Abstract
Renin-angiotensin system inhibitors (RASi), particularly angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin II receptor blockers (ARBs), are commonly used in the treatment of hypertension and are recommended for kidney protection. Uncertainty remains about the effectiveness of RASi being used as first-line antihypertensive therapy on eGFR maintenance compared to its alternatives, especially for those with no or early-stage chronic kidney disease (CKD). We conducted a retrospective cohort study of 19,499 individuals (mean age 64.1, 43.5% males) from primary care in Singapore with 4.5 median follow-up years. The study cohort included newly diagnosed individuals with hypertension (whose eGFR was mainly in CKD stages G1-G2) and initiated on ACEIs, ARBs, beta-blockers (BBs), calcium channel blockers (CCBs) or diuretics (Ds) as first-line antihypertensive monotherapy. We compared the estimated glomerular filtration rate (eGFR) curve before/after the drug initiation over time of patients under different drug classes and analyzed the time to declining to a more advanced stage CKD. Inverse probability of treatment weighting (IPTW) was used to adjust for baseline confounding factors. Two key findings were observed. First, after initiating antihypertensive drugs, the eGFR almost maintained the same as the baseline in the first follow-up year, compared with dropping 3 mL/min/1.73 m2 per year before drug initiation. Second, ARBs were observed to be slightly inferior to ACEIs (HR = 1.14, 95% CI = (1.04, 1.23)) and other antihypertensive agents (HR = 1.10, 95% CI = (1.01, 1.20)) in delaying eGFR decline to a more advanced CKD stage in the study population. Our results showed that initiating antihypertensive agents can significantly maintain eGFR for those newly diagnosed patients with hypertension. However, RASi may not be superior to other antihypertensive agents in maintaining eGFR levels for non-CKD or early stages CKD patients.
Similar content being viewed by others
Introduction
Hypertension affects around 1.2 billion people worldwide1 and is strongly associated with the prevalence of CKD2. In patients with newly diagnosed hypertension, five antihypertensive drug classes [angiotensin-converting enzyme inhibitors (ACEIs), angiotensin II receptor blockers (ARBs), beta-blockers (BBs), calcium channel blockers (CCBs) and diuretics (Ds)] are widely recommended by clinical guidelines as first-line blood pressure lowering drugs, either in combination or as monotherapy3,4,5,6,7,8. Among them, ACEIs and ARBs are recommended by major clinical practice guidelines for their kidney protection benefits4,9, aside from lowering blood pressure and cardiovascular disease protection. These drugs are preferentially prescribed to those with CKD and type-2 diabetes mellitus.
The evidence supporting the superiority of ACEIs and ARBs over other drug classes originated mainly from clinical trials and utilized end-stage kidney disease (ESKD) and doubling of baseline serum creatinine concentration as clinical outcomes10,11,12. Due to the long progression time of chronic kidney disease, ESKD is more likely to be observed in patients already with later stages chronic kidney disease, i.e., eGFR ≤ 45 mL/min/1.73 m2. For those newly diagnosed with hypertension and with eGFR in an early stage of CKD (eGFR > 60 mL/min/1.73 m2), uncertainty remains on whether ACEIs and/or ARBs are superior to other antihypertensive drug classes on kidney protection. Another pertinent question relates to whether ACEIs or ARBs confer better kidney function when they are initiated as monotherapy in patients with hypertension and vascular diseases. The majority of these patients are asymptomatic and are treated in primary care.
In order to determine the effectiveness of ACEIs and ARBs on kidney function, we leverage on a real-world primary care clinical dataset to conduct a comparative analysis of five first-line antihypertensive drug classes as monotherapies on the maintenance of eGFR over time. The study outcomes analyzed included eGFR value change over time after drug initiation and time to eGFR decline to a more advanced CKD stage.
Methods
Study setting and data source
This retrospective cohort study was conducted using electronic medical records (EMR) from SingHealth Polyclinic (SHP), a cluster of public primary care clinics (polyclinics) in the eastern region of Singapore. This cosmopolitan island state at the centre of Southeast Asia had a prevalence of hypertension of 21.5% in 2017 of its adult residents living in a densely populated, urban environment treated by a mixture of private and public primary care clinics13.
SHP includes eight separately located polyclinics, which managed over 2.3 million multi-ethnic Asian patient attendances in 2021. Each polyclinic serves about 450 to 1100 patients across all ages during office hours for each work day, with their clinical care documented in EMR.
All adult patients with a clinical diagnosis of hypertension were identified from the SHP EMR from January 1, 2010 to December 31, 2020. Eligible patients' clinical data, including demographics, laboratory test results, and prescribed medications, were extracted, audited, and de-identified by an approved trusted third party before analysis. All methods were performed in accordance with relevant guidelines and regulations.
Cohort design and data processing
Adult patients with hypertension initiated on a first-line antihypertensive drug as monotherapy from these five drug classes, i.e., ACEIs, ARBs, BBs, CCBs and Ds were identified and extracted from the entire data set. The drugs considered in each class are listed in Table S1.
The study cohort constructed followed a retrospective, comparative new-user design14,15,16. More specifically, we first identified when the individuals were first exposed to an anti-hypertensive drug in the selected classes as monotherapy. Then, we required patients to have at least 1 year of previous EHR observation before first exposure to guarantee the treatment initiation. Last, we required a recorded hypertension diagnosis at or within the date preceding treatment initiation. The diagnosis of hypertension was identified based on International Classification of Diseases 10th Revision (ICD-10) codes. In addition, we only considered adult patients (aged 21 years or older), and the patients should have one baseline eGFR, one control eGFR and at least one follow-up eGFR laboratory test. Patients were divided into three groups according to the drug class: those taking ACEIs, those taking ARBs, and those taking either BBs, CCBs or Ds. Those taking BBs, CCBs and Ds were grouped together as "OTHERS" based on evidence from previous literature showing that they were less effective compared to RASi in kidney protection17. Patients who were prescribed concurrently with Sodium-GLucose coTransporter-2 (SGLT2) inhibitors such as empagliflozin and dapagliflozin in the EMR were excluded because of their known kidney protective effects18,19,20.
The observation period began from the date of drug initiation (index date) to the end date of the same drug prescribed in the EMR. More specifically, the observation would end with any one of the following occurrences: (1) switch to another antihypertensive drug, or (2) addition of another antihypertensive drug, or (3) no refill prescription of the current antihypertensive drug within 1 year from the last prescription.
The eGFR value was computed from serum creatinine values reported from laboratory reports in the EMR using the CKD-EPI Creatinine Equation 202121. Serum creatinine was analyzed using the kinetic Jaffe method on the Cobas c702 analyzer (Roche, Mannheim, Germany). The creatinine assay calibration is traceable to the isotope dilution mass spectrometry (IDMS) reference creatinine method.
The baseline eGFR of each patient was defined as the last eGFR measured within the 1-year period before and including the index date. The control eGFR is defined as the eGFR measured within the 1-year to 2-year period before the index date. The follow-up eGFR sequences are those measured consecutively after the index date to the end of the observation period. In the polyclinics, the eGFR of the patients are tied to a laboratory panel test (which includes kidney function) performed once a year, a single eGFR reading was extracted every year after the index date. In the situation where multiple eGFRs were measured in a single year, we computed the mean eGFR (for control and follow-ups) or the last eGFR (for baseline) as the indicator of kidney function for that year. For lapses in eGFR assessment for any patient, we used linear interpolation to estimate the eGFR value (Fig. 1).
Statistical analysis and outcomes
The effectiveness evaluation was conducted pairwise among ACEIs, ARBs and OTHERS antihypertensive drug classes. Inverse probability treatment weighting (IPTW) was used to control the confounding factors in baseline characteristics. The propensity score was estimated using multivariate logistic regression with l2-regularization. Stabilized weights were used to increase precision by adding the marginal probability to the numerator of the weights. Weights were considered appropriate if the standardized mean difference (SMD) between treatment groups was < 0.1 (Tables S3–S5).
After appropriate weightage was assigned, the following outcomes were estimated. First, the mean and 95% confidence interval of eGFR differences from control to baseline, and also from eGFR at first follow-up value to baseline was computed. This was aimed at evaluating the effectiveness of drug initiation.
Next, patients prescribed the same drug for at least 5 years were selected in each group, and the eGFR differences to baseline over time were compared. In addition, we also estimated the eGFR curve under the case when patients did not initiate the drug at index date by assuming the eGFR would decline with the same slope as that between control and baseline values over time.
Finally, the association between eGFR progression and drug classes was evaluated during the observation period. The eGFR progression was defined as the time from drug initiation to when follow-up eGFR values declined to a more advanced stage (e.g. stage G1 to stage G2 or stage G1 to stage G3) compared to baseline at two consecutive visits. The association was estimated by multivariate Cox proportional hazard regression. The study population was grouped according to their demographic characteristics and clinical status at baseline, including gender, age, systolic blood pressure, eGFR stages, diabetes mellitus, kidney disease, and macrovascular disease. The regression study was repeated in the subgroups of the entire population.
The baseline variables considered in both propensity score estimation and Cox proportional hazard regression were age, gender, ethnic group, laboratory measurements [including HbA1c, low density lipoprotein-cholesterol (LDL-C), systolic and diastolic blood pressure and the body mass index (BMI), and eGFR stages], comorbidity status, complication status, and medication prescription. All analyses were performed using Python 3.8.12.
Ethics approval and consent to participate
Ethics approval was obtained from SingHealth Centralized Institution Review Board (CIRB) in 2019 (SingHealth CIRB Reference: 2019/2604). Requirement of written consent was waived by the SingHealth CIRB as it was deemed impracticable while privacy risks were mitigated through the use of de-identified data.
Results
Study cohort and baseline characteristics
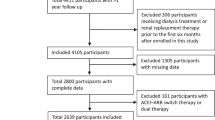
From 124,753 candidate patients newly diagnosed with hypertension, we identified 19,499 patients with mean age 64.1 who satisfied the selection criteria and were included in the study population (Fig. S1), of which 43.5% were male. Among them, 3832(19.6%) patients were initiated with ACEIs, 3171(16.3%) with ARBs and 12,496 (64.1%) with OTHERS drug classes (BBs, CCBs, Ds) as first-line monotherapy treatment for hypertension. The median duration of follow-up was 4.5 (IQR = [2.1, 8.0]) years. The median frequency of eGFR measurement was 1.09 times per year (IQR = [0.96, 1.45]).
The baseline characteristics for patients under different first-line antihypertensive treatments are given in Table 1. Across all three cohorts, patients were similar in the age distribution, baseline laboratory values, diagnosis of hyperlipidaemia and anti-hyperlipidaemic medications. The gender distribution was different between each drug class pair. The majority (> 80%) of patients in each group were in CKD stages G1 and G2, with very few patients (< 1%) in CKD stages G4 and G5. Comparing those initiated with ACEIs or ARBs with those initiated on OTHERS, patients with ACEIs or ARBs were more likely to have diabetes, nephropathy and retinopathy. Thus, more patients with ACEIs or ARBs were on anti-diabetic medications at baseline. The patient profiles of those under ACEIs and ARBs were very similar, except for the cohort with ACEIs, which had slightly more male and diabetic patients.
The pairwise comparison of the baseline characteristics of different study cohorts are given in Tables S3–S5. Among them, some attributes, such as gender and diagnosis of diabetes, were highly unbalanced. Using IPTW to remove the confounding factors, all the SMD values after weighting among different treatment groups are smaller than 0.05.
Drug initiation
For each comparison pair, the differences between the control and the first-year follow-up from the baseline are reported in Table 2. We could see that before initiating the drugs, the eGFR of patients generally declined by about 3 mL/min/1.73 m2 per year, whereas after drug initiation, the eGFR value was almost the same compared to the baseline value in the first follow-up year. The differences between the base and comparator in each pair were not significant. Such results demonstrate the kidney protective effect of antihypertensive drugs on delaying eGFR decline, regardless of class.
eGFR change over time
Figure 2 shows the eGFR changes for each drug class pair over 5 years. The general trend of different drug class pairs were similar. After drug initiation, the eGFR value maintained in the first year, increased around 5 mL/min/1.73 m2 in the second year, and slowly declined until the fifth year. At the end of 5 years, the mean eGFR values were still higher than or near the baseline values, with the differences between drug classes less than 2 mL/min/1.73 m2: 1.67 mL/min/1.73 m2 (ACEIs vs ARBs, P < 0.05), − 1.01 mL/min/1.73 m2 (ACEIs vs OTHERS, P < 0.05), and − 1.96 mL/min/1.73 m2 (ARBs vs OTHERS, P < 0.05), respectively.
Pairwise comparison of eGFR curve over 5 years. The asterisks besides the time label indicate that the corresponding two means of the real eGFR curve are statistically significant different (P < 0.05). The error bar of each data point depicts the 95% confidence interval. Note that this analysis is conducted in a subset of the whole study population, only including those taking the drugs for more than 5 years. The SMD after weighting in all comparative pairs are smaller than 0.1 (Fig. S3).
Compared with the extrapolated control line that resulted in a 15 mL/min/1.73 m2 eGFR reduction over 5 years, consistent intake of the antihypertensive drugs showed significant kidney protection in terms of maintenance of eGFR.
Time to eGFR decline to a more advanced stage
For groups, ACEIs, ARBs and OTHERS, 428 (11.2%), 429 (13.5%) and 941 (7.5%) patients had declining eGFR to a more advanced CKD stage compared with baseline, respectively. The detailed patient numbers of each CKD stage transition group is given in Table S6. In addition, less than 3% of patients in each group had eGFR decline by more than 30% from baseline during the study period (Table S2).
The hazard ratios show no difference between ACEIs and OTHERS in delaying the eGFR to a more advanced CKD stage (HR = 1.03, 95% CI = (0.95,1.12)). However, the ARBs are observed to be slightly inferior to both ACEIs (HR = 1.14, 95% CI = (1.04, 1.23)) and OTHERS (HR = 1.10, 95% CI = (1.01,1.20)) in preventing eGFR decline to a more advanced CKD stage.
In subgroup analysis (Fig. 3), ACEIs were observed to be superior to ARBs in patients with lower risks of vascular complications, such as those aged less than 70 years, systolic BP less than 140 mmHg, in CKD stage G1, without kidney disease or without macrovascular disease. Notably, among diabetic patients, ACEIs were superior to ARBs in delaying the eGFR decline to the next CKD stage. By comparing ARBs vs OTHERS, the only statistically significant subgroup was patients with diabetes (HR = 1.15, 95% (1.02,1.30)). Between ACEIs and OTHERS, no statistically significant differences were observed in the subgroups.
Hazard ratios for eGFR declining to next CKD stage for whole cohort and subgroups divided according to sex, age, systolic blood pressure, CKD stage, diabetes, kidney disease and macrovascular disease. The asterisks besides the labels indicate that the corresponding HR are statistically significant different (P < 0.05).
Discussion
Background and key findings
Leveraging on real-world primary care data, we compared the effectiveness of five common first-line antihypertensive drug classes in maintaining the eGFR of patients who were mostly (> 80%) in CKD stages G1-G2. To the best of our knowledge, such comparison with eGFR value change as an outcome had not yet been investigated. Two main findings were observed in our study. First, for patients with hypertension, initiating an antihypertensive drug in class ACEIs, ARBs, and OTHERS (i.e., BBs, CCBs or Ds) significantly delayed the eGFR decline over time. Second, the ARBs were observed to be slightly inferior to ACEIs and OTHERS in delaying eGFR decline to a more advanced CKD stage, especially for diabetic patients.
Choice of first-line antihypertensive drugs
Among all patients in this study population, ACEIs and ARBs were more likely prescribed to patients with diabetes or nephropathy (Table 1), which is consistent with current clinical guidelines3,4,5,6,7,8.
eGFR changing over time
The natural eGFR decline rate of those with hypertension in our dataset is 3 mL/min/1.73 m2 per year (Table 2), which is significantly higher than 1 mL/min/1.73 m2 per year for the normal population22. The rate of eGFR decline in hypertensive individuals varies across studies due to differing study contexts. Polonia et al. found in a retrospective observational study that the average yearly eGFR decline was 3.3 ± 8.2 mL/min/1.73 m2 for hypertensive individuals with type II diabetes, and 2.4 ± 7.7 mL/min/1.73 m2 for those without type II diabetes23. Collard et al., in a post hoc analysis of clinical trial data, reported yearly eGFR decline rates of 0.95–1.23 mL/min/1.73 m2 for individuals under standard blood pressure targets and 1.14–1.24 mL/min/1.73 m2 for those under intensive targets24. Yu et al., in a prospective cohort study, reported yearly eGFR declines of 1.47 to 1.71 mL/min/1.73 m2 in hypertensive individuals25. Comparatively, our eGFR decline rate aligns closely with the observational study by Polonia et al.23 but is more rapid than rates from clinical trials or the prospective study. Potential reasons for this discrepancy could be irregular eGFR measurements in real-world settings, leading to inexact yearly intervals for estimating decline rates, and our estimation was during a phase when patients did not initiate anti-hypertensive drugs, which may accelerate eGFR decline.
In the fifth-year of follow-up, the mean eGFR value of each drug class was still close to baseline eGFR, demonstrating these drugs' effectiveness on eGFR maintenance. Among them, the OTHERS drug class is observed to have the best eGFR maintenance effect, with a higher mean eGFR 1.01 mL/min/1.73 m2 (P < 0.05) than ACEIs and 1.96 mL/min/1.73 m2 (P < 0.05) than ARBs. Such results are consistent with that in the ALLHAT trial26 which showed that CCB had the highest eGFR maintenance at the fourth follow-up year compared to ACEIs and D. The acute eGFR decline after drug initiation27 was not observed for ACEIs and ARBs; it may be due to the 1-year interval between laboratory testings, while the acute eGFR decline happened in the first 3 months after the drug initiation28.
eGFR increased in all groups in the second year of follow-up
The cause of this is unclear and was not due to a change in BMI (Fig. S2). Improvement in eGFR was seen in various other studies at varying frequencies. In the MDRD study, GFR improved in 11% of patients29 and in a hospital-based nephrology cohort (NephroTest Study Group), measured GFR improved in 15.3% of patients (these patients were less likely to have diabetic glomerulopathy or polycystic kidney disease)30. In a separate nephrology clinic referral cohort, significant improvement in eGFR slope was seen in 48.2% of CKD stage G2 patients31. A rise in eGFR may mean hyperfiltration in a patient with diabetes, or it may signify the potential occurrence of kidney remodelling or regeneration with regression of kidney fibrosis. Remuzzi et al. in an experimental rat model with spontaneous kidney disease, was able to demonstrate glomerular repair using ACEIs32.
eGFR declined to an advanced stage
Our results showed that ARBs were marginally inferior to ACEIs and OTHERS on delaying eGFR to a more advanced CKD stage. Even though the literature on the effect of antihypertensive drugs on eGFR is limited, some of the previous literature has questioned the additional kidney protective effect of RASi compared to OTHERS with ESKD as an outcome. In a meta-analysis of 127 clinical trials of patients with or without diabetes two decades ago10, Casas et al. reported that RASi has a smaller benefit on ESKD than OTHERS with a relative risk of 0.87, CI (0.75, 0.99), but RASi had no additional effect on the eGFR compared to OTHERS during the follow-up period. Such result differences might come from the different drug effectiveness on patients in different CKD stages, considering the patients with no or early-stage CKD have a more challenging time developing ESKD during the follow-up period (Table S2). Moreover, Bangalore et al., in their systematic review and meta-analysis33 on patients with diabetes, have shown that RASi resulted in no significant difference in ESKD compared to other antihypertensive agents (relative risk 0.99, 95% CI (0.78, 1.28)), which is consistent with our observation between ACEIs and OTHERS. In studies comparing antihypertensive drug effectiveness on those with CKD (G3-G5), the ACEIs had shown a superior effectiveness than ARBs in preventing ESKD or doubling of serum creatinine level34,35, which is also consistent with our observation. This study's novelty comes from utilising the eGFR as an outcome and observing that ARBs were marginally inferior to OTHERS in delaying eGFR decline in those with no or early-stage CKD, which have not been reported yet.
Contributions
This study contributes to the knowledge that any antihypertensive drug class can potentially be used for kidney protection in newly diagnosed hypertensive patients with no or early-stage CKD in the primary care setting. First, the results affirm that initiating an antihypertensive drug as monotherapy can reduce eGFR decline and maintain the eGFR over time. Second, the results showed that the RASi were not superior to other antihypertensive drugs in eGFR maintenance of patients with no CKD or early-stage CKD, which may suggest that clinicians can select antihypertensive drugs mainly based on the blood pressure lowering effect and cost, rather than for additional kidney protection. Third, some atypical eGFR trends were observed, such as increased eGFR at the second follow-up year across all drug class groups. The reason for this phenomenon should be further explored.
Limitations
The study has its limitations. Due to the lack of albuminuria data, we could not evaluate the antihypertensive drug effectiveness on albuminuria reduction. Known confounding factors, such as dietary habits (e.g. salt intake), physical activities and other lifestyle behaviours, were not recorded in the EMR for statistical analysis.
The main reason for the lack of albuminuria/proteinuria data is the transition of the semi-quantitative urine test for proteinuria to quantitative microalbuminuria only in 2018. Furthermore, the urine tests are often unavailable due to various reasons: failure to collect urine on the spot at the lab (patients using diapers, incontinence) or inability to quantitate the urine protein/albumin due to concurrent urinary tract infection). Hence, our main health outcome focuses on more objective indicators of kidney function such as eGFR, notwithstanding that this indicator has minor confounders. Regardless of the kidney function status, the clinicians initiate or adjust the anti-hypertensive medications to optimize patients’ blood pressure control based on the local official clinical practice guidelines for hypertension management from the Ministry of Health of Singapore.
Suggestions to real-world practice
The clinicians can leverage the results for shared clinical decision-making with their patients in selecting the appropriate first-line antihypertensive drug for those with no or early-stage CKD. Clinicians can also share with patients the effect of antihypertensive drugs on their kidney function over time to motivate and enhance treatment adherence to maintain their kidney health regardless of the drug class. The study will pave the way for further research to understand the effect of ACEIs, ARBs and OTHERS on albuminuria36.
Conclusions
In conclusion, among patients with no or early-stage CKD, initiating an antihypertensive drug in ACEIs, ARBs, BBs, CCBs or Ds as monotherapy significantly delay the eGFR progression over time, regardless of drug class. However, the superiority of RASi (ACEIs/ARBs) over other antihypertensive drug classes on eGFR maintenance was not observed. This evidence may potentially inform clinical decisions on the choice of antihypertensive therapy for this patient group.
Data availability
The datasets analysed during the current study are not publicly available as they contain information that are sensitive to the study institution. They may be made available from the corresponding author on reasonable request.
Code availability
The code used for the analyses in this study is available for reference in the GitHub repository at https://github.com/GaoQiaoPH/HTN_drug_effectiveness_on_eGFR.
References
NCD Risk Factor Collaboration (NCD-RisC). Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: A pooled analysis of 1201 population-representative studies with 104 million participants. Lancet 398(10304), 957–980 (2021).
Kjeldsen, S. E. Hypertension and cardiovascular risk: General aspects. Pharmacol. Res. 129, 95–99 (2018).
Flack, J. M. & Adekola, B. Blood pressure and the new ACC/AHA hypertension guidelines. Trends Cardiovasc. Med. 30(3), 160–164 (2020).
Unger, T. et al. 2020 International Society of hypertension global hypertension practice guidelines. Hypertension. 75(6), 1334–1357 (2020).
Williams, B. et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 39(33), 3021–3104 (2018).
World Health Organization. Guideline for the Pharmacological Treatment of Hypertension in Adults: Web Annex A: Summary of Evidence (World Health Organization, 2021).
Tay, J. C. et al. Ministry of health clinical practice guidelines: Hypertension. Singap. Med. J. 59(1), 17–27 (2018).
Hua, Q., Fan, L. & Li, J. Joint Committee for Guideline Revision. 2019 Chinese guideline for the management of hypertension in the elderly. J. Geriatr. Cardiol. 16(2), 67–99 (2019).
Pugh, D., Gallacher, P. J. & Dhaun, N. Management of hypertension in chronic kidney disease. Drugs. 79(4), 365–379 (2019).
Casas, J. P. et al. Effect of inhibitors of the renin-angiotensin system and other antihypertensive drugs on renal outcomes: Systematic review and meta-analysis. Lancet. 366(9502), 2026–2033 (2005).
Jafar, T. H. et al. Progression of chronic kidney disease: The role of blood pressure control, proteinuria, and angiotensin-converting enzyme inhibition: A patient-level meta-analysis. Ann. Intern. Med. 139(4), 244–252 (2003).
Jafar, T. H. et al. Angiotensin-converting enzyme inhibitors and progression of nondiabetic renal disease. A meta-analysis of patient-level data. Ann. Intern. Med. 135(2), 73–87 (2001).
Chan, G. C. et al. Hypertension in a multi-ethnic Asian population of Singapore. J. Clin. Hypertens. 23(3), 522–528 (2021).
Ryan, P. B., Schuemie, M. J., Gruber, S., Zorych, I. & Madigan, D. Empirical performance of a new user cohort method: Lessons for developing a risk identification and analysis system. Drug Saf. 36(suppl 1), S59-72 (2013).
Hernán, M. A. & Robins, J. M. Using big data to emulate a target trial when a randomized trial is not available. Am. J. Epidemiol. 183, 758–764 (2016).
Suchard, M. A. et al. Comprehensive comparative effectiveness and safety of first-line antihypertensive drug classes: A systematic, multinational, large-scale analysis. Lancet. 394(10211), 1816–1826 (2019).
Bakris, G. L., Hart, P. & Ritz, E. Beta blockers in the management of chronic kidney disease. Kidney Int. 70(11), 1905–1913 (2006).
Lin, Y. H. et al. Renal and glucose-lowering effects of empagliflozin and dapagliflozin in different chronic kidney disease stages. Front. Endocrinol. 22(10), 820 (2019).
Heerspink, H. J. L. et al. Dapagliflozin in patients with chronic kidney disease. N. Engl. J. Med. 383(15), 1436–1446 (2020).
EMPA-KIDNEY Collaborative Group et al. Empagliflozin in patients with chronic kidney disease. N. Engl. J. Med. 388, 117–127 (2022).
CKD-EPI Creatinine Equation (2021). https://www.kidney.org/content/ckd-epi-creatinine-equation-2021.
Waas, T. et al. Distribution of estimated glomerular filtration rate and determinants of its age dependent loss in a German population-based study. Sci. Rep. 11(1), 10165 (2021).
Polonia, J., Azevedo, A., Monte, M., Silva, J. A. & Bertoquini, S. Annual deterioration of renal function in hypertensive patients with and without diabetes. Vasc. Health Risk Manag. 26(13), 231–237 (2017).
Collard, D. et al. Initial estimated glomerular filtration rate decline and long-term renal function during intensive antihypertensive therapy: A post hoc analysis of the SPRINT and ACCORD-BP randomized controlled trials. Hypertension. 75(5), 1205–1212 (2020).
Yu, Z. et al. Association between hypertension and kidney function decline: The atherosclerosis risk in communities (ARIC) study. Am. J. Kidney Dis. 74(3), 310–319 (2019).
Officers and Coordinators for the ALLHAT Collaborative Research Group. The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA. 288(23), 2981–2997 (2002).
Ku, E. et al. Acute declines in renal function during intensive bp lowering: Implications for future ESRD risk. J. Am. Soc. Nephrol. 28(9), 2794–2801 (2017).
Holtkamp, F. A. et al. An acute fall in estimated glomerular filtration rate during treatment with losartan predicts a slower decrease in long-term renal function. Kidney Int. 80(3), 282–287 (2011).
Hunsicker, L. G. et al. Predictors of the progression of renal disease in the Modification of Diet in Renal Disease Study. Kidney Int. 51(6), 1908–1919 (1997).
Weis, L. et al. Renal function can improve at any stage of chronic kidney disease. PLoS ONE. 8(12), e81835 (2013).
Taskapan, H. et al. Improvement in eGFR in patients with chronic kidney disease attending a nephrology clinic. Int. Urol. Nephrol. 40(3), 841–848 (2008).
Remuzzi, A. et al. ACE inhibition reduces glomerulosclerosis and regenerates glomerular tissue in a model of progressive renal disease. Kidney Int. 69(7), 1124–1130 (2006).
Bangalore, S., Fakheri, R., Toklu, B. & Messerli, F. H. Diabetes mellitus as a compelling indication for use of renin angiotensin system blockers: Systematic review and meta-analysis of randomized trials. BMJ. 11(352), i438 (2016).
Xie, X. et al. Renin-angiotensin system inhibitors and kidney and cardiovascular outcomes in patients with CKD: A Bayesian network meta-analysis of randomized clinical trials. Am. J. Kidney Dis. 67(5), 728–741 (2016).
Zhang, Y. et al. ACE inhibitor benefit to kidney and cardiovascular outcomes for patients with non-dialysis chronic kidney disease stages 3–5: A network meta-analysis of randomised clinical trials. Drugs. 80(8), 797–811 (2020).
Viazzi, F. et al. Apparent treatment resistant hypertension, blood pressure control and the progression of chronic kidney disease in patients with type 2 diabetes. Kidney Blood Press. Res. 43(2), 422–438 (2018).
Acknowledgements
We would like to thank Ms. Tan Wei Ying from Institute of Data Science for her help on pre-processing the data.
Funding
This research is supported by the National Research Foundation, Singapore under its AI Singapore Programme (AISG Award No: AISG-GC-2019-001-2A). Any opinions, findings and conclusions or recommendations expressed in this material are those of the authors and do not reflect the views of National Research Foundation, Singapore.
Author information
Authors and Affiliations
Contributions
Q.G. and N.C.T. conceptualized and designed the study, with input from M.L..L., W.H. and J.C. Q.G. performed the statistical analysis. Q.G., M.L.L. and W.H. contributed to the results analysis from the perspective of data analytics. N.C.T. and J.C. contributed to the results analysis from the perspective of a professional clinician. Q.G. wrote the initial draft of the paper, to which the rest of the authors provided critical review of the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gao, Q., Tan, N.C., Lee, M.L. et al. Comparative effectiveness of first-line antihypertensive drug classes on the maintenance of estimated glomerular filtration rate (eGFR) in real world primary care. Sci Rep 13, 21225 (2023). https://doi.org/10.1038/s41598-023-48427-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-48427-4
- Springer Nature Limited