DS:
-
Down syndrome
- ADL:
-
Activities of daily living
- RM:
-
Repetition maximum
- RoB:
-
Risk-of-bias
- MD:
-
Mean difference
References
Boer, P. H. The effect of 8 weeks of freestyle swim training on the functional fitness of adults with Down syndrome. J. Intellect. Disabil. Res. 64, 770–781 (2020).
Boer, P. H. & de Beer, Z. The effect of aquatic exercises on the physical and functional fitness of adults with Down syndrome: A non-randomised controlled trial. J. Intellect. Disabil. Res. 63, 1453–1463 (2019).
Ruiz-González, L., Lucena-Antón, D., Salazar, A., Martín-Valero, R. & Moral-Munoz, J. A. Physical therapy in Down syndrome: Systematic review and meta-analysis. J. Intellect. Disabil. Res. 63, 1041–1067 (2019).
APTA Guide to Physical Therapist Practice. APTA Guide to Physical Therapist Practice. [citado 19 de abril de 2021]. Disponible en: https://guide.apta.org/.
Boer, P. H. & Moss, S. J. Effect of continuous aerobic vs. interval training on selected anthropometrical, physiological and functional parameters of adults with Down syndrome. J. Intellect. Disabil. Res. 60, 322–34 (2016).
OMS | Actividad física. WHO. World Health Organization; [citado 22 de abril de 2021]. Disponible en: https://www.who.int/dietphysicalactivity/pa/es/.
Gusso, S. et al. The effects of 20 weeks of side-alternating vibration therapy on physical function, bone and muscle health in adolescents with down syndrome. Phys. Occup. Ther. Pediatr. 41(1), 44–55 (2021).
Torres-Nunes, L. et al. Effects of the whole-body vibration exercise on sleep disorders, body temperature, body composition, tone, and clinical parameters in a child with down syndrome who underwent total atrioventricular septal defect surgery: A case-report. Children 10(2), 213 (2023).
Torres-Nunes, L. et al. Effects of whole body vibration exercise on sleep disorders, body composition and clinical parameters in down syndrome children: A study protocol for a randomized cross-over trial. J. Intellect. Disabil. https://doi.org/10.1177/17446295231171197 (2023).
Saquetto, M. B. et al. Effects of whole-body vibration on muscle strength, bone mineral content and density, and balance and body composition of children and adolescents with Down syndrome: A systematic review. Osteoporos Int. 29(3), 527–533 (2018).
Medicine AC of S. Manual ACSM para la valoración y prescripción del ejercicio. Paidotribo; 2019. 500 p.
Shields, N., Taylor, N. F. & Dodd, K. J. Effects of a community-based progressive resistance training program on muscle performance and physical function in adults with Down syndrome: A randomized controlled trial. Arch. Phys. Med. Rehabil. 89, 1215–1220 (2008).
Cowley, P. M. et al. The effect of progressive resistance training on leg strength, aerobic capacity and functional tasks of daily living in persons with Down syndrome. Disabil. Rehabil. 33, 2229–2236 (2011).
Carmeli, E., Kessel, S., Coleman, R. & Ayalon, M. Effects of a treadmill walking program on muscle strength and balance in elderly people with Down syndrome. J. Gerontol. A Biol. Sci. Med. Sci. 57, M106-10 (2002).
Chen, C. C. J. J., Ringenbach, D. R. S. & Snow, M. Treadmill walking effects on grip strength in young men with Down syndrome. Res. Dev. Disabil. 35, 288–293 (2014).
Chen, C. C. J. J., Ringenbach, S. D. R. & Albert, A. R. Assisted cycling exercise improves fine manual dexterity in persons with Down’s syndrome. J. Appl. Res. Intellect. Disabil. 27, 264–272 (2014).
Skiba, A. et al. Evaluation of the effectiveness of Nordic walking training in improving the gait of persons with Down syndrome. BioMed Res. Int. 2019, 6353292 (2019).
Mendonca, G. V., Pereira, F. D. & Fernhall, B. Heart rate recovery and variability following combined aerobic and resistance exercise training in adults with and without Down syndrome. Res. Dev. Disabil. 34, 353–361 (2013).
Mendonca, G. V., Pereira, F. D. & Fernhall, B. Effects of combined aerobic and resistance exercise training in adults with and without Down syndrome. Arch. Phys. Med. Rehabil. 92, 37–45 (2011).
Rimmer, J. H., Heller, T., Wang, E. & Valerio, I. Improvements in physical fitness in adults with Down syndrome. Am. J. Ment. Retard. 109, 165–174 (2004).
Higgins J, Grenn S. Manual Cochrane de revisiones sistemáticas de intervenciones. [citado 22 de abril de 2021]. Disponible en: https://es.cochrane.org/sites/es.cochrane.org/files/public/uploads/Manual_Cochrane_510_reduit.pdf.
Moher D, Liberati A, Tetzlaff J, G. Altman D. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. [citado 11 de mayo de 2021];6(7). Disponible en: https://journals.plos.org/plosmedicine/article?id=https://doi.org/10.1371/journal.pmed.1000097.
Heyward VH. Evaluacion de la aptitud fisica y prescripcion del ejercicio. Ed. Médica Panamericana; 2008. 444 p.
Lesmes JD. Evaluación clínico-funcional del movimiento corporal humano. Ed. Médica Panamericana; 2007.
Palmer LM. Fundamentos de las técnicas de evaluación musculoesquelético. Ed. Paidotribo; 2002.
Living overview of the evidence (L·OVE). [citado 10 de agosto de 2021]. Disponible en: https://app.iloveevidence.com/loves/5d88973d69c00e72a5188eb9?question_domain=5b1dcd8ae611de7ae84e8f14&population=5d8896fa69c00e72a5188eb8&intervention=5d1b60b4daeedf74ca4f4b51&classification=systematic-review.
Rodríguez-Grande EI, Méndez Martínez M. Evidence matrix. https://www.epistemonikos.org/matrixes/5dfd28077db23a0528f5c84e.
RevMan [Internet]. [cited 2022 sep 19]. Available from: /online-learning/core-software-cochrane-reviews/RevMan.
Higgins, J. P. T. Commentary: Heterogeneity in meta-analysis should be expected and appropriately quantified. Int. J. Epidemiol. 37(5), 1158–1160 (2008).
Huedo-Medina, T. B., Sánchez-Meca, J., Marín-Martínez, F. & Botella, J. Assessing heterogeneity in meta-analysis: Q statistic or I2 index?. Psychol. Methods 11(2), 193–206 (2006).
Aguiar, A. S. J. et al. The exercise redox paradigm in the Down’s syndrome: Improvements in motor function and increases in blood oxidative status in young adults. J. Neural Transm. Vienna Austria 1996(115), 1643–1650 (2008).
Perrot, A., Maillot, P., Le Foulon, A. & Rebillat, A. Effect of exergaming on physical fitness, functional mobility, and cognitive functioning in adults with down syndrome. Am. J. Intellect. Dev. Disabil. 126(1), 34–44 (2021).
Cai, W. & Baek, S. Effects of 24-week basketball programme on body composition and functional fitness on adults with Down syndrome. J. Intellect. Disabil. Res. 66(12), 939–951 (2022).
Tsimaras, V. & Fotiadou, E. Effect of training on the muscle strength and dynamic balance ability of adults with Down syndrome. J. Strength Cond. Res. 18(2), 343–347 (2004).
Post, E. et al. The effects of resistance training on physical fitness and neuromotor-cognitive functions in adults with Down syndrome. Front. Rehabil. Sci. 21(3), 927629 (2022).
Silva, V. et al. Wii-based exercise program to improve physical fitness, motor proficiency and functional mobility in adults with Down syndrome. J. Intellect. Disabil. Res. 61, 755–765 (2017).
Davis, W. E. & Sinning, W. E. Muscle stiffness in Down syndrome and other mentally handicapped subjects. J. Mot. Behav. 19, 130–144 (1987).
Ruegsegger, G. N. & Booth, F. W. Health benefits of exercise. Cold Spring Harb. Perspect. Med. 8, a029694 (2018).
Paul, Y., Ellapen, T. J., Barnard, M., Hammill, H. V. & Swanepoel, M. The health benefits of exercise therapy for patients with Down syndrome: A systematic review. Afr. J. Disabil. 8, 576 (2019).
Hardee, J. P. & Fetters, L. The effect of exercise intervention on daily life activities and social participation in individuals with Down syndrome: A systematic review. Res. Dev. Disabil. 62, 81–103 (2017).
Voet, N. B. M., Bleijenberg, G., Padberg, G. W., van Engelen, B. G. M. & Geurts, A. C. H. Effect of aerobic exercise training and cognitive behavioural therapy on reduction of chronic fatigue in patients with facioscapulohumeral dystrophy: Protocol of the FACTS-2-FSHD trial. BMC Neurol. 10, 56 (2017).
Garber, C. E. et al. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 43, 1334–59 (2011).
Harber, M. P. et al. Aerobic exercise training improves whole muscle and single myofiber size and function in older women. Am. J. Physiol. Regul. Integr. Comp. Physiol. 297, R1452–R1459 (2009).
Ozaki, H., Loenneke, J. P., Thiebaud, R. S., Stager, J. M. & Abe, T. Possibility of leg muscle hypertrophy by ambulation in older adults: A brief review. Clin. Interv. Aging 8, 369–375 (2013).
Nelson, A. G., Arnall, D. A., Loy, S. F., Silvester, L. J. & Conlee, R. K. Consequences of combining strength and endurance training regimens. Phys. Ther. 70, 287–294 (1990).
Ozaki, H., Loenneke, J. P., Thiebaud, R. S. & Abe, T. Cycle training induces muscle hypertrophy and strength gain: Strategies and mechanisms. Acta Physiol. Hung. 102, 1–22 (2015).
Pérez de la Cruz, S. Effectiveness of aquatic therapy for the control of pain and increased functionality in people with Parkinson’s disease: A randomized clinical trial. Eur. J. Phys. Rehabil. Med. 53(6), 825–832. https://doi.org/10.23736/S1973-9087.17.04647-0 (2017).
Azd S, Vl I. Effects of dual-task aquatic exercises on functional mobility, balance and gait of individuals with Parkinson’s disease: A randomized clinical trial with a 3-month follow-up. Complement Ther Med. febrero de 2019 [citado 25 de abril de 2022];42. Disponible en: https://pubmed.ncbi.nlm.nih.gov/30670228/.
Shkuratova, N., Morris, M. E. & Huxham, F. Effects of age on balance control during walking. Arch. Phys. Med. Rehabil. 85, 582–588 (2004).
Orr, R. Contribution of muscle weakness to postural instability in the elderly: A systematic review. Eur. J. Phys. Rehabil. Med. 46, 183–220 (2010).
Rodríguez-Grande, E. I., Vargas-Pinilla, O. C., Torres-Narvaez, M. R. & Rodríguez-Malagón, N. Neuromuscular exercise in children with Down Syndrome: A systematic review. Sci. Rep. 12(1), 14988 (2022).
Granacher, U., Gollhofer, A., Hortobagyi, T., Kressig, R. W. & Muehlbauer, T. The importance of trunk muscle strength for balance, functional performance, and fall prevention in seniors: A systematic review. Sports Med. 43, 627–641 (2013).
Berryman, N. et al. Executive functions, physical fitness and mobility in well-functioning older adults. Exp. Gerontol. 48, 1402–1409 (2013).
Ojeda, D. & Wurth, O. Qué es un metaanálisis. Rev. Chil. Anest. 43, 343–350 (2014).
Acknowledgements
The authors express their gratitude to the Doctoral Program in Clinical Epidemiology at Pontificia Universidad Javeriana, Bogotá, Colombia. Special thanks are extended to Jimmy Alexander Valencia Castro for his valuable assistance in screening the studies.
Author information
Authors and Affiliations
Contributions
M.M.-M. performed the research, screening, selection, and analysis of the studies and data, performed the meta-analyses and was a major contributor in writing the manuscript. E.-I.R.-G. confirmed that the studies selected met the study selection criteria, she was an instructor to perform the meta-analyses and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Méndez-Martínez, M., Rodríguez-Grande, EI. Effects of therapeutic exercise on the motor function of adults with Down syndrome: a systematic review and meta-analysis. Sci Rep 13, 21962 (2023). https://doi.org/10.1038/s41598-023-48179-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-48179-1
- Springer Nature Limited