Abstract
The loss of progesterone receptor (PR) often predicts worse biological behavior and prognosis in estrogen receptor-positive (ER +) breast cancer. However, the impact of PR status on inflammatory breast cancer (IBC) has not been studied. Therefore, the purpose of our study was to investigate the influence of PR on IBC. Patients with ER+ and HER2-negative IBC were selected from the Surveillance, Epidemiology and End Results database. Pearson’s χ2 test was used to compare the clinicopathological characteristics between patients with estrogen receptor-positive/progesterone receptor-positive (ER+/PR +) and patients with estrogen receptor-positive/progesterone receptor-negative (ER+/PR−). Univariate and multivariate analyses were performed to investigate the effects of PR status on the breast cancer-specific survival (BCSS) and overall survival (OS) in IBC. Overall, 1553 patients including 1157 (74.5%) patients with ER+/PR+ and 396 (25.5%) patients with ER+/PR− were analyzed in our study. The patients with ER+/PR− were more likely to be high histological grade (p < 0.001) and liver metastasis (p = 0.045) compared to patients with ER+/PR+. Despite higher chance of receiving chemotherapy (83.6% vs 77.3%, P = 0.008), patients with ER+/PR− showed worse BCSS (5-year BCSS rate, 34.3% vs 51.3%, P < 0.001) and OS (5-year OS rate, 31.3% vs 46.1%, P < 0.001) compared with ER+/PR+ phenotype. Multivariate survival analysis showed that patients with ER+/PR− still had worse BCSS (hazard ratios [HR]: 1.764, 95% confidence intervals [CI] 1.476–2.109, P < 0.001) and OS (HR: 1.675, 95% CI 1.411–1.975, P < 0.001) than ER+/PR+ phenotype. Furthermore, patients with ER+/PR− showed worse outcomes than ER+/PR+ phenotype in most subgroups, especially in patients with younger age (≤ 60 years), lower histological grade, lymph node involved and distant metastasis. Patients with ER+/PR− had more aggressive biological behaviors and worse outcomes than patients with ER+/PR+ in IBC. Stronger treatments maybe needed for IBC patients with ER+/PR−.
Similar content being viewed by others

Introduction
Inflammatory breast cancer (IBC) is a rare subtype and accounts for 2–4% of all breast malignant tumors1, but it is characterized by aggressive biological behaviors and accounts for 7% of all breast cancer-related death2. Patients with IBC often present rapid progressive pain, erythema, and edema in the involved breast because lymphovascular spaces were embolized by tumor cells. Due to its aggressive behaviors, 85% of the patients already have lymph node involved and 30% of the patients show distant metastasis at the initial diagnosis of IBC3. Trimodality treatment including chemotherapy, surgery and radiation therapy has become a widely accepted approach and significantly improved the survival for IBC, but the overall survival (OS) rates remain very low (5-year and 10-year OS rates, 55.4% and 37.3%, respectively)4.
As non-IBC, IBC can also be divided into different molecular subtypes according to the status of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor 2 (HER2). Endocrine therapy is recommended for IBC patients with estrogen receptor-positive (ER +) and/or progesterone receptor-positive (PR +) by the National Comprehensive Cancer Network (NCCN) guidelines. Many researches have revealed that the status of PR has great prognostic effect on breast cancer and patients with estrogen receptor-positive/progesterone receptor-positive (ER+/PR +) have better outcomes than patients with estrogen receptor-positive/progesterone receptor-negative (ER+/PR−)5,6,7. However, the role of PR status on IBC has not been illuminated because of the lower incidence of IBC. Actually, IBC is a special subtype and its biologic characteristics are distinct from that of non-IBC. IBC is more likely to be estrogen receptor-negative and HER2-positive compared with non-IBC8,9. Besides, some researches have demonstrated that patients with IBC exhibited higher percentage of progesterone receptor-negative status compared with that of patients with non-IBC (55–56.7% versus 32–46.8%)9,10. While, it is still unknown whether the absence of PR expression will lead to worse prognosis of IBC or not. Thus, the purpose of this study was to estimate the differences of clinicopathologic features and prognosis between ER+/PR− phenotype and ER+/PR+ phenotype in IBC by analyzing the patients from Surveillance, Epidemiology and End Results (SEER) database.
Materials and methods
Patient selection
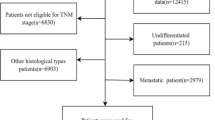
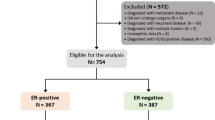
SEER database provides cancer statistics about patients’ demographics, tumor characteristics, methods of treatment and follow-up information, which covers approximately 28% of the United States population. Because the status of HER2 was collected into SEER database since 2010, we used the SEER*Stat version 8.4.0 to identify eligible patients based on the following inclusion criteria: breast cancer, female sex, T4d (cT4d/pT4d), estrogen receptor-positive status, HER2-negtative status and being diagnosed from 2010 to 2018. The excluded criteria were patients with multiple primary tumors, less than 1 month of follow-up, or patients with unknown information about PR status, marital status, lymph node status, distant metastasis and surgery (Fig. 1, flow-chart). Finally, 1553 patients met the criteria and their clinicopathologic data including age, race, marital status, histological grade, lymph node stage, status of PR, sites of distant metastasis, therapeutic regimens and follow-up information were acquired and analyzed. The eligible patients were allocated to two groups (ER+/PR+ phenotype or ER+/PR− phenotype) according the status of PR. After 2010, the status of PR was detected by immunohistochemistry, and negativity was defined as < 1% of malignant cells positively staining for PR11.
Statistical analysis
Pearson’s χ2 test was used to estimate the difference of clinicopathologic factors between ER+/PR− phenotype and ER+/PR+ phenotype in IBC. The endpoints were breast cancer-specific survival (BCSS) and overall survival (OS) in our study. BCSS was defined as the interval from the diagnosis of breast cancer to mortality caused by breast cancer or the final follow-up in censored cases. OS was defined as the interval from the diagnosis of breast cancer to mortality from all causes or the final follow-up in censored cases. Survival curves of the patients with ER+/PR− phenotype or ER+/PR+ phenotype were constructed by the Kaplan–Meier method, and the log-rank test was applied to determine the effect of PR status on BCSS and OS. A Cox proportional hazards model was used for the multivariate analysis and to estimate hazard ratios with 95% confidence intervals (CIs). STATA software (Version 13; Stata Corporation) was applied for all statistical analyses. The forest plot was generated by Microsoft Office Excel (Version 2021; Microsoft Corporation). All tests were two sided and p-value < 0.05 were considered statistically significant.
Ethical approval
This study used previously collected de-identified data, and the need for informed consent had been waived due to the retrospective nature of the study, and was deemed exempt from review by the Ethics Committee of the Affiliated Hospital of North Sichuan Medical College.
Results
Clinicopathologic features
A total of 1553 patients with ER+ and HER2-negative IBC met the criteria and were analyzed in our study. Among them, 1157 (74.5%) patients were ER+/PR+ phenotype and 396 (25.5%) patients were ER+/PR− phenotype. As shown in Table 1, the patients with ER+/PR− phenotype were more likely to be high histological grade (III-IV) compared with ER+/PR+ phenotype (59.8% and 44%, P < 0.001). More bone metastasis happened to patients with ER+/PR+ than patients with ER+/PR− phenotype (29% and 24.5%, P = 0.082), but no statistical difference was reached. While, more liver metastasis happened to patients with ER+/PR− than patients with ER+/PR+ phenotype (11.4% and 8%, P = 0.045). More patients with ER+/PR− phenotype received chemotherapy than patients with ER+/PR+ phenotype (83.6% and 77.3%, P < 0.001). There were no significant differences between ER+/PR− phenotype and ER+/PR+ phenotype in terms of age, race, marital status, lymph node stage, TNM stage, lung metastasis, brain metastasis, surgery and radiation.
Univariate survival analysis
The follow-up time of this cohort ranged from 1 to 107 months, with a median of 25 months. Finally, 724 patients had died and 631 cases of the death were attributed to IBC. The 5-year BCSS and OS rates were 47.3% and 42.5%, respectively. Among the whole cohort, the patients with ER+/PR− phenotype showed worse BCSS (5-year BCSS rate, 34.3% vs 51.3%, P < 0.001) and OS (5-year OS rate, 31.3% vs 46.1%, P < 0.001) compared with patients of ER+/PR+ phenotype (Fig. 2). As shown in Fig. 3, the patients with ER+/PR− phenotype had worse BCSS than patients with ER+/PR+ phenotype in stage III (5-year BCSS rate, 52.8% vs 66.8%, P < 0.001) and stage IV (5-year BCSS rate, 4.4% vs 21.4%, P < 0.001) IBC. After the standard trimodality treatment (chemotherapy, surgery and radiation therapy), the patients with ER+/PR− phenotype still exhibited worse BCSS (5-year BCSS rate, 63.8% vs 72.5%, P = 0.03) and OS (5-year OS rate, 62.2% vs 70.3%, P = 0.014) than patients with ER+/PR+ phenotype in stage III IBC (Fig. 4).
Multivariate survival analysis
Multivariate survival analysis showed that the patients with ER+/PR− phenotype still had worse BCSS (HR: 1.764, 95% CI 1.476–2.109, P < 0.001) and OS (HR: 1.675, 95% CI 1.411–1.975, P < 0.001) than patients with ER+/PR+ phenotype (Table 2). Then, older age (> 60 years), unmarried status, black race, higher histological grade (III–IV), higher lymph node stage (N3) and distant metastasis were independent risk factors for BCSS and OS in patients with ER+ and HER2-ngeative IBC. Breast surgery, radiation and chemotherapy could significantly improve the survival for IBC.
Subgroup survival comparation for ER+/PR+ and ER+/PR− phenotypes
When subgroup analysis was performed by multivariate Cox regression models (Fig. 5), the patients with ER+/PR− phenotype also showed worse outcomes than patients with ER+/PR+ phenotype in most subgroups. Especially in patients with younger age (≤ 60 years) (BCSS, HR: 1.802, 95% CI 1.397–2.323, P < 0.001; OS, HR: 1.928, 95% CI 1.516–2.452, P < 0.001), lower histological grade (BCSS, HR: 2.07, 95% CI 1.455–2.944, P < 0.001), lymph node involved (BCSS, HR: 2.059, 95% CI 1.704–2.487, P < 0.001; OS, HR: 1.944, 95% CI 1.627–2.323, P < 0.001) and distant metastasis (BCSS, HR: 1.846, 95% CI 1.449–2.351, P < 0.001; OS, HR: 1.814, 95% CI 1.436–2.291, P < 0.001), significant worse prognoses were seen in ER+/PR− phenotype compared with ER+/PR+ phenotype.
Discussion
Progesterone receptor (PR) is a downstream gene target of estrogen receptor (ER) and the loss of PR often indicates a poor prognosis in breast cancer7,12,13. Previous studies have shown that ER+/PR− phenotype accounts for 10.5–15% of ER-positive breast cancer6,7,12,14. However, it’s worth noting that 25.5% of the patients were ER+/PR− IBC in our study and this proportion is much higher than that of the whole breast cancer population, which may partly explain the worse outcome of IBC. Keeping with previous study3, 87.8% of the patients already had lymph node involved and 39.3% of the patients were distant metastasis at the initial diagnosis of breast cancer, which demonstrated the aggressive biological behavior of IBC. On accounting of higher proportion of patients with ER+/PR− phenotype and more aggressive biological characteristics, it is very necessary to figure out the effect of PR status on IBC.
As previous studies6,7,15, the loss of PR also predicted unfavorable biological characteristics in IBC. In our study, the patients with ER+/PR− phenotype were prone to be poor histological grade (III-IV), which often predicts worse survival16,17. In addition, the tendency of distant metastasis for ER+/PR− phenotype differed from ER+/PR+ phenotype. Consistent with our previous study15, more liver metastasis happened to patients with ER+/PR− phenotype than patients with ER+/PR+ phenotype in IBC, which indicates greater propensity of visceral metastasis for ER+/PR− phenotype. However, more bone metastasis occurred to patients with ER+/PR+ phenotype than patients with ER+/PR− phenotype, which demonstrated a pattern of bone metastatic spread typically attributed to hormone receptor-positive breast cancer18.
Although the prognosis of breast cancer has been greatly improved with the advent of various systemic treatments, the survival of IBC was still far from satisfaction. Consistent with previous studies19,20, the survival for IBC was very poor in our research (5-year BCSS and OS rates, 47.3% and 42.5%, respectively). Given the poor prognosis for IBC, previous researches tried to find the risk factors and demonstrated many independent predicted factors, such as race, lymph node ratio, AJCC stage, histological grade, ER status, PR status, HER2 status, surgery status, and radiotherapy status16,19. Although those researches have demonstrated that PR-negative status contributes to worse outcome for IBC, the difference of prognosis between ER+/PR− phenotype and ER+/PR+ phenotype is still unknown and needed to be further verified because worse outcome of PR-negative cohort mainly resulted from patients with triple-negative IBC in their study. Therefore, we excluded triple-negative IBC and compared the prognosis between ER+/PR− phenotype and ER+/PR+ phenotype in IBC by those two cohorts. As shown in our study, the patients with ER+/PR− phenotype exhibited significant worse survival than patients with ER+/PR+ phenotype, especially in the patients with younger age, lower histological grade, lymph node involved and distant metastasis. Due to the worse clinicopathologic features and prognosis for patients with ER+/PR− phenotype, the loss of PR has aroused wide attention from scholars. The main researches focused on genomics changes, such as PR promoter hypermethylation or loss of heterozygosity at the PR gene locus21,22. A recent study illuminated that almost 20% of the patients with ER+/PR− and HER2-negative were non-luminal-like and didn’t benefit from sufficient endocrine therapy12, which partly explains the worse outcome for IBC with ER+/PR− phenotype. As mentioned above, higher percentage of patients presented PR loss in IBC and worse survival were seen in those patients. Therefore, the patients with ER+/PR− phenotype belonging to non-luminal-like IBC should also be identified and more effective treatments should be performed on them, such as cyclin-dependent kinase 4/6 inhibitors. In addition to PR status, older age and black race were also prognostic risk factors for patients with ER+ and HER2-negative IBC. Poor histological grade and visceral metastasis were recognized as poor prognostic factors for IBC16,19, which was also demonstrated by our study. As shown above, the patients with ER+/PR− IBC presented poor histological grade and more visceral metastasis, which may also explain the worse outcomes for patients with ER+/PR− phenotype in our study. Chemotherapy, surgery and radiation are the indispensable approaches of trimodality treatment23, and all of these could significantly improve the survival for IBC in our study.
The limitations of this study must be clarified. First, some bias can’t be avoided for the nature of retrospective study. Thus, multivariable Cox proportional hazards model and subgroup analysis were performed to adjust for confounding effects as much as possible. Second, the information about endocrine therapy can’t be acquired from the database, which impeded the further analysis about the effectiveness of endocrine therapy. Nevertheless, most of the patients should have received enough endocrine therapy for the widespread of standard treatment in the United States. Third, because the SEER database didn’t collect the follow-up information about local recurrence and distant metastasis, we can’t analyze the recurrence-free survival and distant disease-free survival. However, our study is the first one that used the relatively large cohort to estimate the influence of PR status on patients with IBC. It illuminated the significant discordance of clinicopathological features and prognosis between ER+/PR− phenotype and ER+/PR+ phenotype in IBC, which indicated the necessity that stronger treatments should be applied to patients with ER+/PR− IBC.
Conclusions
More than 25% of the patients presented loss of PR expression among the ER-positive and HER2-negative IBC. Poor prognostic factors were prone to occur in IBC patients with ER+/PR− phenotype than ER+/PR+ phenotype, such as higher histological grade (III–IV) and liver metastasis. More effective treatments should be applied to IBC patients with ER+/PR− phenotype because significant worse outcomes were seen in those patients compared with ER+/PR+ phenotype.
Data availability
The datasets presented in this study can be found in online repository: the Surveillance, Epidemiology, and End Results (SEER) database (https://seer.cancer.gov).
Abbreviations
- HoR:
-
Hormone receptor
- PR:
-
Progesterone receptor
- ER:
-
Estrogen receptor
- HER2:
-
Human epidermal growth factor receptor 2
- IBC:
-
Inflammatory breast cancer
- BCSS:
-
Breast cancer-specific survival
- OS:
-
Overall survival
- SEER:
-
Surveillance, epidemiology and end results
- CIs:
-
Confidence intervals
- BCS:
-
Breast-conserving surgery
- Ref:
-
Reference
- HR:
-
Hazard ratios
References
Chang, S. et al. Inflammatory breast carcinoma incidence and survival: The surveillance, epidemiology, and end results program of the national cancer institute, 1975–1992. Cancer 82, 2366–2372 (1998).
Hance, K. W. et al. Trends in inflammatory breast carcinoma incidence and survival: The surveillance, epidemiology, and end results program at the national cancer institute. J. Natl. Cancer Inst. 97, 966–975 (2005).
Walshe, J. M. & Swain, S. M. Clinical aspects of inflammatory breast cancer. Breast Dis. 22, 35–44 (2005).
Rueth, N. M. et al. Underuse of trimodality treatment affects survival for patients with inflammatory breast cancer: An analysis of treatment and survival trends from the national cancer database. J. Clin. Oncol. 32, 2018–2024 (2014).
Van Asten, K. et al. Prognostic value of the progesterone receptor by subtype in patients with estrogen receptor-positive, HER-2 negative breast cancer. Oncologist 24, 165–171 (2019).
Dauphine, C. et al. Single Hormone receptor-positive breast cancers have distinct characteristics and survival. Ann. Surg. Oncol. 27, 4687–4694 (2020).
Li, Y. et al. Clinicopathological characteristics and breast cancer-specific survival of patients with single hormone receptor-positive breast cancer. JAMA Netw. Open 3, e1918160 (2020).
Bertucci, F. et al. Gene expression profiling identifies molecular subtypes of inflammatory breast cancer. Cancer Res. 65, 2170–2178 (2005).
Zell, J. A. et al. Prognostic impact of human epidermal growth factor-like receptor 2 and hormone receptor status in inflammatory breast cancer (IBC): Analysis of 2014 IBC patient cases from the California cancer registry. Breast Cancer Res. 11, R9 (2009).
Jiao, D. et al. Comparison of survival in non-metastatic inflammatory and other T4 breast cancers: A SEER population-based analysis. BMC Cancer 21, 138 (2021).
Hammond, M. E. et al. American society of clinical oncology/college of american pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J. Clin. Oncol. 28, 2784–2795 (2010).
Liu, X. Y. et al. Genomic landscape and endocrine-resistant subgroup in estrogen receptor-positive, progesterone receptor-negative, and HER2-negative breast cancer. Theranostics 8, 6386–6399 (2018).
Lv, M. et al. Clinical features and survival of single hormone receptor-positive breast Cancer: A population-based study of 531,605 patients. Clin. Breast Cancer 20, e589–e599 (2020).
Zheng, H. et al. Estrogen receptor-negative/progesterone receptor-positive and her-2-negative breast cancer might no longer be classified as hormone receptor-positive breast cancer. Int. J. Clin. Oncol. 27(7), 1145–1153 (2022).
Luo, Y. et al. Single progesterone receptor-positive phenotype has the similar clinicopathological features and outcome as triple-negative subtype in metastatic breast cancer. Front. Oncol. 13, 1029648 (2023).
Xu, F. et al. Nomograms for estimating cause-specific death rates of patients with inflammatory breast cancer: A competing-risks analysis. Technol. Cancer Res. Treat. 20, 15330338211016372 (2021).
Kridis, W. B. et al. Prognostic factors in inflammatory breast cancer: A single-center study. Breast Dis. 41, 461–469 (2022).
Smid, M. et al. Subtypes of breast cancer show preferential site of relapse. Cancer Res. 68, 3108–3114 (2008).
Zhang, H. et al. Nomogram for predicting cancer specific survival in inflammatory breast carcinoma: A SEER population-based study. PeerJ. 7, e7659 (2019).
Cristofanilli, M. et al. Inflammatory breast cancer (IBC) and patterns of recurrence: Understanding the biology of a unique disease. Cancer 110, 1436–1444 (2007).
Lapidus, R. G., Nass, S. J. & Davidson, N. E. The loss of estrogen and progesterone receptor gene expression in human breast cancer. J. Mammary Gl. Biol. Neoplasia. 3, 85–94 (1998).
Winqvist, R. et al. Loss of heterozygosity for chromosome 11 in primary human breast tumors is associated with poor survival after metastasis. Cancer Res. 55, 2660–2664 (1995).
Newman, A. B. & Lynce, F. Tailoring treatment for patients with inflammatory breast cancer. Curr. Treat Options Oncol. 24, 580–593 (2023).
Acknowledgements
We would like to thank the SEER program for providing open access to the database.
Funding
This research was supported by grant from North Sichuan Medical College Scientific Research and Development Project (CBY22-QNA34) and Guigang Science and Technology Project (GKG2000043).
Author information
Authors and Affiliations
Contributions
Y.L., X.Z., and L.H. conceived and designed this study. Y.L., Q.L., L.Z., and C.P. collected and analyzed the data. Y.L., Q.L., J.F., X.X. and S.Q. organized the manuscript. Y.L, X.Z., and L.H. reviewed the paper and revised the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luo, Y., Li, Q., Fang, J. et al. ER+/PR− phenotype exhibits more aggressive biological features and worse outcome compared with ER+/PR+ phenotype in HER2-negative inflammatory breast cancer. Sci Rep 14, 197 (2024). https://doi.org/10.1038/s41598-023-50755-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-50755-4
- Springer Nature Limited