Abstract
We reported that the full-length width of medial tibial osteophytes comprising cartilage and bone parts correlates with medial meniscus extrusion (MME) in early-stage knee osteoarthritis (OA). However, no data exist on the prevalence of MME and its relationship with osteophytes in the elderly population. 1191 elderly individuals (females 57%; 72.9 years old on average) in the Bunkyo Health Study underwent standing plain radiograph and proton density-weighted MRI on knee joints. MRI-detected OA changes were evaluated according to the Whole-Organ Magnetic Resonance Imaging Score. A new method of assessing the cartilage and bone parts of osteophytes was developed using pseudo-coloring images of proton density-weighted fat-suppressed MRI. Most subjects showed Kellgren-Lawrence grade 1 or 2 radiographic medial knee OA (88.1%), MME (98.7%, 3.90 ± 2.01 mm), and medial tibial osteophytes (99.3%, 3.27 ± 1.50 mm). Regarding OA changes, MME was closely associated with the full-length width of medial tibial osteophytes (β = 1.114; 95% CI 1.069–1.159; p < 0.001) in line with osteophyte width (intraclass correlation coefficient, 0.804; 95% CI 0.783–0.823). Our data revealed that MME and medial tibial osteophytes are observed in the elderly and demonstrate that the degree of MME is consistent with the full-length width of medial tibial osteophytes, suggesting that osteophytes might be implicated in MME.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
In a super-aging society, prevention of motor function deterioration is considered to contribute to the extension of healthy life expectancy. Knee osteoarthritis (OA) is a disease in which the motor function decreases with age1,2. Nearly 30% of individuals older than 45 years old and over 80% of women older than 80 years old are reported to have radiographic knee OA in the United States and Japan, respectively3,4. With the increasing number of elderly people, the number of patients with knee OA is increasing worldwide5. Elucidation of the progression mechanism and development of treatment methods for knee OA will thus be important for extending the healthy life expectancy6,7.
OA has been defined pathologically as an intrinsic degenerative disease of articular cartilage in which biochemical and metabolic alterations result in its breakdown8 and has long been considered a “wear and tear” disease leading to a loss of cartilage9. However, accumulated data from molecular biology and experimental studies have demonstrated that many inflammatory mediators produced by cartilage, subchondral bone, and synovium play a pivotal role in the initiation and perpetuation of OA9. Analyses of knee OA by focusing on OA features and progressors of early-stage knee OA patients are one of the most important topics in this field10,11. After the application of magnetic resonance imaging (MRI) for the diagnosis of knee OA, growing evidence acquired by MRI analyses has indicated that, besides cartilage destruction, alterations in the meniscus, subchondral bone, osteophyte, and synovium are commonly observed in early-stage knee OA12, and the changes proceed asymptomatically many years before the onset of knee OA13,14. Among these, disorders in the meniscus are considered an important progressive factor for early-stage knee OA15,16,17. Meniscal dysfunction preceded by medial meniscus extrusion (MME) is a risk factor for knee OA18,19,20. More than half of the middle-aged patients with early-stage knee OA have been reported to have MME21. Although one of the main causes of MME is meniscus tear22, many patients with early-stage knee OA21 or established knee OA (10–70% of patients)23,24,25,26,27 exhibit MME without meniscus tear. Therefore, an important question that remains to be addressed is how MME develops in early-stage knee OA.
Osteophytes are the most common abnormality among patients with early-stage knee OA who have no radiographic OA12. Synovial and/or periosteal mesenchymal stem cells present near the margin of articular cartilage form osteophytes following a process of endochondral ossification28,29,30,31, thus they are histologically composed of bone tissue capped by cartilage tissue28. In our previous study, we reported that both bone and cartilage parts of osteophytes can be readily detected by T2 mapping MRI in the medial tibia in almost all patients with early-stage knee OA21 and demonstrated that MME is consistent with the full-length width of medial tibial osteophytes (width of cartilage and bone parts)21. In addition, we recently found that anterior meniscus extrusion in elderly subjects is closely associated with the full-length width of the anterior tibial osteophyte32. Based on these data, we hypothesized that MME in early-stage knee OA without meniscal tears may be induced through medial displacement of the medial meniscus by the medial tibial osteophyte21, since the medial meniscus is tightly attached to the medial tibial plateau by the meniscotibial ligament21,33,34. However, our previous study on knee OA was performed in a limited number (n = 50) of patients with early-stage knee OA and joint pain. Little or no information is available on the prevalence of MME and medial tibial osteophytes or their relationship in elderly individuals.
In the present study, elderly individuals in a population-based large cohort, the Bunkyo Health Study (BHS)32,35,36,37,38, were subjected to radiographic and MRI examinations to analyze knee OA changes. We developed a new method to analyze both the cartilage and bone parts of osteophytes by pseudo-coloring the MRI views and studied the correlations between MME and medial tibial osteophyte width along with the prevalence of MME.
Results
Characteristics of the subjects
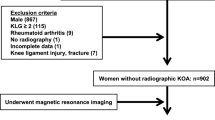
Among 1630 subjects with BHS, 1191 who underwent both radiography in the standing position and MRI of the knee joint were enrolled in the present study (Table 1). While 43.1% (513 of 1191 subjects) of the subjects were male, 56.9% (678 of 1191 subjects) were female. The mean body mass index (BMI) was 22.8 kg/m2 on average. All subjects showed knee joint changes greater than Kellgren-Laurence (K/L) grade 1, and most of the subjects (72.8%, 867 of 1191 subjects) had knee OA with K/L grade 2 on radiography. The femorotibial angle (FTA) was 177.3° on an average. The pain visual analog score (0–100) of the subjects was 8.7 ± 16.3, indicating that the subjects had almost no or weak joint pain.
Application of the pseudo-colored proton density-weighted fat-suppressed (PPDFS) MRI method to osteophyte evaluations
To examine whether or not the PPDFS MRI method could be applied for the evaluation of osteophytes, we compared the findings of osteophytes obtained by proton density-weighted fat-suppressed (PDFS) MRI, PPDFS MRI, T2 mapping MRI, and histology of surgically removed samples from 10 knee OA patients who underwent uni-compartmental arthroplasty (UKA) (Supplemental Table 1). As shown in Supplemental Fig. 1, the cartilage and bone parts of the medial tibial osteophyte could be more clearly detected by the PPDFS MRI view than by the PDFS MRI view. The cartilage-part width and full-length (cartilage and bone parts) width of osteophytes in each patient were separately measured by T2 mapping MRI, PPDFS MRI, PDFS MRI, and histology, the latter of which was performed on the removed osteophytes (Supplemental Table 2). When the osteophyte widths were compared between histology and PPDFS MRI data, histology and T2 mapping MRI data, or PPDFS MRI and T2 mapping MRI data, the interclass correlation coefficient (ICC) was extremely high, with a mean value of > 0.88. In contrast, the values obtained by comparison between histology and PFDS MRI data, PDFS MRI and PPDFS MRI, or PDFS MRI and T2 mapping MRI data were relatively low (ICC: 0.53 to 0.87) (Supplemental Table 3). Based on these results, the PPDFS MRI method was considered suitable for measuring osteophyte width in subjects in this cohort.
Correlations between medial meniscus extrusion (MME) and medial tibial osteophyte width. Associations between MME and the bone-part width of osteophyte (A) and between MME and the full-length (cartilage and bone parts) width of osteophyte (B) were analyzed by the Spearman’s rank correlation analysis. r, correlation coefficient; ICC, intraclass correlation coefficient; CI, confidence interval.
MME and osteophytes in elderly subjects
Using the PDFS MRI and PPDFS MRI data, we analyzed the knee joints of all 1191 subjects in our cohort. As shown in Supplemental Figure 2, MME was determined using PDFS MRI, and osteophyte width was evaluated using PPDFS MRI. MME was observed in almost all subjects (98.7%; 1175 of 1191 subjects), and the mean ± standard deviation (SD) of MME width was 3.90 ± 2.06 mm (Table 2). The prevalence rates of medial femoral and tibial osteophytes (cartilage and bone parts of osteophytes) were 99.1% (1180 of 1191 subjects) and 99.3% (1183 of 1191 subjects), respectively (Table 2). The full-length width of the medial femoral osteophyte was 3.51 ± 1.98 mm, which was calculated by summing the cartilage-part width (1.39 ± 1.05 mm) and the bone-part width (2.13 ± 1.78 mm) of the medial femoral osteophyte in each subject (Table 2). Similarly, the full-length width of the medial tibial osteophyte was 3.27 ± 1.50 mm, and the width of cartilage and bone parts of osteophyte was 1.44 ± 1.09 mm and 1.83 ± 1.26 mm, respectively (Table 2).
MME is most strongly associated with medial tibial osteophyte width among MRI-detected OA structural changes
We scored the OA structural changes observed by PDFS MRI according to Whole-Organ Magnetic Resonance Imaging Score (WORMS) and examined the associations between OA changes and MME (Table 3). All MRI-detected OA structural alterations, including those of cartilage, bone marrow lesions (BMLs), subchondral bone attrition (SBA), subchondral bone cysts (SBCs), osteophytes, meniscus, and the WORMS total score, were associated with MME. Furthermore, the bone-part and full-length widths of osteophytes in the femur were also correlated with MME (r = 0.410 [95% confidence interval [CI] 0.362–0.456] and r = 0.481 [95% CI 0.436–0.524], p < 0.001, respectively). Similarly, the bone-part and full-length width of osteophytes in the tibia were also correlated with MME (r = 0.473 [95% CI 0.428–0.516] and r = 0.809 [95% CI 0.788–0.828], p < 0.001, respectively) (Table 3). A multiple regression analysis indicated that, among these MRI-detected OA structural changes, the full-length width of the medial tibial osteophyte was the most closely associated with MME (β = 0.713; p < 0.001) (Table 4). When the subjects were divided into three subgroups according to the radiographic severity of knee OA, associations between MME and the full-length width of the medial tibial osteophyte were also observed in each subgroup (K/L1: r = 0.711, p < 0.001; K/L2: r = 0.764, p < 0.001; K/L3 and 4: r = 0.820, p < 0.001). These data suggest strong conformity between the MME and medial tibial osteophyte width.
Association of MME with the full-length width of medial tibial osteophytes
Next, we examined the conformity between the MME and the medial tibial osteophyte width. When the bone part of the osteophyte was evaluated, the ICC between the bone part width of the medial tibial osteophyte and the MME was 0.304 (95% CI − 0.079 to 0.576) (Fig. 1A). However, the ICC improved to 0.758 (95% CI 0.578–0.846) when the full-length width of osteophytes was evaluated (Fig. 1B). When the subjects were divided into three groups according to the difference between MME and the full-length width of medial tibial osteophytes, most of the subjects (96.7%; 1152 of 1191 subjects) showed an MME equal to or longer than the full-length width of osteophytes. Furthermore, 66.6% of subjects (793 of 1191 subjects) showed an MME equal to the full-length width of osteophytes ([MME]—[medial tibial osteophyte width] < ± 1 mm), and 30.1% of subjects (359 of 1191 subjects) showed an MME longer than the full-length width of osteophytes ([MME]—[medial tibial osteophyte width] > 1 mm). Only a few subjects (3.3%; 39 of 1191 subjects) showed an MME shorter than the full-length width of osteophytes ([MME]—[medial tibial osteophyte width] < − 1 mm).
Discussion
In the present study, we have demonstrated that MME and medial tibial osteophytes are constantly observed in elderly individuals who complain of no or weak knee joint symptoms, such as pain, and that the degree of MME is consistent with the full-length width of medial tibial osteophytes. Our data in the current study have confirmed the previous finding that radiographic knee OA is commonly observed in elderly people, showing a prevalence of knee OA with K/L grade ≥ 2 in more than 80% of women ≥ 80 years old4, and further provided, to the best of our knowledge, the first evidence that > 98% of elderly individuals exhibit MME and medial tibial osteophytes, which have a direct correlation with each other.
MME is a risk factor for knee OA incidence and progression19,39,40. In addition, we recently reported that among the MRI-detected structural changes of the OA knee joint, MME is a crucial factor for reduced walking speed6, which is one of the determinants of the remaining life span for elderly populations41. Since meniscus tear and/or meniscus degeneration causes elongation of the meniscus42, MME readily develops when knee joints suffer from these meniscal disorders42. However, MME is known to commonly occur without meniscus tear or destruction not only in early-stage knee OA but also in established knee OA patients21,23,24,25,26,27, suggesting a different mechanism for MME development from the meniscus problem. Although no data were available on the prevalence of MME in large-scale population-based cohort studies, our study using conventional PDFS MRI provides the first evidence that MME develops in almost all elderly individuals.
Osteophytes were originally thought to be a secondary process for repair after damage of the articular cartilage in knee OA43, but histological studies on experimental knee OA models demonstrated that osteophyte formation can be seen within two to three days after OA induction at a stage prior to cartilage destruction, indicating a fast process independent of cartilage repair28. Osteophytes play a role in joint stabilization and can be a source of pain and function loss28. In addition, accumulating evidence has revealed that osteophytes are a risk factor for the incidence and progression of knee OA44,45,46,47,48. Osteophytes are formed in the periosteum at the bone-cartilage junction of the joint following the process of the endochondral ossification and are composed of cartilage and bone parts. Thus, the diagnosis of osteophytes using radiographic and conventional PDFS MRI methods is disadvantageous because radiography is unable to detect cartilage tissue, and PDFS MRI is challenging in this regard, resulting in an underestimation of the incidence and size of osteophytes. To overcome this problem, T2 mapping MRI is known to be useful for the evaluation of osteophytes21, since this method can detect cartilage and its degeneration, which is reflected by the water content changes and disarrangement of type II collagen in the tissue. However, because of its relatively time-consuming nature, T2 mapping MRI is demanding for application in large-scale population-based cohort studies. Therefore, in the present study, we developed and applied the PPDFS MRI method to evaluate osteophytes. The PPDFS images were comparable to T2 mapping, showing a higher contrast resolution than T2 mapping images, and are applicable for the evaluation of osteophytes in cohort studies.
One of the important findings in the present study is that, among the OA structural changes, MME was highly consistent with the full-length width of medial tibial osteophytes determined by PPDFS MRI. In our previous study on early-stage knee OA patients, we found a similar correlation between MME and the width of medial tibial osteophytes measured by T2 mapping MRI and hypothesized that osteophytes may contribute to medial displacement of the meniscus, leading to MME21. As illustrated in Fig. 2, the anatomy of the knee joint indicates that the medial meniscus is tightly attached to the medial tibial plateau by the meniscotibial ligament, i.e. the coronary ligament34,49. Thus, once osteophytes are formed in the periosteum at the bone-cartilage junction of the medial tibia, it is plausible to speculate that the local mechanical stretching force occurring during joint movement may displace the medial meniscus to induce MME (Fig. 2). Although our previous study on early-stage knee OA analyzed only 50 cases, in the present study, we demonstrated that MME was equal to or longer than the full-length width of the osteophyte in 96.7% of subjects (1152 of 1191 subjects), and only a few cases (3.3%, 39 of 1191 subjects) showed an MME shorter than the osteophyte width. Since the introduction of MRI for the diagnosis of knee OA, growing evidence obtained from studies using MRI has demonstrated that osteophytes are formed at very early stages of knee OA12,21, suggesting that cartilage degradation and damage occur due to the load concentration onto the articular cartilage uncovered by the meniscus, leading to OA progression39. Taken together, one could speculate that osteophytes may contribute to the development of MME, which promotes meniscus injury and cartilage damage, resulting in OA progression12,21. However, further research is required to provide direct evidence supporting this hypothesis.
A schematic illustration showing the hypothesis on medial meniscus extrusion (MME) induced by osteophyte formed at the bone-cartilage junction of the medial tibia. Osteophytes are formed in the periosteum at the bone-cartilage junction following endochondral ossification and are thus composed of cartilage and bone parts. The most important anatomical characteristic is that the medial meniscus is tightly attached to the medial tibial plateau by the menisco-tibial ligament (coronary ligament). Therefore, the mechanical stretching force generated during joint movement because of the medial tibial osteophyte may contribute to the displacement of the medial meniscus by pressing the menisco-tibial ligament, leading to MME.
Although the present study demonstrated an extremely high incidence of MME and medial tibial osteophytes, both of which are directly correlated, evidence that osteophytes cause MME was not obtained because it was a cross-sectional study. However, since the PPDFS MRI method can be used to monitor changes in the osteophyte and meniscus by re-analyzing the ordinary MRI images already taken in these elderly patients, early-stage knee OA patients, or patients with anterior cruciate ligament injury, longitudinal studies on these subjects or patients, which are now underway in our group, would provide us with data on the relationship. After elucidating the causal relationship between osteophytes and MME, studies on the molecular mechanism of osteophyte formation and the search for their regulators30,31,50,51,52,53,54 might become important to develop disease-modifying therapies that can inhibit the progression of knee OA and/or prevent OA development.
Several limitations associated with the present study warrant mention. First, our cohort included only subjects living in an urban area in Japan. Therefore, the data obtained in the present study may not be applicable to elderly people living in other areas, such as mountainous or coastal areas, and the results cannot be generalized. Second, we developed the PPDFS MRI method by pseudo-colorization of PDFS images obtained from patients with symptomatic knee OA, but not from normal subjects. Therefore, whether or not this method is sufficient to evaluate subtle changes in the knee joints, including osteophytes, in healthy elderly individuals cannot be guaranteed. However, we have shown that the MRI views obtained by PPDFS MRI are almost equivalent to those obtained by T2 mapping MRI, and the widths of the cartilage and bone parts of osteophytes detected by PPDFS MRI were histologically confirmed. Because the method does not take much time for an analysis, it is suitable to evaluate numerous image samples using the ImageJ software program55. The accuracy of this method should be further assessed by analyzing the knee joints of human subjects, including cadavers. Third, the method of evaluating MRI-detected OA changes in the knee joint used in this study was limited. In particular, because the same observers measured both MME and osteophyte width, there is a risk of confirming our own hypothesis. This is one of the biases in the present study. In addition, while the accuracy of the PPDFS MRI method was evaluated using 3.0-Tesla MRI by measuring the osteophyte width of OA patients who received UKA, we measured the osteophyte width of the subjects in our cohort using 0.3-Tesla MRI. Therefore, we cannot exclude the possibility that the different MRI systems used for the analyses may have introduced a certain bias in the present study.
In conclusion, in elderly populations, MME and medial tibial osteophytes are frequently observed, and the degree of MME is consistent with the full-length width of the medial tibial osteophytes.
Methods
Subjects of the cohort study
We organized the BHS, a prospective cohort study over 10 years to identify risk factors for needing long-term care35,36,37,38, and recruited elderly subjects between 65 and 84 years old living in Bunkyo-ku, an urban area in Tokyo, Japan. A total of 1630 subjects participated from November 2015 to September 2018. The exclusion criteria of this cohort were elderly individuals who underwent pacemaker or defibrillator implantation and/or had diabetes requiring insulin therapy.
The study protocol was approved by the ethics committee of Juntendo University in November 2015 (Nos. 2015078, 2016138, 2016131, and 2017121). This study was conducted in accordance with the principles of the Declaration of Helsinki. All participants provided their written informed consent at orientation meetings. Participants were informed that they had the right to withdraw from the trial at any time. The collected data were coded with nonidentifying numbers and stored securely in password-protected files. Accessibility to files was limited to the principal investigators.
The radiographic evaluation of knee OA
The radiographic OA severity was evaluated according to the Kellgren-Lawrence (K/L) classification56 of the weight-bearing anteroposterior radiographs of the femorotibial joint for both knees using the bilateral standing extended view and the weight-bearing posteroanterior radiographs of the femorotibial joint with the knee flexed57. The femorotibial angle (FTA) was evaluated based on weight-bearing anteroposterior radiographs of the lower limbs.
Development of the PPDFS MRI method for evaluation of osteophytes
We previously reported the usefulness of T2 mapping MRI for the analysis of osteophytes, since T2 mapping MRI could detect the cartilage part as well as the bone part of osteophytes, so the full-length width (width of cartilage and bone parts) of osteophytes measured by T2 mapping MRI corresponded well to the width obtained by histology21. However, T2 mapping MRI is not commonly available in clinical situations and is still challenging for a large cohort study because it requires a longer scan time and requires longer post-imaging processing than conventional PDFS MRI. To overcome these limitations, we developed a new method, i.e. PPDFS MRI method32. For this method, MRI views of the knee joints obtained by conventional PDFS MRI were pseudo-colored by selecting “Royal” as the color, which allowed the cartilage to be represented by an intermediate color using the ImageJ software program55.
The accuracy of the PPDFS MRI method in measuring the full-length width of osteophytes was examined using 10 samples from 10 knee OA patients who underwent both PDFS MRI and T2 mapping MRI analyses before the operation and then underwent UKA (Supplemental Table 1). To compare the findings of MRI and histology of osteophytes in the knee joints, the knee joints were analyzed by a 3.0-Tesla MRI system (Siemens Magnetom Verio; Siemens Healthcare, Erlangen, Germany) using the following conditions: coronal T2FS spin-echo images, repetitive time [TR] / echo time [TE] = 2000/25 ms; slice thickness = 3.5 mm and in-plane resolution 0.36 × 0.36 mm; and sagittal T2FS spin-echo images, [TR] / [TE] = 2100/22 ms; slice thickness = 3.5 mm and in-plane resolution 0.36 × 0.36 mm. PPDFS MRI views were prepared by pseudo-coloring PDFS MRI using the ImageJ software program, as described previously32. T2 mapping MRI views were obtained as described in our previous study21 using the following conditions: coronal T2 mapping images, [TR] / [TE] = 1000/13.8, 27.6, 41.4, 55.2, and 69.0 ms; slice thickness, 3 mm; and in-plane resolution, 0.42 × 0.42 mm. The surgically removed tibial tissue was fixed in 10% buffered formalin. After decalcification, the osteophytes were cut vertically in the center from the outer border to the bottom, and serial paraffin sections were stained with hematoxylin and eosin and Safranin O. The width of the osteophytes was determined by measuring the cartilage and bone parts using the cellSens software program (Olympus, Tokyo, Japan).
The study protocol complied with the principles outlined in the Declaration of Helsinki and was approved by the Ethical Committee Review Board of Juntendo University (No. 15-074). Written informed consent was obtained from the patients with knee OA who underwent UKA.
MRI settings in the cohort study
The knee joints of the subjects of the BHS were analyzed using a 0.3-Tesla MRI system (Hitachi AIRIS Vento; Hitachi Medical Corporation, Tokyo, Japan) with a knee coil. Coronal PDFS and PPDFS spin-echo sequences (TR / TE = 1500/21.2 ms; in-plane resolution = 0.39 × 0.39 mm2; section thickness = 3 mm) were used for MME and osteophyte measurement. MRI data were evaluated in the medial femorotibial joint, and OA morphological changes were scored according to the Whole Organ Magnetic Resonance Imaging Score (WORMS)58. Each region of a compartment received its own score, which was added together21. Tears in the medial meniscus were observed in 42% (499/1145) of the subjects, while the remaining 58% (692/1145) showed no damage in the medial meniscus (Kaneko, H., et al. manuscript in preparation).
The evaluation of MME and osteophytes
Single slices of PPDFS MRI images presenting the largest width of the tibial plateau (greatest area of the medial spine) were selected for measurements of the MME and osteophyte width59. Observers who were blinded to the characteristics of the subjects measured MME and osteophyte width from the end of the normal tibial plateau to the outermost edge of the body of the meniscus and osteophyte end, respectively, using the ImageJ software program. The cartilage and bone partsof the osteophytes were measured separately.
Reproducibility of measurements
One observer (TA) assessed all MME and osteophyte widths twice, and the second observer (AA) assessed 154 randomly selected knees. Intra-observer reliability (intra-class correlation coefficient) was 0.94 (95% confidence interval [CI] 0.94–0.95) for MME and 0.92 (95% CI 0.91–0.92) for osteophytes, and inter-observer reliability (inter-class correlation coefficient) was 0.97 (95% CI 0.96–0.98) for MME and 0.96 (95% CI 0.95–0.97) for osteophytes.
Statistical analyses
The associations between MME and other MRI-detected OA structural alterations were analyzed using Spearman's rank correlation and multiple regression analyses. The consistency between MME and medial tibial osteophyte width was also analyzed using the ICC with a two-way random model and absolute agreement type. Statistical significance was set at p values < 0.05. All analyses were performed using the SPSS 27 software program (SPSS Institute, Chicago, IL, USA).
Ethics approval and consent to participate
The study protocol was approved by the ethics committee of Juntendo University in November 2015 (Nos. 2015078, 2016138, 2016131, and 2017121). This study was conducted in accordance with the principles of the Declaration of Helsinki. All participants provided their written informed consent at orientation meetings. Participants were informed that they had the right to withdraw from the trial at any time. The collected data were coded with nonidentifying numbers and stored securely in password-protected files. Accessibility to files is limited to the principal investigators.
Data availability
This study is a longitudinal study, and the data are not publicly available for use by the general public. The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
References
Felson, D. T. Clinical practice. Osteoarthritis of the knee. N. Engl. J. Med. 354, 841–848. https://doi.org/10.1056/NEJMcp051726 (2006).
Katz, J. N., Arant, K. R. & Loeser, R. F. Diagnosis and treatment of hip and knee osteoarthritis: A review. JAMA 325, 568–578. https://doi.org/10.1001/jama.2020.22171 (2021).
Felson, D. T. et al. The prevalence of knee osteoarthritis in the elderly. The Framingham Osteoarthritis Study. Arthritis Rheumatol. 30, 914–918. https://doi.org/10.1002/art.1780300811 (1987).
Yoshimura, N. et al. Prevalence of knee osteoarthritis, lumbar spondylosis, and osteoporosis in Japanese men and women: The research on osteoarthritis/osteoporosis against disability study. J. Bone Miner. Metab. 27, 620–628. https://doi.org/10.1007/s00774-009-0080-8 (2009).
Ishijima, M. et al. Osteoarthritis as a cause of locomotive syndrome: Its influence on functional mobility and activities of daily living. Clin. Rev. Bone Miner. Metab. 14, 77–104. https://doi.org/10.1007/s12018-016-9212-6 (2016).
Arita, H. et al. Medial meniscus extrusion is a determinant factor for the gait speed among MRI-detected structural alterations of knee osteoarthritis. Osteoarthr. Cartil. Open 3, 100176. https://doi.org/10.1016/j.ocarto.2021.100176 (2021).
Ishijima, M. et al. Different changes in the biomarker C-terminal telopeptides of type II collagen (CTX-II) following intra-articular injection of high molecular weight hyaluronic acid and oral non-steroidal anti-inflammatory drugs in patients with knee osteoarthritis: A multi-center randomized controlled study. Osteoarthritis Cartilage 30, 852–861. https://doi.org/10.1016/j.joca.2022.03.003 (2022).
Rosenberg, A. In Robbins Pathologic Basis of Disease 6th edn (eds Cotran, R. S. et al.) 1246–1248 (W.B. Saunders Company, 1999).
Berenbaum, F. Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthritis Cartilage 21, 16–21. https://doi.org/10.1016/j.joca.2012.11.012 (2013).
Luyten, F. P., Denti, M., Filardo, G., Kon, E. & Engebretsen, L. Definition and classification of early osteoarthritis of the knee. Knee Surg. Sports Traumatol. Arthrosc. 20, 401–406. https://doi.org/10.1007/s00167-011-1743-2 (2012).
Mahmoudian, A., Lohmander, L. S., Mobasheri, A., Englund, M. & Luyten, F. P. Early-stage symptomatic osteoarthritis of the knee—time for action. Nat. Rev. Rheumatol. 17, 621–632. https://doi.org/10.1038/s41584-021-00673-4 (2021).
Guermazi, A. et al. Prevalence of abnormalities in knees detected by MRI in adults without knee osteoarthritis: Population based observational study (Framingham Osteoarthritis Study). BMJ 345, e5339–e5339. https://doi.org/10.1136/bmj.e5339 (2012).
Ishijima, M., Kurosawa, H., Kaneko, H. & Kaneko, K. In Arthritis: Pathophysiology, Prevention, and Therapeutics (eds Bagchi, D. et al.) 39–54 (CRC Press, 2011).
Hada, S. et al. The degeneration and destruction of femoral articular cartilage shows a greater degree of deterioration than that of the tibial and patellar articular cartilage in early stage knee osteoarthritis: A cross-sectional study. Osteoarthritis Cartilage 22, 1583–1589. https://doi.org/10.1016/j.joca.2014.07.021 (2014).
Englund, M. et al. Incidental meniscal findings on knee MRI in middle-aged and elderly persons. N. Engl. J. Med. 359, 1108–1115. https://doi.org/10.1056/NEJMoa0800777 (2008).
Roemer, F. W. et al. MRI-detected subchondral bone marrow signal alterations of the knee joint: Terminology, imaging appearance, relevance and radiological differential diagnosis. Osteoarthritis Cartilage 17, 1115–1131. https://doi.org/10.1016/j.joca.2009.03.012 (2009).
Englund, M., Roemer, F. W., Hayashi, D., Crema, M. D. & Guermazi, A. Meniscus pathology, osteoarthritis and the treatment controversy. Nat. Rev. Rheumatol. 8, 412–419. https://doi.org/10.1038/nrrheum.2012.69 (2012).
Gale, D. R. et al. Meniscal subluxation: association with osteoarthritis and joint space narrowing. Osteoarthritis Cartilage 7, 526–532. https://doi.org/10.1053/joca.1999.0256 (1999).
Sharma, L. et al. Relationship of meniscal damage, meniscal extrusion, malalignment, and joint laxity to subsequent cartilage loss in osteoarthritic knees. Arthritis Rheumatol. 58, 1716–1726. https://doi.org/10.1002/art.23462 (2008).
Hunter, D. J. et al. The association of meniscal pathologic changes with cartilage loss in symptomatic knee osteoarthritis. Arthritis Rheumatol. 54, 795–801. https://doi.org/10.1002/art.21724 (2006).
Hada, S. et al. Association of medial meniscal extrusion with medial tibial osteophyte distance detected by T2 mapping MRI in patients with early-stage knee osteoarthritis. Arthritis Res. Ther. 19, 201. https://doi.org/10.1186/s13075-017-1411-0 (2017).
Snoeker, B. A. M. et al. Are structural abnormalities on knee MRI associated with osteophyte development? Data from the Osteoarthritis Initiative. Osteoarthritis Cartilage 29, 1701–1708. https://doi.org/10.1016/j.joca.2021.06.012 (2021).
Costa, C. R., Morrison, W. B. & Carrino, J. A. Medial meniscus extrusion on knee MRI: is extent associated with severity of degeneration or type of tear?. AJR Am. J. Roentgenol. 183, 17–23. https://doi.org/10.2214/ajr.183.1.1830017 (2004).
Lerer, D. B., Umans, H. R., Hu, M. X. & Jones, M. H. The role of meniscal root pathology and radial meniscal tear in medial meniscal extrusion. Skelet. Radiol. 33, 569–574. https://doi.org/10.1007/s00256-004-0761-2 (2004).
Crema, M. D. et al. Factors associated with meniscal extrusion in knees with or at risk for osteoarthritis: The Multicenter Osteoarthritis study. Radiology 264, 494–503. https://doi.org/10.1148/radiol.12110986 (2012).
Park, H. J. et al. Medial meniscal root tears and meniscal extrusion transverse length ratios on MRI. Br. J. Radiol. 85, e1032-1037. https://doi.org/10.1259/bjr/26261821 (2012).
Yoon, S. J., Ahn, J. M., Kang, Y., Kim, B. R. & Kang, H. S. Morphological changes in the superficial medial collateral ligament on knee MR imaging: association with medial meniscal extrusion and posterior root medial meniscus abnormality. Skelet. Radiol. 51, 1399–1405. https://doi.org/10.1007/s00256-021-03978-3 (2022).
van der Kraan, P. M. & van den Berg, W. B. Osteophytes: relevance and biology. Osteoarthritis Cartilage 15, 237–244. https://doi.org/10.1016/j.joca.2006.11.006 (2007).
Kaneko, H. et al. Reference intervals of serum hyaluronic acid corresponding to the radiographic severity of knee osteoarthritis in women. BMC Musculoskelet. Disord. 14, 34. https://doi.org/10.1186/1471-2474-14-34 (2013).
Sadatsuki, R. et al. Perlecan is required for the chondrogenic differentiation of synovial mesenchymal cells through regulation of Sox9 gene expression. J. Orthop. Res. 35, 837–846. https://doi.org/10.1002/jor.23318 (2017).
Roelofs, A. J. et al. Identification of the skeletal progenitor cells forming osteophytes in osteoarthritis. Ann. Rheum. Dis. 79, 1625–1634. https://doi.org/10.1136/annrheumdis-2020-218350 (2020).
Adili, A. et al. Anterior meniscus extrusion is associated with anterior tibial osteophyte width in knee osteoarthritis—The Bunkyo Health Study. Osteoarthr. Cartil. Open 5, 100364. https://doi.org/10.1016/j.ocarto.2023.100364 (2023).
De Maeseneer, M. et al. Normal and abnormal medial meniscocapsular structures: MR imaging and sonography in cadavers. AJR Am. J. Roentgenol. 171, 969–976. https://doi.org/10.2214/ajr.171.4.9762977 (1998).
Smigielski, R., Becker, R., Zdanowicz, U. & Ciszek, B. Medial meniscus anatomy-from basic science to treatment. Knee Surg. Sports Traumatol. Arthrosc. 23, 8–14. https://doi.org/10.1007/s00167-014-3476-5 (2015).
Someya, Y. et al. Skeletal muscle function and need for long-term care of urban elderly people in Japan (The Bunkyo Health Study): a prospective cohort study. BMJ Open 9, e031584. https://doi.org/10.1136/bmjopen-2019-031584 (2019).
Someya, Y. et al. Reduced muscle strength of knee extensors is a risk factor for silent lacunar infarcts among Japanese elderly people: The Bunkyo Health Study. JCSM Clin. Rep. 5, 79–85. https://doi.org/10.1002/crt2.24 (2020).
Someya, Y. et al. Insulin resistance and muscle weakness are synergistic risk factors for silent lacunar infarcts: The Bunkyo Health Study. Sci. Rep. 11, 21093. https://doi.org/10.1038/s41598-021-00377-5 (2021).
Otsuka, H. et al. Associations of exercise habits in adolescence and old age with risk of osteoporosis in older adults: The Bunkyo Health Study. J. Clin. Med. 10, 5968. https://doi.org/10.3390/jcm10245968 (2021).
Roemer, F. W. et al. Co-localisation of non-cartilaginous articular pathology increases risk of cartilage loss in the tibiofemoral joint–the MOST study. Ann. Rheum. Dis. 72, 942–948. https://doi.org/10.1136/annrheumdis-2012-201810 (2013).
Emmanuel, K. et al. Quantitative measures of meniscus extrusion predict incident radiographic knee osteoarthritis—data from the Osteoarthritis Initiative. Osteoarthritis Cartilage 24, 262–269. https://doi.org/10.1016/j.joca.2015.08.003 (2016).
Studenski, S. et al. Gait speed and survival in older adults. JAMA 305, 50–58. https://doi.org/10.1001/jama.2010.1923 (2011).
Bhatia, S., LaPrade, C. M., Ellman, M. B. & LaPrade, R. F. Meniscal root tears: significance, diagnosis, and treatment. Am. J. Sports Med. 42, 3016–3030. https://doi.org/10.1177/0363546514524162 (2014).
Felson, D. T. et al. Osteophytes and progression of knee osteoarthritis. Rheumatology (Oxford) 44, 100–104. https://doi.org/10.1093/rheumatology/keh411 (2005).
Dieppe, P. A., Cushnaghan, J. & Shepstone, L. The Bristol “OA500” Study: Progression of osteoarthritis (OA) over 3 years and the relationship between clinical and radiographic changes at the knee joint. Osteoarthritis Cartilage 5, 87–97. https://doi.org/10.1016/S1063-4584(97)80002-7 (1997).
Boegard, T., Rudling, O., Petersson, I. F. & Jonsson, K. Correlation between radiographically diagnosed osteophytes and magnetic resonance detected cartilage defects in the tibiofemoral joint. Ann. Rheum. Dis. 57, 401–407. https://doi.org/10.1136/ard.57.7.401 (1998).
Felson, D. T. The sources of pain in knee osteoarthritis. Curr. Opin. Rheumatol. 17, 624–628. https://doi.org/10.1097/01.bor.0000172800.49120.97 (2005).
Zhu, Z. et al. Association between MRI-detected osteophytes and changes in knee structures and pain in older adults: A cohort study. Osteoarthritis Cartilage 25, 1084–1092. https://doi.org/10.1016/j.joca.2017.01.007 (2017).
Zhu, Z. et al. MRI-detected osteophytes of the knee: Natural history and structural correlates of change. Arthritis Res. Ther. 20, 237. https://doi.org/10.1186/s13075-018-1734-5 (2018).
El-Khoury, G. Y., Usta, H. Y. & Berger, R. A. Meniscotibial (coronary) ligament tears. Skeletal. Radiol. 11, 191–196. https://doi.org/10.1007/BF00349493 (1984).
Kaneko, H. et al. Synovial perlecan is required for osteophyte formation in knee osteoarthritis. Matrix Biol. 32, 178–187. https://doi.org/10.1016/j.matbio.2013.01.004 (2013).
Shiozawa, J. et al. Implication of HYBID (hyaluronan-binding protein involved in hyaluronan depolymerization) in hyaluronan degradation by synovial fibroblasts in patients with knee osteoarthritis. Am. J. Pathol. 190, 1046–1058. https://doi.org/10.1016/j.ajpath.2020.01.003 (2020).
Momoeda, M. et al. Deletion of hyaluronan-binding protein involved in hyaluronan depolymerization (HYBID) results in attenuation of osteoarthritis in mice. Am. J. Pathol. 191, 1986–1998. https://doi.org/10.1016/j.ajpath.2021.07.008 (2021).
Shiozawa, J. et al. Expression and regulation of recently discovered hyaluronidases, HYBID and TMEM2, in chondrocytes from knee osteoarthritic cartilage. Sci. Rep. 12, 17242. https://doi.org/10.1038/s41598-022-22230-z (2022).
Negishi, Y. et al. Interleukin-6 reduces spheroid sizes of osteophytic cells derived from osteoarthritis knee joint via induction of apoptosis. Am. J. Pathol. S0002-9440(23)00411-X. https://doi.org/10.1016/j.ajpath.2023.10.005 (2023).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675. https://doi.org/10.1038/nmeth.2089 (2012).
Kellgren, J. H. & Lawrence, J. S. Radiological assessment of osteo-arthrosis. Ann. Rheum. Dis. 16, 494–502. https://doi.org/10.1136/ard.16.4.494 (1957).
Buckland-Wright, J. C., Macfarlane, D. G., Williams, S. A. & Ward, R. J. Accuracy and precision of joint space width measurements in standard and macroradiographs of osteoarthritic knees. Ann. Rheum. Dis. 54, 872–880. https://doi.org/10.1136/ard.54.11.872 (1995).
Peterfy, C. G. et al. Whole-organ magnetic resonance imaging score (WORMS) of the knee in osteoarthritis. Osteoarthritis Cartilage 12, 177–190. https://doi.org/10.1016/j.joca.2003.11.003 (2004).
Zhang, F. et al. Risk factors for meniscal body extrusion on MRI in subjects free of radiographic knee osteoarthritis: Longitudinal data from the Osteoarthritis Initiative. Osteoarthritis Cartilage 24, 801–806. https://doi.org/10.1016/j.joca.2015.12.003 (2016).
Acknowledgements
We express our deep appreciation to the members of the Laboratory of Molecular and Biochemical Research, Research Support Center, Juntendo University Graduate School of Medicine for their technical assistance. This study was supported in part by a Grant-in-Aid for Scientific Research from the Japanese Society for the Promotion of Science (JSPS) to MI (15K10494 and 18K09082), HK (15K20019, 18K09083, and 21K09259), SH (16K20069 and 23K08705), TA (19K17209), JS (20K18075 and 23K15750), MM (22K16774), and YO (16H05454 and 22K07029). This study was also funded in part by a High Technology Research Center Grant and the Program for the Strategic Research Foundation at Private Universities (2014-2019) from the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT) and by the Center of Innovation (COI) program, one of the main funding programs under the Center of Innovation Science and Technology based Radical Innovation and Entrepreneurship Program (COI STREAM), which was launched in 2013 by MEXT and operated by the Japan Science and Technology Agency (JST), to Juntendo University. This study was also supported in part by the Strategic International Collaborative Research Program (SICORP) from the Japan Agency for Medical Research and Development (AMED) and the NordForsk (the Academy of Finland, the Research Council of Norway, and the Swedish Research Council), and by the Japanese Orthopaedic Association (JOA)-Subsidized Science Project Research 2022-2024 to MI.
Author information
Authors and Affiliations
Contributions
Y.N., H.K., Y.S., Y.T., and M.I. conceived and designed the study. Y.N., T.A., L.L., A.A., H.A., M.M., H.H., J.T., S.W., and J.S. collected and registered the data. Y.N., H.K., T.A., L.L., A.A., S.H., M.K., S.A., H.W., R.K., T.N.K., Y.O., and M.I. played major roles in the analysis and interpretation of the data and contributed to drafting the report. H.A., H.K., L.L., Y.O., and M.I. supervised the statistical analysis. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Negishi, Y., Kaneko, H., Aoki, T. et al. Medial meniscus extrusion is invariably observed and consistent with tibial osteophyte width in elderly populations: The Bunkyo Health Study. Sci Rep 13, 22805 (2023). https://doi.org/10.1038/s41598-023-49868-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-49868-7
- Springer Nature Limited